Abstract
T
In situ hybridization (ISH) analysis of TCRβ rearrangement is problematic for several reasons. A major difficulty is that standard ISH for either TCRβ RNA or DNA would not be likely to detect the low copy number of the respective nucleic acids (Nuovo 1997). Second, even if sensitivity is not an issue, the analysis requires many probes and tissue sections to demonstrate which of the 25 distinct TCR Vβ families are rearranged (Robinson et al. 1993). This would not be possible on the small core biopsies often obtained in the clinical work-up of a possible T-cell neoplasm. Each of these problems can be circumvented using an in situ amplification system. This report describes a reverse transcriptase (RT) in situ polymerase chain reaction-based assay (RT in situ PCR), which was used to demonstrate clonality in paraffin-embedded, formalin-fixed materials diagnostic of a T-cell neoplasm and polyclonality in reactive lymph nodes. The report also shows the clinical utility of the RT in situ PCR assay for evaluation of TCRβ gene rearrangements in tissue sections for which a definitive diagnosis of T-cell vs B-cell lymphoma could not be made on the basis of the clinical, histological, immunohistochemical, and flow cytometric data.
Materials and Methods
Tissue Preparation
Fifteen formalin-fixed, paraffin-embedded tissues were retrieved from the surgical pathology archives of the Department of Pathology at Ohio State University Medical Center. The tissues included three lymph nodes obtained from resections of melanoma, breast cancer, and colon cancer. Metastastic melanoma was evident in the lymph node from the first case, whereas there was no evidence of metastastic disease in the latter two cases. We also analyzed seven tissues from five patients diagnosed with T-cell lymphoma on the basis of the clinical, histological, immunohistochemical and, when available, flow cytometric data. These seven tissues consisted of an HTLV-1-positive T-cell leukemia/lymphoma that had infiltrated the bone marrow, an angioimmunoblastic T-cell lymphoma in a lymph node and in the bone marrow from the same patient, one case of peripheral T-cell lymphoma of the lung, two biopsies of mycosis fungoides from the same patient (although the second biopsy after therapy was deemed suggestive but not diagnostic of mycoses fungoides), and one lymphomatoid papulosis of the skin. In addition, we evaluated five cases of lymphoma in which the distinction of T-cell-rich B-cell lymphoma vs B-cell-rich T-cell lymphoma could not be made on the basis of the clinical and pathological data.
RT In Situ PCR
Our RT in situ PCR protocol has been previously described (Nuovo et al. 1995, 1999; Nuovo 1997). In brief, optimal protease digestion was determined using nonspecific direct incorporation of the reporter nucleotide after protease digestion times that varied from 20 to 60 min. Optimal digestion time was defined as that producing an intense nonspecific signal in all cell types that was eliminated by overnight digestion at 37C in RNase-free DNase (10 U per tissue section) (Boehringer Mannheim; Indianapolis, IN). The EZ RT-PCR system (Perkin-Elmer; Norwalk, CT) was employed. The RT/amplifying solution contained the EZ rTth buffer, 200 μM each of dNTP, 10 μM of the reporter nucleotide digoxigenin dUTP, 400 μg/ml of bovine serum albumin to minimize adsorption of the rTth on the glass slide, 40U RNasin, 20 μM of the 5′ and 3′ primers, 2.5 mM of MnCl2, and 5 U of rTth. The sequences of the different primers employed for TCRβ RNA analysis are listed in Table 1 (Clontech; Palo Alto, CA). After an initial incubation at 65C for 30 min, and a denaturation of 94C for 3 min, 20 cycles were performed at 60C for 1 min and 94C for 30 sec. After a high-stringency wash (0.2 × SSC and 2% bovine serum albumin at 60C for 10 min), the amplified cDNA was complexed with the anti-digoxigenin–alkaline phosphatase conjugate (Boehringer Mannheim), which reacted with the chromogens nitroblue tetrazolium and 5-bromo-4-chloro-3-indolyl-phosphate (NBT/BCIP) (Enzo Biochemicals; Farmingdale, NY), yielding a blue signal. The counterstain nuclear Fast Red causes the negative cells to stain light pink. Negative controls included omission of the rTth, use of irrelevant (human papillomavirus–HPV-specific) primers, because HPV cannot infect T-cells (Nuovo et al. 1995), and predigestion in RNase as previously described (Nuovo 1997). The control conditions were done on the same slides as the experimental conditions to ensure uniformity of possible confounding variables.
Oligoprimers used for detection of TCRb mRNAs a

All sequences are in the 5′ to 3′ orientation.
Immunohistochemistry
Our immunohistochemical protocol has been previously published (Nuovo et al. 1995, 1999). Tissues were tested with monoclonal antibodies against CD3, CD15, CD20, CD30, CD45, CD45RO, and ALK-1.
Results
To determine whether the histological distribution of TCRβ expression was equivalent to the distribution of reactive T-cells, as defined by CD3 positivity, three lymph nodes were examined that were obtained from patients as part of the surgical treatment of their malignancy (breast and colon carcinoma and melanoma). The total polyclonal T-cell population in the lymph nodes would be expected to demonstrate each of the different Vβ rearrangments of the TCRβ (Robinson 1991; Lynas and Howe 1997). In each lymph node, a reactive histological pattern was evident and immunohistochemisty showed the expected geographical distribution of the CD3+ T-cells (primarily interfollicular zone), CD20+ B-cells (primarily germinal center and mantle zone), and CD21+ interfollicular dentritic cells (data not shown). Metastastic tumor was evident in the lymph node in the melanoma case. To simplify the initial part of the analysis, five groups of five consecutive TCRβ oligoprimers that corresponded to adjacent Vβ families (e.g., Group 1 Vβ1,2,3,4,5.1, Group 2 Vβ5.2,6,7,8,9, and so on) were each paired with the consensus Cβ region primer. In this way, the analysis of five tissue sections, which could be placed on two glass slides with an additional section remaining for a control, was sufficient to screen the entire repertoire of TCRβ expression. Any one set of five consecutive Vβ primers that yielded a signal could then be re-analyzed with the corresponding five individual component Vβ primers using two additional glass slides. Further, because adjacent serial sections that are 4 μm apart were used, one could examine the distribution of each of the 25 Vβ families in the same areas in adjacent tissue sections.
For each of the three lymph nodes, a signal was evident with each of the five oligoprimer groups that in total comprised the entire TCRβ sequence. More detailed analysis showed that TCRβ mRNA could be demonstrated with each of the 25 individual Vβ family-specific primers. The histological distribution of the T-cell receptor mRNAs was the same as that of the T-cell marker CD3. The percentage of T-cells that expressed a given TCRβ rearrangement was equivalent (3–7% of the total T-cells) for each of the 25 Vβ families for the lymph nodes from the patients with non-metastatic breast and colon cancer. However, in the lymph node with metastatic melanoma, certain transcription segments, specifically Vβ5.1 and Vβ16, were present in disproportionately greater numbers relative to the other Vβ families (four to five times as many positive cells), although each of the 25 was present (data not shown). The cells expressing the TCRβ Vβ16 and Vβ5.1 transcripts were in close proximity to the metastatic melanoma, and therefore their relative increase may represent oligoclonal expansion induced by tumor-related antigens.
We next tested seven tissues for which in each case an unequivocal diagnosis of T-cell lymphoma was made on the basis of the clinical, histological, and immunophenotypic analysis. This was done to determine whether the RT in situ PCR assay could demonstrate the expected monoclonality in these samples. The analyses were done blindly with respect to the histological diagnosis and which cases corresponded to the same patient (i.e., Cases 1 and 4 each had two biopsies taken at different time points; Table 2). In Cases 1 and 3, independent confirmation of T-cell clonality was demonstrated at outside laboratories using TCR7 analysis and HTLV-1 clonal insertion, respectively. These data are compiled in Table 2. In each case, a signal was evident in over 95% of the CD3-positive cells with only one of the 25 TCRβ Vβ family-specific primers. In these seven cases of unequivocal T-cell lymphoma, no other Vβ family primer produced a signal in more than 1% of the T-cells, presumably reflecting reactive non-neoplastic T-cells among the malignant monoclonal T-cells. A representative case of angioimmunoblastic T-cell lymphoma is presented in Figure 1. Note that a signal was evident in the neoplastic cells in the lymph node and bone marrow specimen, taken several months apart from the same patient, using the TCRβ Vβ2-specific primers. In addition, the monoclonal rearrangement was localized in the bone marrow to the neoplastic T-cells and not to the adjoining normal hematopoetic elements. This serves as an internal control for the specificity of the RT in situ PCR assay. Furthermore, the signal was lost if HPV specific primers were employed or if RT in situ PCR was done with the Vβ2 primer set after a 30-min pretreatment with RNase (Figure 1), demonstrating that the signal was RNA-based. Also note that in Case 4 the same T-cell receptor rearrangement (Vβ22) was noted in the skin biopsy of mycoses fungoides and the subsequent biopsy done after therapy, in which the atypical lymphoid infiltrate was deemed suggestive but not diagnostic of mycoses fungoides due to the lack of epidermal invasion, presumably as a consequence of therapy.
Correlation of the Vβ expression pattern, as determined by RT in situ PCR, with the pathological findings
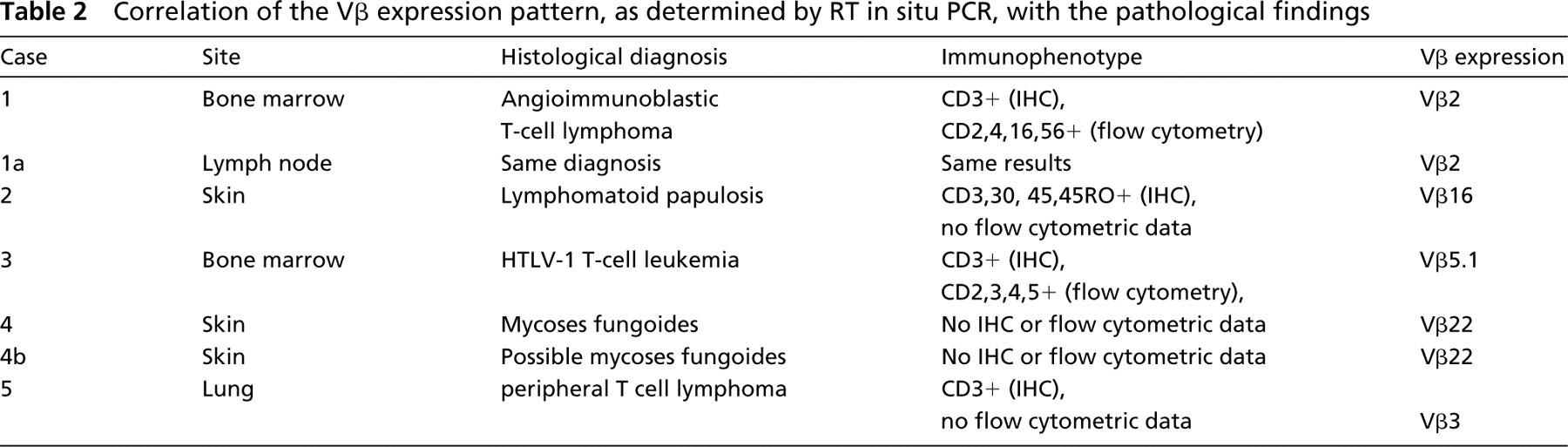
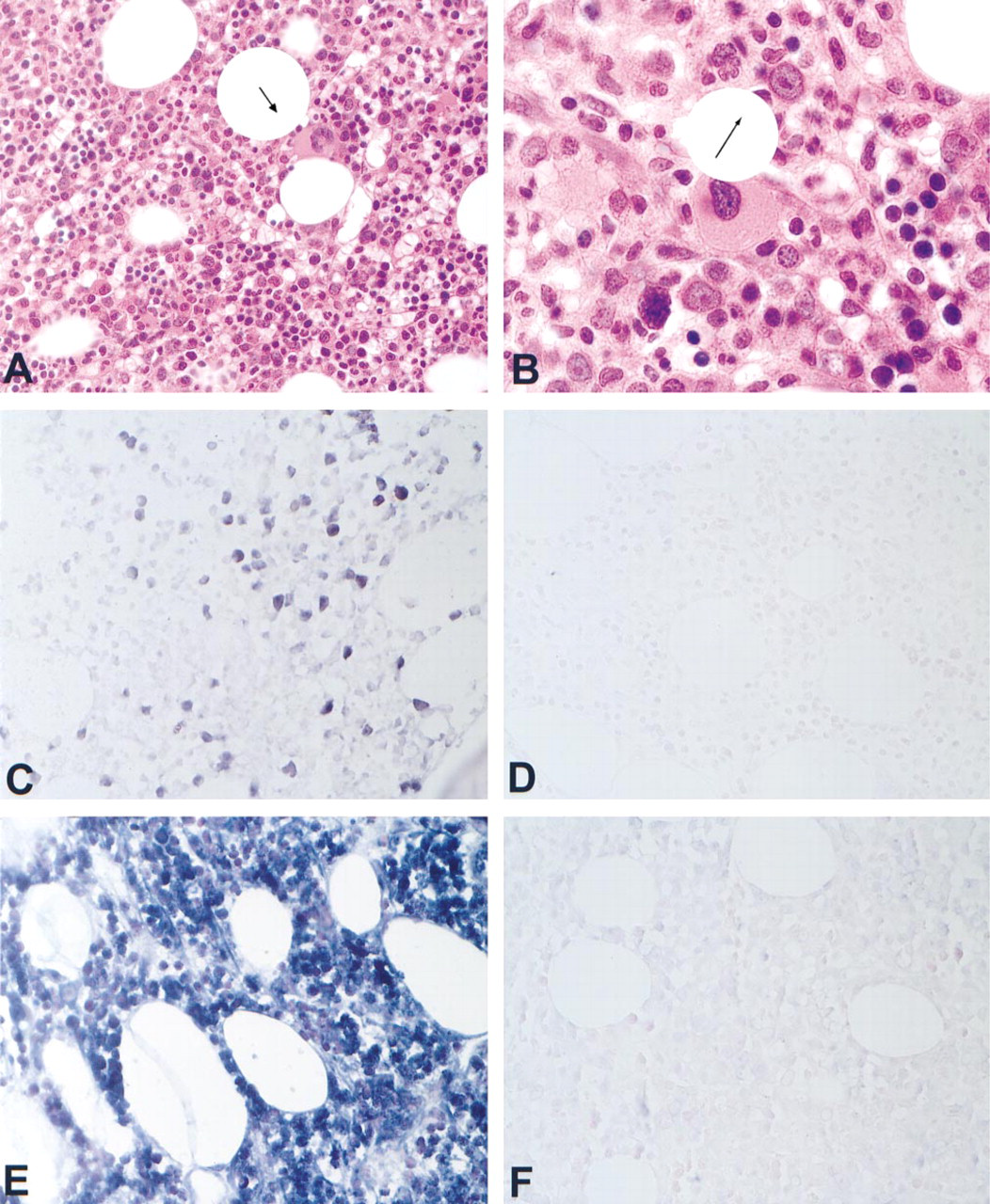
Expression patterns of TCRβ in an angioimmunoblastic T-cell lymphoma. The patient had a diagnosis of angioimmunoblastic T-cell lymphoma from biopsies of the bone marrow and a lymph node done several months apart (Case 1, Table 2). (
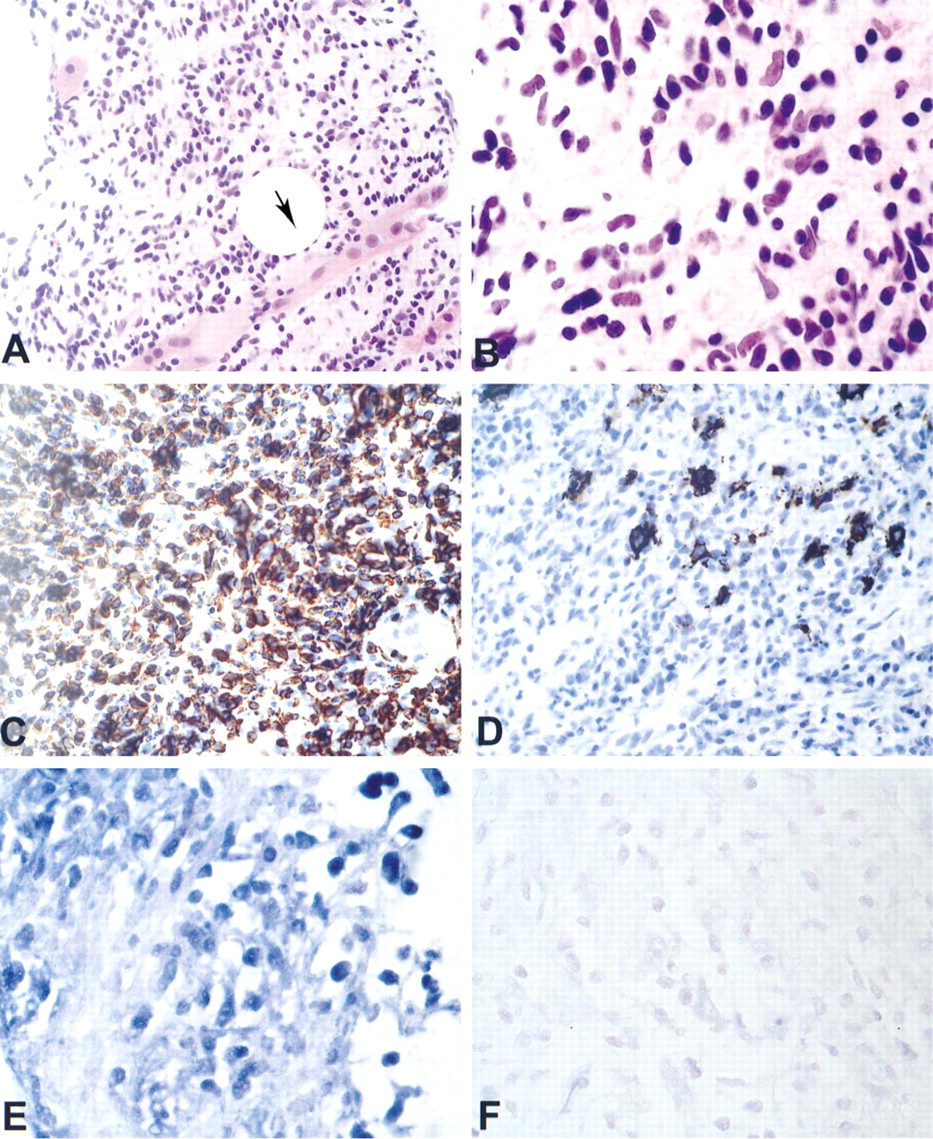
Expression patterns of TCRβ in a retroperitoneal tumor. This core biopsy from a retroperitoneal mass (Case 5, Table 2), showed an infiltrate of intermediate to large lymphoid cells that was invading skeletal muscle (
The final part of the study was use of the RT in situ PCR assay in five cases for which the clinical and pathological data were unequivocal for lymphoma but for which a definitive distinction of B-cell-rich T-cell lymphoma vs T-cell-rich B-cell lymphoma could not be made because of the presence of atypical CD20-and CD3-positive cells. In three of the cases, a monoclonal T-cell proliferation consistent with a T-cell lymphoma (Vβ1, Vβ12, Vβ16, respectively, Figure 2) was seen after RT in situ PCR, whereas in the other two cases a polyclonal T-cell population was identified, consistent with a T-cell-rich B-cell lymphoma; these latter two cases showed monoclonal expression of Ig light chain kappa by RT in situ PCR (unpublished data). Co-labeling experiments in the former three cases confirmed that the cells that showed a monoclonal expression pattern of TCRβ did co-label with CD3 (data not shown).
Discussion
This study showed that the RT in situ PCR assay for TCRβ rearrangment could detect transcripts from each of the 25 Vβ families in cells from lymph nodes removed as part of the surgical management of carcinomas or melanoma. Furthermore, the assay could demonstrate the specific monoclonal TCR Vβ rearrangement present in seven archival paraffin-embedded, formalin-fixed tissues diagnostic of a T-cell neoplasm. In two of these cases (Case 1 and Case 4; Table 2), the two separate biopsies from the same patient done blindly each showed exactly the same TCRβ rearrangement. Importantly, the assay could differentiate those cases of lymphoma for which the histological and immunohistochemical data could not distinguish between a B-cell and a T-cell lymphoma.
It is well documented that there may be limited gene usage of both the variable α and β TCR sequences in T-lymphocytes in reactive inflammatory processes, such as multiple sclerosis (Ben-Nun et al. 1991) and rheumatoid arthritis (Witzens et al. 1997). Moreover, it has been shown that carcinomas may be associated with expansions of multiple T-cell clones as defined by rearrangment of one of the TCR chains, which likely represents the induced proliferation of restricted T-cell populations secondary to tumor-specific antigenic stimulation (Puisieux et al. 1994; Baier et al. 1998). These observations are consistent with what we noted in the case of melanoma metastatic to a lymph node. Furthermore, because the RT in situ PCR technique can localize signal to specific cells, it was observed that the expansion of certain T-cell clones in the case of metastatic melanoma was strictly defined by the proximity of these cells to the metastatic melanoma cells. Whether this actually represents stimulation of certain clones due to tumor-specific antigens awaits further study.
The surgical pathologist must often deal with biopsies in which a T-cell lymphoma is considered in the differential diagnosis. The standard practice in such instances is to perform immunohistochemistry for a variety of T- and B-cell markers. However, even the demonstration of a predominant T-cell phenotype does not rule out a reactive non-neoplastic process nor the presence of a T-cell-rich B-cell lymphoma. Indeed, one of the cases in this study that was shown by RT in situ PCR to be a T-cell lymphoma was initially diagnosed as T-cell-rich B-cell lymphoma by an expert hematopathologist from an outside laboratory, underscoring the difficulty in such cases when clonality data are not available because of insufficient tissue. The RT in situ gene rearrangment analysis showed a monoclonal T-cell population and a polyclonal B-cell infiltrate in three of the five cases, demonstrating that these lesions were B-cell-rich T-cell lymphomas; the distinction has important clinical implications (Armitage et al. 1999). The RT in situ PCR assay for TCRβ rearrangement can be done in half a day using paraffin-embedded tissue as small as 2 mm; such samples are inadequate for either flow cytometry or Southern blotting analysis of TCRβ DNA or RNA patterns. The RT in situ PCR assay for TCRβ rearrangement may be of assistance in elucidating the clonality patterns of Hodgkin's disease and in better understanding the mechanisms of how certain conditions, such as celiac sprue and EBV infection, may predispose to T-cell lymphomas, by examining the relationship of such conditions to the expression patterns of the TCRβ (Armitage et al. 1999).
