Abstract
Conventional bioactive glasses enhance mineralization in glass-ionomer cements (GICs), but their high sodium content adversely affects mechanical performance, limiting clinical utility. To address the limitations of bioactive glass, this study explores a novel sodium-free, fluoride- and magnesium-enriched bioactive glass (J-BAG) in GICs. The mechanical performance, ion release, antibacterial efficacy against multispecies cariogenic biofilms, and mineralization capacity were investigated. The J-BAG (38.3SiO2–6P2O5–43.9CaO–6.8CaF2–5MgO) and two experimental ionomer glasses were synthesized using the melt-quench technique. A new ionomer glass, LG99Sr-Mg-Zn, with molar composition (4.5SiO2–3Al2O3–1.5P2O5–3SrF2–0.5SrO–1MgO–0.5ZnO), was developed from a previously established LG99Sr (4.5SiO2–3Al2O3–1.5P2O5–3SrF2–2SrO), with Fuji IX (GC Corporation, Tokyo, Japan) serving as the reference material. Incorporating J-BAG at 5 and 10 weight% into the experimental Mg-Zn and reference GICs resulted in significant improvements in compressive and flexural strength as well as microhardness, compared with unmodified GICs, with further gains noted postmaturation. However, mechanical properties declined at 15% loading, especially with finer J-BAG particles. Therefore, the 5% J-BAG–loaded LG99Sr-Mg-Zn (LG5) and 10% J-BAG–loaded Fuji IX (F10) formulations were selected for further testing. LG5 and F10 exhibited significantly enhanced fluoride release, along with elevated magnesium and zinc release from LG5. Antibacterial assays revealed biofilm inhibition and reduced live biomass (P < 0.05) compared with controls. Mineralization studies confirmed that higher J-BAG concentrations and longer immersion in media led to greater mineral deposition. In addition, LG5 and F10 facilitated mineral deposition on demineralized dentine surfaces and partially occluded dentinal tubules. In conclusion, the incorporation of J-BAG at 5% in Mg-Zn–fortified GIC and 10% in Fuji IX simultaneously enhanced mechanical properties, ion release, mineralization, and antibacterial performance. This multifunctionality supports the therapeutic and restorative roles of GICs in clinical dentistry. It offers a promising solution for atraumatic restorative treatments and long-term tooth rehabilitation in patients at high risk of caries.
Keywords
Introduction
The dental field is undergoing a transformative shift, driven by the innovative development of restorative materials. One notable direction is the remarkable transformation fueled by advancements in bioactive materials (Schmalz et al 2023) and their restorative and reconstructive applications. Bioactivity, once defined narrowly as the capacity to induce mineralization, is now recognized as a dynamic multifaceted phenomenon involving complex biological and chemical interactions (Schmalz et al 2023). As highlighted by Imazato et al (2023), the FDI World Dental Federation emphasizes that bioactive functions transcend mere mineralization. Recent expert consensus underscores the need for contemporary restorative materials to actively promote calcium phosphate nucleation, support tissue repair, and modulate microbial activity, an evolving paradigm particularly relevant to caries management and improved clinical outcomes (Price and Roulet 2020; Ferracane et al 2023; Schmalz et al 2023).
Glass-ionomer cement (GIC) remains a valuable restorative material due to its chemical interaction with hard tissues and its controlled release of fluoride and other therapeutic ions, offering notable, albeit temporary, antibacterial effects. Their intrinsic adhesive qualities foster ionic bonding with enamel and dentine (Toledano et al 2021; Nicholson et al 2024). This predilection for ion release and chemical bond formation makes them particularly attractive for atraumatic restorative treatments in pediatric and special care dentistry under challenging moisture management conditions. Despite their clinical advantages, GICs exhibit inherent mechanical limitations that restrict use in high-stress posterior restorations (Hill 2022; Makanjuola et al 2022). Recent research trends aim to enhance antibacterial efficacy and mechanical performance simultaneously. While natural additives (eg, chitosan, propolis) and synthetic agents (eg, metallic dopants, quaternary ammonium salts) show promise, excessive concentrations often compromise strength and other properties (Ching et al 2018; Makanjuola and Deb 2023). We recently developed LG99Sr-Mg-Zn, a biocompatible ionomer glass leveraging the synergistic antibacterial effects of zinc and magnesium, meeting ISO standards while maintaining mechanical integrity (Makanjuola et al 2025).
Incorporating conventional bioactive glass (BAG) into GICs compromises mechanical properties due to diminished Al³+ availability, which hinders the development of robust 3-dimensional crosslinking networks. The original BAG, 45S5 Bioglass®, developed by Larry Hench, comprises of 46.1 mol% SiO2, 24.4 mol% Na2O, 26.9 mol% CaO, and 2.6 mol% P2O5 (Hench 2006; Jones 2015). The high sodium content in 45S5 enhances BAG solubility but limits polyacrylate crosslinking; thus, omitting Na2O and using alternative modifiers while maintaining network connectivity (~2.10) is essential (Chen et al 2017). The integration of bioactive materials exhibiting intrinsic antibacterial properties into GICs presents a promising strategy for augmenting their clinical performance. These additives not only bolster the native fluoride-mediated caries inhibition characteristic of GICs but also release essential ions, calcium and phosphate, inducing mineralization. Such innovations can enhance the long-term stability of the tooth-restoration interface and promote regenerative outcomes in demineralized dental tissues. This study formulated a sodium-free, phosphate-rich, fluoride- and magnesium-containing BAG termed J-BAG. The optimal particle size and concentration of J-BAG to improve the mechanical properties, ion release, antibacterial activity, and mineralization of LG99Sr-Mg-Zn were determined. The results were benchmarked against the modification of a well-established high-viscosity GIC, Fuji IX GP (GC Corporation, Tokyo, Japan).
Materials and Methods
Glass Synthesis
Two experimental ionomer glasses and a newly engineered sodium-free BAG, J-BAG, were synthesized using a conventional melt-quench technique. LG99Sr-Mg-Zn, with a molar ratio of 4.5SiO2-3Al2O3-1.5P2O5-3SrF2-0.5SrO-1MgO-0.5ZnO, was developed from its internal control LG99Sr (4.5SiO2-3Al2O3-1.5P2O5-3SrF2-2SrO) (Guida et al 2002). J-BAG of novel composition, 38.3SiO2-6P2O5-43.9CaO-6.8CaF2-5MgO, was also synthesized through the melt process. Reagents (Sigma-Aldrich, Gillingham, UK) were mixed in 200-g batches and melted in a platinum–rhodium crucible using a furnace (EHF 17/3, Lenton, Hope Valley, UK) at 1,450 °C for ionomer glasses (1 h) and 1,500 °C for J-BAG (2 h). The molten glass was quenched in cold water to obtain clear frits and then vacuum dried for 12 h. A 100-g batch of frit was vibratory milled twice for 7 min/cycle (Gyro Mill, Glen Creston, London, UK). Ionomer glasses were sieved (<45 µm), while J-BAG was sequentially fractionated using 38-, 45-, 60-, and 90-µm meshes (Endecotts Ltd., London, UK) to obtain particle size ranges of <38, 38 to 60, and 45 to 90 µm.
Particle Size Analysis
Particle size was measured in triplicate using a laser diffraction analyzer (Malvern 2000, Worcestershire, UK) based on the spatial distribution of a monochromatic light beam.
X-Ray Diffraction
X-ray diffraction (XRD) patterns of glass powders were obtained using Panalytical Empyrean (Almelo, The Netherlands) with Cu-Kα1 radiation (1.54Å) at 4 kV and 40 mA. Scans ranged from 10° to 70° 2θ, with a step size of 0.02° and a scanning speed of 0.04°/min.
Interaction of J-BAG Powder in Tris Buffer Solution
Tris buffer solution was prepared by dissolving 15.09 g of tris(hydroxymethyl)aminomethane in 800 mL deionized water, then adding 44.2 mL 1M HCl, and incubating at 37 °C on a shaker. The pH was adjusted to 7.30 using 1M HCl, and the final volume was adjusted to 2,000 mL with deionized water (Brauer et al 2011). Thereafter, 75 mg of J-BAG was incubated in 50 mL of the solution and agitated at 60 rpm for 3 to 772 h. Following centrifugation and particle retrieval, the supernatant’s pH was measured, and dried samples underwent attenuated total reflectance–Fourier transform infrared spectroscopy (FTIR) and XRD analysis.
Evaluation of Physical Properties
Specimen preparation
LG99Sr and its Mg-Zn variant were prepared using a 2.4:1 powder-to-liquid ratio, as previously established (Makanjuola et al 2025). To minimize variability, Fuji IX liquid was used across all formulations, with the reference GIC mixed at 3.6:1, per manufacturer guidelines. Homogeneous incorporation of J-BAG (0 to 15 wt%) was achieved via vortexing and agate mixing. After establishing an optimal powder-to-liquid ratio, 24-h mechanical testing assessed the influence of J-BAG particle size and concentration. Selected BAG-GIC groups were matured in deionized water at 37 °C before further mechanical evaluation.
Setting time determination
Setting times of the different BAG-GICs (n = 5 per group) were measured at ambient temperature, 23.0 ± 1 °C, using a 453.6-g Gillmore needle and 1.06-mm indenter (Impact Test Equipment Ltd, Stevenston, UK) and a digital timer according to ISO 9917–1:2007 (see Appendix).
Compressive strength
Compressive strength (CS) tests (n = 8) were performed on cylindrical specimens, 6 mm × 4 mm, at each time point, according to ISO 9917–1:2007, as performed in our previous study (Makanjuola et al 2025).
Flexural strength
At each time point, flexural strength (FS) was determined by conducting a 3-point bending test on specimens measuring 25 mm × 2 mm × 2 mm (n = 8), according to ISO 4049:2019.
Vickers microhardness
Polished disc-shaped specimens (10 mm × 2 mm) were subjected to indentations (n = 6 × 5 indentations/disc) 100-µm apart across the surface by applying a 50-g load for 10-s dwell time using Durascan G5 Hardness Tester (Emco-Test, Kuchl-Salzburg, Austria).
Fluoride release
Ten specimens (6 mm × 4 mm) were immersed in 50 mL of deionized water at 37 °C and agitated at 60 rpm. The medium was refreshed daily for 1 wk, then weekly for 1 mo. Fluoride release was measured using DX219-F and DX200 electrodes after adding 5 mL of total ionic strength adjustment buffer III to each eluent (further details are given in the Appendix).
Inductively Coupled Plasma–Mass Spectroscopy (ICP-MS) Analysis
Mg2+ and Zn2+ release from BAG-GIC specimens was measured on days 7, 14, 21, and 30 following storage in acid-treated Eppendorf tubes containing ultrapure water, using NexION 5000 Triple Quadrupole inductively coupled plasma–mass spectroscopy analysis (PerkinElmer, Waltham, Massachusetts, USA) and multielement standards (see Appendix).
Mineralization Studies
Specimen discs (10 mm × 2 mm) per group were immersed in Tris buffer solution and simulated body fluid (SBF). Preparation of SBF solutions followed the established protocol described by Kokubo and Takadama (2006). BAG-GIC discs (J-BAG ranging from 5% to 15%) were separately immersed in each medium, refreshed every 2 d, retrieved after 28 d, rinsed, and dried in a desiccator. Dried BAG-GIC discs were mounted and sputter coated with carbon to improve conductivity. Microstructural analysis was conducted using a scanning electron microscope (JCM-6000 Plus, JEOL, Tokyo, Japan) to identify mineralized layers, followed by energy-dispersive X-ray spectroscopy (EDS) for elemental analysis. In addition, ethical approval (IRAS 331785; REC: 25/EE/0020) was obtained prior to assessing the mineralization induction capacity of LG0, F0, LG5, and F10. Three premolars per group were demineralized using 17% ethylenediamine tetra-acetic acid (Coltene/Whaledent, Langenau, Germany), approximated with BAG-GIC blocks (6 × 6 × 4 mm³) and immersed in SBF for 14 d. After immersion, dentine surfaces were rinsed, fixed in glutaraldehyde, and prepared for scanning electron micrograph imaging using standard protocols, and the precipitates formed on the BAG-GIC blocks were quantitatively analyzed using EDS (see Appendix).
Antibacterial Studies
Biofilm inhibition test
Biofilms were established using four bacterial species, each selected for their relevance to either cariogenic or endodontic origins: Streptococcus mutans NCTC 10449, Lactobacillus paracasei NCTC 2649, Streptococcus mitis/oralis, and Cutibacterium acnes (retrieved from a study by Niazi et al 2010). Selected bacterial strains were used to develop a multispecies biofilm model that simulates the complexity of oral microbial ecosystems. Each strain was revived from −80 °C glycerol stocks, plated on Fastidious Anaerobe Agar supplemented with 5% defibrinated horse blood and cultured anaerobically at 37 °C in a Don Whitley A95 TG workstation (80% N2, 10% H2, 10% CO2). Starter cultures were prepared in modified fluid universal medium (mFUM) and adjusted to an optical density of 0.5 at 540 nm (~107 cells/mL). Biofilms were grown on ultraviolet-sterilized BAG-GIC and presterilized hydroxyapatite control (3D Biotek, New Jersey, USA) discs (n = 4), according to the benchmark protocol by Niazi et al (2014). The discs were incubated anaerobically in 1 mL of culture medium for 24 h, replenished daily with mFUM for 7 d and then subjected to 7 d of nutrient starvation. Viable counts were obtained by vortexing discs in Brain Heart Infusion broth (Oxoid, Thermo Fisher Scientific, Hampshire, UK), followed by five serial dilutions and plating for enumeration after 7 d. Following the single-species experiment, hydroxyapatite (HA) discs were inoculated with 300 µL (3 × 106 cells) of each strain for the multispecies validation. After 24 h, the identical incubation and starvation protocol was applied, followed by quantitative viable count evaluation. After validation, the multispecies biofilm test for the different groups (n = 4) was executed by repeating the steps performed during the multispecies validation. Further details are provided in the Appendix.
Live/dead bacterial viability
BAG-GIC and HA discs (n = 4), cultured with 14-d multispecies biofilms, were rinsed with sterile phosphate-buffered saline. Biofilms were stained with 50 µL of resuspended SYTO-9 and propidium iodide (LIVE/DEAD™ BacLight™, ThermoFisher Scientific, Eugene, OR, USA). Postincubation, confocal imaging (DM-IRE2, Leica Microsystems, Germany) was performed at 63× magnification. Five random z-stacks per disc were acquired, and biomass was quantified using ImageJ software (v1.54 g).
Data Analysis
Two-way analysis of variance and Tukey–Kramer honestly significant difference post hoc tests were performed using GraphPad Prism 9.5.1 (733) (GraphPad Software, San Diego, CA, USA) to determine the differences in physical and bioactive properties between the groups. Statistical significance was set at P < 0.05.
Results
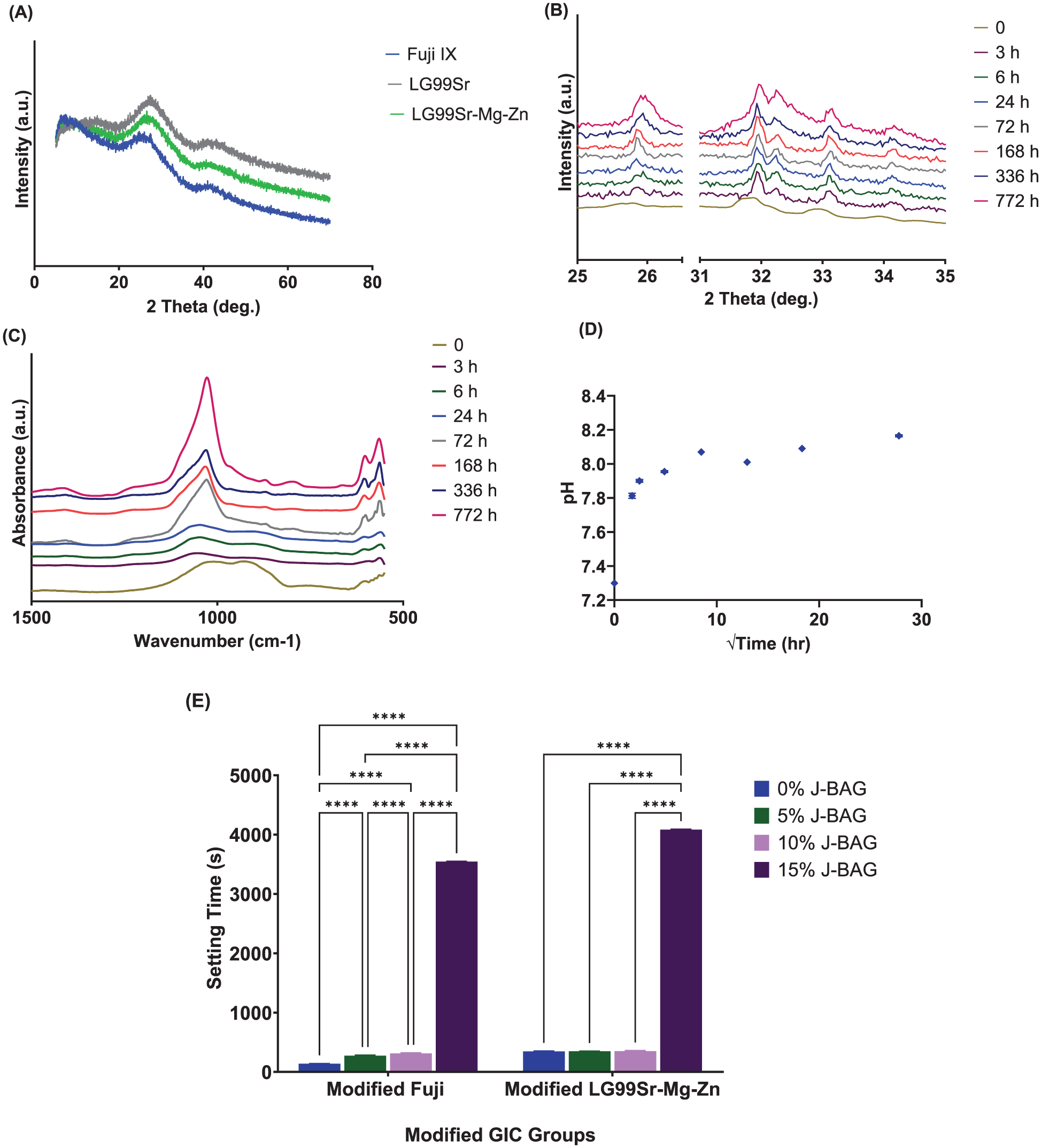
Fuji IX, LG99Sr, and LG99Sr-Mg-Zn powders had comparable average particle sizes, and the particle size distribution of J-BAG powders is shown in Table A. The optimal powder-to-liquid ratios of the BAG-GICs are shown in Table B. XRD results confirmed the amorphous nature of the ionomer glasses (Fig 1A). J-BAG post-immersion in tris buffer exhibited progressive XRD peak intensification between 26°–35° 2θ, indicating apatite formation over time (Fig 1B). FTIR analysis revealed the loss of vibrational bands around 1,060 and 950 cm−1 attributed to nonbridging oxygens, while characteristic apatite split bands at 560 and 600 cm−1 and a phosphate band near 1,050 cm−1 emerged and developed over time (Fig 1C). The pH increased from 7.3 to 8.0, stabilizing at about 8.17 after 772 h (Fig 1D). Incorporating more than 10% J-BAG into LG99Sr-Mg-Zn and Fuji IX significantly altered setting time beyond ISO standards (P < 0.05) (Fig 1E).
(
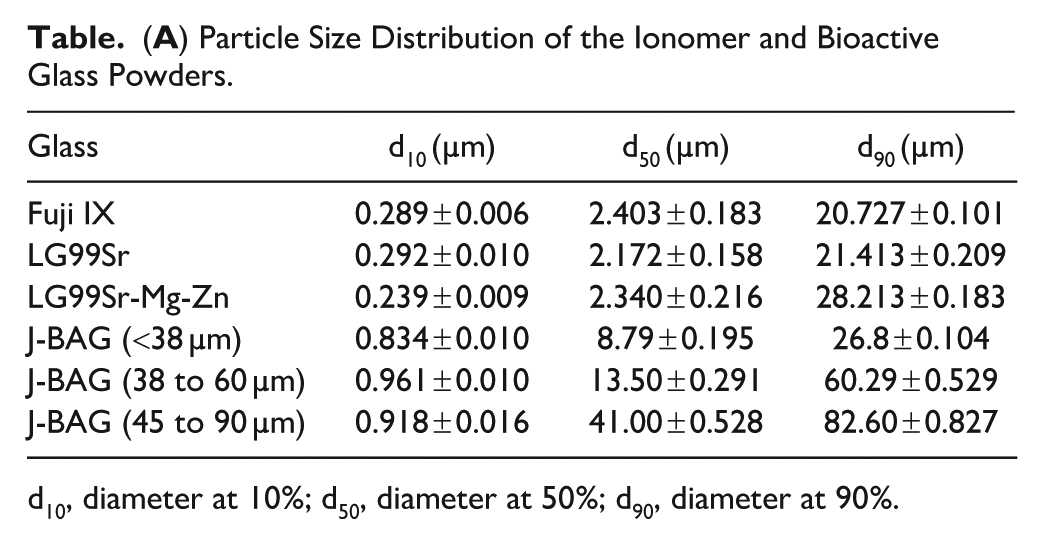
d10, diameter at 10%; d50, diameter at 50%; d90, diameter at 90%.
(
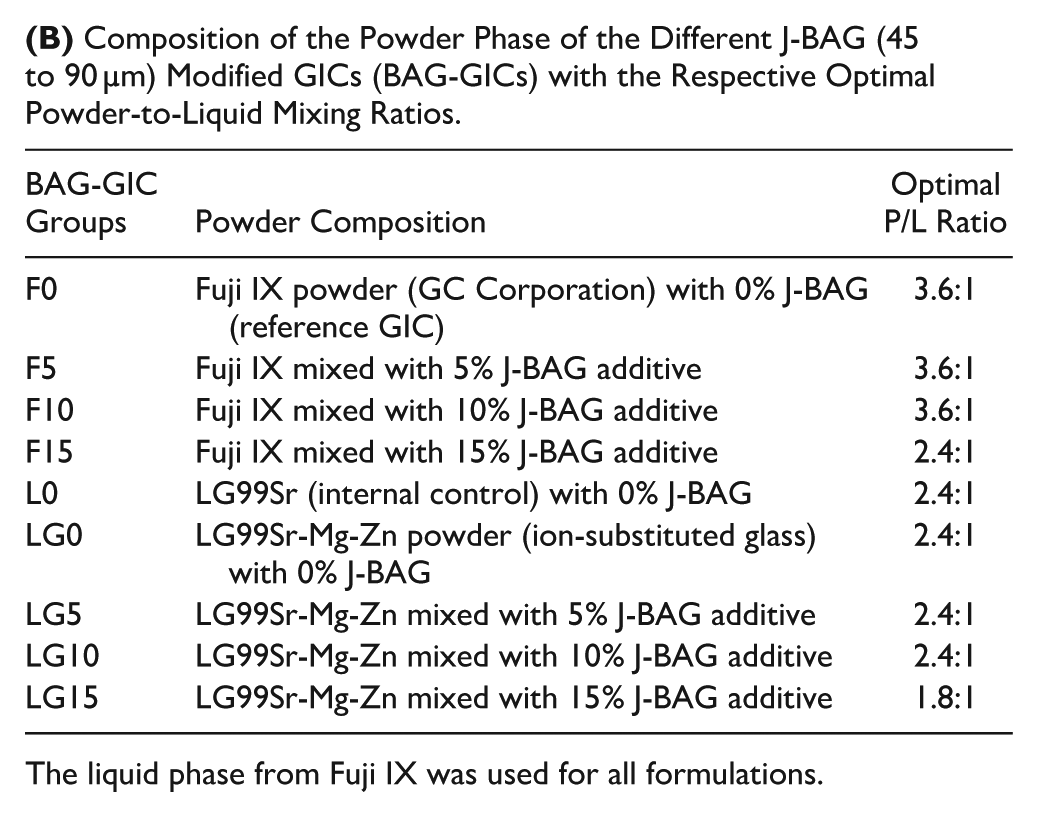
The liquid phase from Fuji IX was used for all formulations.
(
Adding J-BAG of varying sizes into LG99Sr-Mg-Zn at 5% and 10% by weight led to a significant reduction in CS (P < 0.05), except for the 45- to 90-µm fraction at 5%. Conversely, incorporating J-BAG into Fuji IX resulted in a notable enhancement of CS (P < 0.05), especially in the specific size fraction (Fig 2.1A and B). FS was significantly higher (P < 0.05) in J-BAG–modified Fuji IX and LG99Sr-Mg-Zn (5% and 10%, various sizes) than their unmodified counterparts (Fig 2.1C and D). Microhardness measurements (Fig 2.1E and F) revealed significant improvements in the LG5 and F10 groups (P < 0.05). Data indicated that incorporating 15% J-BAG negatively affected the mechanical properties of BAG-GICs. Notably, LG5 and F10 modified with 45 to 90 µm J-BAG exhibited superior mechanical properties, further enhanced after 1 mo of maturation (Fig 2.2A–C). The BAG-GIC groups showed similar fluoride release patterns, with LG5 and F10 exhibiting significantly elevated fluoride release compared with their unmodified counterparts. ICP-MS analysis confirmed that magnesium and zinc release increased substantially (P < 0.05) by loading LG99Sr-Mg-Zn with 5% J-BAG, and magnesium was also released from F10 (Fig 2.3A and B).
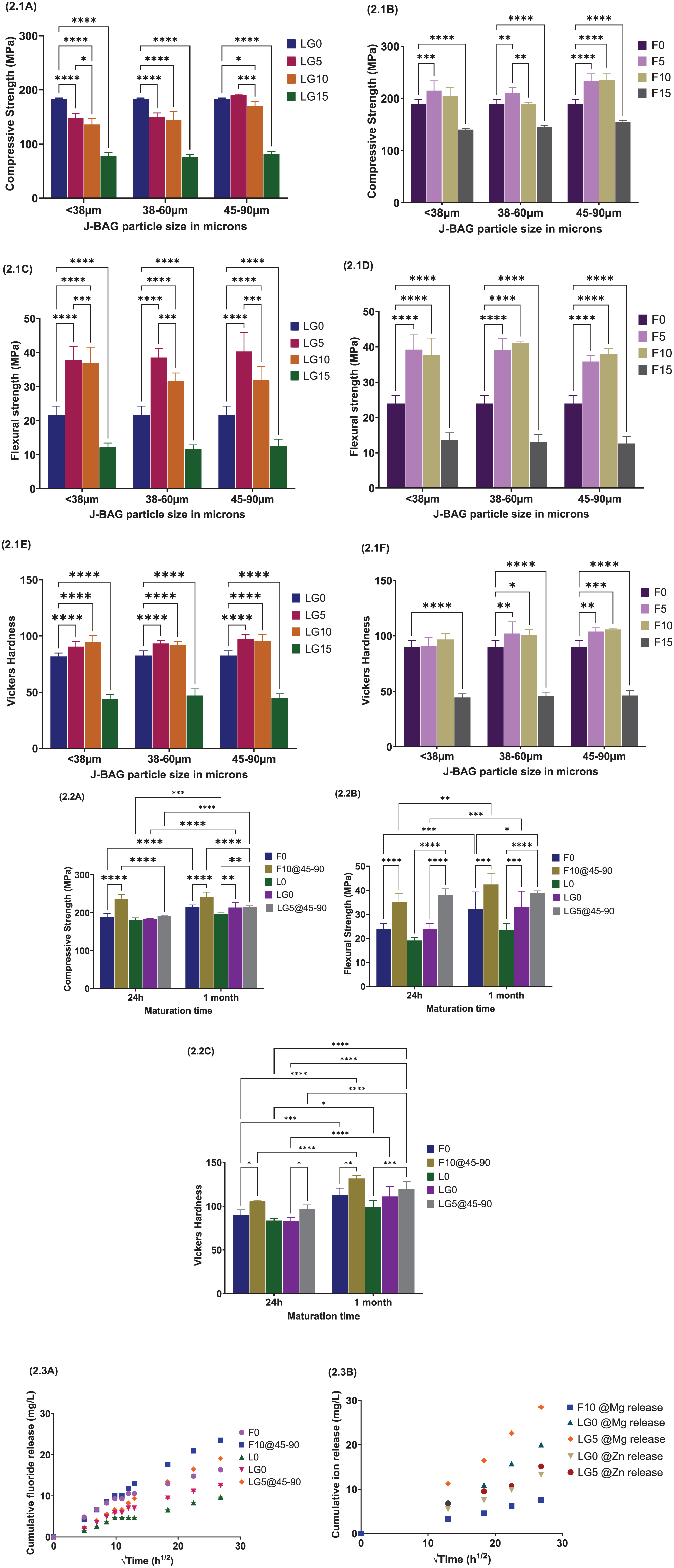
Mechanical properties and ion release of the bioactive glass (BAG)–glass-ionomer cement (GIC) groups.
The micrographs (Fig 3.1 to 3.2) display a significant increase in mineralized deposits as J-BAG concentrations rise from 5% to 15% after 28 d of immersion in different media (Fig 3.3). SEM revealed that F10 and LG5 facilitated mineral deposition on demineralized dentine surfaces and partially occluded tubules, whereas F0 and LG0 showed no evident remineralization and retained open tubules after 14 d of SBF immersion (Fig 3.3). Quantitative analysis (Fig 3.4 to 3.5) reveals a significant increase (P < 0.05) in Ca/P weight ratio from 0.17 (LG5) and 0.28 (F10) preimmersion to 1.73 and 1.94, respectively, postimmersion. Quantitative viable count demonstrated a significant reduction (P < 0.05) in colony-forming units (CFUs) for S. mitis/oralis, L. paracasei, and C. acnes in F10 compared with F0. LG5 exhibited significantly lower CFUs than L0 for L. paracasei and C. acnes, with counts also lower than LG0 in the C. acnes group (Fig 4.1A). F10 exhibited significantly reduced (P < 0.05) multispecies CFUs compared with F0; LG0 and LG5 showed lower counts than L0 (Fig 4.1B). The confocal images revealed reduced biofilm growth on F10 and LG5 compared with their respective controls (Fig 4.2 and 4.3). A marked reduction in live biomass (P < 0.05) was observed on F10 and LG0 relative to their respective internal controls, with further reduction in the magnesium–zinc variant GIC when 5% J-BAG was added (LG5) (Fig 4.4).
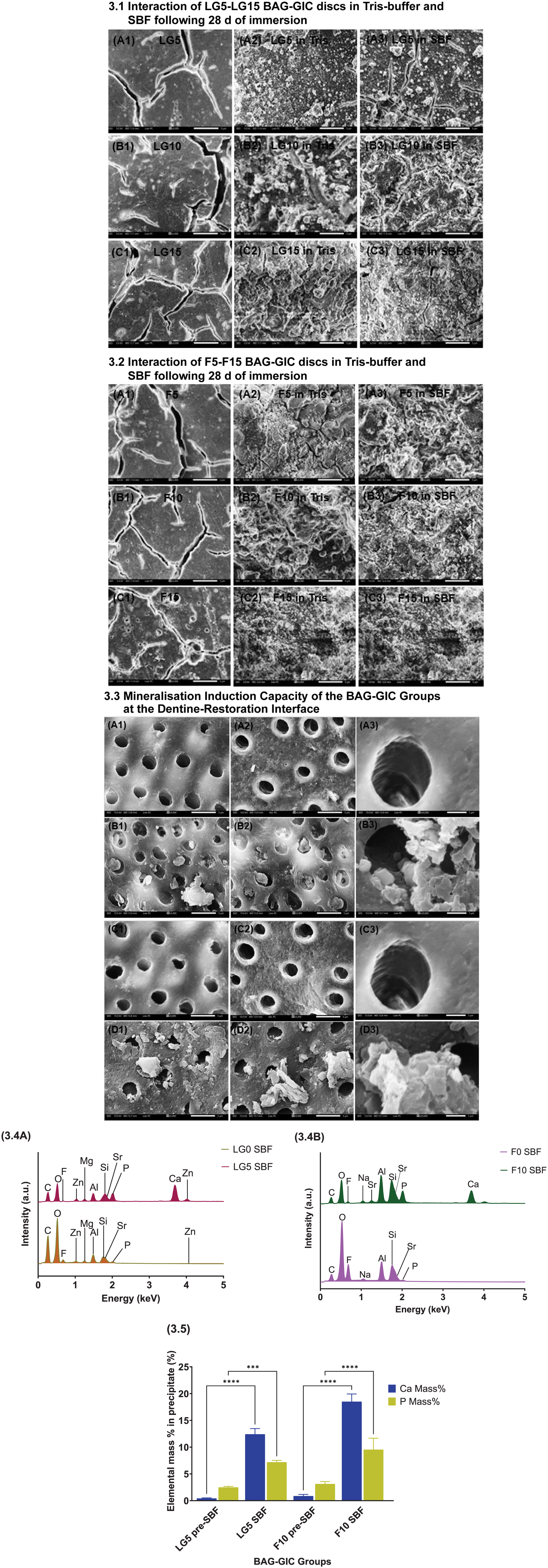
Results of mineralization studies.
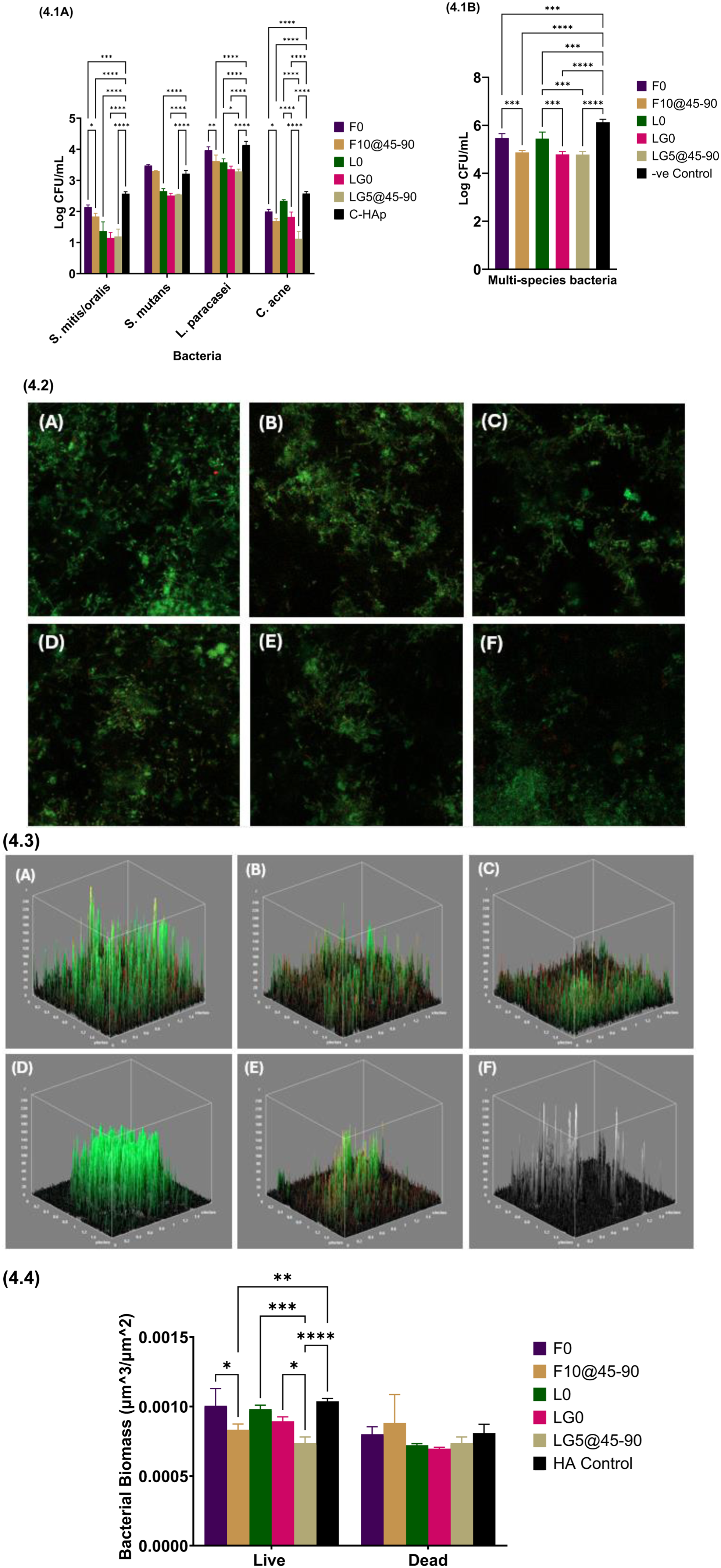
Results of antibacterial studies.
Discussion
BAG, originally designed for orthopedic applications, forms a bonelike apatite layer when in contact with bone. Its uses have expanded in dental materials as fillers, particularly for GICs and composites. While BAG promotes apatite nucleation and remineralization (Ana and Anggraeni 2021; Kim et al 2021; Tiskaya, Gillam, et al 2021; Tiskaya, Shahid, et al 2021), its incorporation into GICs poses challenges related to the detriment of mechanical properties (Yli-Urpo et al 2005; Valanezhad et al 2016; De Caluwé et al 2017). The effective mineralization of GICs requires the conversion of BAG to apatite-like phases while maintaining mechanical integrity. Contrary to previous misconceptions, past studies have demonstrated that sodium in the BAG is not a prerequisite for mineralization (Mitchell et al 2011; Davis et al 2014; Chen et al 2014, 2017), and degradation rates are more closely tied to the silicate network’s low polymerization (Chen et al 2017).
Effective BAG design requires a network connectivity of approximately 2.10 to optimize material properties. Replacing Na2O with CaF2, coupled with an increased phosphate content, can regulate degradation and enhance mineralization (Brauer et al 2010). The sodium-free BAG developed in this study, enriched with phosphate and fluoride, markedly improved mineralization, supporting findings by Mneimne et al (2011). FTIR and XRD analyses confirmed apatite formation within 3 h in Tris buffer solution, a solution devoid of apatite-forming ions. The reduction in nonbridging oxygens and the characteristic FTIR band splitting indicate Si–OH condensation and silica gel formation, critical for apatite nucleation. The elevated phosphate, with CaO and MgO as network modifiers, facilitates orthophosphate charge balance, further supporting rapid mineral deposition (Al-Khafaji et al 2019).
Incorporating J-BAG above 10 wt% altered powder consistency and increased viscosity, hindering homogeneous mixing and necessitating a reduced powder-to-liquid ratio at 15 wt%. This negatively affected mechanical properties. The elevated viscosity stems from BAG’s high silica content, promoting self-agglomeration. In addition, its lower cationic release compared with fluoro-alumino-silicate glass disrupts the acid–base reaction essential for GIC setting (Kim et al 2017). Sodium in conventional BAG increases BAG solubility and disrupts the essential 3-dimensional cross-linking of polyacrylic acid with Al³+ ions. Na+ cannot cross-link with COO–, thereby weakening the ionic interactions with COO– groups (De Caluwé et al 2017). High concentrations of BAG also contribute to poor particle dispersion, increased porosity, surface crack tensions, stress concentrations, and moisture-induced bond deterioration (Valanezhad et al 2016; Martins et al 2022). In contrast, the innovative sodium-free J-BAG effectively addressed these issues. It enhanced physical and mineralizing properties at optimal loadings, 5% in LG99Sr-Mg-Zn and 10% in Fuji IX. Its macro-granular size (45 to 90 µm) minimized premature bioglass–polyacid interaction, reducing excessive Ca²+ release that competes with Al³+ for cross-linking with polycarboxylic acid while preserving phosphate availability essential for sustained mineralization and long-term cement durability (Mannaa 2021). SEM/EDS analyses confirmed progressive surface mineralization postimmersion. Notably, J-BAG promoted nucleation and mineral deposition on demineralized dentine, partially occluding tubules within 14 d. The resulting stoichiometric Ca/P ratios indicate apatite formation, demonstrating the potential of BAG-modified GICs to support interfacial dentine repair under conditions of severe mineral loss.
Targeting cariogenic bacteria is essential in developing bioactive restorative materials that prevent biofilm-associated oral diseases, particularly secondary caries adjacent to restorations. This study focused on four endodontic pathogens linked to restoration failure. Oral biofilms compromise restoration longevity through dysbiosis-driven lactic acid production and extracellular polymeric substance secretion. This modulates substance diffusion and creates diverse microenvironments (Wang et al 2016), underscoring the importance of monitoring bacterial colonization and metabolism for effective caries-control strategies. Magnesium’s antibacterial effect is attributed to its ability to elevate local alkalinity, although the precise mechanisms of action remain less defined. Zinc disrupts bacterial metabolism by binding proteins, interfering with DNA, and inhibiting Streptococcus mutans activity, thereby reducing acidogenicity and plaque mineralization (Nizami et al 2021). SEM/EDS identified mineral deposits on BAG-GIC surfaces. This was aided by phosphate, enhancing P-O-P and Al-O-P linkages, potentially promoting fluorapatite formation and reducing bacterial activity (Mannaa 2021). In this study, the enhanced ion release, alongside improved mineralization, was associated with the observed antibacterial efficacy, suggesting a synergistic effect at the tooth-restoration interface (AlSahafi et al 2022). Although statistically significant differences in biofilm inhibition were observed between the control and experimental groups, log CFU reductions were limited to 1 to 2 orders of magnitude. Further investigations using confocal imaging demonstrated a marked reduction in biofilm on LG5 and F10 compared with internal controls. While these findings underscore the potential clinical relevance of the new material, further investigations using additional clinically representative models are needed to establish its efficacy.
Beyond GICs, this innovative J-BAG exhibits potential as a versatile filler in composites, fissure sealants, root-end materials, toothpastes, and for bone regeneration. This multifaceted applicability warrants further research on its effectiveness for clinical applications. Overall, the sodium-free, phosphate-rich J-BAG, fortified with fluoride and magnesium, significantly enhanced mineralization, mechanical, ion-releasing, and antibacterial properties of GICs without compromising setting kinetics if added in controlled concentrations. Ongoing studies are evaluating the impact of J-BAG on shear bond strength, positioning it as a durable posterior restorative for in vivo use.
Conclusions
The particle size of BAG influences the mechanical properties of GICs. Incorporation of the newly designed, sodium-free J-BAG (45 to 90-µm) significantly enhanced mechanical properties, ion release, and mineralization, while achieving a statistically significant antibacterial effect, albeit moderate. While the J-BAG–modified GICs demonstrated significant reductions in live biomass, the modest 1- to 2-log CFU reductions observed during colony evaluation indicate that clear evidence of clinical efficacy remains elusive and warrants further investigation. Optimal properties were achieved with 5% J-BAG in the Mg-Zn GIC and 10% in Fuji IX. Although loading J-BAG at 15% improved mineralization, it exceeded ISO setting time limits and compromised strength. Overall, these BAG-GIC composites demonstrate clinical potential, offering multifunctional benefits that could expand their application in restorative dentistry and improve treatment outcomes.
Author Contributions
J.O. Makanjuola, contributed to conception and design, data acquisition, analysis, and interpretation, drafted and critically revised the manuscript; R.G. Hill, S.A. Niazi, contributed to design, data analysis and interpretation, critically revised the manuscript; J. Aduse-Opoku, contributed to design, data acquisition, analysis, and interpretation, critically revised the manuscript; S. Banerji, S. Deb, contributed to conception and design, data analysis and interpretation, critically revised the manuscript. All authors gave final approval and agreed to be accountable for all aspects of the work, ensuring integrity and accuracy.
Supplemental Material
sj-docx-1-jdr-10.1177_00220345251392515 – Supplemental material for Exploring the Multifunctional Potential of Bioactive Glass-Ionomer Cements
Supplemental material, sj-docx-1-jdr-10.1177_00220345251392515 for Exploring the Multifunctional Potential of Bioactive Glass-Ionomer Cements by J.O. Makanjuola, R.G. Hill, S.A. Niazi, J. Aduse-Opoku, S. Banerji and S. Deb in Journal of Dental Research
Footnotes
Acknowledgements
The authors would like to thank Professor John Nicholson and Professor Owen Addison for their invaluable contributions to the study design. We also extend our gratitude to Dr Leanne Cleaver for her expertise and technical support during the confocal laser scanning microscopy imaging. In addition, we appreciate the assistance of Dr Magdalena Flak and Dr Ahmed Almaroof with the microbiome studies. Special appreciation to Dr Natalia Karpukhina for her significant contributions during the data analysis and interpretation. We also acknowledge Drs Aaron Le Blanc and Shannon Stockdale, along with the staff of the Centre for Ultrastructural Imaging, for their support during imaging procedures. Our thanks also extend to Dr Daniel White, Peter Pilecki, and Steven Gilbert for their technical support during the research work.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Tertiary Education Trust Fund, Nigeria, under the award number TETF/ES/UNIV/LAGOS/TSAS/2020; King’s Careers and Employability; King’s Doctoral College and King’s College London. The funders had no role in the study design, data collection and analysis, decision to publish, or manuscript preparation.
A supplemental appendix to this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
