Abstract
The fluorescent agent Fluoro Jade was applied to cortical brain sections obtained from human patients at early postnatal ages and in patients with Alzheimer's disease, and from a Cebus apella monkey after mechanical lesioning of the cerebral cortex. Fluoro Jade labeled reactive astrocytes and early differentiating astroglial cells.
N
This study was conducted on human brain samples obtained from various accredited institutional sources under approved protocols: Institute of Brain Aging and Dementia (Irvine, CA); Brain and Tissue Banks for Developmental Disorders (U. of Maryland, Baltimore, MD, and University of Miami, Miami, FL), the Harvard Brain Tissue Resource Center (McLean Hospital, Belmont, MA), and the Alzheimer Tissue Center at Northwestern University (Chicago, IL). Brain samples underwent neuropathological assessment at the original institutions. For this study, human samples from newborn (ages 16 and 28 days old) (n=2) and aged (73 years old) (n=1) patients with no known neuropathological disease and from AD cases (ages 70–77 years old) (n=5) were used.
Moreover, to have a controlled injury condition, an adult male Cebus apella (from another experimental protocol) born indoors at the institutional (CEMIC) monkey colony, maintained according to the NIH Guide for the Care and Use of Laboratory Animals (1985), was included in this study. This animal received a unilateral surgical ablation of striatal cerebral cortex. This procedure was performed after ketamine immobilization, tracheal intubation, and halothane anesthesia. After a limited craniotomy, a U-shaped knife was used under stereotaxic coordinates to excise a cortical slab. After surgery the animal received proper antibiotic and analgesic medication for 3 days. Three months later the brain was perfused transcardially with buffered paraformaldehyde 4% under deep barbiturate anesthesia. After postfixation in the same fixative, the brain was sucrose-embedded in preparation for cryostat sectioning at 40–60 μm.
Fluoro Jade (Histo-Chem; Jefferson, AR) staining procedures followed exactly those reported by Schmued et al. (1997). The basic steps involved mounting sections on gelatin-coated slides, 15-min exposure to 0.06% K-permanganate, followed by rinsing and exposure for 30 min to a 0.001% Fluoro Jade solution in 0.1% acetic acid, followed by air-drying and coverslip mounting. Stained sections were analyzed and photographed on an Axiovert Zeiss microscope equipped for epifluorescence, using a filter for fluorescein.
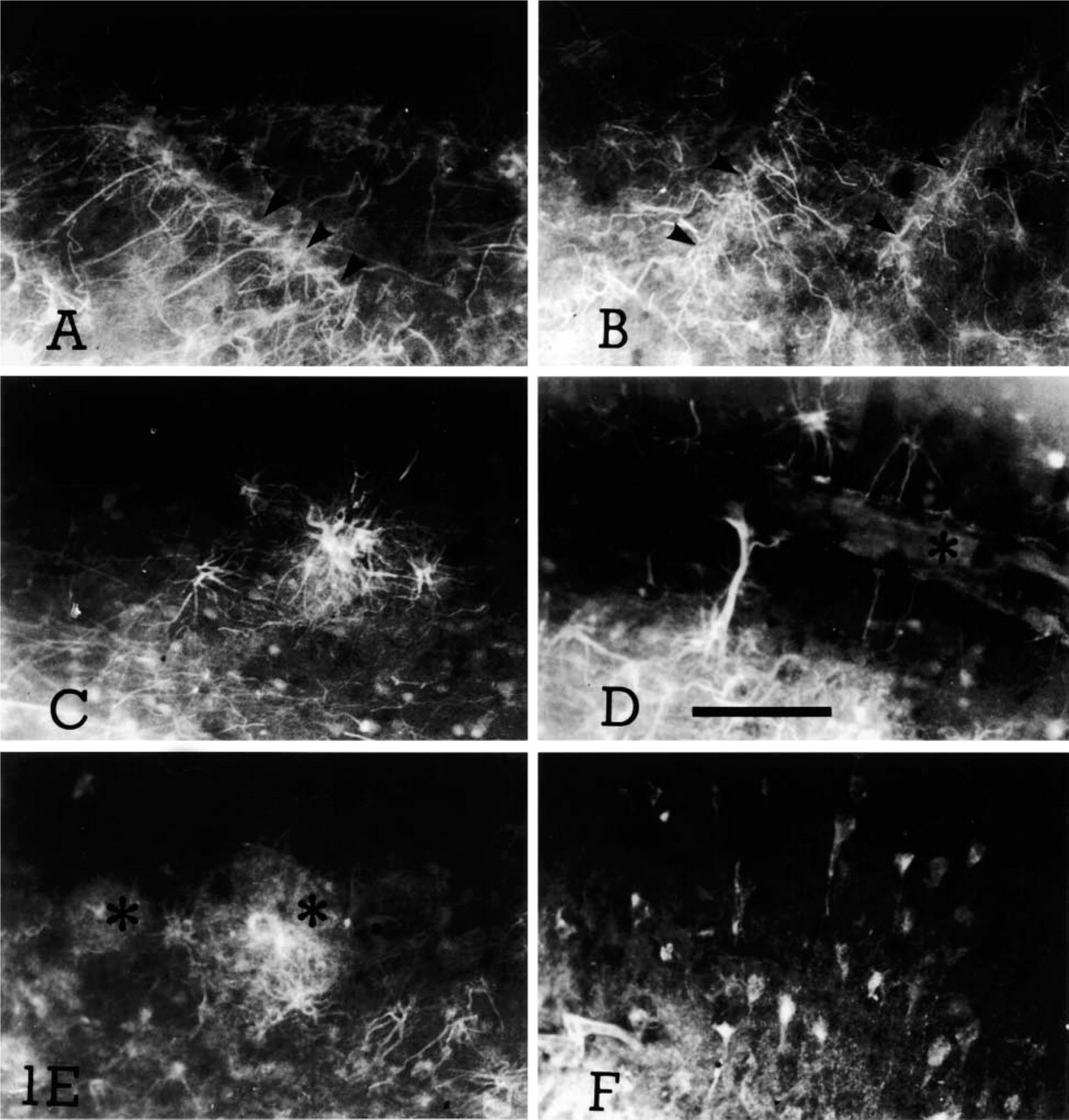
Fluoro Jade staining of several samples of primate cerebral cortex. (
In general, background staining was faint, although neuronal cell bodies and blood vessels occassionally appeared with their profiles partially visible at a comparatively low fluorescence intensity level. Glia limitans and leptomeninges usually appeared well stained. Cortical samples from newborn infants showed well-characterized labeled astrocytes and/or their processes projecting to blood vessels (Figures 1A and 1B). This population of stained astrocytes may belong to those present during the period in which middle lamina astrocytes are transitorily reactive to the glial fibrillary acidic protein (GFAP) after transformation from radial glia. In fact, whereas in the adult animal GFAP immunoreactivity is mostly limited to superficial and deep cortical lamina, in early postnatal human cortex there is a transient phase in which generalized GFAP immunoreactivity takes place. In sections obtained from the aged control case, lamina I glial processes were occasionally observed. This staining was more intense in AD cases and in the cortical sample obtained from the surgically lesioned monkey. In addition, reactive-like astrocytes were clearly stained in several cortical lamina in AD brain samples, characterized by thick processes and enlarged overall appearence (Figures 1C and 1D). In several cases these astrocytes were located close to plaque-like structures (Figure 1E) or blood vessels (Figure 1D). A mesh of astrocytic processes was often visible in lamina I–II in AD cases, suggestive of the ongoing astrogliosis characteristic of this neurodegenerative disease (Schechter et al. 1981; Mancardi et al. 1983; Martin et al. 1994; Esiri et al. 1997).
The cerebral cortex in the lesioned monkey showed at the lesion site many intensely fluorescent glial cells, appearing to “barricade” the lesion. Some neuronal (pyramidal) cells were also stained (Figure 1F). This image was not mimicked on the contralateral side, where essentially negative staining was observed.
These observations are compatible with the fact that in the primate brain Fluoro Jade appears to clearly stain non-neuronal elements of the normal (early postnatal) and pathological cerebral cortex, as observed in cases with AD and in mechanically lesioned monkey's cerebral cortex.
At present, the possible reason for the differences from other reports, in which only rat hippocampus and entorhinal cortex observations were reported, is not clear. In these cases, staining of degenerating neurons reportedly appeared to be rather selective. Based on the present observations, Fluoro Jade appears to be a more ubiquitous labeling agent than was originally suspected. Hence, a word of caution should be raised concerning the presumed ability of this agent to selectively stain degenerating neurons, regardless of cortical brain region and/or animal species.
Footnotes
Acknowledgements
Supported by Fondation Jerome K. Lejeune (France), FONCYT (PICT # 01–03465), Fundación Conectar (Argentina), Chevron-San Jorge S.A., Emprendimientos San Jorge S.A., Fundación René Baron, Fundación Bunge y Born, CEMIC, CONICET, Corpomédica S.A., and the Alexander von Humboldt Foundation (Germany).
We wish to acknowledge the provision of human brain samples by the following institutions: National Neurological Research Specimen Bank, VAMC, Los Angeles, CA (sponsored by NINDS/NIMH, National Multiple Sclerosis Society, VA Greater Los Angeles Healthcare System, and Veterans Health Services and Research Administration, Department of Veterans Affairs); and Banco de Tejidos Neurológicos (U. Complutense, Madrid, Spain).
