Abstract
Apoptosis-associated speck-like protein containing a caspase recruitment domain (ASC) is a pyrin N-terminal homology domain (PYD)- and caspase recruitment domain (CARD)-containing a proapoptotic molecule. This molecule has also been identified as a target of methylation-induced silencing (TMS)-1. We cloned the ASC cDNA by immunoscreening using an anti-ASC monoclonal antibody. In this study, we determined the binding site of the anti-ASC monoclonal antibody on ASC and analyzed the expression of ASC in normal human tissues. ASC expression was observed in anterior horn cells of the spinal cord, trophoblasts of the placental villi, tubule epithelium of the kidney, seminiferous tubules and Leydig cells of the testis, hepatocytes and interlobular bile ducts of the liver, squamous epithelial cells of the tonsil and skin, hair follicle, sebaceous and eccrine glands of the skin, and peripheral blood leukocytes. In the colon, ASC was detected in mature epithelial cells facing the luminal side rather than immature cells located deeper in the crypts. These observations indicate that high levels of ASC are abundantly expressed in epithelial cells and leukocytes, which are involved in host defense against external pathogens and in well-differentiated cells, the proliferation of which is regulated.
A
ASC, composed of an N-terminal pyrin N-terminal homology domain (PYD) and a C-terminal CARD, oligomerizes and enhances anti-cancer drug-induced apoptosis (Masumoto et al. 1999). ASC was also identified as target of methylation-induced silencing (TMS)-1, because methylation of CpG islands causes loss of TMS-1 expression (Conway et al. 2000). The oligomerization of TMS-1 is accompanied by activation of caspase-9 (McConnell and Vertino 2000).
Although we initially cloned ASC cDNA by immunoscreening using an anti-ASC monoclonal antibody, which domain of ASC is recognized by the antibody has not been determined. In this study we determined the site on ASC recognized by the anti-ASC monoclonal antibody and analyzed the cellular distributions of ASC in various tissues to obtain information about the cells in which ASC functions. Here we report that ASC is differentially expressed in human tissues in a manner dependent on both maturation and cell type.
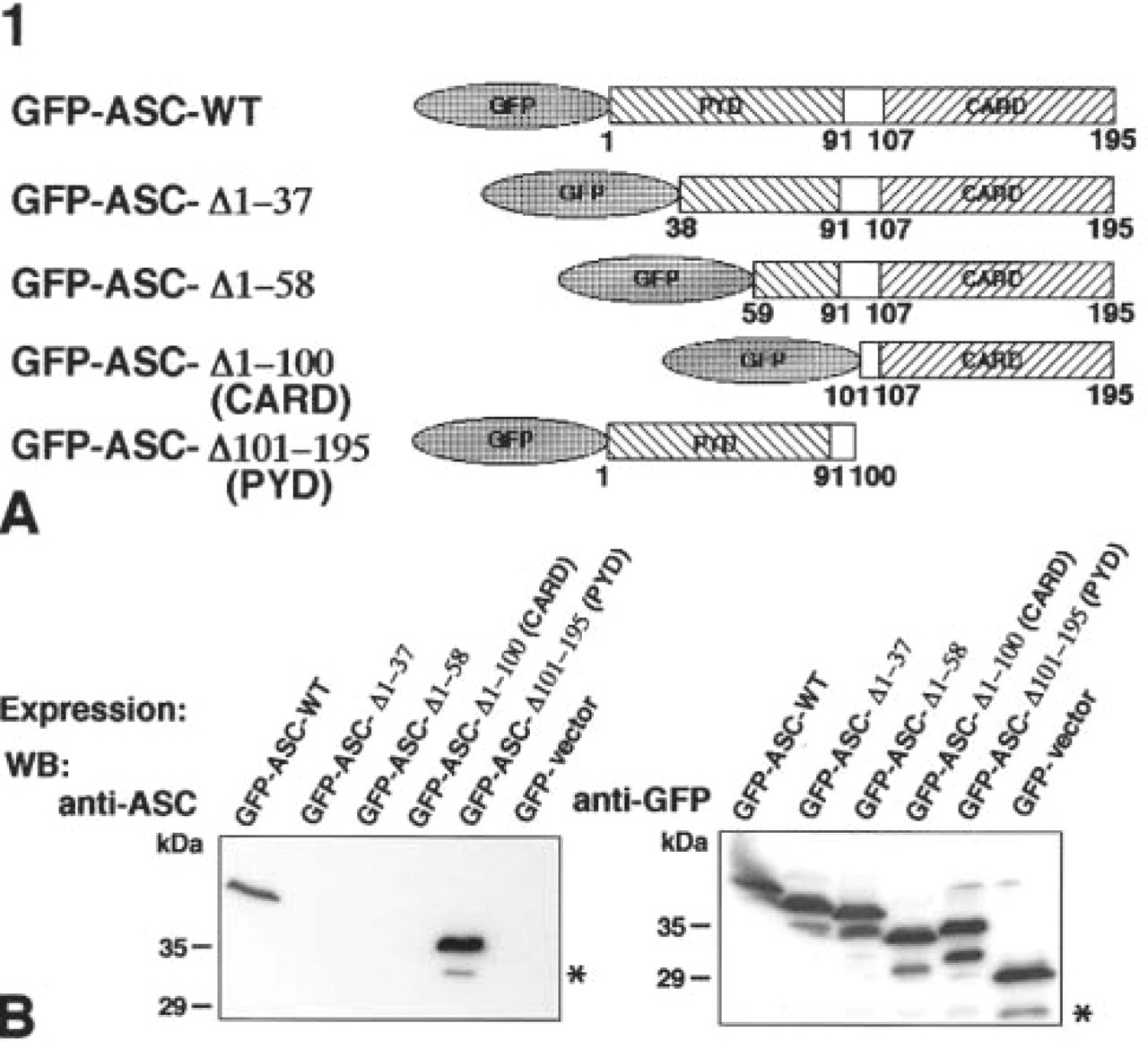
Determination of the sites on ASC reactive with the anti-ASC monoclonal antibody. (
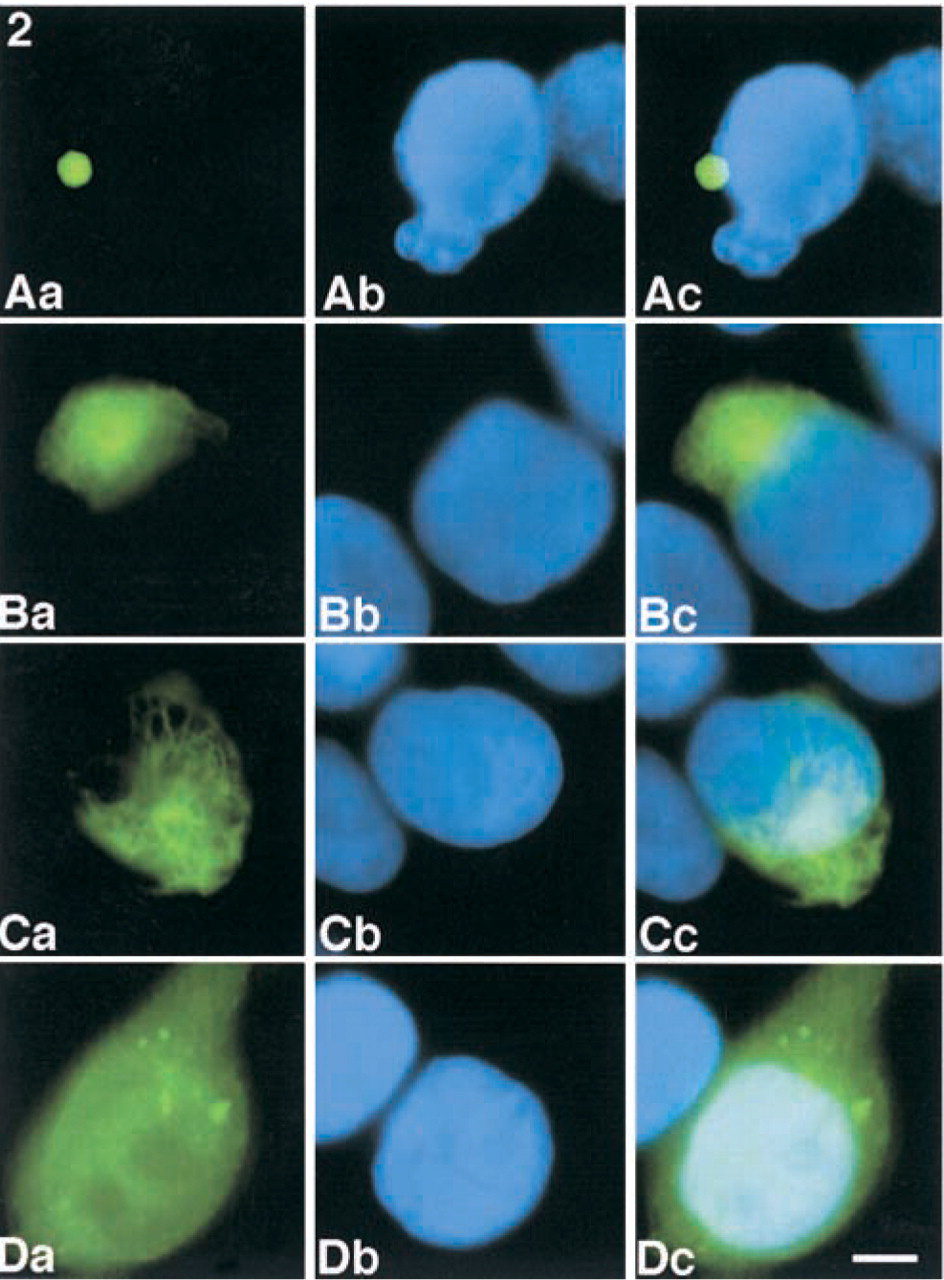
Subcellular localization of GFP-fused deletion mutants of ASC. COS-7 cells were transiently transfected with pEGFP-WT, pEGFP-ASC-Δ1–100 (CARD), pEGFP-ASC-Δ101–195 (PYD), or pEGFP-C2 (vector control). After 24 hr, green fluorescence signals in the COS-7 cells were detected by immunofluorescence microscopy. (
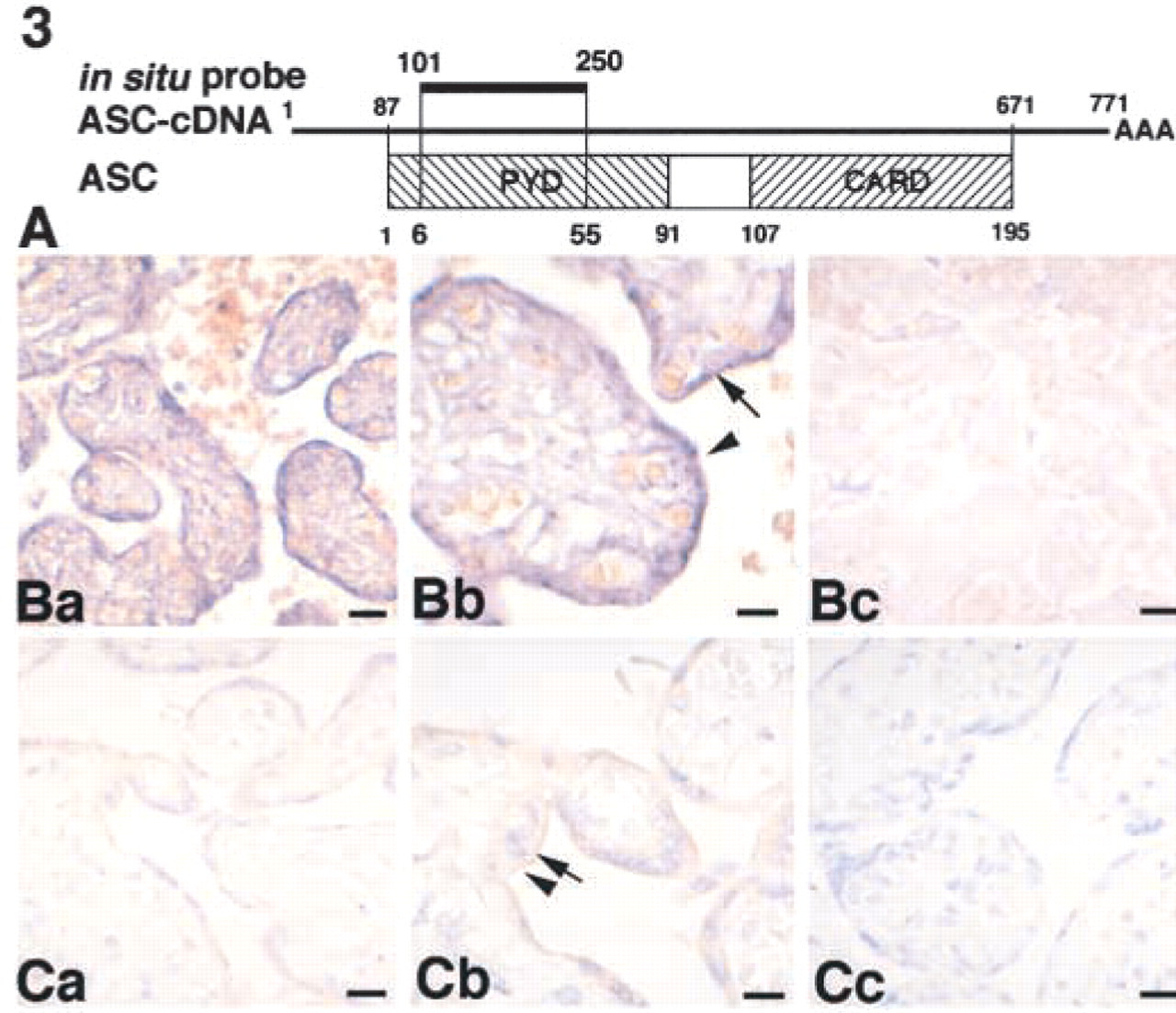
Identification of ASC expression in the human placenta by two different methods: ISH analysis and histochemistry. (
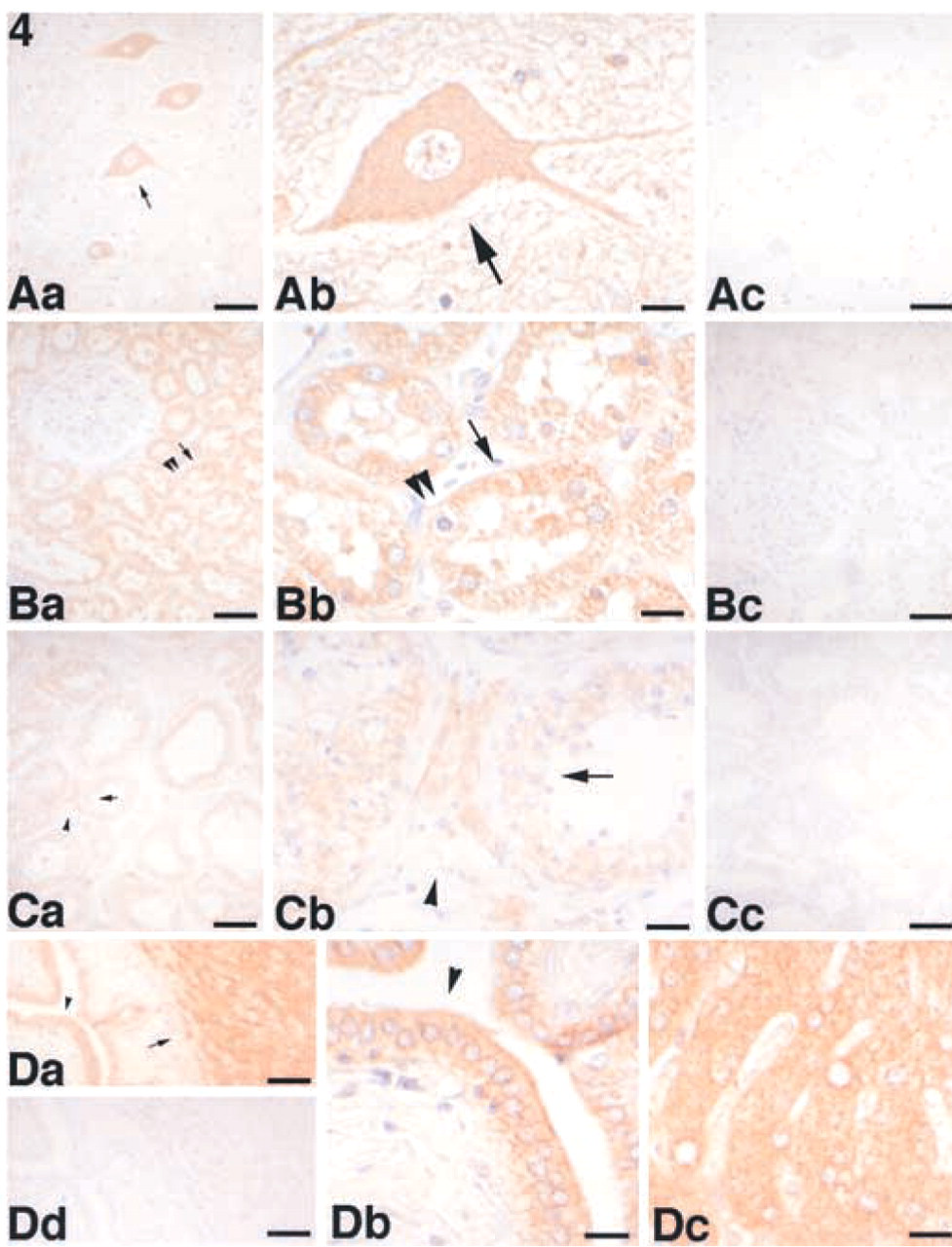
Expression of ASC in various cells of normal human tissues. (
Materials and Methods
Construction of Expression Plasmids
The entire open reading frame of ASC was inserted into the EcoRI and SalI sites of pEGFP-C2 (CLONTECH; Palo Alto, CA) to produce pEGFP-ASC. Deletion mutants of pEGFPASC-Δ1–37, pEGFP-ASC-Δ1–58, pEGFP-ASC-Δ1–100 (CARD), and pEGFP-ASC-Δ101–195 (PYD) were constructed by polymerase chain reaction (PCR) using including a λgt11 clone of the entire open reading frame of ASC (Masumoto et al. 1999) as a template after insertion into the EcoRI and SalI sites of pEGFP-C2.
Transfection, Expression of GFP-fused Proteins and Determination of the Binding Site of the Anti-ASC Monoclonal Antibody
106 COS-7 cells were transfected with expression plasmids using LipofectAMINE-PLUS reagent (Life Technologies; Rockville, MD) according to the manufacturer's instructions. Localizations of GFP-ASC-WT, GFP-ASC-Δ1–37, GFP-ASC-Δ1–58, GFP-ASC-Δ1–100 (CARD), and GFP-ASC-Δ101–195 (PYD) in transfected COS-7 cells were analyzed by immunofluorescence microscopy (Zeiss; Oberkochen, Germany). Transfected COS-7 cells were lysed with SDS sample buffer (Laemmli 1970), and detected by Western blotting using the anti-ASC monoclonal antibody or an anti-GFP monoclonal antibody.
In Situ Hybridization of ASC Transcripts and Immunohistochemistry of ASC
To detect ASC transcripts in normal human placenta, in situ hybridization was carried out using a non-radioactively labeled RNA probe. Paraffin-embedded blocks of normal human placenta fixed for 48 hr in 20% buffered formalin (pH 7.4) were selected from the pathology files of the Central Clinical Laboratories, (Shinshu University Hospital, Matsumoto, Japan). An ASC-specific nucleotide sequence (nucleotides 101–250) was amplified by PCR using upstream primer (5′-GC
Isolation of Polymorphonuclear Leukocytes, Monocytes, T-lymphocytes, and B-lymphocytes from Human Peripheral Blood
Human peripheral leukocytes obtained from ourselves and our colleagues with their informed consent were isolated by Ficoll–Paque (Amersham Pharmacia Biotech; Uppsala, Sweden) centrifugation of heparinized peripheral blood under conditions described previously (Berkow et al. 1983). Red blood cells were lysed with 33 mM NaCl solution added to the same volume of 270 mM NaCl solution, followed by separation of leukocytes. The leukocytes were suspended in RPMI 1640 medium containing 10% (v/v) FBS and incubated at 37C. FITC-conjugated CD3, PE-conjugated CD20, and FITC-conjugated CD14 were purchased from Beckton–Dickinson (Mountain View, CA). Cells were purified with a fluorescence-activated cell sorter. Peripheral blood mononuclear cells were collected in plastic tubes and incubated with appropriately diluted FITC- or PE-conjugated monoclonal antibodies. The cells were washed twice, then analyzed and sorted with a FACS Vantage SE flow cytometer (Beckton–Dickinson). Viable cells were gated according to their forward light-scatter characteristics (FSCs) and side-scatter characteristics (SSCs).
Results
Determination of the Epitope for the Anti-ASC Monoclonal Antibody
Several deletion mutants of ASC or intact ASC fused with GFP (Figure 1A) were overexpressed in COS-7 cells, and the GFP-fusion proteins were detected by Western blotting using an anti-ASC MAb or an anti-GFP MAb (Figure 1B). Then, both GFP-ASC-WT and GFP-ASC-Δ101–195 (PYD) were detected using the anti-ASC MAb, whereas GFP-ASC-Δ1–37, GFP-ASC-Δ1–58, and GFP-ASC-Δ1–100 (CARD) were not detected (Figure 1B, left panel). Expression of GFP constructs was also confirmed by Western blotting using the anti-GFP MAb (Figure 1B, right panel).
Subcellular Localizations of the Deletion Mutants of ASC Fused to GFP Were Analyzed by Fluorescence Microscopy
The subcellular localizations of deletion mutants of ASC presented in Figure 1A were examined. GFP-ASC-WT appeared as perinuclear speck-like aggregates in the transfected COS-7 cells (Figures 2Aa-2Ac). GFP-ASC-Δ1–37 (data not shown), GFP-ASC-Δ1–58 (data not shown), GFP-ASC-Δ1–100 (CARD) (Figure 2Ba–2Bc), and GFP-ASC-Δ101–195 (PYD) (Figure 2Ca–2Cc) appeared as filament-like aggregates in the transfected COS-7 cells. GFP, used as a control, was diffusely localized in the cytoplasm in transfected COS-7 cells (Figure 2Da–2Dc).
ASC Expression in the Human Placenta Was Identified by ISH Analysis and IHC
Because we had no data about the distribution of ASC in individual cells, we chose the placenta because it includes a mixture of various types of cells. We analyzed ASC expression in the placenta by two independent experimental methods. We examined whether the data regarding ASC expression obtained by histochemical analysis using the anti-ASC MAb were the same as those for ASC gene expression determined by ISH analysis with an ASC-specific DNA probe (Figure 3A). By ISH, definite signals for ASC mRNA were demonstrated in syncytiotrophoblasts and cytotrophoblasts (Figure 3Ba–3Bc), in which ASC protein was actually detected by IHC with the anti-ASC MAb (Figure 3Ca–3Cc). These results established that the placental trophoblasts lining chorionic villi synthesize ASC and that the anti-ASC MAb specifically recognizes this particular protein.
ASC Expression in Normal Human Tissues Was Demonstrated by IHC
We examined the tissue distribution of ASC in normal human tissues using the anti-ASC MAb in paraffin-embedded sections from a variety of tissues. ASC was detected in a variety of cells, including those of the placenta, anterior horn cells of the spinal cord (Figure 4Aa–4Ac), renal tubules of the kidney (Figure 4Ba–4Bc), seminiferous tubules and Leydig cells of the testis (Figure 4Ca–4Cc), hepatocytes and interlobular bile ducts of the liver (Figure 4Da–4Dd), squamous epithelium of the tonsil (Figure 5Aa–5Ac) and skin (Figure 5Ba–5Bc), hair follicles, sebaceous and eccrine glands of the skin (Figure 5Ba–5Bc), epithelial cells in the colon (Figure 6Aa–6Ac), and peripheral blood leukocytes (data not shown). No significant ASC expression was detected in the ciliated epithelium of the trachea (data not shown), glomeruli of the kidney (Figure 4Ba), cardiac muscle, alveolar epithelium of the lung, or lymphocytes (the lymphatic follicle of the tonsil is shown in Figure 5Ab). The results are summarized in Table 1. We also observed that ASC appeared as speck-like aggregates in peripheral blood leukocytes separated and incubated for 24 hr (data not shown) similar to those observed in apoptotic cells (Masumoto et al. 1999; McConnell and Vertino 2000). Although the ASC aggregates might also be present in apoptotic tissues, these speck-like aggregates were difficult to detect in the thin tissue sections. However, cytoplasmic ASC staining was redistributed to the cellular periphery in tubular epithelial cells with chromatin condensation (Figure 4Bb, double arrowheads).
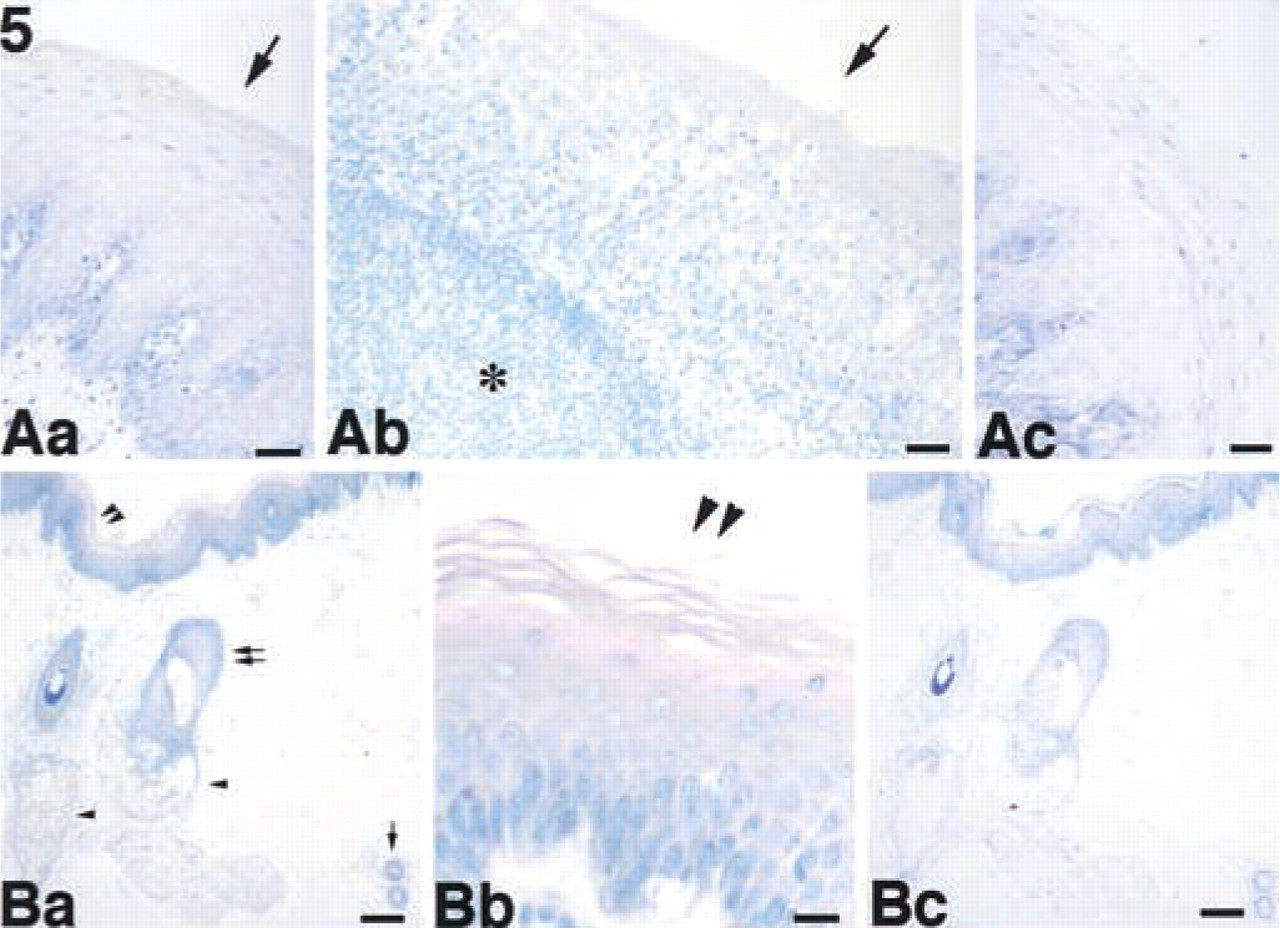
Expression of ASC in the tonsil and skin. (
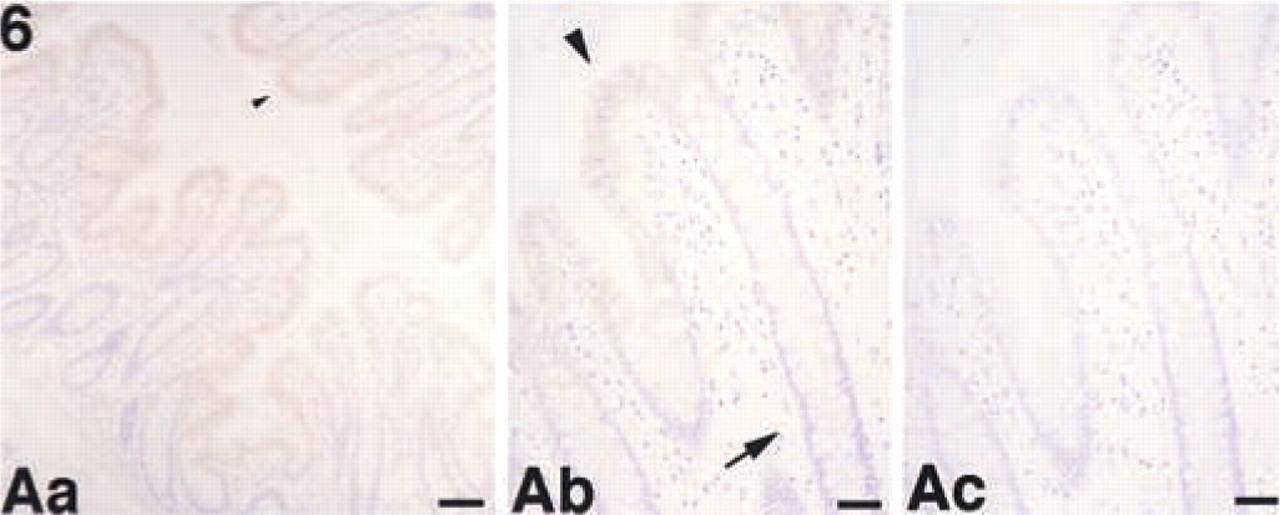
Expression of ASC in the colon. (
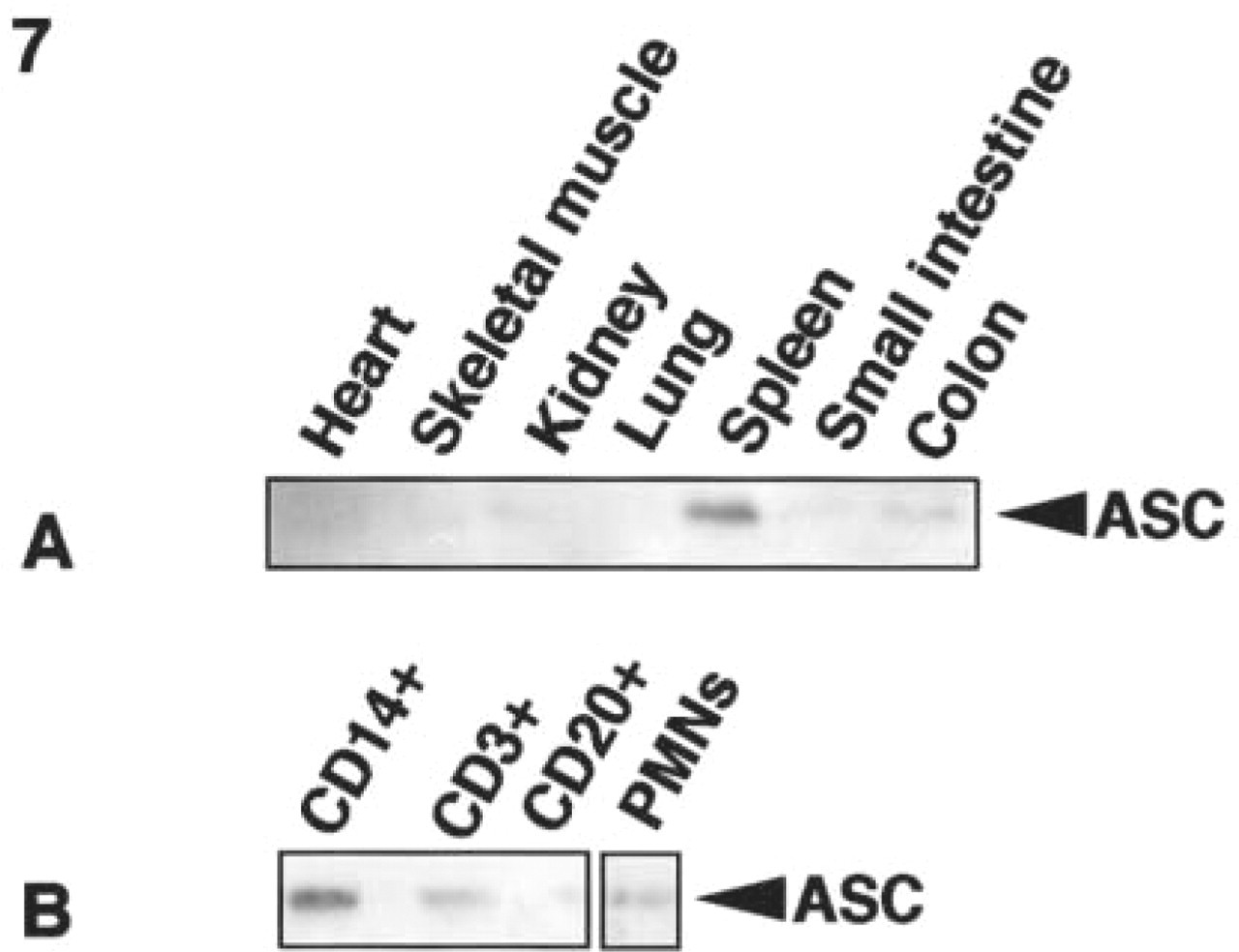
Protein expression of ASC in normal human tissues and separated peripheral leukocytes analyzed by Western blotting. (
ASC Expression in Normal Human Tissues Was Analyzed by Western Blotting
We also examined the expression of ASC using the anti-ASC MAb in normal human tissues from frozen samples and in CD14-positive monocytes, CD3-positive T-lymphocytes, CD20-positive B-lymphocytes, and polymorphonuclear leukocytes (PMNs) separated by cell sorting. ASC expression was detected in the spleen, small intestine, and colon. A trace amount of ASC expression was detected in the kidney (Figure 7A). In peripheral blood, a high level of ASC expression was detected in CD14-positive monocytes, and low levels were detected in PMNs and CD3-positive T-lymphocytes (Figure 7B).
Discussion
Anti-ASC MAb Bound to PYD of ASC
The anti-ASC MAb utilized in this study was used for the initial immunoscreening to identify ASC (Masumoto et al. 1999). However, its binding site on ASC had not been determined. Figure 1 shows that the anti-ASC MAb reacts with a region around the N-terminal 37 residue of ASC. In addition, two ASC cDNA clones of 425 and 725 base pairs, which were obtained by initial immunoscreening with the anti-ASC MAb, encoded the entire PYD domain (Masumoto et al. 1999). These results indicated that the anti-ASC MAb reacted with the PYD of ASC. Furthermore, the anti-ASC MAb did not crossreact with the murine orthologue of ASC (mASC) (Masumoto et al. 2001a). Because the N-terminal 15 residues of mASC are entirely conserved, the anti-ASC MAb is believed to react with a region including residues 16–30 of the PYD of ASC.
Summary of the cellular distributions of ASC in various tissues

+ and – denote significant staining intensities compared with negative controls. We cannot exclude the possibility that trace amounts of ASC were present in some cells judged negative.
Is ASC a Possible Key Adaptor Molecule Between PYD-containing Proteins and CARD-containing Proteins?
The speck-like aggregation of GFP-ASC-WT was different from the filament-like aggregation of GFP-ASC-Δ1–100 (CARD) or GFP-ASC-Δ101–195 (PYD). This was believed to be due to an intramolecular heterophilic interaction between PYD and CARD, which is involved in the oligomerization of ASC (Masumoto et al. 2001b). The PYD domain has been referred to as the PYRIN, DAPIN, PAAD, and pyrin domain, and a number of PYD-containing proteins were reported while this article was in preparation (Bertin and DiStefano 2000; Martinon et al. 2001; Pawlowski et al. 2001; Staub et al. 2001). The recently identified proapoptic molecule DEFCAP/NAC/NALP1/CARD7 (Bertin and DiStefano 2000; Chu et al. 2001; Hlaing et al. 2001; Martinon et al. 2001), a member of the mammalian Ced-4 protein family that includes Apaf-1 and Nod1, which contain an N-terminal PYD domain and leucine-rich repeats (LRR), binds to ASC, referred to as pycard by Martinon et al. (2001), via the PYD. The PYD domain was reported to be present in the N-terminus of the zebrafish caspases Caspy and Caspy2 (Inohara and Núñez, 2000). The PYD domains of Caspy and Caspy2 correspond to CARD or DED, which are involved in protein–protein interactions that result in effector proximity (Hofmann 1999). Therefore, the ASC may be a key adaptor molecule that plays important roles in interaction with other PYD-containing molecules through its PYD and CARD domains.
Protein Expression of ASC Was Detected in the Epithelial Surface and in Some Differentiated Functional Cells for Which Proliferation is Regulated
The expression patterns of ASC associated with differentiation in squamous epithelium in the skin (Figure 5Ba–5Bc), tonsil (Figure 5Aa–5Ac), and intestinal mucosal epithelium (Figure 6Aa–6Ac), were very similar (Chu et al. 2001), consistent with the binding of ASC to DEFCAP/NAC/NALP1/CARD7 (Martinon et al. 2001). On the other hand, significant ASC expression was not detected in the ciliated epithelium of the trachea, glomeruli of the kidney, cardiac muscle, alveolar epithelium of the lung, or lymphocytes (Table 1), although these cell types are also believed to be well-differentiated. DEFCAP/NAC/NALP1/CARD7 is a multiple leucine-rich repeat (LRR)-containing mammalian Ced4 family protein that is believed to interact with some pathogens. Other members of the mammalian Ced4 protein family, including human Nod1/CARD4 and Nod2, confer responsiveness to bacterial lipopolysaccharides (Inohara et al. 2001; Ogura et al. 2001). By interaction with DEFCAP/NAC/NALP1/CARD7 or other mammalian Ced4 members, ASC may activate downstream effector proteins involved in apoptosis and host defense against external pathogens.
We previously reported high levels of ASC mRNA expression in the spleen, which contains large numbers of lymphocytes (Masumoto et al. 1999), but no significant ASC expression was detected in individual lymphocytes. Our fractionation indicated that ASC was primarily expressed in CD14-positive monocytes in peripheral blood leukocytes (Figure 7). Therefore, we speculated that this signal in the spleen might have been due to large numbers of splenic monocytes/follicular dendritic cells and accumulation of trace levels of ASC expressed in individual CD3-positive T-lymphocytes. It is also noteworthy that ASC was expressed in epithelial cells located in the upper region of the colon mucosa rather than those in deeper regions (Figure 6Aa–6Ac). Cell number in the gastrointestinal tract is regulated by apoptosis occurring in the upper region of the mucosa (Hall et al. 1994). This result suggested that ASC might be involved in maturation of colon epithelial cells and host defense against surface pathogens.
Recently, ASC was independently identified as TMS-1 and was shown to confer lack of ASC expression through methylation-mediated silencing, a survival advantage (Conway et al. 2000; McConnell and Vertino 2000). Therefore, ASC expression may be involved in suppression of cell growth to regulate normal differentiation and responses to some pathogens. Although the precise functions of ASC in the tissues are not yet clear, ASC was differentially expressed in human tissues in a manner dependent on both maturation and cell type.
Footnotes
Acknowledgements
Supported by a Grant-in-Aid 12670109 from the Ministry of Education, Science and Culture, Japan.
We thank Drs Hiroshi Zenda and Shin Ohta (Department of Pharmacy, Shinshu University Hospital) for encouragement during this work and Masanobu Momose (Central Clinical Laboratory, Shinshu University Hospital) for technical assistance.
