Abstract
The proteoglycan-rich extracellular matrix (ECM) directly associated with the cells of articular cartilage is anchored to the chondrocyte plasma membrane via interaction with the hyaluronan receptor CD44. The cytoplasmic tail of CD44 interacts with the cortical cytoskeleton. The objective of this study was to determine the role of the actin cytoskeleton in CD44-mediated matrix assembly by chondrocytes and cartilage matrix retention and homeostasis. Adult bovine articular cartilage tissue slices and isolated chondrocytes were treated with latrunculin or cytochalasin. Tissues were processed for histology and chondrocytes were examined for CD44 expression and pericellular matrix assembly. Treatments that disrupt the actin cytoskeleton reduced chondrocyte pericellular matrix assembly and the retention of proteoglycan within cartilage explants. There was enhanced detection of a neoepitope resulting from proteolysis of aggrecan. Cytoskeletal disruption did not reduce CD44 expression, as monitored by flow cytometry, but detergent extraction of CD44 was enhanced and hyaluronan binding was decreased. Thus, disruption of the cytoskeleton reduces the anchorage of CD44 in the chondrocyte membrane and the capacity of CD44 to bind its ligand. The results suggest that cytoskeletal disruption within cartilage uncouples chondrocytes from the matrix, resulting in altered metabolism and deleterious changes in matrix structure.
C
The cortical cytoskeleton of chondrocytes helps to maintain phenotypic stability, organize the distribution of the cell organelles, and interact with the membrane proteins. On ligand binding, the interaction of certain membrane receptors with the cytoskeletal proteins sometimes brings about a change in the organization of the cytoskeleton. The punctate peripheral staining of actin observed in chondrocytes increased in organization with matrix assembly during culture in agarose culture (Idowu et al. 2000). The possible regulation of the cytoskeleton by differences in the dynamic loading within the layers of articular cartilage has also been proposed (Langelier et al. 2000). Modulating the association between the cytoskeleton and CD44 may alter the function of CD44. Our results show that hyaluronan binding to chondrocytes upregulates the phosphorylation of CD44 (Knudson et al. 1997), which could influence its interaction with cytoskeletal components (Isacke 1994). Using differential detergent extraction, CD44 was localized predominantly in the cytoskeleton-associated pool from matrix-intact chondrocytes. In matrix-depleted chondrocytes, a decrease in this pool, with a parallel increase in the detergent-soluble pool, was detected (Nofal and Knudson 1999). In addition to the retention of proteoglycans of the pericellular matrix to the plasma membrane, CD44–hyaluronan binding may stabilize chondrocyte cell shape by the interaction of CD44 with the cortical cytoskeleton. Conversely if cytoskeletal elements regulate CD44–hyaluronan binding, disruption of the cytoskeleton may perturb matrix retention. To examine this hypothesis, cartilage matrix assembly and retention were monitored in the presence or absence of cytochalasin (Brown and Benya 1988) or latrunculin (Spector et al. 1989). Cytochalasin binds to the (+) of actin filaments while latrunculin binds actin monomers. Both cause destabilization and depolymerization of the actin network.
The objective of this study was to determine the role of the actin cytoskeleton in CD44–hyaluronan-mediated matrix assembly by chondrocytes and cartilage matrix retention and homeostasis. Adult bovine articular chondrocytes cultured in alginate beads and adult bovine articular cartilage tissue slices in explant cultures were treated with latrunculin A or dihydrocytochalasin B. CD44 expression determined by flow cytometry, the detergent solubility of CD44, and hyaluronan binding were examined. Pericellular matrix assembly was monitored and histological assessment of cartilage was performed. Although disruption of the cytoskeleton did not alter cell surface CD44 expression, detergent extraction of CD44 increased. In addition, there was a reduction in hyaluronan binding, pericellular matrix assembly, and cartilage matrix retention. Therefore, disruption of the cytoskeleton is apparently another mechanism by which uncoupling chondrocytes from the matrix results in changes in chondrocyte metabolism and deleterious changes in matrix structure. The direct interaction of chondrocytes with their matrix is vital for maintenance of cartilage homeostasis.
Materials and Methods
Cell Culture
Bovine cartilage was collected from the metacarpophalangeal joints of 18-month-old animals obtained from a local slaughterhouse. For explant cultures, the cartilage was dissected into ∼1-cm2 slices; 25 mg was the average wet weight per tissue slice. Alternatively, the chondrocytes were isolated with a sequential 1-hr treatment with 0.2% Pronase (Calbiochem; La Jolla, CA) followed by an overnight treatment with 0.025% collagenase P (Boehringer-Mannheim; Indianapolis, IN) in Ham's F-12 medium (Cellgro; Mediatech, Washington, DC) supplemented with 5% FBS (Summit Biotechnology; Fort Collins, CO) (Aydelotte and Kuettner 1988). The cell suspension was then passed through a 70-μm nylon cell strainer (Falcon 2350; Becton-Dickinson, Lincoln Park, NJ) to obtain a single cell suspension. The chondrocytes were cultured in alginate beads. A 1.2% solution of alginate (Kelco; Chicago, IL) was prepared in 0.15 M NaCl and a suspension of 4 × 106 cells/ml was loaded into a syringe. The alginate-cell suspension was then passed through a 22.5-gauge needle into a 102 mM CaCl2 solution to initiate alginate polymerization and generate beads containing 4.4 × 104 cells per bead. The beads were kept in this solution for 7 min, then washed three times with 0.15 M NaCl. The chondrocytes were cultured in Dulbecco's modified Eagle's medium (DMEM)–F-12 (50:50 mix) (Mediatech), supplemented with 10% FBS and 2.5 μg/ml ascorbic acid for 5 days before any experimental manipulation. To release the chondrocytes, the beads were transferred to 55 mM sodium citrate in 150 mM NaCl for 45 min at RT, which results in the depolymerization of the alginate (Hauselmann et al. 1992).
Effects of Latrunculin A and Dihydrocytochalasin B Treatment on Cell Surface CD44 Expression
Bovine chondrocytes in alginate beads were cultured either with 10 μg/ml dihydrocytochalasin B (DHCB; Sigma, St Louis MO) or 0.2 μg/ml latrunculin A (Calbiochem) or the vehicle buffer for various time periods: 3 hr, 1, 2, 5, 6, and 7 days. After the respective incubation period, the cells were fixed with 1% paraformaldehyde (Fisher; Pittsburgh, PA) in PBS, pH 7.4, for 1 hr at 4C. The alginate beads were then depolymerized with 55 mM sodium citrate and the cells were rinsed twice with PBS, incubated with 0.2 M glycine-PBS for 5 min, and blocked with 4% non-fat dry milk in PBS for 1 hr. The cells were then probed for CD44 with biotinylated monoclonal antibody IM7.8.1 (Pharmingen; San Diego, CA) 2.5 μg/ml per 106 cells, for 1 hr at 4C, which was detected with 2.5 μg/ml streptavidin-phycoerythrin (Gibco BRL; Life Technologies, Rockville, MD). The cells were then analyzed by flow cytometry as described (Aguiar et al. 1999) using an Epics Profile Analyzer II (Coulter; Hialeah, FL) with Profile 2.6 software. Immunofluorescence was detected using excitation at 565 nm and detection of the emission at 578 nm. All of the cells were analyzed using the fixed parameters for positive CD44 staining relative to the background peak of the secondary reagent only. Cells were gated according to size [forward scatter (FS)] and granularity [light side angle scatter (LSS)]. Log fluorescence channel vs cells per channel was plotted for 10,000 cells.
Detergent Extraction of CD44 on Cytoskeletal Disruption
Bovine chondrocytes were plated into 24-well plates (Falcon 3047) at 2 × 105cells per well. The cells were then incubated with either 0.5 μg/ml latrunculin A, 10 μg/ml DHCB, or the vehicle, for 1 hr at 4C. After the incubation period, the cells were extracted with 0.1% Nonidet P-40 (NP-40; Calbiochem) for 30 min on ice. The NP-40 buffer consisted of 50 mM Tris-HCl (Bio-Rad; Hercules, CA), 150 mM NaCl, 5 mM EDTA, 100 mM ∊-amino-N-caproic acid, 2 ng/ml pepstatin A, 10 μg/ml leupeptin, 10μg/ml aprotinin, and 5 mM benzamidine (all from Sigma) at a pH of 7.4 (Camp et al. 1991). The cells were then fixed with buffered 1% paraformaldehyde for 30 min at RT and subsequently blocked with 4% normal goat serum in PBS overnight at 4C. Half of the cells were sequentially probed for CD44 with 2.5 μg/ml IM7.8.1 for 3 hr at RT, followed by the secondary reagents provided in the Rat Vectastain Elite ABC kit (Vector Laboratories; Burlingame, CA) at the recommended dilutions for 1 hr at RT. Detection of peroxidase was performed with SIGMA FAST DAB Peroxidase Substrate Tablet Set (Sigma). Parallel cultures not incubated with the anti-CD44 antibody were probed only with the secondary reagents. The cells were viewed and photographed under identical conditions using a Zeiss Axiovert 25 microscope.
Hyaluronan Binding by Chondrocytes
Fluorescein hyaluronan (fl-HA) was prepared as previously reported (Hua et al. 1993). Briefly, to 60 ml of 1 mg/ml of hyaluronan (Sigma, grade I) in 30% dimethyl sulfoxide (DMSO) (Calbiochem) in water was added 500 μl of DMSO that contained 25 mg fluoresceinamine, 25 μl acetaldehyde, and 25 μl cyclohexylisocyanide (all from Sigma). The pH was adjusted to 4.5 and the solution was incubated for 5 hr at RT in the dark. The hyaluronan was precipitated three times with 1.3% C2H3O2K in 95% ethanol, dialyzed extensively against distilled water at 4C, lyophilized, and stored at −20C.
Bovine chondrocytes were released from the alginate beads, suspended 5 × 105 cells/ml, and treated with 5 U/ml Streptomyces hyaluronidase (Sigma) in DMEM supplemented with 10% FBS for 60 min at 37C to remove endogenous ligand, hyaluronan, and expose total CD44 receptors. After the enzyme treatment, the cells were rinsed and then incubated for 3 hr at 37C with 10 μg/ml DHCB, 1 μg/ml latrunculin A, or vehicle buffer. At the end of this pre-incubation period, the cells were incubated for 80 min at 4C with 200 μg/ml fl-HA cells in the continued presence of the above agents. The cells were rinsed twice, counted, and resuspended in DMEM to give a final cell concentration of 3.3 × 105 cells/ml. These cells were then transferred into a 96-well plate at a volume of 200 μl per condition in triplicate for analysis on a fluorescent plate reader (Bio-Tek FL500; Winooski, VT). In parallel, cell surface CD44 expression on pretreated chondrocytes was determined by flow cytometry as described above.
Pericellular Matrix Retention or Assembly on Cytoskeletal Disruption
Chondrocytes were cultured in alginate for 5 days and then treated either with 0.2 μg/ml latrunculin A, 10 μg/ml DHCB, or vehicle buffer for 15 hr. After release from the alginate beads, the cells in suspension were “splatted” onto a six-well plate (Falcon 3046) at a density of 1 × 105 cells per well by centrifugation at 550 × g for 10 min (Maleski and Knudson 1996) and the pericellular matrix visualized with the particle exclusion assay. Alternatively, chondrocytes were cultured in alginate beads for 5 days, released from the beads, and plated overnight into 35-mm dishes (Falcon 3001) at 1 × 105 cells per dish. The cells were initially treated with 5 U/ml Streptomyces hyaluronidase for 70 min at 37C, rinsed three times, and allowed to recover in the presence or absence of 0.5 μg/ml latrunculin A or 10 μg/ml DHCB in DMEM with 10% FBS at 37C for 1–3 days. To visualize the pericellular matrix by the particle exclusion assay (Clarris and Fraser 1968), the medium is removed and 750 μl of a suspension of formaldehyde-fixed horse erythrocytes at 108 particles/ml in 0.1% BSA/PBS is added. The particles settle within 15–20 min and reveal the halo of matrix surrounding the living chondrocytes (Knudson and Toole 1985). The relative size of the pericellular matrix was calculated as the ratio of the matrix area to the cell area as previously reported (Knudson et al. 1995). Data are reported as the average percentage of cells for which the matrix:cell area ratios were either >2.0, between 1.5 and 2.0, or <1.5.
Maintenance of the Extracellular Matrix in Cartilage Explants on Cytoskeletal Disruption
Safranin O Staining of Cartilage Tissue. Bovine cartilage slices were allowed to equilibrate for 24 hr in DMEM with 10% FBS, transferred into individual wells of a 96-well plate (Falcon 3072), and cultured in the presence or absence of 0.2 μg/ml latrunculin A or 10 μg/ml DHCB. Each well contained the explant cartilage and 200 μl of the respective medium condition, which was changed daily. Explants were collected after 1, 2, 4, 5, 7, 8, 12, or 13 days in culture. Some of the cultures after 7 days of treatment were cultured for an additional 3 days in the absence of latrunculin A or DHCB. The cartilage was fixed in Perfix histological fixative (Fisher) containing 0.1% safranin O (Fisher) for 2 hr at 4C. After fixation the tissue was rinsed twice with PBS and quenched in PBS containing 2 mM glycine, 5% glycerol, and 1% BSA. Explants were then embedded in paraffin. Ten-μm sections were stained with fast green and safranin O (Williams et al. 1993). After mounting, the sections were photographed under identical conditions (Knudson et al. 2000) with a Zeiss Axiovert 25 microscope.
Detection of the NITEGE Neoepitope in Cartilage Tissue. The same cartilage explants were also used to stain for the NITEGE neoepitope of aggrecan (Lark et al. 1997). After the explants were sectioned, they were treated with 0.1 U/ml chondroitinase ABC (Sigma) for 2 hr at 37C (Nishida et al. 2000a). The sections were then blocked with 2% normal goat serum for 24 hr, followed by a 24-hr incubation with a 1:500 dilution of the anti-NITEGE antiserum (Lark et al. 1997), kindly provided by Dr. D. Visco (Merck Research Laboratories; Rahway, NJ). The secondary reagents from an anti-rabbit Vectastain Elite kit were then used at the recommended dilutions. Detection of peroxidase was performed with SIGMA FAST DAB Peroxidase Substrate Tablet Set. The negative control consisted of omission of the primary antibody. The sections were viewed and photographed under identical conditions (Knudson et al. 2000) using a Zeiss Axiovert 25 microscope.
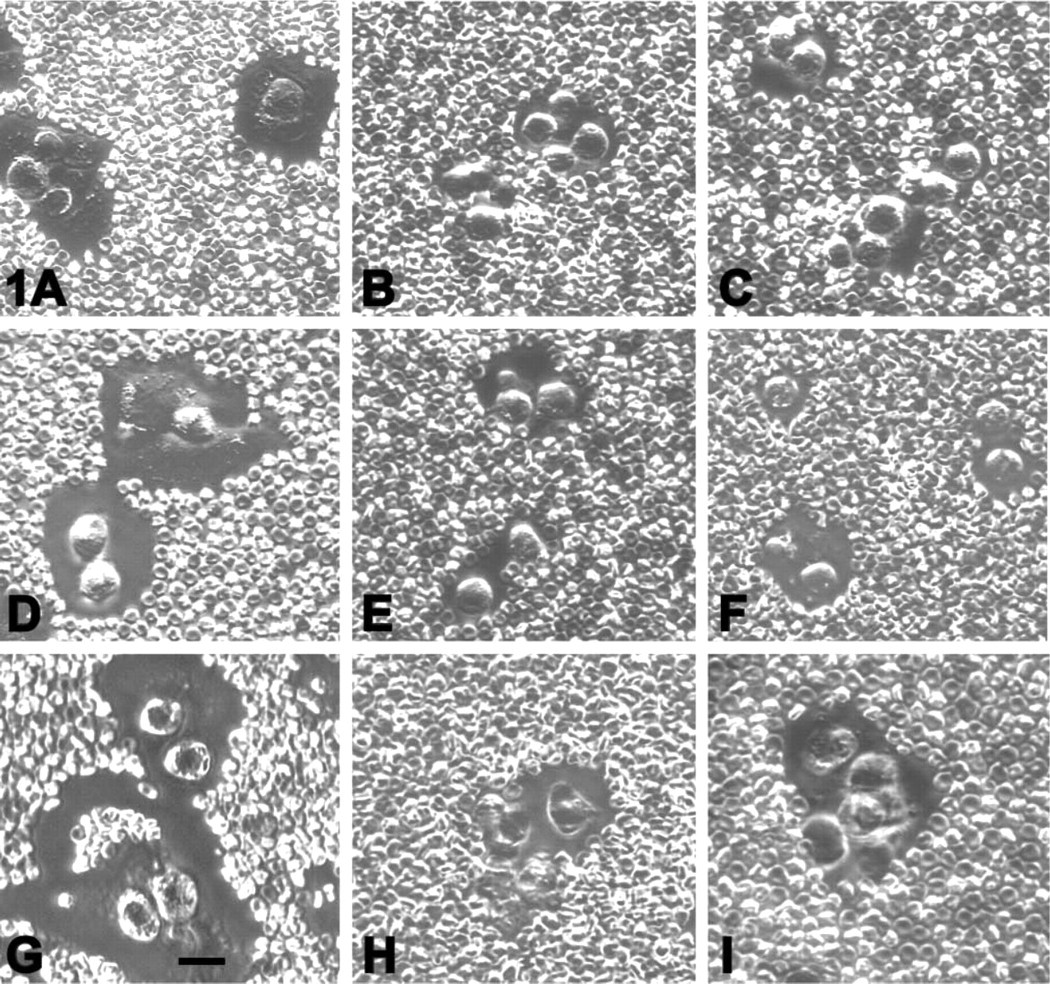
Pericellular matrix assembly by chondrocytes after cytoskeletal disruption. Chondrocytes were treated with Streptomyces hyaluronidase and then incubated in the absence (
Cell Viability Assay
Cell viability was determined by co-incubation of chondrocytes in alginate beads or cartilage slices in 2 μM calcein AM and 4 μM ethidium homodimer-1 (EthD-1) in PBS (LIVE/DEAD Kit, L-3224; Molecular Probes, Eugene, OR) for 40 min, followed by rinsing in Hanks' balanced salt solution (GIBCO BRL.) Chondrocytes were released from the alginate beads and resuspended in a small volume of 10% glycerol in PBS, which was placed on a glass slide and cover-slipped. Cartilage was embedded in OCT (EM Sciences; Ft Washington, PA), and frozen sections placed on glass slides and warmed before viewing with a Nikon Eclipse E600. Uncultured cartilage slices were examined as a control. Dead cells (red nuclear fluorescence) were evaluated by the uptake of EthD-1 (ex/em = 495 nm/635 nm). Living cells were visualized by the fluorescence (green) of the calcein (ex/em = 495 nm/515 nm) as a result of endogenous esterase activity.
. Disruption of the cytoskeleton results in reduced matrix assembly a
After 3 days of matrix assembly in control medium or medium containing DHCB or latrunculin, the observed chondrocytes were grouped into three categories: cells with a matrix area:cell area ratio >2.0, cells with a matrix:cell area ratio between 1.5 and 2.0, and cells with a matrix:cell area ratio <1.5. The results represent the average percentage of cells in each of the three ranges, determined from three separate cultures for each culture condition (control n=195; +DHCB n=188; +latrunculin n=130).
Results
Chondrocyte Pericellular Matrix Assembly and Retention
Bovine articular cartilage chondrocytes were cultured for 5 days in alginate beads, in which the cells maintain a round morphology and synthesize an ECM. The pericellular matrix is dependent on a scaffold of hyaluronan and is removed by treatment with Streptomyces hyaluronidase (Knudson et al. 1996).
The ability of the cells after disruption of the cytoskeleton to re-assemble a pericellular matrix following Streptomyces hyaluronidase treatment was examined with the particle exclusion assay. Figure 1 shows the pattern of matrix assembly monitored at day 1 (Figures 1A–1C), day 2 (Figures 1D–1F), and day 3 (Figures 1G–1I) after Streptomyces hyaluronidase treatment. The progressive increase in pericellular matrix seen in control cells (Figures 1A, 1D, and 1G) was reduced in the presence of either cytochalasin (Figures 1B, 1E, and 1H) or latrunculin (Figures 1C, 1F, and 1I). After washout of the treatment reagents, matrix assembly progressed to that exhibited by controls (data not shown.) A morphometric analysis of the matrix:cell area ratio was determined at day 3 of matrix assembly (Table 1). Whereas only 7% of the control chondrocytes had a smaller pericellular matrix (ratio <1.5), ∼50% of the chondrocytes in the DHCB- or latrunculin-treated cultures exhibited a matrix:cell area ratio of <1.5. Conversely, whereas nearly 60% of the control chondrocytes exhibited a matrix:cell area ratio of >2.0, less than 10% of the chondrocytes in either experimental group exhibited these larger matrices. Chondrocyte viability after 3 days of treatment (Figure 2) was similar between DHCB-treated (Figure 2A), latrunculin-treated (Figure 2B), and untreated (Figure 2C) chondrocytes or after 5 days (data not shown) as revealed by the LIVE/DEAD cell assay.
Cell viability after DHCB or latrunculin A treatment. Chondrocytes were cultured for 3 days in the absence (

The ability of chondrocytes to maintain a pericellular matrix on cytoskeletal disruption. After 5 days of culture, chondrocytes were maintained in control medium (
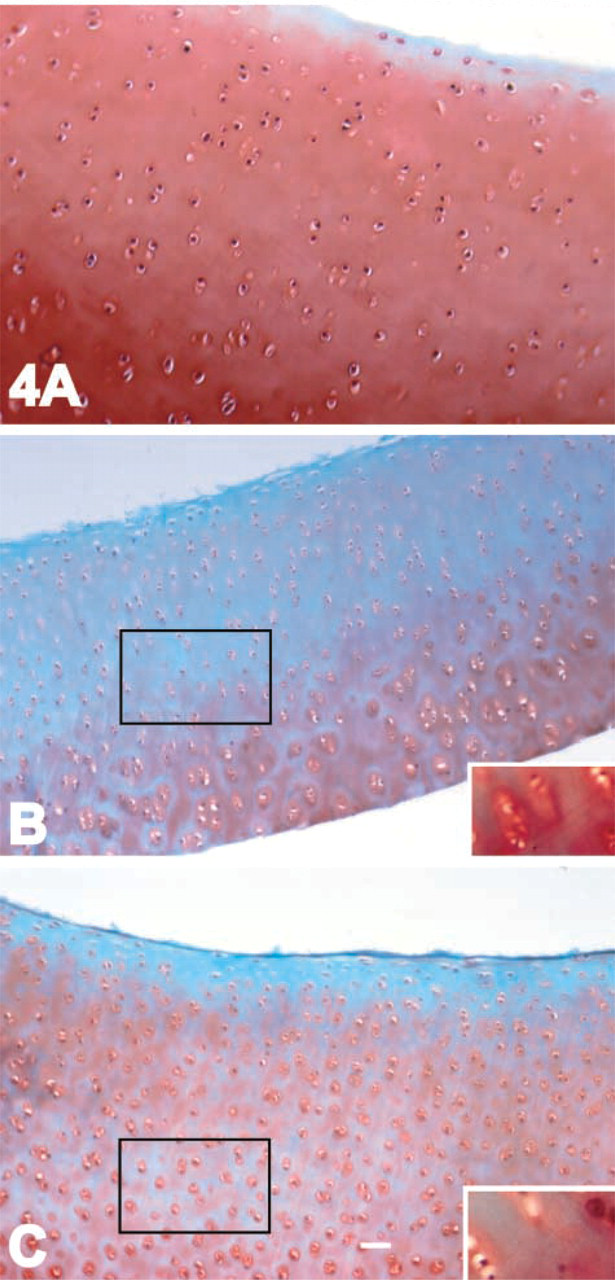
Proteoglycan retention in cartilage explants after cytoskeletal disruption. Bovine articular cartilage explants were cultured in the presence or absence of latrunculin A or DHCB. (
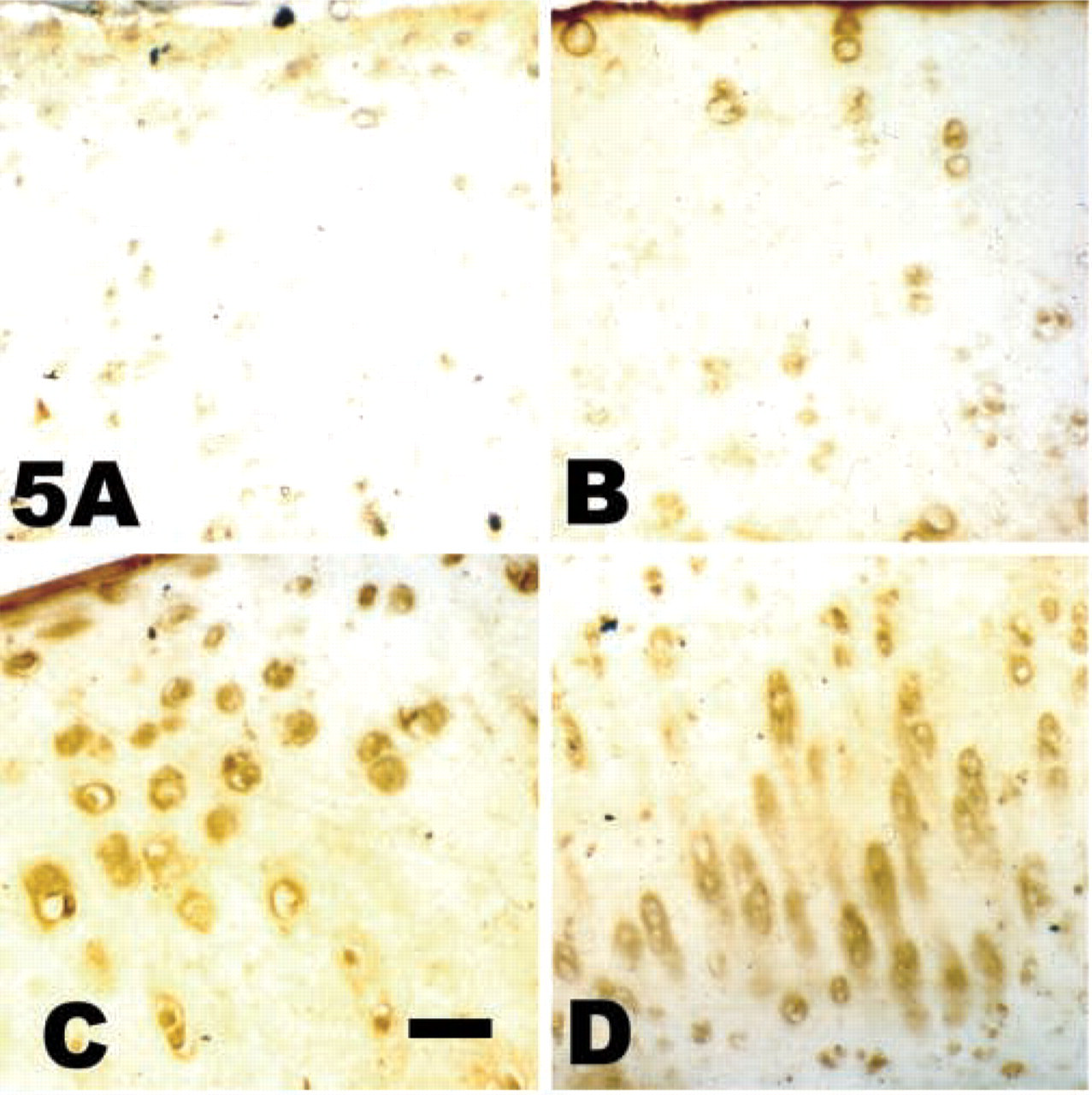
Detection of the NITEGE neoepitope in cartilage explants. Cartilage explants incubated in the presence or absence of DHCB or latrunculin A were analyzed for the presence of the NITEGE neoepitope. (
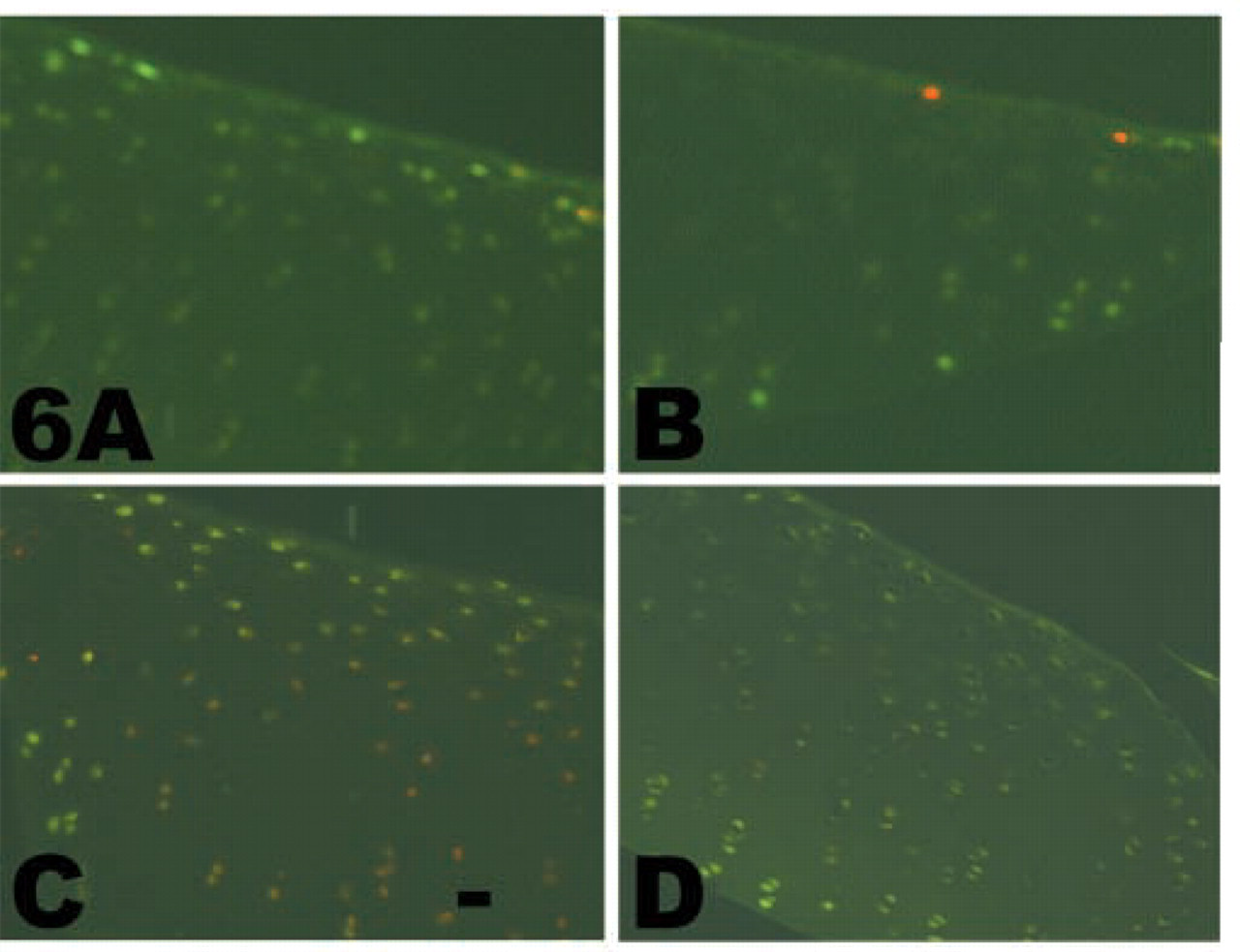
Cell viability in cartilage explants. Cartilage was analyzed for cell viability (green fluorescence). Red nuclear fluorescence reveals dead cells. (
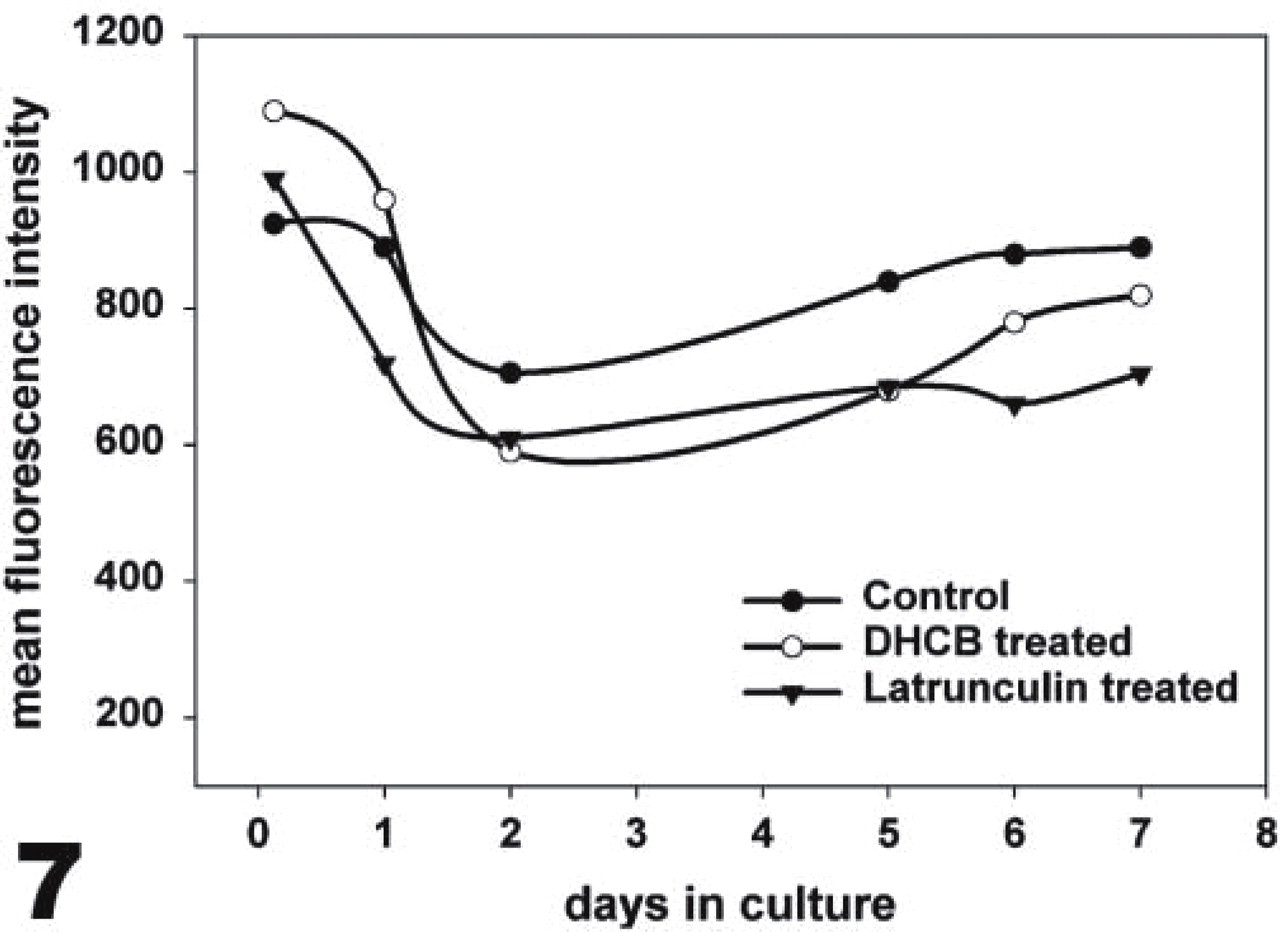
CD44 expression by bovine chondrocytes after cytoskeletal disruption. Chondrocytes were cultured in alginate beads in the absence (closed circles) or presence of DHCB (open circles) or latrunculin A (triangles) for 3 hr up through 7 days. CD44 was detected by flow cytometry; for each time interval, 10,000 cells were analyzed. This graph represents CD44 detection by the mean channel values at each of the conditions at the respective time points.
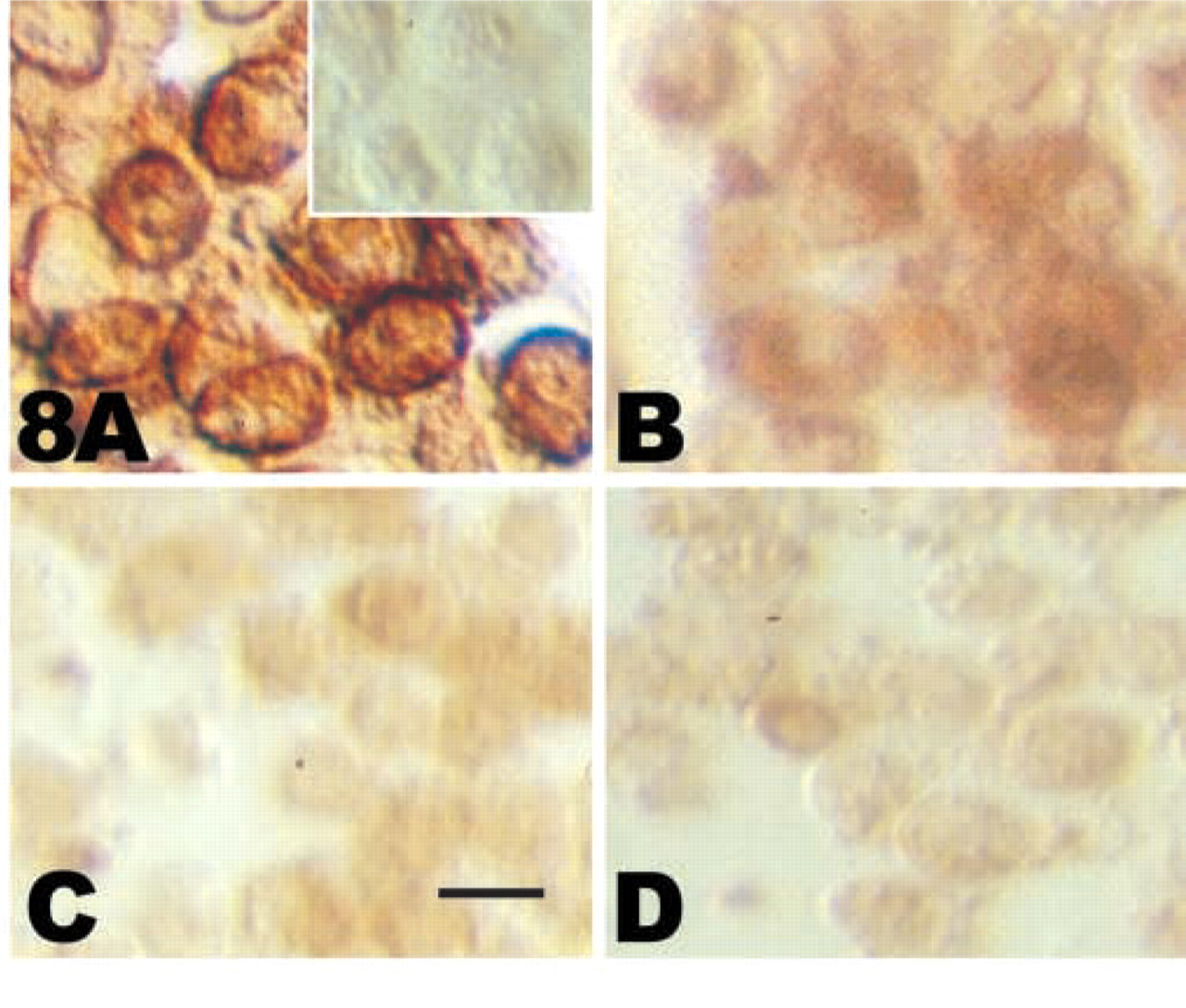
Immunodetection of the cytoskeleton-associated pool of CD44 after detergent extraction. Total CD44 on untreated control chondrocytes (
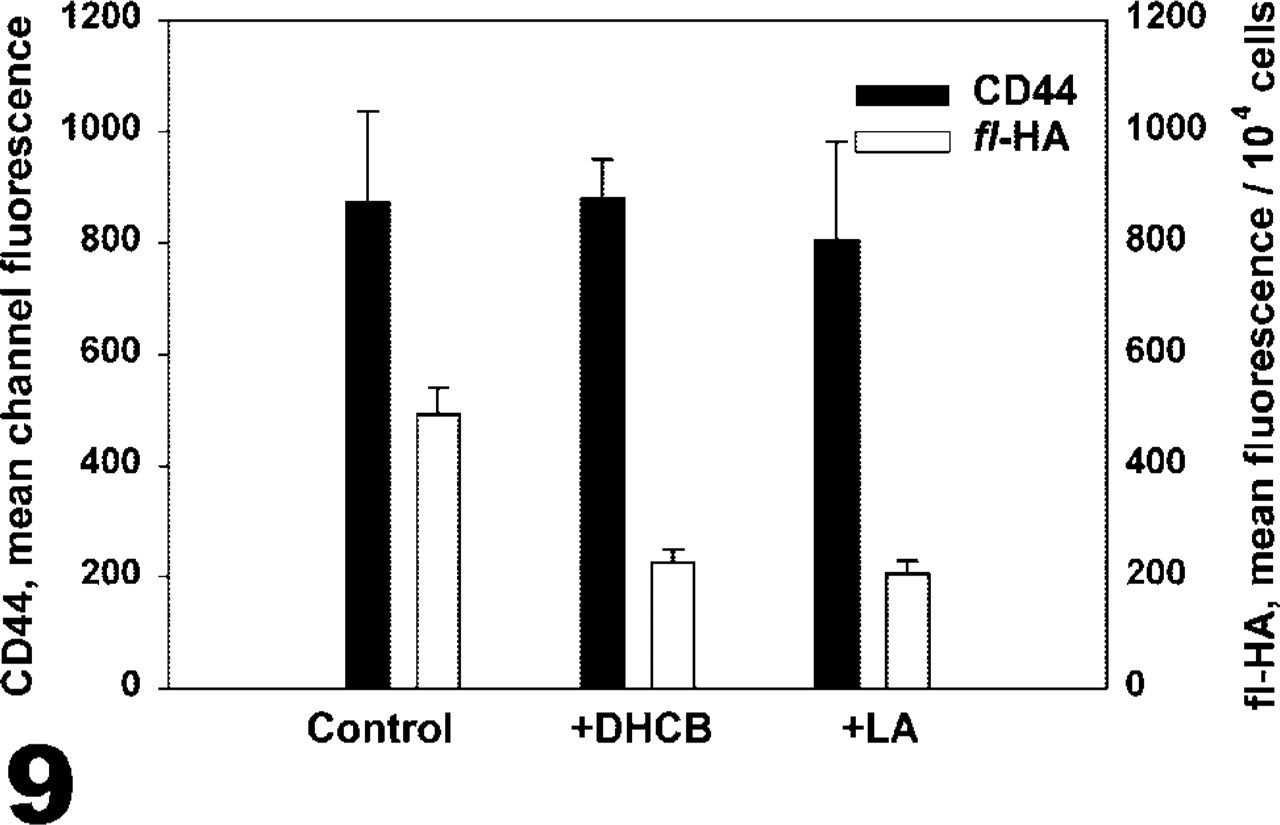
Fl-HA binding to bovine chondrocytes and CD44 expression. Chondrocytes were incubated in the presence or absence of DHCB or latrunculin A (LA) for 3 hr and CD44 cell surface expression was detected by flow cytometry (black bars). These cells were also incubated with fl-HA, and this binding was detected by fluorescence spectrometry (white bars). Although little change in CD44 expression was detected, fl-HA binding decreased after cytoskeletal disruption.
The ability of chondrocytes to maintain the existing pericellular matrix after treatment to disrupt the cytoskeleton was analyzed. Chondrocytes released from the beads after depolymerization of the alginate exhibit a matrix that can be visualized using the particle exclusion assay (Knudson et al. 1999). Figure 3A shows the pericellular matrix assembled during a 5-day culture period in alginate beads and retained by the untreated control cells. After 5 days of culture in alginate beads, the chondrocytes were treated for 15 hr with cytochalasin or latrunculin. The exclusion area around the control cells (Figure 3A) is greater than that around the cytochalasin-treated (Figure 3B) or latrunculin-treated (Figure 3C) cells. This indicates that the disruption of the cytoskeleton reduces pericellular matrix retention.
Cytoskeletal Disruption of Cells in Articular Cartilage
The results with isolated chondrocytes suggested that an intact actin cytoskeleton was critical to maintain chondrocyte-matrix interactions. To expand these studies to intact cartilage, latrunculin and cytochalasin treatments of explant cultures were evaluated. The chondrocytes in the latrunculin- or cytochalasin-treated cartilage appeared more compact. There was a decrease in the safranin O staining of both cytochalasin-treated (Figure 4B; 2 μg/ml, 2 days) and latrunculin-treated (Figure 4C; 10 μg/ml, 5 days) cartilage, indicating the loss of proteoglycan from these tissues compared to cultured controls (Figure 4A, 5 days). When the explants were allowed to recover for 3 days after a 5-day treatment, there appeared to be an increase in safranin O staining, especially in the area right around the cells (insets in Figures 4B and 4C shown at higher magnification, but from the middle zone of the cartilage indicated by the rectangles).
A decrease in safranin O staining could be due to decreased proteoglycan synthesis or increased turnover owing to the activity of matrix metalloproteinases. To investigate this later possibility, sections from the treated and untreated control explants were stained for the NITEGE neoepitope of aggrecan. Figures 5A and 5B are the untreated control tissue, with Figure 5A showing negative control for the immunohistochemistry and Figure 5B the NITEGE epitope. Figure 5C shows the DHCB-treated tissue and Figure 5D shows the latrunculin-treated tissue. There was a detectable level of NITEGE staining in all of the tissue sections, indicative of endogenous “aggrecanase” activity (Lark et al. 1997). The staining in the control cartilage is predominantly at the superficial layer of the cartilage. When the treated cartilage was compared to the control cartilage, NITEGE staining in the areas directly around the chondrocytes was detected.
Cell viability in the freshly isolated, uncultured cartilage slices (Figure 6A) was quite high, as detected by green cytosolic fluorescence. Cartilage explants cultured for 3 or 5 days were also evaluated for chondrocyte viability. Although some dead cells (red nuclear fluorescence) could be detected in all sections, no increase in the number of dead cells was detected at day 3 (data not shown) or day 5 of culture in the absence (Figure 6B) or presence of DHCB (Figure 6C) or latrunculin (Figure 6D).
CD44 Expression, Association with the Cytoskeleton, and Hyaluronan Binding
Because CD44 is a critical receptor in chondrocyte-matrix interactions, cell surface expression of CD44 was monitored by flow cytometry. Five-day alginate bead cultures of chondrocytes were cultured in the presence or absence of either DHCB or latrunculin A for an additional 3 hr to 7 days. It appears that both of the agents used had similar minimal effects on the level of cell surface CD44 (Figure 7). The treated cells gave, at almost any given incubation time, less then a 25% decrease in the levels of cell surface CD44 expression. For DHCB the average decrease in cell surface CD44 was 9.2% and for latrunculin the average decrease in cell surface CD44 was 19.4%.
Immunocytochemistry combined with the differential detergent extraction protocol was used to visualize the cytoskeleton-associated pool of CD44 after disruption of the cytoskeleton. Figure 8A shows representative results. Figure 8A shows staining of total cell surface CD44 on untreated control chondrocytes. The inset in Figure 8A shows the negligible background staining with the omission of the primary antibody and using the secondary reagents only. No discernible differences in total cell surface staining between untreated control, cytochalasin-treated, or latrunculin-treated chondrocytes were noted (data not shown.) Figure 8B shows the residual CD44 on untreated control cells after extraction with 0.1% NP-40. There is a decrease in epitope staining due to the extraction of CD44 by the detergent, and the residual staining represents the cytoskeleton-associated pool of CD44. Chondrocytes treated with DHCB (Figure 8C) or latrunculin (Figure 8D) for 3 hr were then extracted with 0.1% NP-40 before CD44 detection. The staining intensity for the cytoskeleton-associated pool of CD44 was reduced after treatments with these agents, suggesting that in the absence of an intact actin cytoskeleton, CD44 is more readily extracted from the plasma membrane with detergent. These results help to verify that there are in fact two pools of CD44 in the chondrocytes and that the cytoskeleton-associated pool of CD44 is dependent on the stability of the cytoskeleton.
The relationship between CD44–cytoskeletal interactions and the functional aspect of hyaluronan binding by CD44 was explored. After the cytoskeleton of the chondrocytes was disrupted for 3 hr, the cells were incubated with fl-HA and the amount of binding to these chondrocytes was determined in comparison to untreated controls. As shown in Figure 9, there was a decrease in the amount of fl-HA that was bound after cytoskeletal disruption compared to the control cells. The relative fluorescence values reflect a 54% decrease in the amount of bound fl-HA in the DHCB-treated cells and a 58% decrease with the latrunculin-treated cells. When cells from the same three populations were analyzed by flow cytometry, there was little difference in CD44 detected after cytoskeleton disruption compared to untreated control (Figure 9). These results indicate that the association between CD44 and the cytoskeleton plays a role in the capacity for the interaction between CD44 and its extracellular ligand hyaluronan.
Discussion
Chondrocyte-matrix interactions and the regulation of ECM assembly are key to cartilage matrix homeostasis. Initial matrix assembly occurs in a zone immediately adjacent to the chondrocyte cell surface. Cell-matrix interactions, mediated by receptors, promote optimal assembly and signal feedback regulation for biosynthesis and/or turnover. This cell-associated matrix can be visualized as a distinct structure within sections of cartilage (Schenk et al. 1986; Hunziker 1992) and surrounding individual chondrocytes grown in vitro (Knudson and Toole 1985; Knudson et al. 1996). Previously we reported a cell isolation protocol in which a brief treatment of embryonic cartilage with purified collagenase in serum-containing medium liberates chondrocytes from the tissue. These freshly isolated, non-cultured chondrocytes retain matrix, as visualized by the particle exclusion assay (Maleski and Knudson 1996), thus suggesting that chondrocyte-matrix interactions occur in cartilage tissue and that a cell-associated matrix exists as an entity tethered to the chondrocyte in vivo, resistant to collagenase treatment and centrifugation. We have demonstrated that hyaluronan binding sites participate in the assembly and retention of the hyaluronan-dependent aggrecanrich portion of the pericellular matrix on chondrocytes in cell culture (Knudson 1993; Knudson et al. 1996). Treatment with Streptomyces hyaluronidase releases the pericellular matrix assembled in cell culture (Knudson 1993) or in vivo (Maleski and Knudson 1996) indicating a major structural role for hyaluronan.
Hyaluronan hexasaccharides (HA6) are a useful tool for probing cell surface hyaluronan receptor function. HA6 do not mediate or disrupt proteoglycan aggregate formation (Hardingham and Muir 1973; Hascall and Heinegard 1974) but, binding of HA6 to cell surface hyaluronan receptors does compete with binding by large molecular weight hyaluronan (Underhill and Toole 1979; Nemec et al. 1987; Lesley et al. 2000). The ability of the HA6 to “disengage” chondrocytes from their matrix in embryonic tibial cartilaginous anlagen (Maleski and Knudson 1996) showed that a chondrocyte in cartilage tissue is “engaged” with its ECM via hyaluronan-hyaluronan receptor interactions. In our previous studies, the effects of matrix-chondrocyte uncoupling by HA6 were examined in slices of intact adult articular cartilage (Knudson et al. 2000). Safranin O staining and immunohistochemistry detected loss of proteoglycan from HA6-treated cartilage tissues as well as aggrecanase activity, as detected by the increase of the aggrecanase neoepitope NITEGE (Lark et al. 1997). The data suggest that uncoupling chondrocytes from their matrix, outside-in signaling, results in critical changes in overall metabolism and loss of cartilage homeostasis.
Although disruption of the cytoskeleton did not alter cell surface CD44 expression, there was a reduction in hyaluronan binding, pericellular matrix assembly, and cartilage matrix retention. Even at the tissue level, the ability of the chondrocytes to retain the ECM was hindered when the cytoskeleton was disrupted. Therefore, change in the cytoskeleton is apparently another mechanism to uncouple chondrocytes from the matrix, resulting in modulation of chondrocyte metabolism and deleterious changes in matrix structure. However, after removal of the latrunculin or cytochalasin, the cells, both isolated and in cartilage, were able to reconstruct the matrix.
Maintenance of the chondrocyte phenotype, as well as initial cartilage differentiation, may depend on the close association of the cell with its matrix and on cell shape as modulated by the actin cytoskeleton (Daniels and Solursh 1991). Another paradigm is that the presence and/or composition of the ECM may regulate the interactions of receptors with elements of the cytoskeleton. There are two pools of CD44 on chondrocytes, as shown here, one that can be easily extracted with detergent and a second pool that is cytoskeleton-associated. Disruption of the f-actin components of the cytoskeleton reduced the proportion of CD44 retained in the cytoskeleton-associated pool. In other studies using this differential extraction technique, we observed that a greater proportion of CD44 in the plasma membrane was anchored to the cytoskeleton in matrix-intact chondrocytes as compared to matrix-depleted cells (Nofal and Knudson 1999). Therefore, it is possible that CD44 interaction with matrix enhances receptor clustering which, in turn, enhances interaction of CD44 with the intracellular cytoskeleton. The enhanced cytoskeletal linkage may lead to stabilization of CD44 at the cell surface, thus retarding its internalization.
Our previous studies demonstrated an increased turnover of CD44 from the chondrocyte cell surface after depletion of the hyaluronan-rich matrix using the enzyme Streptomyces hyaluronidase. In the reverse experiment, when high molecular weight hyaluronan was added back to the matrix-depleted chondrocytes, a partial inhibition of CD44 internalization resulted, returning the CD44 half-life to near baseline levels as seen on matrix-intact chondrocytes (Aguiar et al. 1999). These results suggest that CD44 occupancy with hyaluronan can affect CD44 residence time at the cell surface on chondrocytes. Similar results were observed for the expression of integrin α5β1 on fibroblasts. This integrin was internalized more rapidly in the absence of the fibronectin, while subsequent attachment to fibronectin substrate was found to stabilize the expression of cell surface α5β1 integrin (Dalton et al. 1995).
Our results show that destabilization of the actin network in chondrocytes results in decreased matrix assembly or retention. The spatial organization of CD44 at the cell surface, controlled via cytoskeletal interactions, may function to establish or regulate the structure of the pericellular matrix dependent on a hyaluronan scaffolding for aggrecan. On cytoskeletal disruption, the stability of the CD44 molecules at the cell surface and/or the pattern of distribution on the cell surface may be altered. These results are in accord with previous studies in which cytochalasin treatment of bovine endothelial cells (which express CD44v10) reduced the capacity of these cells to bind hyaluronan (Lokeshwar et al. 1996). Taken together, these data suggest that enhanced matrix assembly by CD44 increases receptor stability at the cell surface. Thus, CD44 interaction with the hyaluronan present within cartilage, decorated with many aggrecan monomers as well as link protein, may be strongly retained at the cell surface and display little internalization (Embry and Knudson 2001). The anchorage of the cytoplasmic tail of CD44 to the cytoskeleton may stabilize the retention of proteoglycan aggregates in the pericellular matrix.
It becomes apparent that disruption of cytoskeleton of chondrocytes brings about a metabolic response. A preliminary survey of the effects of treatment with DHCB and latrunculin A on the message levels for hyaluronan synthase 2 (HAS-2) and aggrecan indicate that the decrease in matrix assembly and retention on cytoskeletal disruption is not due to a decrease in aggrecan or hyaluronan synthesis because there was little difference in their message levels between the control and treated cells (data not shown). These observations lead us to speculate that signaling events are initiated by the disruption of the cytoskeleton.
Our results suggest that the integrity of microfilament-dependent cytoskeletal structures can be a source of regulatory signals, as suggested previously by others (Benya et al. 1988; Daniels and Solursh 1991). Chondrocytes exhibit a cortical network of microfilaments detected with phalloidin (Langelier et al. 2000; Svoboda 1998) as well as vimentin filaments (Langelier et al. 2000). An extended microtubular architecture has also been described in chondrocytes (Brown and Benya 1988; Langelier et al. 2000). Chondrogenic differentiation of limb mesenchymal cells involves the control of cell shape by the actin cytoskeleton but not the microtubular system (Zanetti and Solursh 1984). Cell rounding induced by cytochalasin stimulated aggrecan synthesis by cultured articular chondrocytes (Newman and Watt 1988). The specificity of DHCB in affecting principally the microfilaments and the reorientation of these filaments inducing a re-expression of the chondrocyte differentiated phenotype has also previously been reported (Brown and Benya 1988).
Other reports indicate that the use of cytoskeleton-disrupting agents, specifically DHCB, leads to an increase in matrix metalloproteinases (Aggler et al. 1984). If this were the case in cartilage as well, then the decrease in safranin O staining would be enhanced due to the activity of the enzymes and not merely to the disruption in the CD44–cytoskeletal interaction. One way to detect the presence of enzyme activity directed towards aggrecan is by NITEGE neoepitope expression in the cartilage (Lark et al. 1997). The treated explants that exhibited decreased safranin O staining show NITEGE staining in the pericellular zones of the chondrocytes with diffusion of the stain into the extended matrix, whereas the control explants show significant staining only in the superficial layer of the tissue. These findings are indicative of an increase in “aggrecanase” activity, inside-out signaling, on treatment with DHCB and latrunculin A.
The interaction of CD44 with the cytoskeleton and the matrix enables us to speculate that both inside-out and outside-in communication patterns are occurring within the chondrocytes via CD44. Chondrocytes may exhibit metabolic and physical changes through the interaction with the matrix, which might induce matrix degeneration, remodeling, or repair. Our results indicate that destabilization of the cytoskeleton brings about a change in the matrix. In addition to artificial cytoskeleton-disrupting agents, such as cytochalasins and latrunculins, natural agents such as NO, which is elevated during degenerative cartilage diseases (Stefanovic-Racic et al. 1993), have also been shown to inhibit actin polymerization (Frenkel et al. 1996).
Footnotes
Acknowledgments
Supported by NIH grants AR39507 and AR39239 and by a grant from the National Arthritis Foundation.
This work was submitted by G.A.N. in partial fulfillment of the requirements for the PhD from Rush University, Chicago, IL. We thank Roma A. Andhare for her assistance in obtaining the images in Figures 2 and ![]() .
.
