Abstract
The structure and symmetry of chinchilla outer hair cell (OHC) stereocilia side and attachment links were investigated by transmission electron microscopy using tannic acid and Cuprolinic blue histochemical procedures. The side links run laterally between and across the rows of the stereocilia and connect the stereocilia together within the bundle. Attachment links form a crown-like array around the tips of only the tallest OHC stereocilia and attach these stereocilia to the Type B fibrils of the tectorial membrane. Computer averaging of the side links from tannic acid-treated tissues showed a central dense region of the link between adjacent stereocilia and a smaller dense portion at the plasma membrane end of the link. Computer averaging of Cuprolinic blue-treated tissues showed low electron density of the central region of the link, and the plasma membrane ends of the link were electron dense. After tannic acid treatment, the attachment links showed a diffused radial distribution around the tips of the tallest OHC stereocilia. After Cuprolinic blue treatment, the attachment links appeared as electron-dense, membrane-bound granular structures arranged with radial symmetry. The central regions of the side links are reactive to tannic acid. These regions appear to contain neutral and basic residues of proteins and participate in side-by-side association of the side links in regular aggregates. Cuprolinic blue-reactive regions of the side and attachment links appear to contain acidic sulfated residues of glycoproteins or proteoglycans, which may be involved in the attachment of these links to the stereocilium membrane.
D
OHC stereocilia in mammals are arranged in bundles with pseudo-hexagonal symmetry in each bundle and are connected with several types of crosslinks. There are three types of extracellular links in OHC stereocilia (Tsuprun and Santi 1998). The first type of link is the tip link which extends from the tip of shorter stereocilia and attaches to the shaft of the adjacent taller stereocilia of the next row (Osborne et al. 1984,1988; Pickles et al. 1984,1989; Comis et al. 1985; Furness and Hackney 1985; Hackney et al. 1988; Osborne and Comis 1990). In the tip-link model for transduction, the tip links act as gating springs for mechanically sensitive ion channels (Hudspeth 1989; Pickles and Corey 1992). Results from electron microscopy recently showed that the tip link consists of many globular protein structures periodically arranged into two or three filaments with a helical symmetry (Kachar et al. 2000; Tsuprun and Santi 2000). These globular structures may correspond to individual protein subunits or may represent repeating identical domains of one polypeptide (Tsuprun and Santi 2000). The coiled filaments of the tip link may provide a rigid structure for force transmission to open the ion channels during hair cell mechanical stimulation (Kachar et al. 2000; Tsuprun and Santi 2000). Cuprolinic blue treatment also revealed electron-dense anionic regions at both ends of the tip link that may serve to anchor this link to the stereocilium membrane (Tsuprun and Santi 1998).
The second set of stereocilia links is the side links, which run laterally between and across the rows of the stereocilia and connect the stereocilia to one another to maintain the morphological integrity of the bundle (Pickles et al. 1984; Osborne et al. 1984; Furness and Hackney 1985). A model for the mechanics of the stereocilia bundle (Pickles 1993) predicts that the tip links are in an appropriate position to detect stereocilia deflections, whereas the main role for the side links may be to couple the deflections among the stereocilia. TEM shows a regular distribution of the side links between adjacent stereocilia with a period of about 20 nm (Tsuprun and Santi 1998).
A third type of the link is called the attachment link (Tsuprun and Santi 1998). Scanning electron microscopy (Jongebloed et al. 1998) demonstrated the presence of filamentous structures only at the tip of the tallest row of the OHC stereocilia of each bundle. Only the tallest rows of the OHC stereocilia are attached to the overlying acellular tectorial membrane (TM), as evidenced by imprints of the stereocilia on the under-surface of the TM (Kimura 1966; Lim 1986). The undersurface of the TM overlying the OHC is covered by a layer containing thin (Type B) fibrils (Tsuprun and Santi 1998) composed of proteins called tectorins (Legan et al. 1997) and keratan and/or chondroitin sulfate proteoglycans (Munyer and Schulte 1994). Attachment links form a crown-like array around the tip of tallest stereocilium of the OHCs and appear to attach these stereocilia to the Type B fibrils of the TM (Tsuprun and Santi 1998).
The molecular composition of the extracellular components associated with the mammalian stereocilia is poorly understood. The plasma membrane of the stereocilium is covered with a coat of evenly distributed glycoconjugates (Flock et al. 1977; Slepecky and Chamberlain 1985; Lim 1986; Prieto and Merchan 1986; Santi and Anderson 1987; Takumida et al. 1989; Hozawa et al. 1993; Katori et al. 1996b; Jongebloed et al. 1998). The side and tip links are digested with elastase (Osborne and Comis 1990). However, no immunoreactivity was found in hair bundles with antibodies to elastin (Katori et al. 1996a).
The purpose of the present work was to investigate the structure, distribution, and symmetry of the stereocilia side and attachment links in chinchilla OHC and the relationship between the structural organization of these links and OHC stereocilia properties. TEM using tannic acid and Cuprolinic blue histochemical procedures and computer image analysis were used in this study.
Materials and Methods
Samples of cochlear tissue were obtained from our tissue bank from normal chinchillas approximately 1 year old. The animals were deeply anesthetized with 0.2 ml ketamine/xylazine and then sacrificed by decapitation. Cochleas were removed from temporal bones and fixed by perfusion via round and oval windows. The care and use of the animals for this study were reviewed and approved by the University of Minnesota's Institutional Animal Care Committee.
Tannic acid and Cuprolinic blue treatments were used for a TEM study to obtain histological and structural information on the OHC stereocilia links. Tannic acid reacts with proteins irrespective of their electrical charge (Hayat 1989). However, the cationic dye Cuprolinic blue “at high critical electrolyte concentration” (Scott 1980,1985) selectively binds to negatively charged sulfated residues of glycoconjugates.
Tannic acid treatment was used as previously described (Tsuprun and Santi 1999). Six cochleas were fixed for 1 hr in a solution containing 2% paraformaldehyde in 0.1 M sodium cacodylate buffer containing 4 mM calcium chloride and 2% tannic acid (pH 5.3). The tissues were postfixed in 1% osmium tetroxide in 0.1 M sodium cacodylate buffer at pH 6.8 for 1 hr at room temperature. For Cuprolinic blue treatment, eight cochleas were fixed using the “high critical electrolyte concentration” procedure (Scott 1980,1985; van Kuppevelt et al. 1987) and as previously described (Santi et al. 1990). Cochleas were fixed for 1 hr in solution containing 2.5% glutaraldehyde, 0.2% Cuprolinic blue (BDH; Poole, UK) and 0.2–0.5 M magnesium chloride buffer (pH 5.6). After rinsing in the same solution without Cuprolinic blue for 20 min, the cochleas were immersed for 2 hr in buffer containing 2.5% glutaraldehyde, 0.5% sodium tungstate, and 0.2–0.5 M magnesium chloride. After fixation, the cochleas were rinsed in the same solution without sodium tungstate for 20 min. The cochleas were immersed for 2 hr in 2.5% glutaraldehyde containing 0.2% Cuprolinic blue, 0.2–0.5 M magnesium chloride, and 0.5% sodium sulfate, rinsed for 20 min in 1% sodium tungstate in 30% ethanol and then in 1% sodium tungstate in 50% ethanol. Other connective tissues in the cochlea known to contain electron-dense proteoglycans (Santi et al. 1990; Tsuprun and Santi 1997,1999) were used as a positive control. As a negative control, some of the tissues were treated with all of the reagents specified except that they did not contain Cuprolinic blue. The cochleas were dehydrated, embedded in epoxy resin, and sectioned on an LKB Nova ultramicrotome as previously described (Santi et al. 1990).
Cochlear sections were examined using a JEOL 1010 electron microscope at 60 kV and at magnifications of ×5000–15,000. Prints of micrographs were digitized using a flatbed scanner with a pixel size of 0.6 nm at specimen level. Image analysis was carried out on an SGI Onyx computer using SPIDER (System for Processing Image Data in Electron Microscopy) software. This software system was developed by Frank and co-workers and has been used for 2D and 3D image analysis of a large number of biological macromolecules in many laboratories (Frank et al. 1981,1996).
Some micrographs show adjacent stereocilia that contain images of the side links in the same projection in the section. Because of image noise, the projected structure of the link cannot be determined visually from these micrographs with high reliability. The averaging of a number of repeated noisy images of the side links improves the signal-noise ratio to visualize more objectively its structural details. Individual links between stereocilia can deviate from ideal periodic lattice positions or have some changes in the rotational orientation, and their images must be aligned by relative translation and rotation before averaging. The procedure that determines shifts and angles and, accordingly, brings two particles into a common position of alignment using SPIDER software, uses the cross-correlation function for translational alignment and auto-correlation function for orientational alignment (Frank et al. 1981,1996). Using one particle as a reference image for alignment, many particle images can be averaged (Frank et al. 1981). However, it has been shown that the use of a reference particle or model for alignment of population of particles can bias the result of averaging towards the reference image (Boekema et al. 1986). To exclude bias, a reference free alignment procedure (Penczek et al. 1992) implemented within the SPIDER software system was used in this study. In this procedure, a reference image is not used and the result of the alignment no longer depends on the choice of any particular reference image. At the beginning of reference-free alignment, two images are randomly picked from the whole set. These two images are added according to the orientation parameters found, thereby providing the first approximation of the average with improved signal-noise ratio compared with a single image. Then this average can be used to find the position of the third image (again picked randomly) which, again, is added to the sum of the two initial images. This procedure continues using in each step an “improved” average until all images from the whole set are included. The algorithm is able to align even images that share only a small number of common features.
Micrographs of OHC stereocilia after tannic acid treatment. (
Micrographs of OHC stereocilia after Cuprolinic blue treatment. (
(
(
Image fragments of the same size (more than one period), containing individual links quasi-regularly distributed between adjacent stereocilia, were selected interactively for averaging from the images displayed on the monitor screen. Before averaging a circular mask was imposed on selected fragments to remove unnecessary background, and each selected image was floated within this mask using the average density and Gaussian filter (Frank et al. 1981). The final step involved reference-free alignment and averaging of selected fragments.
Results
Side Links
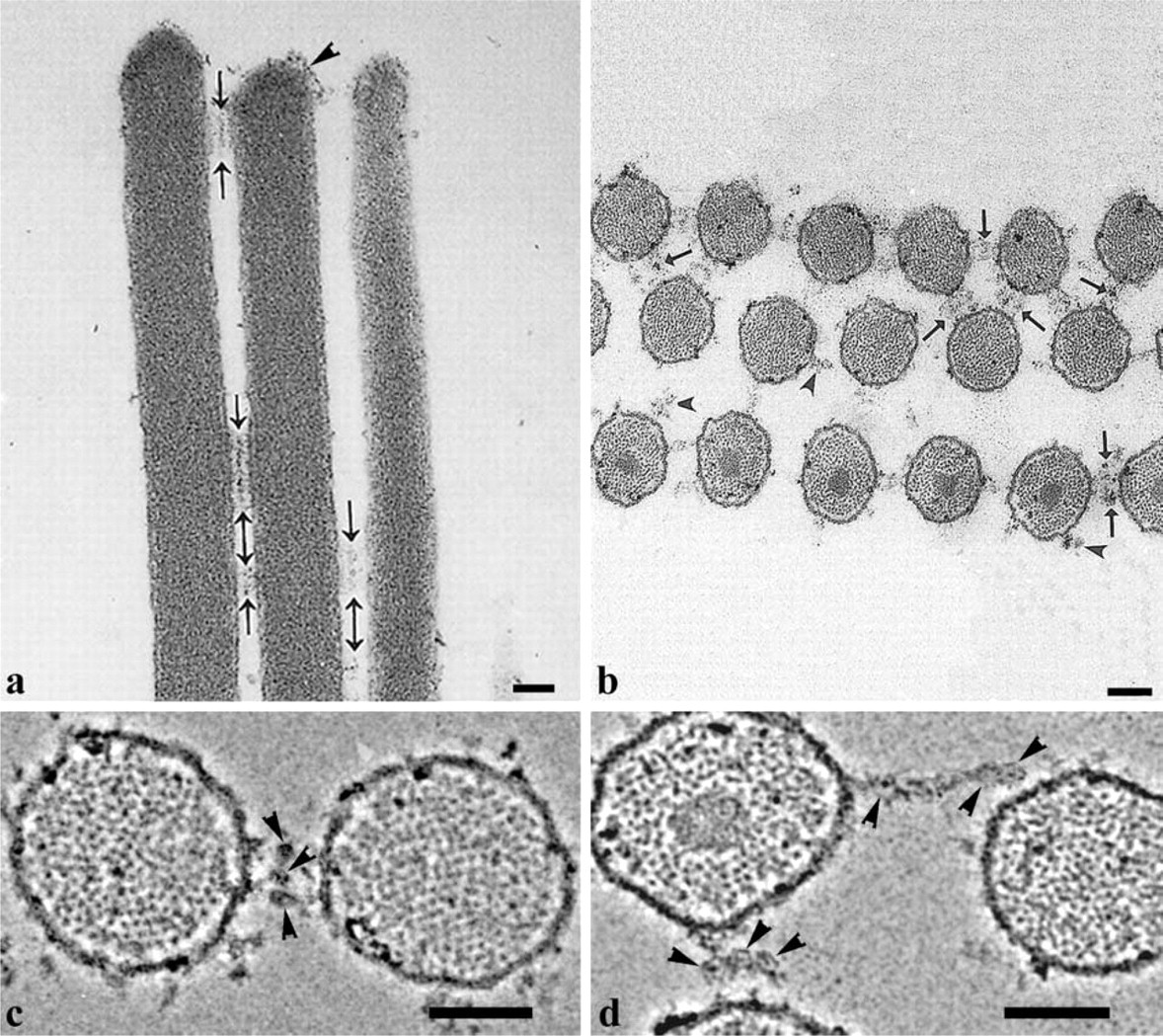
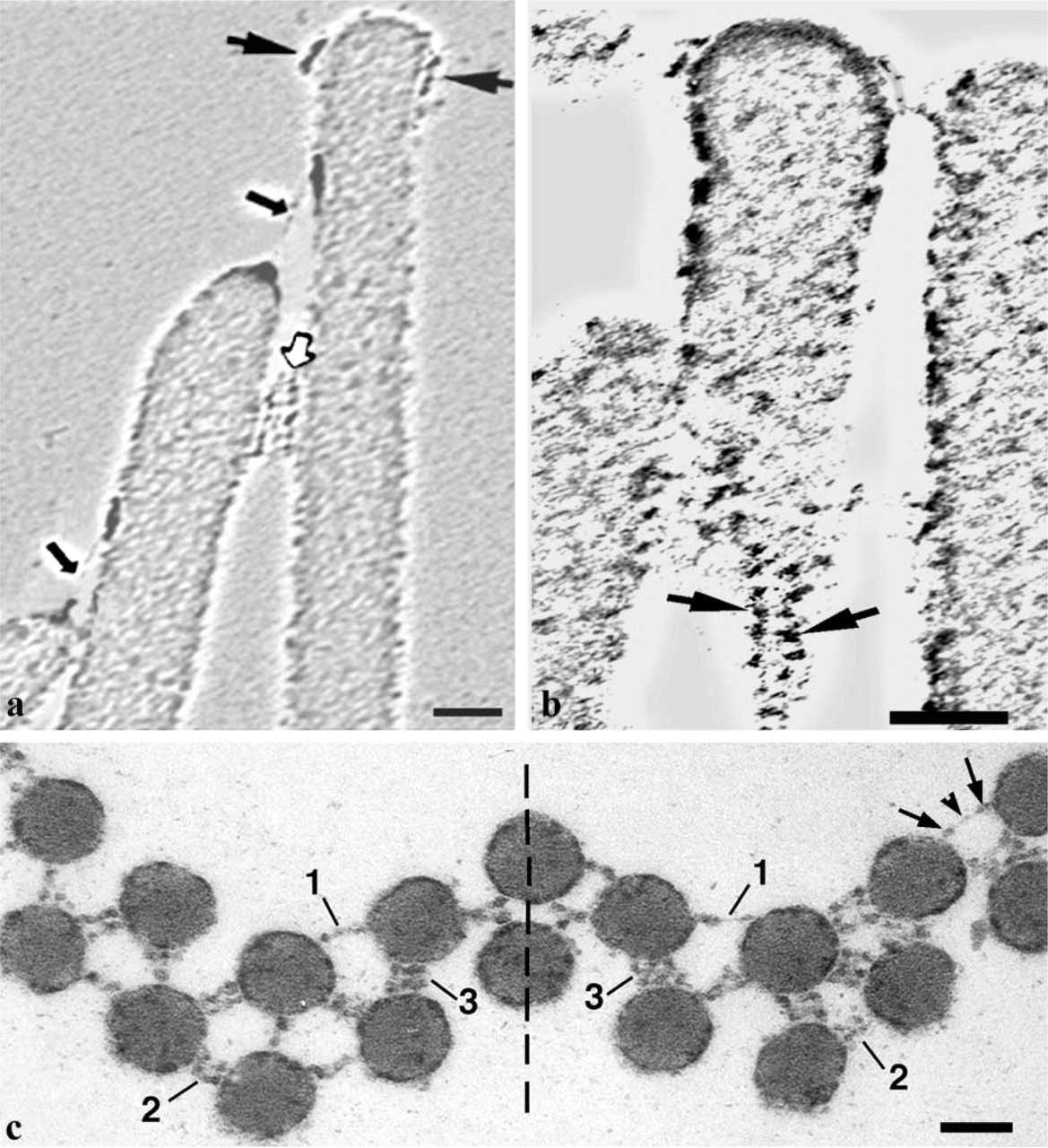
Longitudinal sections along a row of OHC stereocilia treated with tannic acid, show discrete bands of side links between adjacent stereocilia, and some diffuse material is visible over the stereocilia tips (Figure 1a). Figure 1b shows a horizontal section through a bundle of streocilia. An electron-dense core of compact actin filaments, called the rootlet (as they insert into cuticular plate), was observed only in the stereocilia corresponding to the longest row of the bundle in which the stereocilia have the longest rootlets. However, spotlike projections of actin filaments in each stereocilium indicate that this section crosses the stereocilia perpendicular to their axes. In horizontal sections, the side links formed a pseudo-hexagonal lattice. The length of the links varied and corresponded most frequently to the minimal distance between surfaces of adjacent stereocilia, as shown by arrows in Figure 1b. Some of the side-link strands projecting from the cell membrane have free ends and did not appear to connect to adjacent stereocilia. Most of the side links are composed of one to three strands, each containing central globular domains about 20 nm in diameter. High-power images show that these domains contact each other between adjacent stereocilia (Figure 1c and 1d) or sometimes form linear aggregates (Figure 1d).
Tip, side, and attachment links can also be observed in the longitudinal sections crossing the rows of stereocilia after Cuprolinic blue treatment (Figure 2a). Cuprolinic blue treatment provided an inverse staining pattern of the side links compared to the tannic acid treatment (Figure 2a). Darkly stained granular, membrane-associated structures were located, with spacing of about 20 nm between them. No central dense region, like that revealed after tannic acid treatment, was observed. In one of the rare cases, longitudinal section of the bundle showed two parallel rows of electron-dense cross-sections of the side links arranged with about 20-nm periodicity along the stereocilium axis (Figure 2b).
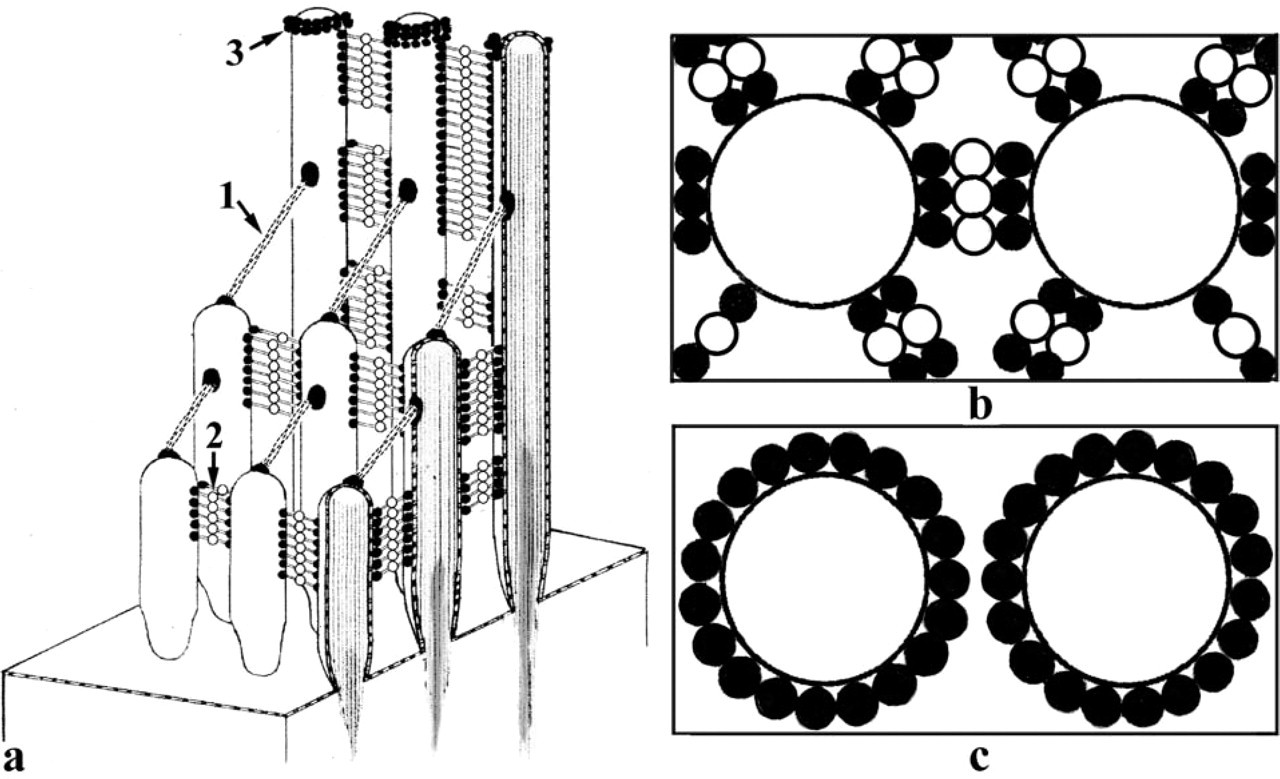
Symmetric organization and histochemical reactivity of OHC stereocilia links in the chinchilla. (
Figure 2c shows a typical horizontal section of an OHC stereocilia bundle after Cuprolinic blue treatment and demonstrates that the distribution of the side links appears very close to mirror symmetry. The projections of the side links consist of one, two, or three strands. Membrane-bound ends of the links are seen as electron-dense, globular regions and the central portions of the strands were only slightly stained or are not stained at all.
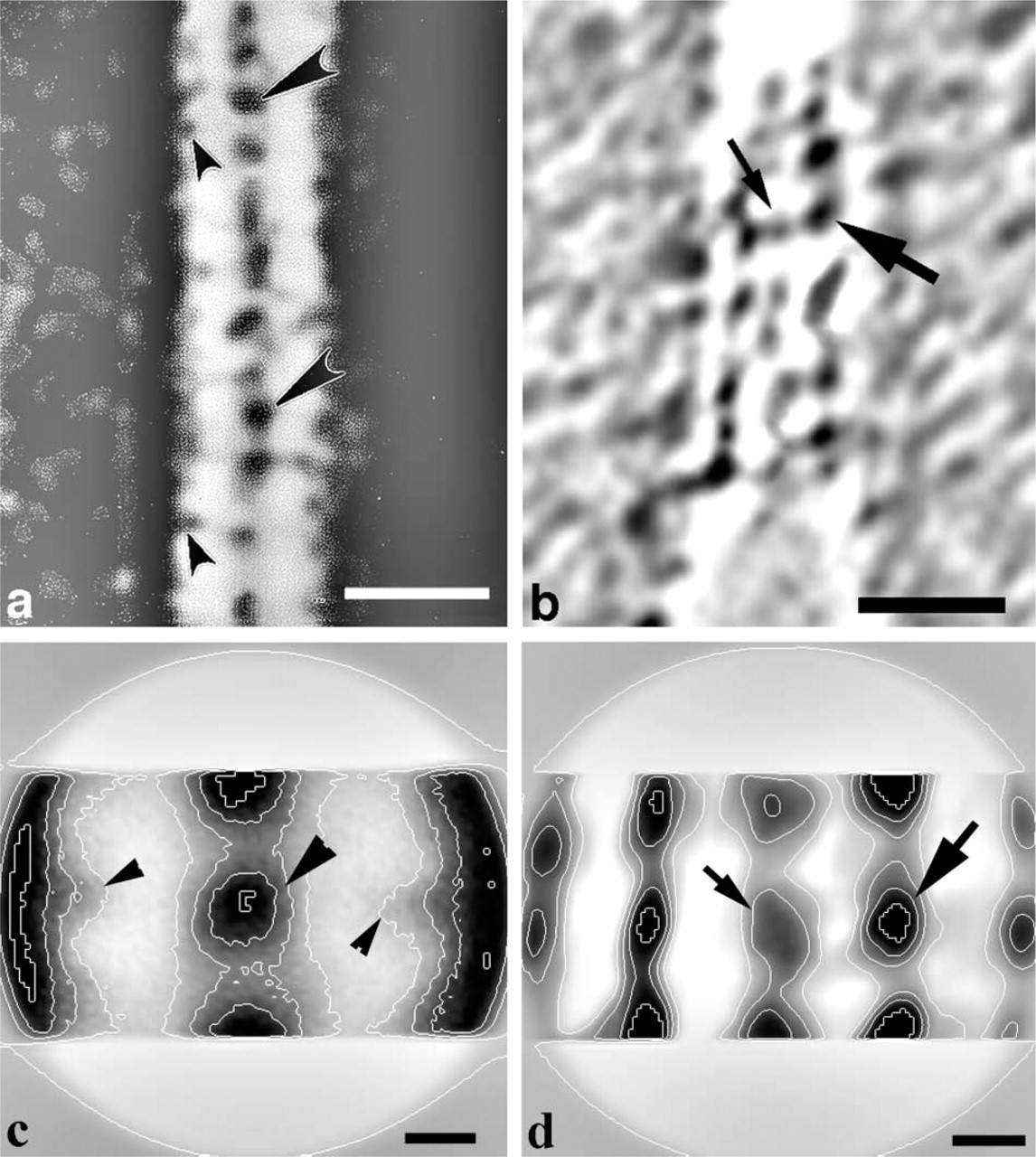
High-power images of longitudinal sections of the OHC stereocilia after tannic acid (Figure 3a) and Cuprolinic blue (Figure 3b) treatments show linear arrays of the side links between adjacent stereocilia in their rows, with a quasi-regular spacing of about 20 nm along the shaft of stereocilium. Some variations of staining and spacing between individual side-link images were observed after both procedures. For image enhancement of the side-link projections, repeated noisy fragments between stereocilia were aligned and averaged using SPIDER software.
For tannic acid treatment, we selected 10 image fragments of an original image from the area between adjacent stereocilia (Figure 3a), with each fragment of about 40-nm height and containing about two individual side-link images. The average of these fragments (Figure 3c) was similar to that of the original image and clearly showed the large central dense region (large arrowhead) and smaller low-density regions (small arrowheads) of the side link located near the surface of the stereocilium plasma membrane.
Images of the side links after Cuprolinic blue treatment were also computer-aligned and averaged. The average of six individual fragments (Figure 3d) showed that the distribution of electron density of the link was inverse to that obtained after tannic acid treatment (Figure 3c). Membrane-bound ends of the link were seen as regions of high electron density, whereas, the central portion of the link demonstrated much lower electron density.
Attachment Links
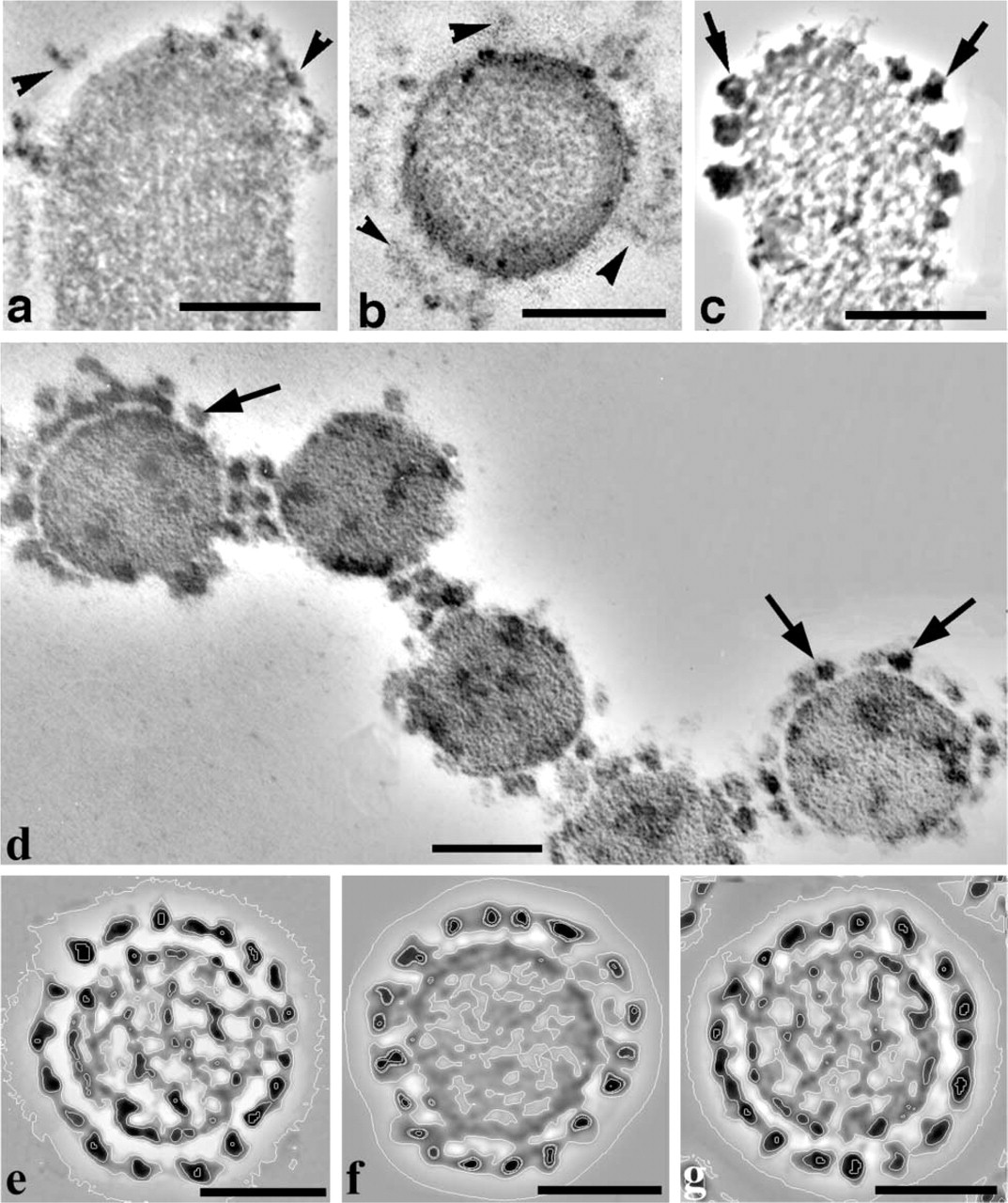
At high magnification, the presence of attachment links around the tips of the tallest stereocilia was observed in longitudinal sections after tannic acid treatment, with a bald area at the extreme tip of the stereo-cilium (Figure 4a). In horizontal sections (Figure 4b), the attachment links showed a radial distribution at some distance from the stereocilium membrane (Figure 4b). A different staining pattern was produced after Cuprolinic blue treatment. Membrane-associated granular, electron-dense structures 15–25 nm in diameter were observed around the tips of the tallest stereocilia in longitudinal (Figure 4c) and horizontal sections (Figure 4d). Sometimes the attachment links could be seen together with the side links in horizontal sections, and both link types had a similar morphological appearance (Figure 4d). However, the attachment links demonstrated a radial distribution around the tips of only the tallest OHC stereocilia, whereas, the side links were arranged with a pseudo-hexagonal symmetry between stereocilia of the whole bundle (Figure 1b and 2c).
Horizontal sections of some stereocilia showed a strong radial symmetry of granular structures (Figure 4e and 4f). Two of these images, with the best correlation coefficients between them, were aligned (within internal and external radiuses around location of granular structures) and averaged using one image as reference. The average (Figure 4g) was very similar to the original images (Figure 4e and 4f) and clearly showed a radial symmetry in distribution of electron-dense, granular projections.
Discussion
A diagram summarizing symmetric organization of OHC stereocilia links and their histochemical reactivity in the chinchilla is shown in Figure 5. The helical structure of the tip links has been described previously (Kachar et al. 2000; Tsuprun and Santi 2000). The helical organization of the tip link is stabilized by a large number of contacts between repeating protein units and may provide rigidity to the tip link during sound-induced stereocilia deflections. Our data suggest that the side and attachment links form symmetric molecular arrays. However, the structure, symmetry, and histochemical properties of these links in the chinchilla are different from those of the tip link and each other. The central and membrane-bound portions of the side links observed after tannic acid and Cuprolinic blue treatments are regularly distributed between the shafts of adjacent stereocilia. From one to three laterally aggregated projections of the side links can be seen in the horizontal sections of the OHC bundles, with most of these projections aligned along hexagonal lattice lines of the bundles. The side-link arrays between adjacent stereocilia appear to consist of one to three rows of links arranged periodically along the stereocilium shaft, with a tetragonal symmetry. Side-link arrays appear to form a great number of repeating bonds between each other and the stereocilia to provide morphological integrity of the bundle. The tallest row of the OHC stereocilia showed tannic acid- and Cuprolinic blue-reactive structures around their tips. These structures were previously termed attachment links because of their attachment to the TM in the area consisting of thin Type B fibrils (Tsuprun and Santi 1998). Cuprolinic blue treatment showed membrane-associated granular, electron-dense projections with strong radial symmetry around stereocilia in some horizontal sections. The radial density distribution in horizontal sections distinguishes them from the narrow bands of side links directed mostly along the centers of adjacent stereocilia.
Computer image analysis of tannic acid-treated tissues showed the central dense regions of the side links between adjacent stereocilia and much smaller dense portions of the links attached to the plasma membrane. Some of the side links appeared to consist of linear aggregates of globular domains (Figure 1d), which may correspond to the central regions of the side links. Computer image analysis of OHC stereocilia after Cuprolinic blue treatment showed electron-dense regions on both ends of the side links, whereas their central areas were only lightly stained. The different staining patterns after tannic acid and Cuprolinic blue treatments may indicate compositional differences between central and membrane-associated regions of the side links. Tannic acid contains phenolic and carboxyl groups and, when used in combination with glutaraldehyde and osmium tetroxide, it interacts with many different types of proteins, glycoproteins, and other components, regardless of their electrical charge (Birembaut et al. 1982; Hayat 1989). Cuprolinic blue has very high affinity for acidic sulfated residues and was successfully applied for visualization of glycosaminoglycan chains of proteoglycans in different connective tissues (Scott 1980,1985). Central regions of the side links, which are highly reactive to tannic acid and only very slightly reactive to Cuprolinic blue, appear to contain predominantly neutral and basic residues of proteins. Strong Cuprolinic blue reactivity of both ends of the side links and their light staining with tannic acid suggests their high concentration of negatively charged groups of sulfated glycoproteins or proteoglycans. We have previously suggested that Cuprolinic blue-reactive regions of the links may serve as anchoring structures to attach these links to the stereocilium membrane (Tsuprun and Santi 1998). In the side and attachment links, these regions appear to be similar to cell surface, membrane-bound glycoproteins or proteoglycans, which play a major role in cell-matrix and cell-cell attachment.
The results of this study may be compared with the data on composition, structure, and properties of stereocilia links obtained for other species. Different morphological and compositional types of crosslink have been found between the stereocilia in avian hair cells. A 275-kD protein, hair cell antigen (HCA), was demonstrated in avian sensory hair cells (Richardson et al. 1990), and immunogold labeling showed its association with stereocilia side links (Goodyear and Richardson 1992). The side links were arranged with hexagonal symmetry in the avian hair bundles and showed regular distribution between adjacent stereocilia (Goodyear and Richardson 1992), similar to the side links of OHC stereocilia bundles in chinchilla. Both types of links may serve to maintain the structure of stereocilia bundles. TEM study of the sensory epithelia from fish (Neugebauer and Thurm 1987) has shown that not only the crosslinks between stereocilia but also the surface charges of the stereocilium membrane are important factors for determining the shape of the hair bundle and the spatial interrelation of the stereocilia. However, polycation-induced fusion of stereocilia has been observed in frog (Flock et al. 1977) and fish (Neugebauer and Thurm 1987), but not in chinchilla or a lizard (Slepecky and Chamberlain 1985). It appears that side links in the chinchilla and lizard possess considerable longitudinal stiffness to prevent stereocilia from coming too close to each other.
Footnotes
Acknowledgements
Supported by 1R03 DC 04464–01A1 and NS12125 grants from NIDCD.
The computations were performed on an SGI Origin 3800 computer at the Supercomputer Institute of the University of Minnesota.
