Abstract
Trials that use a randomization process are preferred for evaluating causal effect. However, these interventional studies are not always feasible due to ethical, time, and cost constraints. Additionally, they are often criticized for not accurately representing patients seen in clinical practice. Observational studies can help bridge this gap, but they are often affected by several sources of bias. A growing framework known as target trial emulation (TTE) is increasingly being applied to real-world data to minimize these common biases in observational studies and, under certain conditions, allow for causal interpretation. In this study, we assessed the 5-y effect of thiazide monotherapy, an antihypertensive medication known to cause xerostomia, on the risk of developing dental caries. We applied the target trial framework to build a TTE cohort using an active comparator new user design. We compared thiazide vs nonthiazide monotherapy using electronic health records from the All of Us Research Program. Furthermore, we developed 2 naïve cohorts: a prevalent user cohort, which included existing medication users, and a nonaligned cohort, which was affected by immortal time bias. These cohorts helped illustrate the advantages of TTE. In the TTE cohort, we found no significant difference in the 5-y risk of dental caries between groups, with a risk difference of 0.29% (95% CI, −0.36% to 0.95%). In contrast, the naïve cohorts showed directionally opposite effects, with 5-y risk differences of 0.65% (95% CI, 0.22% to 1.09%) in the prevalent user cohort and −0.24% (95% CI, −0.55% to 0.07%) in the nonaligned cohort, highlighting the impact of design-related biases on the observed outcomes. We demonstrated how the TTE framework can help avoid common biases in observational research, enabling researchers to answer important oral health questions.
Keywords
Introduction
Inferences from randomized trials (hereafter, trials) are preferred because random assignment of strategies under study supports causal interpretation (Hariton and Locascio 2018). However, trials are not always feasible because of cost, time constraints, and ethical considerations. Moreover, trials may not adequately represent the target populations for whom the interventions are intended (Rothwell 2005). As a result, observational studies play a crucial role in bridging the gap between the limited generalizability of trial findings and real-world clinical practice.
The increasing availability of real-world data (RWD), including electronic health records (EHRs), has expanded the potential of observational research to complement trials and support clinical and policy decision making. While observational studies may better reflect the target population’s characteristics, they are susceptible to various sources of bias, such as confounding, selection bias, and prevalent user bias (Supplemental Material 1; Nguyen et al 2021), which have been discussed in dental observational studies (Celeste 2020; Jamieson 2020; Kaaber et al 2025)
The target trial emulation (TTE) framework has been proposed to address these limitations. TTE provides a structured approach for designing and analyzing observational data to estimate causal effects by mimicking the design of a hypothetical trial (the “target trial” [TT]) that could have been conducted to address the same research question (Hernan and Robins 2016; Hernan 2021). In practice, TTs emulated with RWD are inherently pragmatic trials in which neither placebo nor blinding of treatment assignment and outcome ascertainment is possible (Hernan 2021).
TTE has been widely applied by several methodological approaches, including the active comparator new user design, the clone-censor-weight design, and the sequential trial emulation (Fu 2023). Regardless of the chosen design, a TT protocol must specify the same components as a trial: eligibility criteria, treatment strategies, treatment assignment, timing of follow-up, outcomes, causal contrast, and statistical analysis (Hernan and Robins 2016).
A key strength of TTE is its ability to prevent avoidable biases by ensuring that 3 essential components of the TT protocol are aligned at baseline (time zero): 1) all eligibility criteria must be met, 2) treatment strategies are assigned, and 3) follow-up begins. Failure to align these components introduces biases such as immortal time bias and selection bias (Supplemental Material 1; Hernan et al 2016, 2025).
TTE has been successfully applied across various fields, demonstrating that emulating a hypothetical trial with observational data can yield valid causal estimates and benchmark results from an existing trial (Scola et al 2023; Wang et al 2023). Importantly, this approach also enables the use of RWD to evaluate the safety and efficacy of interventions in populations often underrepresented in traditional trials (Fu 2023).
Although this methodology has gained rapid traction in the medical literature, its adoption in a dental research question remains limited. Consequently, dental researchers may still be unfamiliar with the concepts, advantages, and practical implementation of TTE. Bond et al (2025) outlined a protocol applying TTE in oral epidemiology; however, limitations of the selected data source prevented its emulation. Therefore, the aim of this article is to summarize the key components of TTE and demonstrate its implementation using EHRs. Additionally, we compare the results of the TTE with those obtained from 2 flawed cohort study designs (naïve cohorts) to highlight the impact of unnecessary researcher-inflicted biases on study results.
Motivating Example: 5-y Effect of Thiazide Monotherapy on Dental Caries Risk in Patients with Hypertension
Dental caries is a multifactorial disease influenced by several risk factors, including the flow and composition of saliva (Pitts et al 2017). Reduced salivary flow (ie, hyposalivation) may lead to xerostomia, the subjective feeling of dry mouth, although xerostomia may also occur with normal salivary flow (Guggenheimer and Moore 2003). Hyposalivation and xerostomia have been associated with several demographic and clinical factors, including autoimmune diseases, polypharmacy, age, and sex (Barbe 2018). The relationship between xerostomia and medication use has primarily been investigated through observational studies, particularly among older adults, who are more likely to experience hyposalivation and are often prescribed multiple medications (ie, polypharmacy; Aliko et al 2015; Villa et al 2015).
A systematic review on medication inducing salivary dysfunction and xerostomia found that over half of the medication (n = 36) acts on the nervous system (Wolff et al 2017). Antihypertensive treatments, including thiazide diuretics, beta-blocking agents, calcium channel blockers, and angiotensin-converting enzyme inhibitors, have also been linked to xerostomia and hyposalivation (Villa et al 2015; Wolff et al 2017). However, evidence on these associations remains limited (Ramirez Martinez-Acitores et al 2020), and only a few studies have examined the potential association between antihypertensive medications and dental caries (Streckfus et al 1985, 1990; Thomson et al 1995; Thomson et al 2002), with no studies comparing the effects across different antihypertensive classes. This unclear evidence may be attributed to the methodological limitations of observational studies, which often carry a high risk of bias.
Considering that thiazide diuretics remain among the most commonly prescribed first-line therapies for hypertension, representing about 20% to 25% of antihypertensive prescriptions (Natov and Schwartz, 2025), and that nearly 90% of US adults aged 20 to 64 y have experienced dental caries (Centers for Disease Control and Prevention 2019), further investigation into whether thiazide use influences caries risk is warranted. To this end, we aimed to emulate a pragmatic clinical trial—that is, a clinical trial designed to reflect real-world clinical practice—evaluating the comparative safety of thiazide monotherapy versus other commonly used first-line antihypertensive monotherapies regarding the risk of dental caries.
Material and Methods
Target Trial Emulation
The TTE framework aims to address causal questions of interventions that could have been answered by conducting a trial, where treatment strategies are assigned at random. In this context, our causal question is as follows: Among people with hypertension, does thiazide monotherapy increase the 5-y risk of developing dental caries as compared with nonthiazide monotherapy (ie, angiotensin II receptor blocker, angiotensin-converting enzyme inhibitor, calcium channel blocker, or beta-blocking agent)?
In this study, we outline the protocol of the TT and its emulation to guide the design process (Table 1). This helps identify and align critical protocol components at time zero, such as eligibility criteria, treatment assignment, and start of follow-up. Notably, the protocol design is an iterative process that benefits from multidisciplinary collaboration to refine design choices and deliver clinical relevance and methodological rigor. This study adhered to the RECORD-PE guidelines, an extension of the STROBE statements for pharmacoepidemiologic studies (Langan et al 2018).
Abbreviated Protocol of the Pragmatic Target Trial and Its Emulation using the All of Us Data from 1990 to 2017.
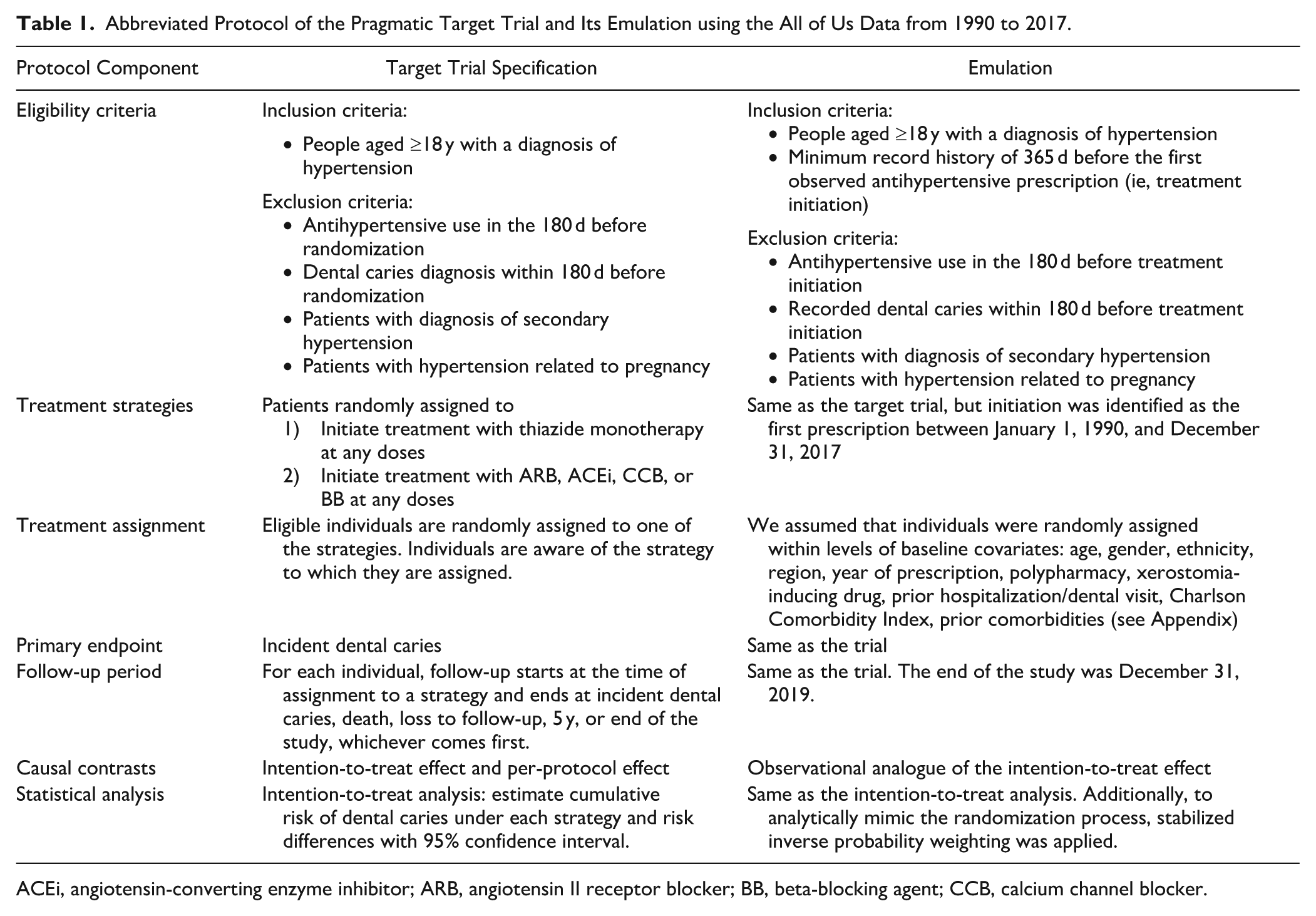
ACEi, angiotensin-converting enzyme inhibitor; ARB, angiotensin II receptor blocker; BB, beta-blocking agent; CCB, calcium channel blocker.
Data Source
In a trial, the recruitment process is carefully planned to ensure enrollment of an adequate and appropriate study population. In TTE, RWD sources replace the recruitment and data collection process. However, this substitution poses several challenges, as the selected data source should represent the target population and have an adequate distribution of patients’ characteristics, interventions, and outcomes under study.
Leveraging RWD is now a widely adopted practice in medical research. Yet, dentistry encounters unique challenges, as routine data sources often lack the same level of standardization, granular information, completeness, and longitudinal follow-up, making their use more complex or infeasible (Benoit et al 2022; Tenuta et al 2022).
For this case study, we relied on the All of Us Research Program, a national US initiative that aims to gather information from >1 million people living across the country. The database contains individuals’ EHRs, including information on diagnoses, procedures, laboratory tests, dental care, and demographics. Additional information is provided in Supplemental Material 2 (see also All of Us Research Program Investigators et al 2019; Ramirez et al 2022).
Eligibility Criteria
Participants in the TT should represent the target population and meet all the eligibility criteria at inclusion. Common eligibility criteria include demographic characteristics, clinical conditions, and measures relevant to the research question. RWD may be insufficient to emulate a specific criterion, as trials often rely on laboratory tests or measurements that may not be routinely collected in clinical practice. In such cases, researchers may need to consider simplifying or omitting them to ensure emulation feasibility. Note that eligibility criteria should generally rely only on information available at baseline, not on future data.
Table 1 shows the eligibility criteria of our TT and its emulation, while Figure 1 illustrates the emulation timeline. See Supplemental Material 3 for the condition and medication concept IDs used.
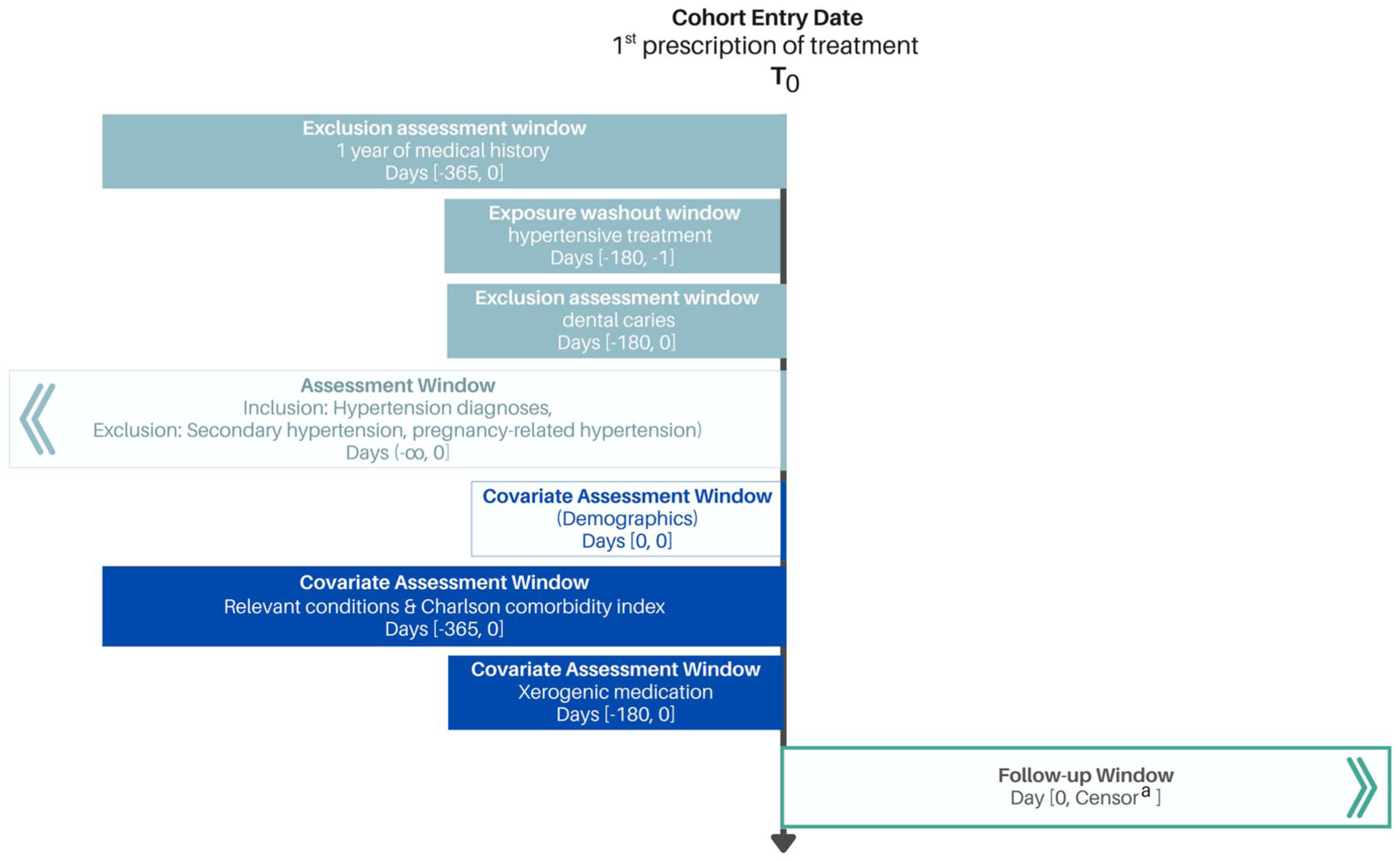
Target trial emulation cohort design. T0, time zero–baseline.
Treatment Strategies
Defining treatment strategies for emulation with existing data can be challenging. Trials often assess specific medications at specific doses or intervals, which can be unfeasible in a TTE due to variability in prescribing practices in routine care, as influenced by guidelines, physician discretion, or budget. Thus, TTs are typically pragmatic, comparing strategies in real-world settings where patients and health care workers are aware of the assigned strategy (Hernan and Robins 2016). Additionally, evaluating newly marketed medications can be difficult due to limited RWD.
In our design, we focused on 5 antihypertensive treatments commonly prescribed as first-line monotherapy (Whelton et al 2018). We defined 2 strategies: 1) initiation of thiazide monotherapy and 2) initiation of nonthiazide monotherapy with an angiotensin II receptor blocker, angiotensin-converting enzyme inhibitor, calcium channel blocker, or beta-blocking agent. Each strategy covers treatment at any dose or frequency.
Although beta-blocking agents have been second-line since 2014, they were previously first-line (James et al 2014); thus, we included them given our study period. We identified medications using their corresponding OMOP concept IDs (Supplemental Material 3.2).
Assignment Procedure
In the TT, patients are randomly assigned to a treatment strategy, ensuring exchangeability between the groups by design. Its emulation with EHRs requires mimicking this randomization process analytically. We can accomplish this by adjusting for baseline variables using methods such as propensity score matching, stratification, standardization, or inverse probability of treatment weighting (Hernan and Robins 2020).
In our emulation, we assumed exchangeability between thiazide initiators and nonthiazide initiators conditional on baseline characteristics (Table 2). It is important to note that individuals can be assigned to only the strategy consistent with their observed data. For instance, patients initiating thiazide could be assigned to only the thiazide initiator group.
Characteristics of the Cohort with Hypertension in the Target Trial Emulation According to the All of Us Program.
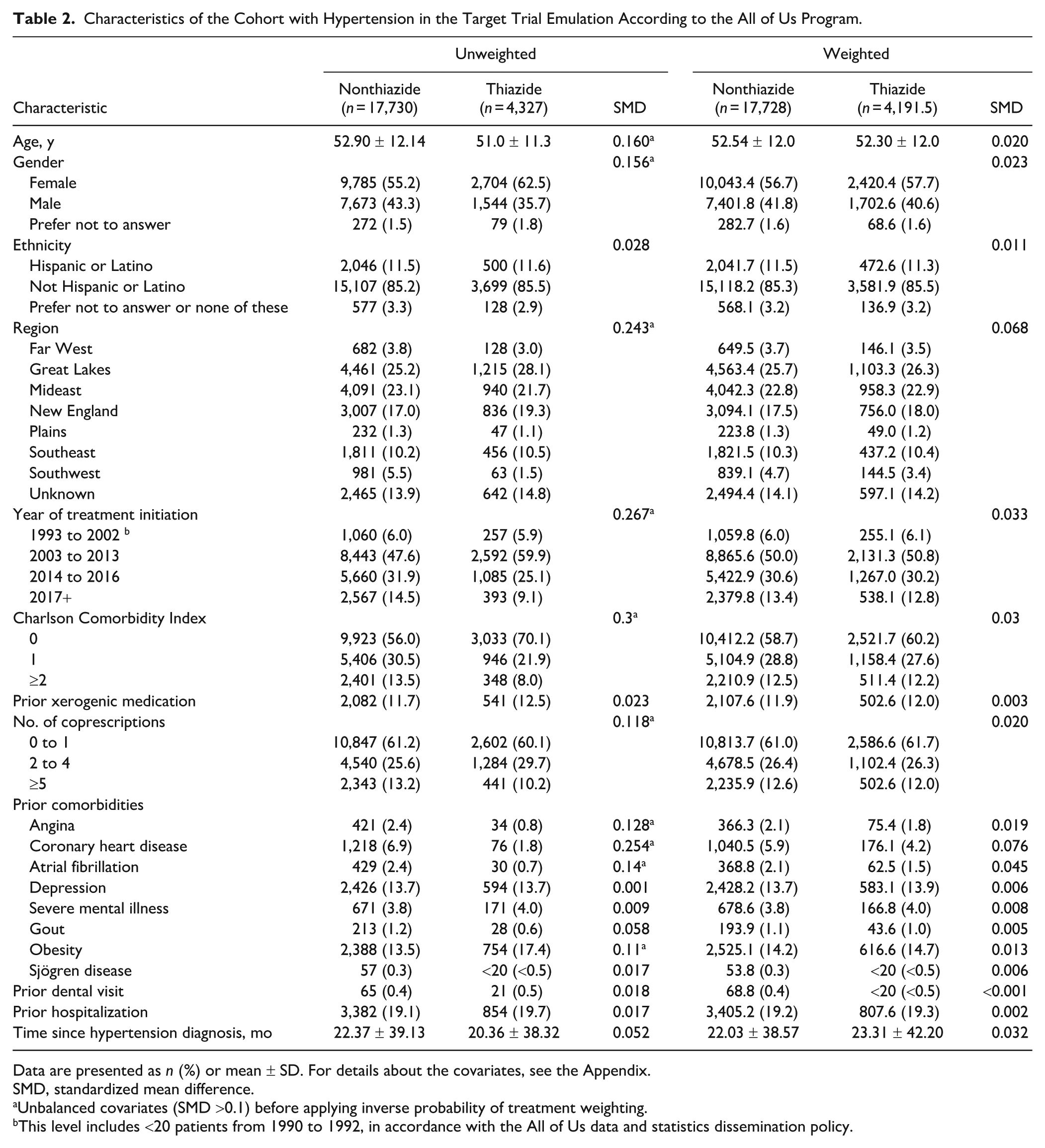
Data are presented as n (%) or mean ± SD. For details about the covariates, see the Appendix.
SMD, standardized mean difference.
Unbalanced covariates (SMD >0.1) before applying inverse probability of treatment weighting.
This level includes <20 patients from 1990 to 1992, in accordance with the All of Us data and statistics dissemination policy.
Outcomes
EHRs provide valuable opportunities for outcome assessment but have important limitations. Certain outcomes, particularly those requiring specialized tests or clinical assessments, may not be routinely recorded. Additionally, sufficient follow-up time is crucial to observe the relevant event within the study period.
In our TT, the outcome was incident dental caries diagnosed by a dental practitioner. In its emulation, we identified dental caries using the corresponding condition concept ID (Supplemental Material 3.3).
Follow-up Period
In the TT, follow-up begins at time zero (the time of the intervention) and continues until the earliest occurrence of a dental caries diagnosis, death, loss to follow-up, 5 y of follow-up, or the end of the study period. In its emulation, time zero was set as the date of the first prescription of the regarding medication. At this point, eligibility criteria had to be met, and treatment was assigned. Follow-up ended according to the TT protocol, and loss to follow-up was defined as the date of the last recorded health care encounter, if there were no further contacts after year 5.
Causal Contrast of Interest
In trials, 2 causal contrasts are often estimated: 1) the intention-to-treat (ITT) effect, which measures the impact of being randomized to a treatment strategy, regardless of whether the individual adhered to the strategy; 2) the per-protocol effect, which evaluates the treatment effect based on those who followed the study protocol as specified (Hernan et al 2013; Murray et al 2021). Our emulation aimed to estimate the analogue ITT effect, which is the effect of initiating treatment at baseline.
Although not implemented in our emulation, an observational analogue of a per-protocol analysis could be conducted by accounting for treatment adherence, making use of prescription refill data (Hernan and Hernandez-Diaz 2012; Hernan and Robins 2020).
Statistical Analysis
The TT protocol includes a tailored statistical analysis plan to address the causal question appropriately. Unlike the TT, the emulation requires an additional analytical method to mimic randomization and minimize confounding. In our emulation, we used stabilized inverse probability of treatment weighting (Supplemental Material 5).
Once both intervention groups had comparable (balanced) pretreatment characteristics, we fit a weighted pooled logistic regression to estimate cumulative incidence curves and risk differences over a 5-y follow-up period (Supplemental Material 8.2). We applied standardization based on the fitted outcome model to obtain marginal effects under each treatment strategy. Nonparametric bootstrapping with 500 replicates was used to derived t-based 95% confidence intervals (Hernan and Robins 2020; Murray et al 2021).
We calculated the E-value for the main analysis to assess how strongly an unmeasured confounder would need to be associated with treatment initiation and dental caries to explain away the observed association after adjustment for all measured confounders (VanderWeele and Ding 2017).
We conducted complete case analyses assuming that data were missing at random. All analyses were conducted in R version 4.5.0.
Naïve Cohorts
Prevalent User Cohort
The prevalent user cohort consisted of individuals who met the TT eligibility criteria, except that it also included those already on antihypertensive treatment before time zero (ie, prevalent users). This deviation in the eligibility criteria introduces prevalent user bias (Fig. 2). In this cohort, time zero was correctly specified.
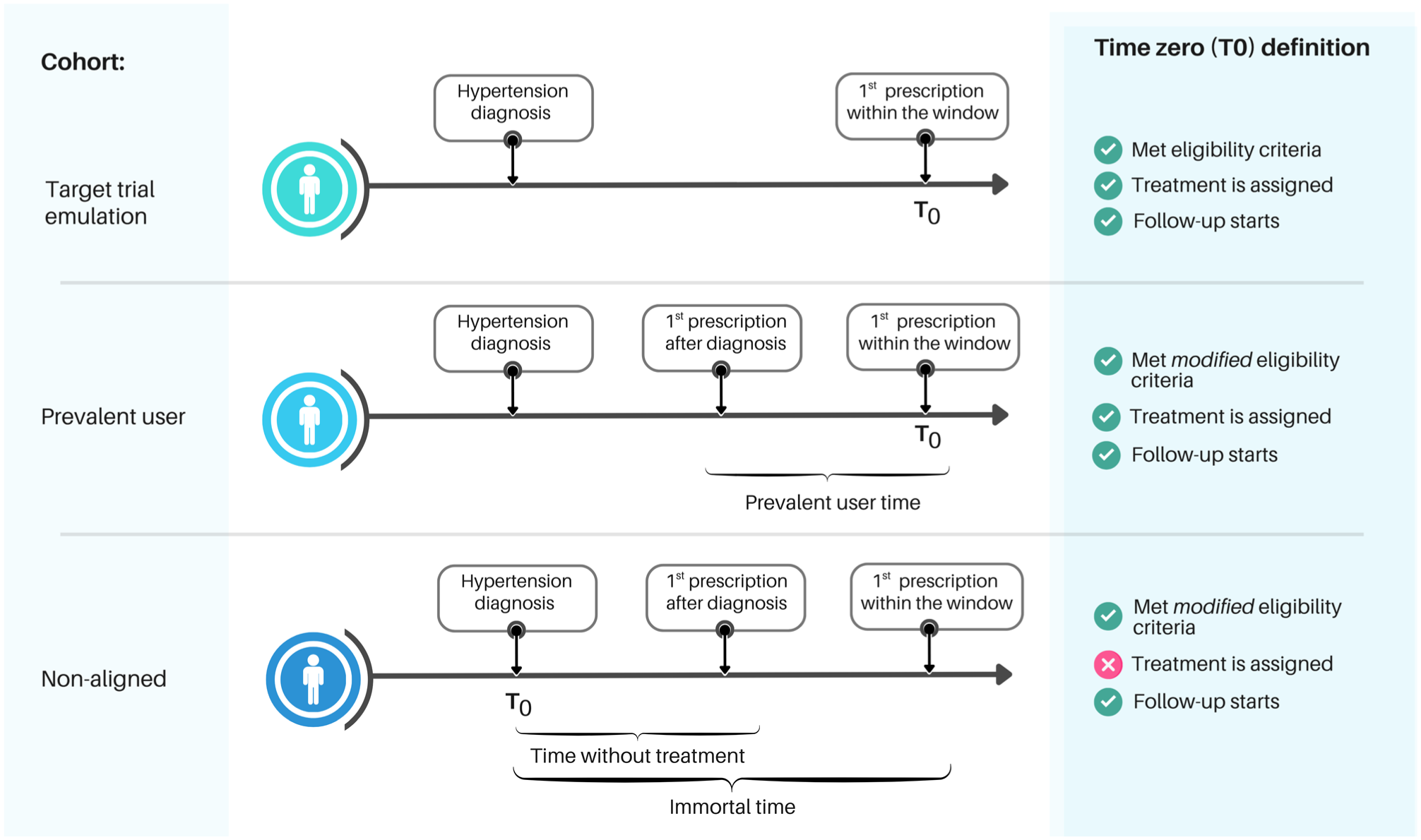
Time zero across the 3 cohorts. In the target trial emulation cohort (top panel), time zero was correctly aligned with treatment assignment—first prescription within the established window—and eligibility was confirmed at that point. Follow-up began at the same time, avoiding immortal time bias. Prevalent users were excluded, minimizing prevalent user bias. The prevalent user cohort (middle panel) included prevalent users: individuals who had initiated treatment before the start of the window, introducing prevalent user bias. Follow-up of the nonaligned cohort (bottom panel) started at hypertension diagnosis, introducing immortal time bias. The only difference between the “met eligibility criteria” and “met modified eligibility criteria” is the inclusion of prevalent users in the latter.
Nonaligned Cohort
The nonaligned cohort was defined similarly to the prevalent user cohort, except that follow-up began at the hypertension diagnosis, regardless of treatment initiation. This misaligned time zero introduces immortal time bias and reclassifies some prevalent users as untreated during the pretreatment period. Consequently, treatment-free periods add bias by misallocating time (Fig. 2).
We applied the same statistical analysis used in the TTE to the naïve cohorts to evaluate how the improved cohort definitions under the TTE framework reduce bias and enable causal inference.
Results
The final TTE cohort included 22,057 patients. Table 2 presents baseline characteristics stratified by the monotherapy initiated. The majority of participants were female (56.6%), with a mean age of 52.5 y (SD, 12.0). Most identified as not Hispanic or Latino (85.3%). Within the cohort, 4,327 patients (20%) initiated thiazide monotherapy between 1990 and 2017 (see Appendix Fig. 1 for initiation trends). When compared with the nonthiazide group, patients in the thiazide group showed a lower Charlson Comorbidity Index and fewer cardiovascular comorbidities.
At baseline, 10 of 19 covariates were unbalanced. After weighting, covariate balance was achieved across all variables (Table 2, Appendix Fig. 2). During the 5-y follow-up, 785 patients experienced dental caries. The adjusted 5-y risk was 3.75% in the thiazide group and 3.46% in the nonthiazide group, yielding a risk difference of 0.29% (95% CI, −0.36% to 0.95%; Fig. 3). The E-value risk ratio was 1.38 (Appendix Section 6).
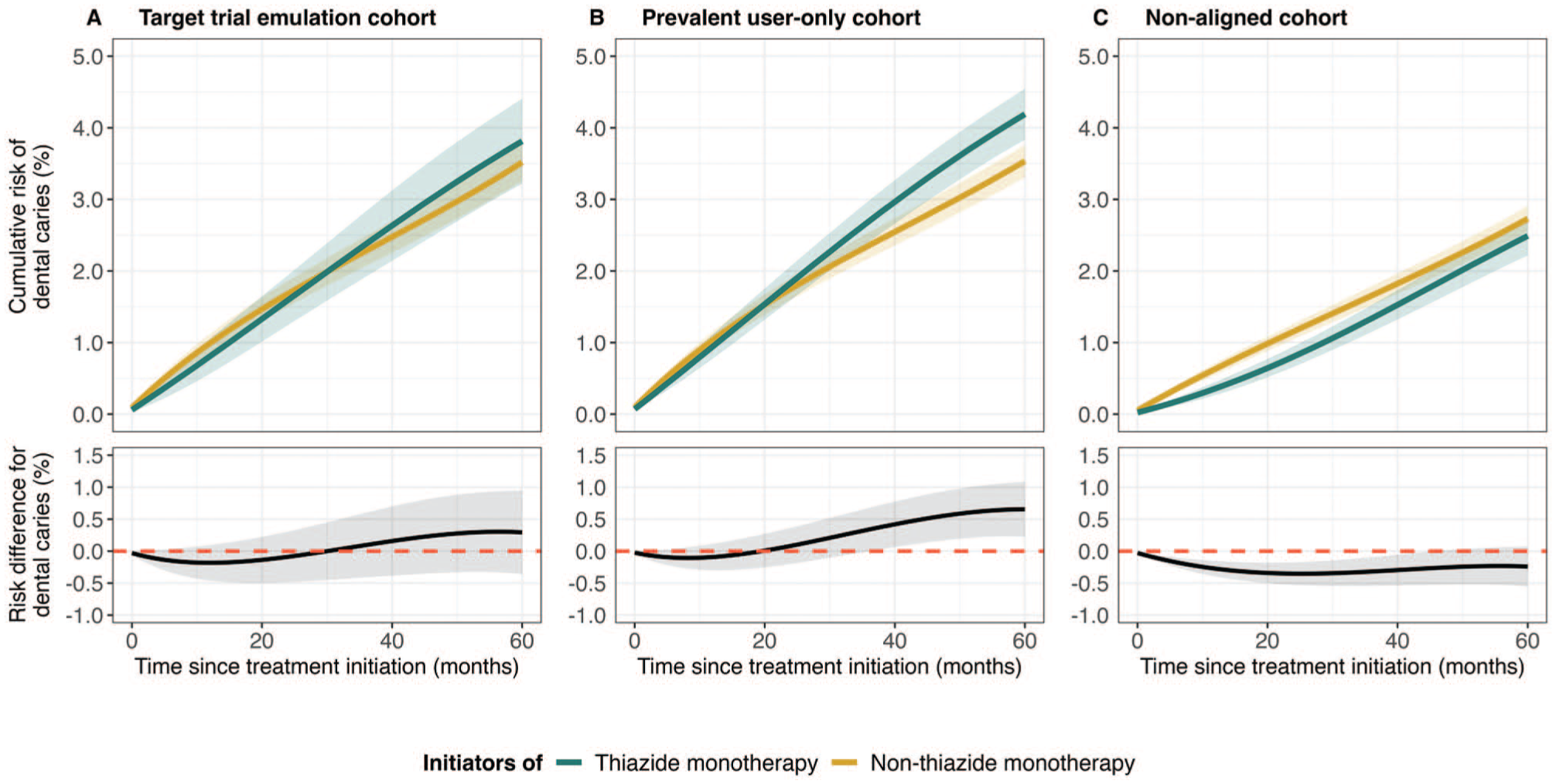
Cumulative risk and risk difference among patients with hypertension initiating monotherapy. Each panel shows the cumulative risk curves (top) for thiazide initiators (green line) and nonthiazide initiators (yellow line). The lower section displays the estimated risk differences (black line), with the red dashed line indicating the null effect. Shaded areas represent t-based 95% confidence intervals derived from 500 bootstrap samples. (
The prevalent user cohort included 39,879 patients, while the nonaligned cohort comprised 40,534 patients. Their baseline characteristics were generally similar to those of the TTE cohort (Appendix Tables 1 and 2). Within the prevalent user cohort, 1,482 patients developed dental caries during follow-up. The adjusted 5-y risk was 4.13% in the thiazide group and 3.48% in the nonthiazide group, resulting in a risk difference of 0.65% (95% CI, 0.22% to 1.09%). In the nonaligned cohort, which recorded 1,067 events, the adjusted 5-y risk was 2.44% in the thiazide group and 2.68% in the nonthiazide group. This resulted in a risk difference of −0.24% (95% CI, −0.55% to 0.07%; Fig. 3).
Discussion
We aimed to demonstrate how the TTE framework can help address common biases in observational studies. In this study, we evaluated a safety concern regarding the potential association between thiazide use and the development of dental caries using EHRs. Our findings showed no difference in the risk of dental caries between individuals who initiated thiazide monotherapy and those who initiated nonthiazide monotherapy over the 5-y follow-up in the TTE cohort.
Thiazides are frequently cited as xerogenic medications, with the potential to impair salivary function. Although previous literature has associated thiazide and other antihypertensive medications with xerostomia and/or hyposalivation, this association remains inconclusive. Consequently, the proposed biological mechanism—whereby thiazide use alters salivary flow and composition, leading to increased dental caries—remains hypothetical. Some cross-sectional studies have reported a negative association between antihypertensive treatment and dental caries (Streckfus et al 1990; Thomson et al 2002); however, the designs employed in existing studies were susceptible to confounding factors and other sources of bias. Furthermore, no studies have examined the comparative effect of the different antihypertensive medications on dental caries.
To illustrate the importance of proper study design, we created 2 naïve cohorts and demonstrated how common biases, such as immortal time bias and prevalent user bias, can distort research findings.
In the prevalent user cohort, where time zero was correctly specified but included prevalent users, risk differences were initially close to the null. However, these differences increased over time for those who initiated thiazide. This delayed divergence may reflect the influence of prevalent users, who, due to longer cumulative exposure, are more likely to develop dental caries, a long latency outcome, during later stages of follow-up. Among those classified as thiazide initiators, 65.1% had prior use of any studied antihypertensive (ie, were prevalent users), as compared with 46.2% among nonthiazide initiators.
In the nonaligned cohort, follow-up started on the date of hypertension diagnosis, which preceded the correct time zero (ie, first prescription within the specified period) for some individuals (22.5% thiazide vs 36.0% nonthiazide initiators). This introduced immortal time bias, leading to a lower risk estimate in both groups, with thiazide initiators showing an even greater reduction in risk of caries over the follow-up period. This greater reduction may be attributed to a longer “immortal” period for thiazide initiators (53 vs 36 mo). The observed results are expected because immortal time bias artificially lowers the incidence of the outcome by including a period during which an individual must remain event-free to qualify for treatment (Hernan et al 2025). Moreover, significant effect estimates were observed at specific follow-up times.
The 5-y risk of dental caries showed opposite effect directions among the naïve cohorts. In contrast, the prevalent user cohort and the TTE cohort displayed a similar pattern during the follow-up, with some periods being statistically significant in the prevalent user cohort. The observed variation and significant estimates likely reflect the presence of prevalent users in the naïve cohort and its large sample size. Notably, prevalent users may be more susceptible to future events, particularly given the long latency period associated with dental caries.
The TTE framework provides a strong structure that compels researchers to define crucial aspects of study design and analysis explicitly, enhancing the validity of causal estimates (Hernan and Robins 2016; Hansford et al 2023; Wang et al 2023) and facilitating an informed discussion of bias caused by various design choices. In this study, we mitigated immortal time bias and prevalent user bias by aligning protocol components that are inherently synchronized in trials but often overlooked in observational research. Additionally, we ensured that all participants in the emulated TT were classified as new users of the treatment drugs.
While our TTE offers a robust approach for estimating causal effects using routine data, several limitations should be acknowledged. First, as with all observational studies, confounding remains a concern, especially due to unmeasured covariates. However, the TTE used an alternative antihypertensive as an active comparator, thereby controlling for confounding by design. In addition, we incorporated available proxy variables for some unmeasured confounders (Appendix Section 4). After adjusting for observed confounders, it is unlikely that initiators of different antihypertensive classes differ meaningfully in terms of unmeasured confounders.
Some important prognostic variables for dental caries (eg, exposure to fluoridation) were not present in the EHRs and were therefore not included in the propensity score model. To the extent that these factors are truly prognostic factors for dental caries, their omission would be expected to primarily reduce precision (Brookhart et al 2006).
Second, additional limitations inherent to EHRs, such as incomplete information and coding inaccuracies, may affect our findings. These issues are particularly relevant for dental data, which is often fragmented across providers, and the infrequency of routine checkups increases the risk of outcome misclassification. The measurement of dental caries presents specific challenges. Observational studies often rely on indices such as DMFT (decayed, missing, filled teeth) or DMFS (decayed, missing, filled surfaces) to define dental caries burden; however, these indices are rarely recorded directly, and their components are often inconsistently documented in structured EHRs, limiting study comparisons (White et al 2019). Nevertheless, there is no reason to believe that clinicians’ likelihood of diagnosing or recording dental caries would vary depending on whether a patient was treated with thiazide or nonthiazide antihypertensives, and any resulting misclassification of dental caries would likely be nondifferential, thus biasing the results toward the null.
Finally, it is important to note that our results may be generalized only to populations with characteristics comparable to those of the participants in this TTE.
Conclusion
Although trials remain the gold standard for evaluating treatment efficacy and safety, the TTE framework offers a strong alternative for generating robust evidence. We illustrated how dental research can benefit from applying this approach.
Future work should extend the use of this framework to other oral health topics, leveraging enriched datasets and linkage to dental records to strengthen observational causal inference in dental research.
Author Contributions
I. Leiva-Escobar, contribution to conception and design, data acquisition, analysis and interpretation, drafted and critically revised the manuscript; A. Puzhakkara Chennas, S.L. Reckelkamm, L. Gelberg, contribution to conception and design, critically revised the manuscript; S.-E. Baumeister, M. Nolde, contribution to conception and design, data acquisition and interpretation, drafted and critically revised the manuscript. All authors gave final approval and agreed to be accountable for all aspects of the work.
Supplemental Material
sj-docx-1-jdr-10.1177_00220345261429938 – Supplemental material for Target Trial Emulation to Assess Oral Drug Safety
Supplemental material, sj-docx-1-jdr-10.1177_00220345261429938 for Target Trial Emulation to Assess Oral Drug Safety by I. Leiva-Escobar, A. Puzhakkara Chennas, S.L. Reckelkamm, L. Gelberg, S.-E. Baumeister and M. Nolde in Journal of Dental Research
Footnotes
Acknowledgements
We gratefully acknowledge the All of Us participants for their contributions, without whom this research would not have been possible. We also thank the National Institutes of Health’s All of Us Research Program for making available the participant data examined in this study.
A supplemental appendix to this article is available online.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Deutsche Forschungsgemeinschaft (grant 552087827).
