Abstract
Cytokeratins (CKs) are a group of 20 antigenically distinct intermediate filaments, generally confined to epithelia and their neoplasms. Immunostaining for CKs, in particular coordinate staining for CK7 and CK20, has become a useful tool in diagnostic pathology. Although studies defining CK distribution in neoplasms identify 0–7.7% of renal cell carcinomas (RCCs) positive for CK20, none has described the incidence of CK20 immunopositivity in renal oncocytomas (ROs). Distinction between RCC and RO may be difficult but this distinction is clinically significant, prompting us to establish the incidence of CK20 positivity in RO. We selected fifteen surgical cases of RO from our archives and studied their immunoreactivity for CKs including CK7 and CK20; 12/15 (80%) were positive for CK20, with variation in the number of cells staining. There was also variation in the distribution of CKs within the cells, including diffuse cytoplasmic, perinuclear, and a punctate or dot-like pattern. Such punctate staining corresponds to cytoplasmic balls of intermediate filaments and has been described with CAM 5.2 in RO and CK20 in Merkel cell carcinomas. Our findings suggest that CK20 immunohistochemistry is a useful tool for distinguishing RCCs from ROs.
I
Renal tubule neoplasms include renal cell carcinomas (RCCs), by definition malignant, and renal oncocytomas (ROs), which are benign (Amin et al. 1997). The distinction between these lesions is therefore significant. Oncocytoma cells demonstrate abundant eosinophilic cytoplasm due to abnormally numerous mitochondria that enlarge the cell. Unfortunately, subtypes of RCC may have a similar appearance. Molecular and ultrastructural studies that may distinguish RCC from RO are not always readily available, prompting the study of immunochemical methods of distinction.
Although studies describe an almost complete lack of CK20 positivity in RCC (Moll et al. 1992; Miettinen 1995; Wang et al. 1995; Chu et al. 2000), we are aware of no series studying the pattern of CK20 immunostaining in RO, prompting our investigation.
Fifteen cases of RO were selected from our files. Cases not definitively identified as oncocytomas were excluded. Tissue sections were formalin-fixed, paraffin-embedded, and stained with hematoxylin–eosin. All paraffin-embedded tissues were immunostained using the avidin–biotin–peroxidase technique with diaminobenzidine as a chromogen, on an automated immunostainer (Ventana ES; Ventana Medical Systems). We used antibodies against the following antigens: cytokeratin 7 (clone K72, prediluted; protease digestion; Ventana); cytokeratin 20 (clone K5 20.8, prediluted; protease digestion; Ventana); cytokeratins 8 and 18 (clone CAM 5.2, prediluted; protease digestion; Becton–Dickinson, San Jose, CA); and vimentin (clone V9, diluted 1:80; Biogenex, San Ramon, CA). All slides were counterstained with hematoxylin, dehydrated, and mounted. Microwave heat epitope retrieval was performed in a low-pH citrate buffer. A representative case was selected for ultrastructural study. Fresh tissue was fixed in 2.5% glutaraldehyde, then osmicated and processed for conventional embedding in Epon 812. Ultrathin sections were mounted on copper grids and stained with uranyl acetate followed by lead citrate before examination in a Philips CM10 electron microscope.
A total of 12/15 (80%) of cases were positive for CK20, 12/15 (80%) for CK7, 15/15 (100%) for CAM5.2, and 4/15 (27%) for vimentin. Regarding coordinate expression, 9/15 (60%) were CK7+/CK20 +, 3/15 (20%) CK7+/CK20–, 3/15 (20%) CK7–/CK20 +, and 0/15 (0%) were CK7–/CK20–. The extent of staining with all three antikeratins varied by case, ranging from less than 10% to over 80% of cells positive. In cases staining for vimentin, greater than 50% of cells were positive.
A peculiar variation in distribution of keratins was noted in the cells, including diffuse cytoplasmic staining, perinuclear rings, and punctate or dot-like staining (Figure 1A). One, two, or all three patterns were identified in individual cases. Noted in some cases was a zonal staining quality, i.e., regions of tumor demonstrated diffuse cytoplasmic staining, with other distinct regions showing the dot-like or the perinuclear pattern, possibly representing a clonal phenomenon. Vimentin-positive cases always showed a diffuse cytoplasmic pattern. Dot-like positivity for CAM 5.2 has been previously described to occur in 73% of ROs but in 0% of RCCs, and therefore if present distinguishes between these lesions (Bonsib et al. 1991). The staining corresponds to single large cytoplasmic condensations of intermediate filaments referred to as globular filamentous bodies (Figure 1B). Similar staining for CK20 is seen in Merkel cell carcinoma of skin (Miettinen 1995). Other benign and malignant neoplasms may demonstrate such aggregates, which vary in their intermediate filament composition (Bonsib et al. 1991). To our knowledge, these filament aggregates are decribed only in pathological processes; the molecular basis for their formation remains to be elucidated.
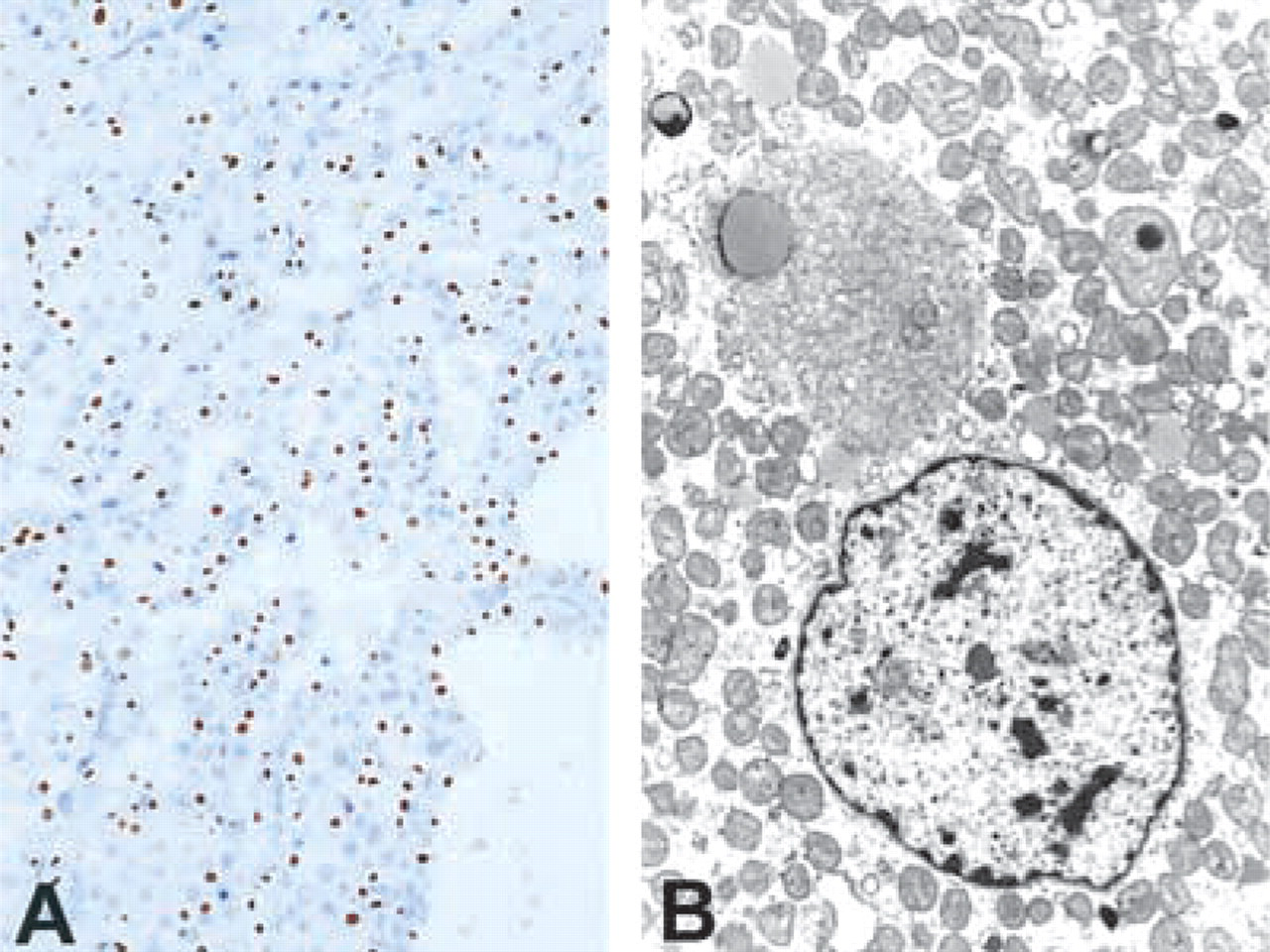
Renal oncocytoma. (
Of the clear-cell, papillary, and chromophobe variants of RCC, the latter presents the greatest difficulty in diagnostic distinction from oncocytoma. The published series we have reviewed show from 0% to 7.7% of RCCs positive for CK20 (Moll et al. 1992; Miettinen 1995; Wang et al. 1995; Chu et al. 2000). Only one series provided the percent positivity of each variant (Moll et al. 1992). We hope to obtain examples of the chromphobe variant to study for CK20 expression.
Given the degree of CK20 positivity we have identified in RO and the low level of expression reported in RCC, we conclude that CK20 immunostaining is a useful diagnostic tool for distinguishing between these lesions.
