Abstract
The development of caries management materials has concentrated on the dual objectives of inhibiting cariogenic microorganisms and promoting remineralization. In this study, 2 dual-functional ionic liquid (IL) varnishes, exhibiting both antibacterial and remineralization capabilities, were synthesized as novel anticaries agents. The ionic liquids (ILs) were prepared by modifying 1-hexylimidazolium IL with 3-chloropropyltriethoxysilane, followed by anion exchange with F− and a coordination reaction with Sr2+, respectively. The ILs were characterized using energy-dispersive spectroscopy (EDS), ion chromatography (IC), inductively coupled plasma optical emission spectrometry (ICP-OES), and Fourier transform infrared spectroscopy (FTIR). The antibacterial efficacy of the ILs was evaluated through colony counting, live/dead staining, and scanning electron microscopy (SEM). Subsequently, the ILs were blended with rosin to form IL varnishes. The remineralization potential of the IL varnishes was assessed through microhardness test, acid resistance test, SEM, EDS, and X-ray diffraction. In addition, in vivo anticaries treatment with the IL varnishes was conducted using a dental caries animal model. Histopathological and oral microbiome analyses were performed to evaluate the in vivo biocompatibility of the materials. The comprehensive analysis by EDS, IC, ICP-OES, and FTIR collectively confirmed the successful synthesis of the ILs. Antibacterial assays revealed that ILs at concentrations as low as 25 µM eliminated more than 80% of cariogenic bacteria within 60 min and significantly decreased viable bacteria in biofilms within 24 h. Following a 7-d treatment with the IL varnishes, SEM analysis of acid-etched enamel demonstrated reduced interspace depth, along with substantially increased microhardness and significantly improved acid resistance versus the negative control group. As compared with the fluoride varnish, the IL varnishes are more effective in preventing dental caries in rats, without harming oral buccal mucosa, major organs, or microbiota diversity. In conclusion, the IL varnishes developed in this study are not only straightforward to synthesize but also exhibit significant potential against dental caries.
Introduction
Dental caries is a multifactorial, biofilm-mediated chronic disease characterized by progressive destruction of dental hard tissues (Machiulskiene et al 2020). Classified by the World Health Organization as 1 of 3 major global noncommunicable diseases (Ballantine et al 2018; Van Chuyen et al 2021), it affects approximately 2.4 billion adults with untreated permanent caries and 532 million children with deciduous caries, imposing substantial health care burdens (Chan et al 2023; Spatafora et al 2024). Without intervention, it may progress to pulp–periapical lesions or maxillofacial infections, severely compromising life quality (Spatafora et al 2024).
Fluorides remain the primary anticaries agents by forming acid-resistant fluoroapatite through hydroxyl substitution in hydroxyapatite (HA), inhibiting demineralization while promoting remineralization (Zeng et al 2023; Veneri et al 2024). However, their dose-dependent antibacterial effects and poor oral retention due to salivary clearance limit sustained efficacy. Likewise, chlorhexidine mouthwashes disrupt oral microbiota and induce bacterial resistance with prolonged use (Niu et al 2021; Brookes, McCullough, et al 2023; Brookes, Teoh, et al 2023). Therefore, it is essential to develop materials that integrate antibacterial properties with remineralization capabilities (Zhou et al 2021).
In the pursuit of innovative anticaries materials, ionic liquids (ILs), particularly alkylimidazolium ILs, have attracted considerable attention due to their potent antibacterial activity, low propensity to induce drug resistance, excellent structural tunability, and favorable biocompatibility (Curreri et al 2021; Hassan et al 2022). These characteristics render them promising candidates for addressing the limitations inherent in conventional anticaries materials. The antibacterial mechanism of alkylimidazolium ILs involves an initial electrostatic attraction between positively charged ILs and negatively charged bacterial surfaces, followed by the insertion of hydrophobic alkyl chains into the phospholipid bilayer. This process results in membrane disruption, cytoplasmic leakage, and ultimately bacterial cell death (Fang et al 2022; Avirdi et al 2023). This nonspecific physical disruption of the membrane effectively mitigates the issue of bacterial resistance commonly associated with traditional antibiotics, thereby providing a robust foundation for the application of alkylimidazolium ILs in caries prevention. However, translating the advantages of ILs into clinically viable anticaries materials necessitates the resolution of 2 critical challenges: (1) ensuring prolonged retention of ILs on the enamel surface within the complex oral microenvironment to mitigate rapid loss and (2) imparting ILs with the capability to promote enamel remineralization. The inherent structural tunability of alkylimidazolium ILs presents a promising approach for the development of a dual-functional system that combines sustained antibacterial activity with active remineralization.
To address these challenges, we engineered a composite system using natural rosin as a carrier, leveraging its exceptional film-forming and sustained-release properties (Danışman-Kalındemirtaş et al 2022). Alkylimidazolium ILs incorporated within this matrix underwent targeted silanization to enhance their retention on the enamel surface (Khongwong et al 2016; Nihei 2016; Matinlinna et al 2018). Simultaneously, Sr2+ and F− were incorporated into the system respectively to use their properties in promoting mineral deposition and enhancing the acid resistance of mineralized crystals (Dai et al 2019; Wang et al 2019; Dai et al 2021). Through the synergistic integration of the ILs’ potent antibacterial efficacy, enamel retention enhanced by silanization, Sr2+/F− remineralization potential, and the rosin matrix’s sustained-release characteristics, we successfully synthesized 2 dual-functional IL anticaries varnishes. The antibacterial efficacy of the ILs and the enamel remineralization potential of the varnishes were systematically evaluated through in vitro studies. In addition, in vivo studies were conducted to thoroughly assess the overall anticaries efficacy and biocompatibility of the varnishes. The central hypothesis posits that the synthesized dual-functional IL varnishes deliver potent antibacterial activity through alkylimidazolium ILs and promote enamel remineralization via the respective release of F− or Sr2+.
Materials and Methods
All experimental procedures, including the preparation of enamel blocks, synthesis and characterization of ILs, evaluation of the ILs’ antibacterial activity, preparation and characterization of IL varnishes, assessment of the IL varnishes’ remineralization efficacy, cytocompatibility and hemocompatibility testing of IL varnishes, and the in vivo evaluation of the IL varnishes’ anticaries efficacy, are detailed in the Appendix Materials and Methods. All animal experiments were approved by the Animal Ethics Committee of the School of Stomatology, the Fourth Military Medical University, and conformed to the ARRIVE (Animal Research: Reporting of In Vivo Experiments) guidelines.
Statistical Analysis
SPSS software (version 26.0) was used for statistical analysis (see Supplemental Appendix for details).
Results
Characterization of ILs
Energy-dispersive spectroscopy (EDS; Hitachi S-4800) analysis (Fig 1A) confirmed the elemental profiles of the compounds: 1-hexyl-3-propyltriethoxysilane imidazolium chloride (IL-Cl) (C, N, O, Si, Cl), 1-hexyl-3-propyltriethoxysilane imidazolium fluoride (IL-F) (C, N, O, Si, F), and 1-hexyl-3-propyltriethoxysilane imidazolium strontium chloride complex (IL-Sr) (C, N, O, Si, Sr, Cl), aligning with their intended compositions. Ion chromatography (IC; Thermo Fisher ICS-1100) and inductively coupled plasma optical emission spectrometry (ICP-OES; Agilent Technologies Agilent 5800) revealed F and Sr mass fractions of 3.88% ± 0.38% and 13.19% ± 0.97%, respectively (Fig 1B), closely matching the theoretical values of 5.05% for F and 15.96% for Sr. Fourier transform infrared spectroscopy (FTIR; Shimadzu Corporation IRTracer-100) spectra (Fig 1C) showed peaks at 3,200 to 3,000 cm−1 (=C-H stretch in imidazole), 3,000 to 2,800 cm−1 (alkane C-H stretch), 1,628 cm−1 (imidazole C=N stretch), 1,563 cm−1 (aromatic C-C stretch), and 1,041 to 984 cm−1 (Si-O stretch). EDS, IC, ICP-OES, and FTIR collectively verified successful IL synthesis.
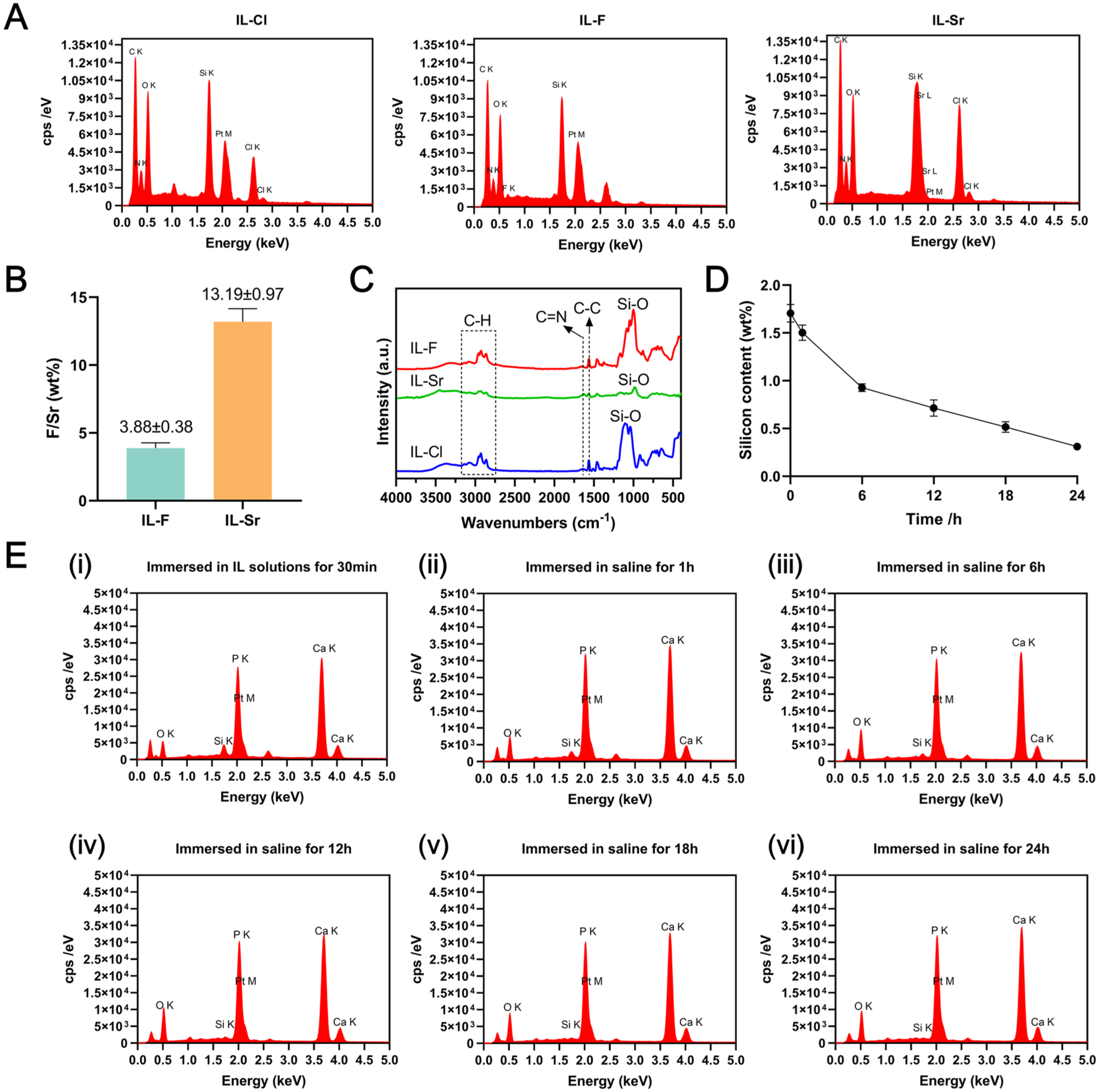
Material characterization results of ionic liquids (ILs). (
Results from the retention experiment of ILs on the enamel surface demonstrated that the content of Si (a characteristic element of ILs) on the tooth surface exhibited a time-dependent decrease (Fig 1D, E). However, detectable Si signals persisted after 24 h of hourly saline replacement, confirming the sustained retention of ILs on the enamel surface.
Antibacterial Properties of ILs
The results of the antibacterial assays indicated that the growth of Streptococcus mutans, Streptococcus sanguinis, and Actinomyces viscosus was unaffected in the negative control (NC), NaF, and SrCl2 groups, whereas it was significantly inhibited after treatment with 25 µM ILs (Supplemental Appendix Fig 1). After a 60-min exposure to IL treatments, the survival rates of S. mutans, S. sanguinis, and A. viscosus were significantly decreased to 1.34%, 0.20%, and 16.31%, respectively (Fig 2A). To further investigate the antibacterial mechanism of the ILs, we observed morphological changes in S. mutans using scanning electron microscopy (SEM; Hitachi S-4800). No significant structural damage was observed in S. mutans within the NC, NaF, and SrCl2 groups. In contrast, intervention with ILs led to a disruption of normal cellular morphology, compromised the integrity of the cytoplasmic membrane, and resulted in the leakage of cytoplasmic contents (Fig 2B). In addition to inhibiting planktonic growth, ILs exhibited significant efficacy against biofilms. Live/dead staining assays verified that ILs, at a concentration of 25 µM, effectively killed live bacteria within mature biofilms after just 24 h of treatment (Fig 2C). SEM analysis of treated S. mutans biofilms revealed a melting-like breakdown of the extracellular matrix and morphological changes in bacterial cells, including shrinkage and cytoplasmic leakage (Fig 2D).
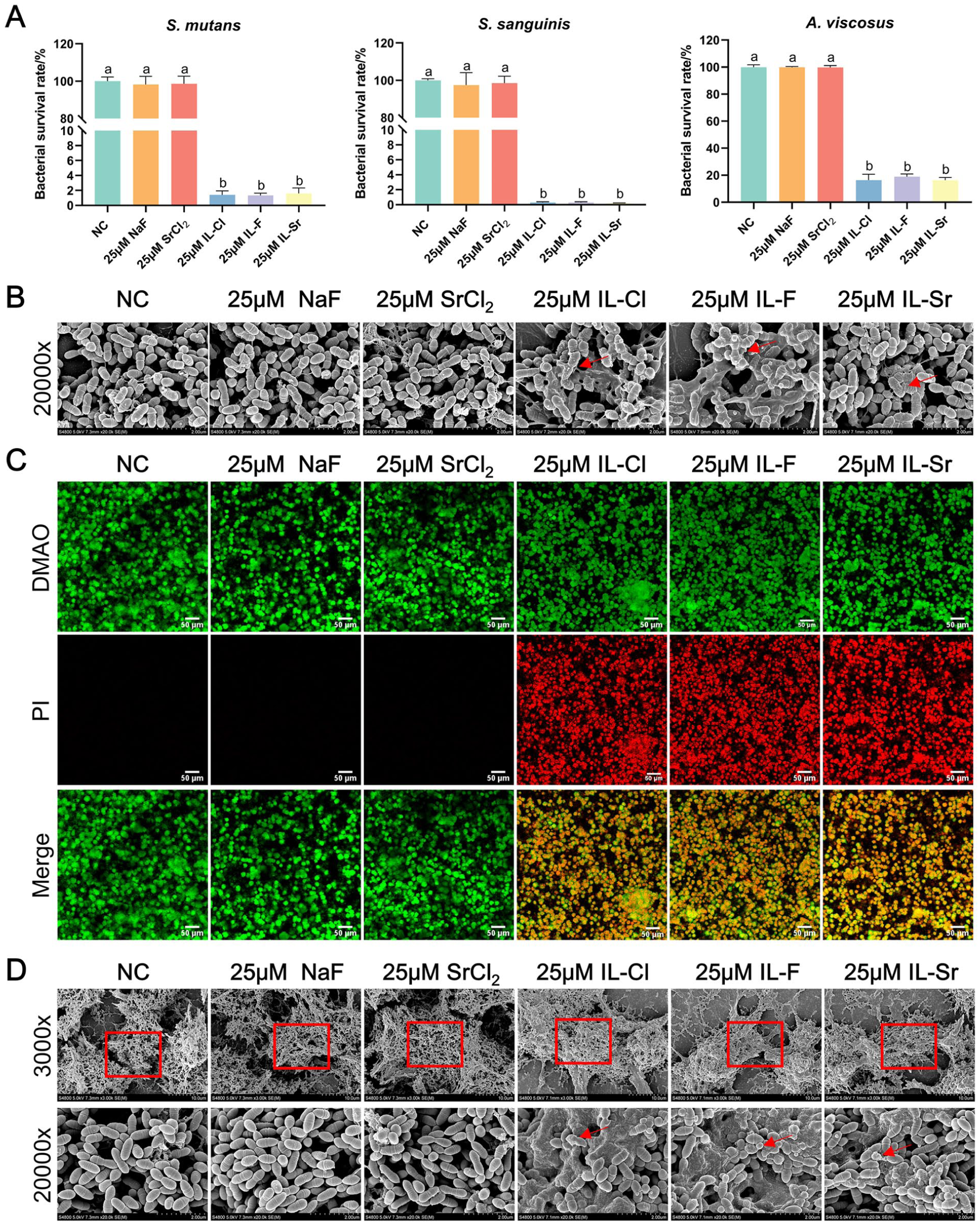
Antibacterial activity of different materials. (
Characterization and Remineralization Efficacy of IL Varnishes
Stereomicroscopy (Leica M205FA) analysis of varnishes immersed in double-distilled water (DDW) for 6 h revealed that both IL varnishes developed a uniform multiscale porous structure, contrasting with fluoride varnish’s fewer, sparser pores (Fig 3A). Subsequent ion release profiling demonstrated that the release of F− from the IL-F varnish and that of Sr2+ from the IL-Sr varnish demonstrated a gradual increase over a 12-h period, ultimately reaching a plateau. Throughout the experimental duration, the release of Sr2+ from the IL-Sr varnish was comparable to that of F− from the fluoride varnish; however, the release of F− from the IL-F varnish was significantly greater than both that of Sr2+ from the IL-Sr varnish and that of F− from the fluoride varnish (Fig 3B). SEM (Fig 3C) revealed that after acid etching (to expose the prismatic “fish scale” structure of enamel) followed by a 7-d remineralization period, minimal mineral deposition was observed in the DDW–NC group. In contrast, significant mineral deposition, which notably reduced the depth of the interprismatic spaces, was evident in both the fluoride varnish group and IL varnish groups. Among these, the fluoride varnish and IL-F varnish demonstrated superior efficacy compared with the IL-Sr varnish. The surface microhardness recovery rate results (Fig 3D) demonstrated that all varnish groups demonstrated a statistically significant increase in surface microhardness compared with the NC group (P < 0.05). The IL-F varnish group exhibited the greatest enhancement (52.14% ± 5.90%), followed by the fluoride varnish group (41.23% ± 4.49%) and IL-Sr varnish group (30.17% ± 5.97%). Acid resistance testing (Fig 3E) revealed significantly lower mineral dissolution in all varnish-treated groups compared with the NC group (P < 0.05). Among the varnish treatments, the IL-F varnish group demonstrated superior acid resistance (Ca: 0.32 ± 0.05 mg/L; P: 0.16 ± 0.04 mg/L), surpassing the performance of both the fluoride varnish (Ca: 0.47 ± 0.04 mg/L; P: 0.27 ± 0.04 mg/L) and the IL-Sr varnish (Ca: 0.62 ± 0.04 mg/L; P: 0.38 ± 0.05 mg/L), the latter of which exhibited the highest ion loss. EDS detected Ca, P, O, and F in remineralized deposits in the fluoride varnish group and IL-F varnish group (Fig 3F). X-ray diffraction (XRD; Bruker D8 Advance) (Fig 3G) showed peaks at 2θ = 25.8° (002), 28.1° (102), 31.8° (211), 32.2° (112), 34.0° (202), and 49.5° (213), matching HA (JCPDS 09-0432) and fluorhydroxyapatite (FHA; JCPDS 15-0876), suggesting an HA/FHA mixture. For the IL-Sr varnish group, EDS identified Ca, P, O, and Sr (Fig 3F). XRD (Fig. 3G) revealed peaks at 2θ = 25.8° (002), 28.1° (102), 31.7° (300), 32.2° (112), 34.0° (202), and 49.5° (213), corresponding to HA and Sr-HA (JCPDS 33-1348), indicating an HA/Sr-HA mixture.
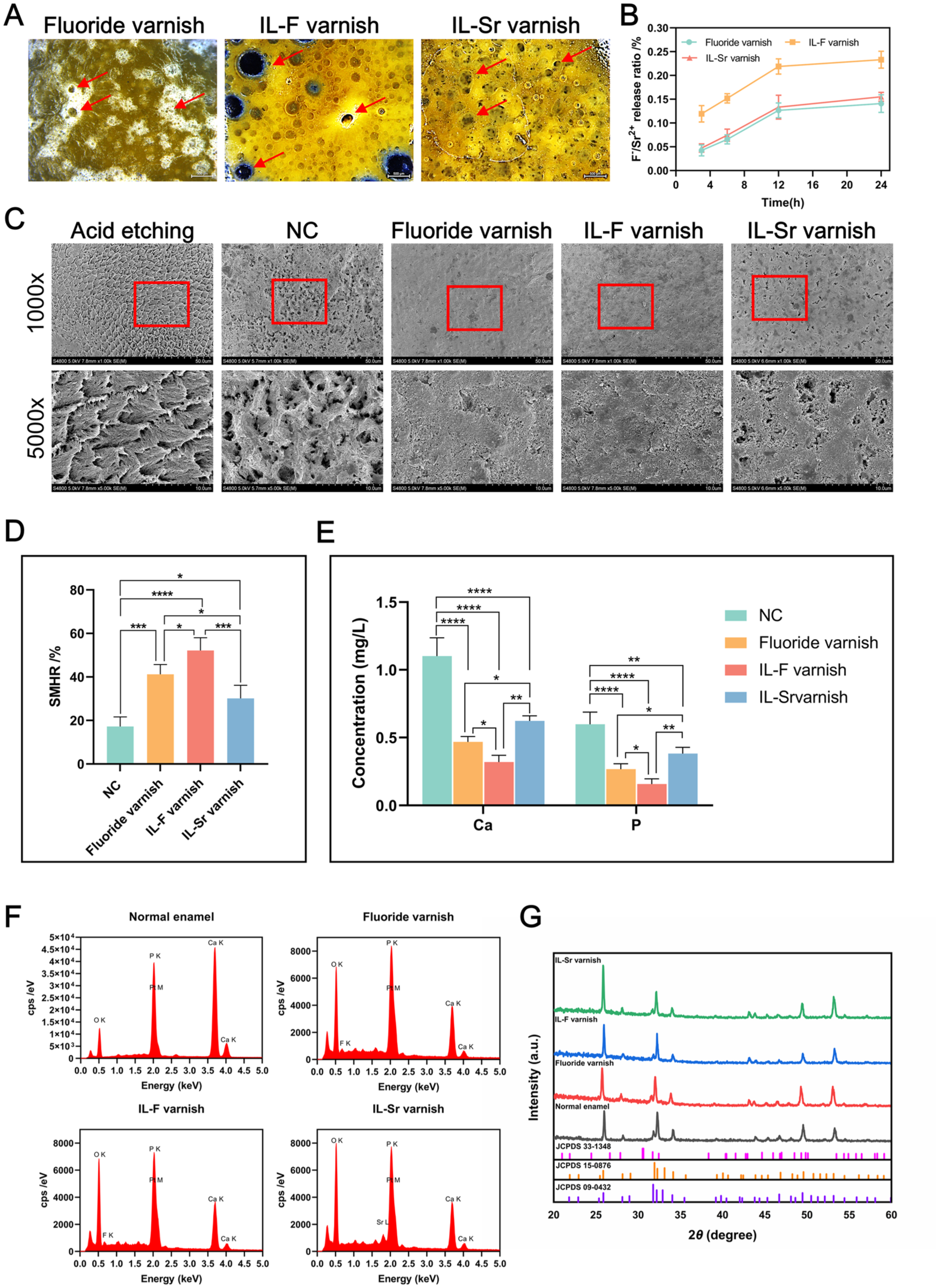
Mineralization capacity of different materials. (
In Vivo Efficacy of IL Varnishes
Statistical analysis revealed that all 3 varnish treatments significantly reduced the E scores on smooth surfaces compared with the NC group (caries model group), with the IL-F varnish group exhibiting the lowest E scores. Furthermore, since early caries predominantly occur in pits and fissures, we quantified the severity of pit-and-fissure caries lesions by establishing 4 categories: total lesions (E + Ds + Dm + Dx), enamel caries (E), mild dentinal lesions (Ds), and moderate dentinal lesions (Dm). Total lesions represent caries incidence, while the other categories reflect lesion severity. The study showed that all varnish groups had a significantly lower caries incidence than the NC group did (P < 0.05), with the IL varnish groups being more effective against caries than the fluoride varnish group (Fig 4A, B). Micro–computed tomography (Siemens Inveon MM) revealed discontinuous enamel and low-density radiolucent zones in molars from the NC and fluoride varnish groups, whereas these carious features were absent or minimal in the IL varnish groups (Fig 4C). Hematologic parameters, including red blood cell, white blood cell, aspartate transaminase, and creatinine levels, were observed to remain within normal physiological ranges across all groups (Fig 4D). Histopathological examination with hematoxylin and eosin staining revealed no significant abnormalities in the buccal mucosa or major organs (Fig 4E).
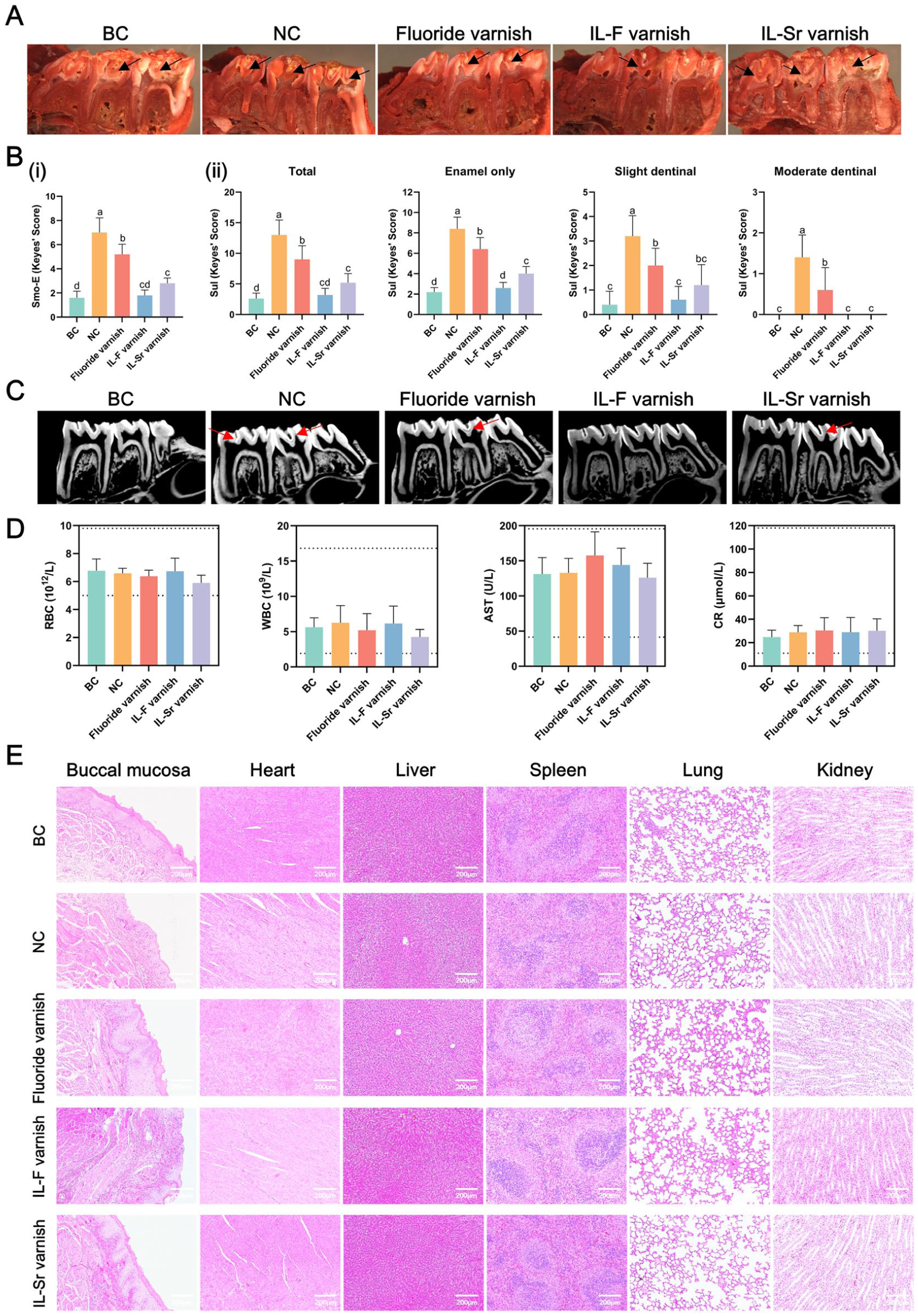
Caries-preventive effects and biocompatibility of different materials in vivo. (
Effects of IL Varnishes on Oral Microflora
Comprehensive analysis of oral microbiota was conducted using high-throughput 16S rRNA sequencing. The Venn diagram revealed that the oral microbiota of rats in the healthy blank control (BC), NC, fluoride varnish, IL-F varnish, and IL-Sr varnish groups shared a total of 592 identical operational taxonomic units (OTUs), along with unique OTUs of 1,006, 698, 1,000, 1,376, and 1,310, respectively. These results demonstrate that the IL varnish intervention significantly increased the OTU number in the oral microbiota (Fig 5A). The ACE, Chao1, Shannon, and Simpson indices indicated that treatments with IL varnishes, particularly the IL-F varnish, significantly enhanced the α-diversity of the oral microbiota (Fig 5B). Principal coordinate analysis (PCoA) indicated that the groups treated with IL varnishes, particularly the IL-Sr varnish, showed a higher degree of compositional similarity to the BC group (Fig 5C). The subsequent comparison focused on the relative abundance of predominant bacteria across groups at the phylum level (Fig 5D). The relative abundance of Firmicutes showed no statistically significant differences across all experimental and control groups. The NC group had a significantly lower abundance of Bacteroidota than the BC group did, while the IL varnish intervention increased Bacteroidota levels, aligning with the BC group (Fig 5F). At the genus level, the relative abundance of Akkermansia exhibited an increasing trend following the IL varnish intervention, particularly after IL-Sr varnish administration (Fig 5E, G).
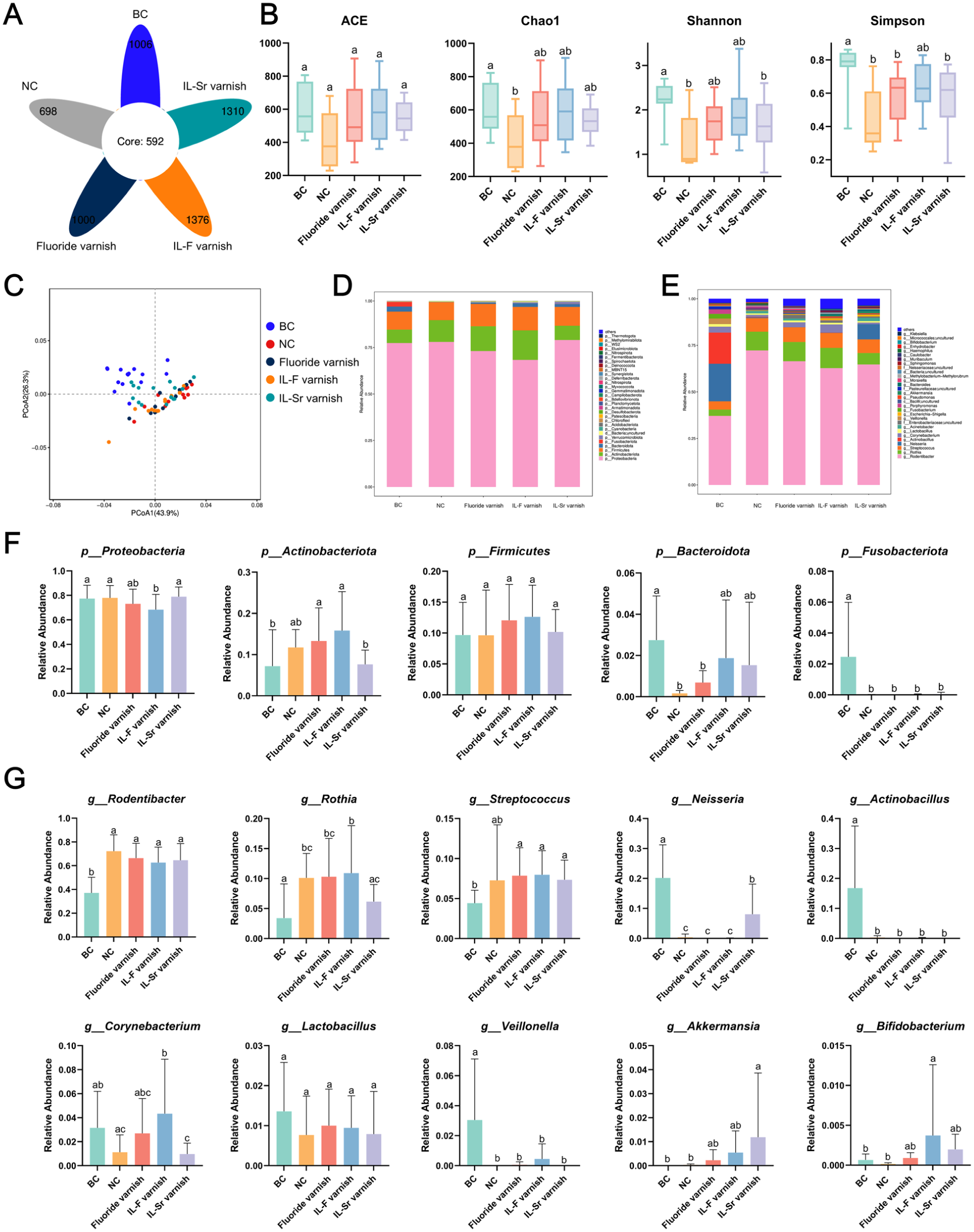
Effect of different treatments on oral microbiota in rats. (
Discussion
Ideal anticaries materials must integrate 2 essential functionalities: robust antibacterial activity and effective remineralization capacity (Zhi et al 2024). To achieve this, we used the broad-spectrum antimicrobial efficacy, minimal resistance induction risk, structural designability, and biocompatibility of ILs to develop 2 dual-functional silane-imidazolium ILs. Silane coupling agents act as “molecular bridges,” with their organic termini forming covalent bonds with IL molecules, while their silane termini establish strong covalent linkages with the hydroxyl groups prevalent on enamel surfaces. This mechanism enables the ILs to bind to the enamel surface. EDS analysis demonstrated that detectable IL retention persisted on enamel after 24 h of hourly saline replacement (simulating oral rinsing), confirming good retention capacity. This provides the foundation for sustained anticaries efficacy (Xu et al 2020).
Considering the fundamental role of cariogenic plaque biofilms in the pathogenesis of dental caries, characterized by their 3-dimensional structure that protects bacteria and hinders antimicrobial penetration due to an extracellular polysaccharide barrier, it is essential to prioritize effective biofilm disruption (Yin et al 2023; Han et al 2025). ILs exhibited potent antimicrobial activity against S. mutans and S. sanguinis, achieving near-complete eradication (>98%) at a concentration of 25 µM. While their efficacy against A. viscosus was comparatively lower, they still reduced the viability of this bacterium to less than 20%. For S. mutans biofilms, treatment with 25 µM ILs effectively eradicated most viable bacteria within mature biofilms, indicating their capacity for deep penetration and structural disruption. This potent antibiofilm activity highlights their potential for caries prevention.
To prolong the retention time of ILs on the enamel surface, facilitate the sustained release of ions that promote mineralization, and ultimately improve anticaries efficacy, this study used rosin as the matrix due to its superior adhesiveness, film-forming capability, and drug sustained-release properties (Danışman-Kalındemirtaş et al 2022; Madhavi et al 2023). Consequently, 2 IL varnishes were successfully developed by incorporating ILs with rosin. Stereomicroscopy revealed that the IL varnishes formed a porous surface structure in DDW, facilitating F− and Sr2+ release, which was quantitatively confirmed by ion release experiments: the IL-F varnish exhibited significantly higher cumulative F− release (0.25% ± 0.02%) than fluoride varnish did (0.14% ± 0.02%) after 24 h, while the IL-Sr varnish showed less Sr2+ release (0.16% ± 0.01%), potentially due to diffusion constraints from Sr2+’s larger ionic radius. SEM and microhardness testing demonstrated that all varnish-treated groups significantly enhanced remineralization of demineralized enamel compared with the NC group, with the IL-F varnish group showing the greatest efficacy and the IL-Sr varnish group the least. Acid resistance testing further revealed that all varnish groups exhibited significantly superior performance compared with the NC group. The overall efficacy of the varnish groups followed the hierarchy: IL-F varnish group > fluoride varnish group > IL-Sr varnish group. Collectively, both IL varnishes effectively promoted enamel remineralization. The superior performance of IL-F varnish is attributable to the small ionic radius of F−, which enables efficient substitution for hydroxyl groups in HA to form stable, acid-resistant FHA (Peng et al 2012). Sr2+ can also improve acid resistance and increase enamel surface microhardness, which is consistent with previous reports (Dai et al 2019; Wang et al 2019; Dai et al 2021). However, our experimental results demonstrate that the effectiveness of Sr2+ in promoting enamel remineralization is significantly lower than that of F−. This is likely because the larger ionic radius of Sr2+ leads to the formation of Sr-HA with lower crystal structure stability compared with FHA.
In vivo experiments further validated the anticaries efficacy of IL varnishes. The IL varnish–treated groups exhibited significantly lower Keyes’ scores than the fluoride varnish group did, with the IL-F varnish group showing the lowest scores and optimal caries prevention effect. These findings not only confirm the crucial role of fluoride in caries management but also highlight the potential advantages of the dual synergistic “antibacterial-remineralization” strategy over conventional monofluoride approaches (Gupta et al 2020; Shi et al 2025). In addition, biocompatibility assessments revealed physiologic hematologic parameters and no significant pathological changes in buccal mucosa or major organs, indicating excellent biocompatibility of the IL varnishes and providing a solid foundation for further clinical translation. Given the potential risk of dysbiosis induction by potent antimicrobial agents, the impact of IL varnishes on the rat oral microbiome was assessed via 16S rRNA gene sequencing. The results indicate that the topical application of IL varnishes does not disrupt the ecological balance of the oral microbiota. Venn diagrams showed significantly increased OTUs; ACE, Chao1, Shannon, and Simpson indices indicated partial restoration of α-diversity. PCoA analysis revealed that the IL varnish intervention induced structural convergence of the oral microbiota toward the BC group configuration. At the phylum level, the IL varnish intervention resulted in an increased abundance of Bacteroidota and a reduced Firmicutes/Bacteroidota ratio, a shift that has been documented in the literature to elevate salivary pH and buffering capacity, potentially reducing the risk of caries (Brookes et al 2021; Zhang et al 2022). At the genus level, IL varnish treatment significantly increased the relative abundance of Akkermansia, a key commensal bacterium known to enhance mucosal barrier integrity, modulate immune responses, suppress inflammation, and correlate with improved oral health outcomes (Anderson et al 2024). Nevertheless, the long-term ecological impact and precise mechanisms warrant further investigation through extended studies and deeper multiomics analyses.
In summary, this study successfully synthesized 2 silane-imidazolium ILs containing F− and Sr2+, respectively. In vitro experimental results demonstrated that both ILs exhibited excellent antibacterial activity. These ILs were subsequently blended with rosin to develop IL varnishes. By using the carrier properties of rosin, a sustained release of F− and Sr2+ was achieved, thereby preserving the antibacterial efficacy of the ILs and promoting enamel remineralization. In vivo studies showed that applying IL varnishes topically was more effective than fluoride varnish in preventing caries in rats, without causing any negative effects on oral tissues, major organs, or the diversity of the oral microbiome. This novel strategy provides a promising approach for caries management, indicating considerable clinical translational potential.
Supplemental Material
sj-docx-1-jdr-10.1177_00220345251397916 – Supplemental material for Dual-Functional Ionic Liquid Varnishes for Dental Caries Management
Supplemental material, sj-docx-1-jdr-10.1177_00220345251397916 for Dual-Functional Ionic Liquid Varnishes for Dental Caries Management by H.-W. Chen, Y.-F. Yuan, C.-L. Wang, D.-Y. Wang, Z.-C. Zhou, L. Fan, Q.-X. Zhang, Y.-N. He, W.-K. Jiang and S.-C. Wang in Journal of Dental Research
Author Contributions
H.-W. Chen, Y.-F. Yuan, C.-L. Wang, contributed to data acquisition, analysis, and interpretation, drafted and critically revised the manuscript; D.-Y. Wang, Z.-C. Zhou, L. Fan, Q.-X. Zhang, Y.-N. He, contributed to design, data acquisition and interpretation, critically revised the manuscript; W.-K. Jiang, S.-C. Wang, contributed to conception and design, data interpretation, critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of the work.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (82270973), the China Postdoctoral Science Foundation (2024M764325), the general project from State Key Laboratory of Oral & Maxillofacial Reconstruction and Regeneration (2024MS03), the project from National Clinical Research Center for Oral Diseases (LCC202203), and the Shaanxi key research and development program (2025YF-07; 2025SYS-SYSZD-024).
Data Availability Statement
A supplemental appendix to this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
