Abstract
Bone fracture healing requires coordinated interactions between immune cells and skeletal tissues, with flat bones exhibiting unique biomechanical and physiologic characteristics as compared with long bones. The mandible, the lower jawbone that supports the oral cavity, has distinct features due to its proximity to the oral mucosa, which is enriched in immune cells and microbiota, and its exposure to masticatory forces, making it a clinically relevant model for studying immune–skeletal interactions during fracture repair. Moreover, mandibular fractures are common maxillofacial injuries, posing challenges owing to functional and aesthetic considerations, as well as the risk of infection and impaired healing. Recent studies have highlighted conflicting roles for interleukin 17 (IL-17) and γδT cells in bone fracture or defect healing. As the primary source of IL-17 in the oral mucosa, γδT cells play a critical role in alveolar bone remodeling and respond to environmental factors such as the microbiota and age. We thus sought to investigate their role in the repair process of a mandibular defect. Here, we developed a murine model where a 1.5-mm drill hole defect spontaneously healed within 3 wk. Analysis revealed rapid leukocyte infiltration at the defect site by day 3, predominantly neutrophils and inflammatory monocytes, with γδT cells and adaptive leukocytes accumulating later at days 7 and 14. Depletion of γδT cells via Tcrd-GDL mice or genetic ablation of IL-17 in Il17af-/- mice accelerated the kinetics of mandibular healing. Bulk RNA sequencing and immunologic analysis revealed that while early recruitment of neutrophils and monocytes was unaffected in Il17af-/- mice, later inflammatory responses were diminished, resulting in accelerated repair. These findings suggest that IL-17–producing γδT cells (γδ17T cells) delay mandibular fracture repair by inhibiting inflammation resolution, thus prolonging the reparative phase. Targeting γδ17T cells or IL-17 may thus represent therapeutic strategies to enhance bone regeneration, particularly in challenging clinical settings involving mandibular fractures.
Introduction
Bone fracture healing involves complex interactions between the immune system and skeletal tissues (Baht et al 2018). While extensive research has focused on immune mechanisms in long bone fractures, flat bone fractures require more focused investigation due to their unique biomechanical and physiologic characteristics. As a representative of flat bones, the calvaria has been instrumental in advancing our understanding of bone regeneration mechanisms, revealing challenges in bone repair, and serving as a platform for testing various therapeutic strategies (Torres-Guzman et al 2023). However, other flat bones, such as the mandible, are located near the oral cavity, a specialized tissue that presents a distinct and clinically relevant site for studying fracture healing. Indeed, mandibular fractures are among the most common facial fractures, significantly affecting vital physiologic functions (Afrooz et al 2015). The unique features of the mandible, including its proximity to the oral microbiome, the abundant presence of resident leukocytes, and exposure to masticatory forces, distinguish it from the calvaria (Kotagudda Ranganath et al 2022; Salunkhe et al 2022). Furthermore, its susceptibility to oral diseases and higher infection risk make it a clinically relevant model for studying immune–skeletal interactions during bone healing (Kim and Ku 2023).
Recent studies have underscored the importance of interleukin 17 (IL-17) and γδT cells in bone fracture healing. However, the results have been conflicting, with some studies suggesting beneficial effects and others revealing detrimental roles for IL-17 and/or γδT cells in the healing process (Colburn et al 2009; Kim et al 2014, 2020; Ono et al 2016; Dixit et al 2017; Dar et al 2023). The discrepancies are not attributed to the type of bone (ie, long vs flat bones), indicating the complexity of the role of these factors in bone repair. Notably, the unique location of the mandible near the oral mucosa exposes it to an environment abundant in γδT cells. Oral γδT cells, predominantly of the Vγ6+ subset, are a major source of IL-17, termed γδ17T cells (Wilharm et al 2019). γδ17T cells are also involved in bone remodeling in other settings, such as natural bone loss, periodontitis, dental implants, and orthodontic tooth movement (Krishnan et al 2018; Wilharm et al 2019; Wald et al 2021; Barel et al 2022). Furthermore, oral γδT cells respond to the microbiota and are influenced by age—both factors that can affect fracture healing (Wilharm et al 2019). Given these considerations, oral γδT cells will likely play a key role in mandibular fracture repair. By using a spontaneously healing mandibular drill hole model, this study shows that oral γδ17T cells impair bone repair by delaying the reparative phase.
Material and Methods
Mice
C57BL/6 (B6), Tcrd-GDL, and Il17a/f-/- mice were bred and maintained in the central animal facility at the Hebrew University Faculty of Medicine (Jerusalem, Israel). The mice were maintained under specific pathogen–free conditions. All animal protocols were approved by the Hebrew University Institutional Animal Care and Use Committee (permit MD-21-16468-5). The study conforms to the ARRIVE guidelines.
Results
Development of the Mandibular Drill Hole Model
To investigate the immunologic mechanisms involved in the healing of a mandibular defect, we developed a drill hole model in mice (Fig. 1A). Analysis of the mandible by micro–computed tomography at various time points postsurgery revealed spontaneous healing, with nearly complete recovery observed in the third week following the procedure (Fig. 1B, C). Circulating levels of the bone resorption marker C-terminal telopeptide of type I collagen (CTX-1) were elevated 7 d after the procedure and remained high throughout the healing period (Fig. 1D). The levels of the bone formation marker N-terminal propeptide of type I procollagen showed an increase on days 7 and 14 as compared with the third day after the procedure. To further characterize the healing process, hematoxylin and eosin histologic cross sections were prepared from the bone and soft tissue of the defect site. As depicted in Figure 1E, the uninjured bone (day 0) appears as normal organized bone tissue. By day 3, leukocyte infiltration indicates early inflammation, and granulation tissue begins forming. Leukocyte infiltration increases by day 7 as granulation tissue increases. On day 14, leukocytes decrease, showing reduced inflammation, while new woven bone starts to form. By day 21, inflammation subsides, and new bone formation is well established. This drill hole model was subsequently used to investigate the immune mechanisms involved in the spontaneous healing of the mandible.
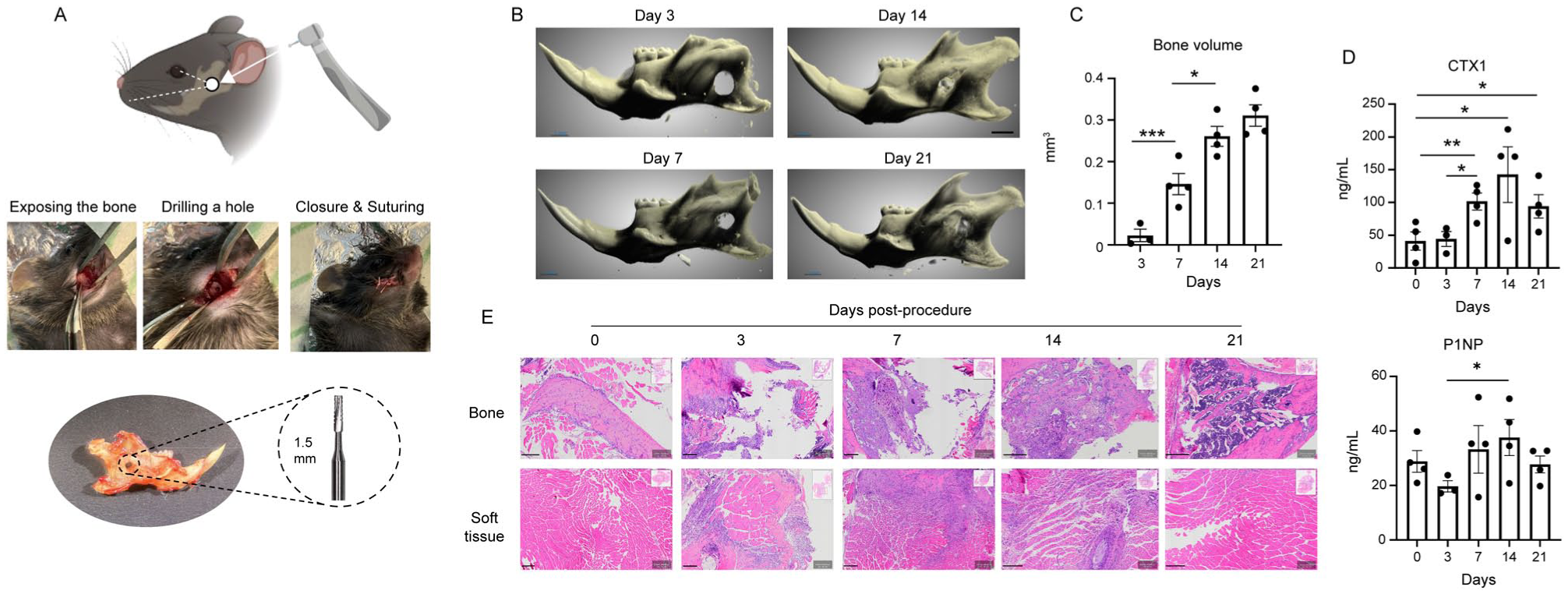
Kinetics of bone healing in a murine mandibular drill hole model. (
Characterization of Leukocyte Infiltration into the Bone Defect Site
To gain insights into the leukocytes infiltrating the mandibular bone defect site, the soft tissues surrounding the drilled hole were collected and analyzed by flow cytometry (Appendix Fig. 1A). As shown in t-SNE flow cytometry plots and graphs, a substantial population of leukocytes was present in the bone defect area from day 3 to day 14, with a significant reduction observed by day 21 postprocedure (Fig. 2A, B). The majority of infiltrating leukocytes were neutrophils and Ly6Chi monocytes, indicative of inflammatory monocytes (Fig. 2C, D). Circulating Ly6Clow monocytes were also detected, albeit in lower numbers. A population of antigen-presenting cells (APCs) was evident on days 3 and 7 postprocedure. Regarding lymphocytes, CD4+ and CD8+ T cells, as well as B cells, exhibited a gradual increase in numbers, peaking between days 7 and 14. γδT cells showed a significant elevation in frequency and absolute number on day 7, remaining elevated on day 14 but decreasing in number by day 21 (Fig. 2C, D). Next, the leukocytes in the bone marrow of the injured mandible were examined (Appendix Fig. 1B, C). No significant change was found in total leukocytes during the examined times; nevertheless, an increase in CD11b+ cells was found and attributed to changes in the neutrophil and monocyte subsets. The lymphocytes represented a small fraction of the leukocytes, with CD3 cells relatively reduced on day 3, whereas the γδT-cell subset increased moderately. By day 14, all the examined bone marrow leukocytes returned to their steady-state levels. To further characterize local immune responses, the expression of immunologically relevant genes was analyzed by quantitative polymerase chain reaction (qPCR). As shown in Figure 2E, the levels of mRNA coding for the cytokines Il17a, Tnfa, Ifna, and Ifng and for the transcription factor Foxp3 displayed a biphasic pattern, peaking on days 3 and 14. The level of Il1b, however, was constantly upregulated throughout this period. Regarding chemokine expression, Cxcl2 displayed a biphasic pattern, whereas Cxcl1 and Ccl2 levels were persistently upregulated until day 21. These findings suggest that mandibular bone defects trigger the rapid infiltration of neutrophils and inflammatory monocytes, which persist in the tissue until healing progresses. In contrast, adaptive leukocytes infiltrate later in smaller numbers, potentially explaining the biphasic expression pattern of cytokines and chemokines produced by these cells.
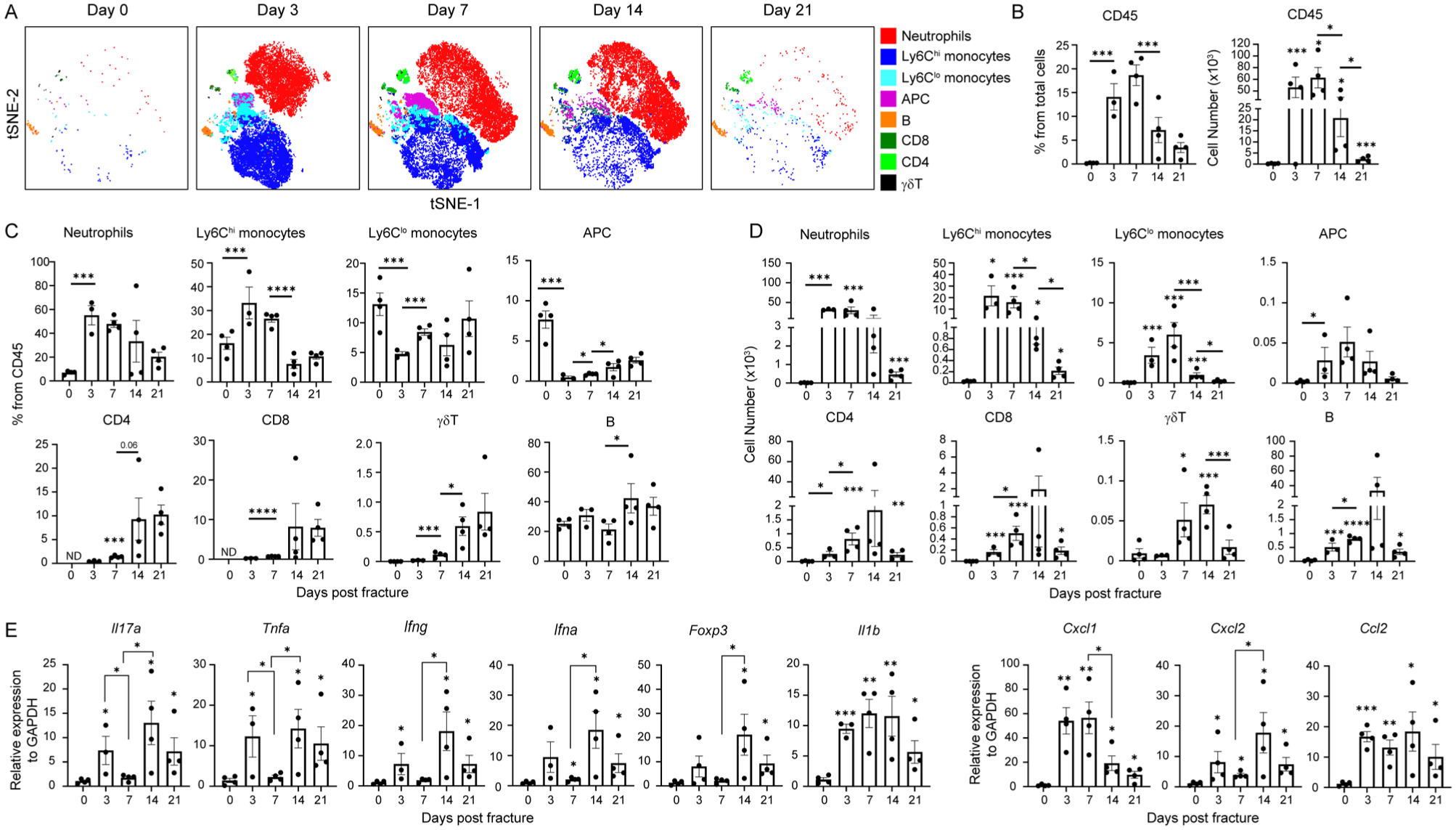
Longitudinal characterization of the immune response at the site of the drilled hole. (
Accelerated Mandibular Bone Defect Healing in the Absence of γδT Cells and IL-17
To investigate the role of γδT cells in the mandibular drill hole model, we first analyzed the subsets of γδT cells present at the defect site 10 d postprocedure. Flow cytometric analysis revealed a significant increase in γδT-cell frequencies in the injured mandible as compared with the intact control (Fig. 3A). Among these cells, approximately half of the γδT cells were Vγ6+, the other half were Vγ4+, whereas Vγ1+ cells were not detected (Fig. 3A). Next, to determine whether IL-17 production is mediated by γδT cells, we used Tcrd-GDL mice, which express green fluorescent protein, diphtheria toxin (DT) receptor, and luciferase under the control of a successfully rearranged δ TCR chain (Sandrock et al 2018). DT was administered before and after the drill hole procedure to deplete γδT cells, and soft tissues were collected after 7 d. qPCR analysis showed a significant reduction in Il17a expression in DT-treated mice (γδT cell depleted) as compared with phosphate-buffered saline (PBS)–treated controls (Fig. 3B). In contrast, the absence of γδT cells did not alter the expression of Ifng, Areg, Igf1, or Kgf, genes that are associated with these cells (Appendix Fig. 2). To directly assess IL-17–producing cells, intracellular cytokine staining was performed on tissue isolated 7 d postprocedure. As depicted in Figure 3C, gating on Thy1+ leukocytes and further on αβT cells, γδT cells, and non–T cells revealed that only γδT cells secreted IL-17. To examine the impact of γδT-cell depletion on bone healing, we compared mandibular drill hole repair in DT- and PBS-treated Tcrd-GDL mice (Fig. 3D). Micro–computed tomography analysis revealed that DT-treated mice exhibited higher bone volume in the defect site as compared with PBS-treated controls. Additionally, the bone surface density and bone-specific surface area were increased in DT-treated mice, indicating structural alterations in the newly formed bone. However, bone mineral density was similar between the groups, demonstrating that the quality of bone mineralization remained unchanged. To further examine the role of IL-17, we performed the mandibular bone defect procedure in Il17af-/- mice. As shown in Figure 3E, the bone volume within the defect site was significantly higher in Il17af-/- mice as compared with littermate controls. Moreover, bone analysis in Il17af-/- mice mirrored the findings in DT-treated Tcrd-GDL mice, except for the bone mineral density, which was increased in Il17af-/- mice. Analysis of intact mandibles revealed altered bone structure in Il17af-/- mice (Appendix Fig. 3), indicating that the increased bone mineral density at the healed site reflects an intrinsic effect of IL-17 deficiency. To determine whether the absence of γδT cells affects the healing of a drill hole defect in long bones, we created a defect in the femur and assessed the healing at days 14 and 21 postprocedure. As demonstrated in Appendix Figure 4, no differences were observed in the level of bone generated at the fracture site. Collectively, these results suggest that γδT cells impair the healing of mandibular bone defects through IL-17 production.
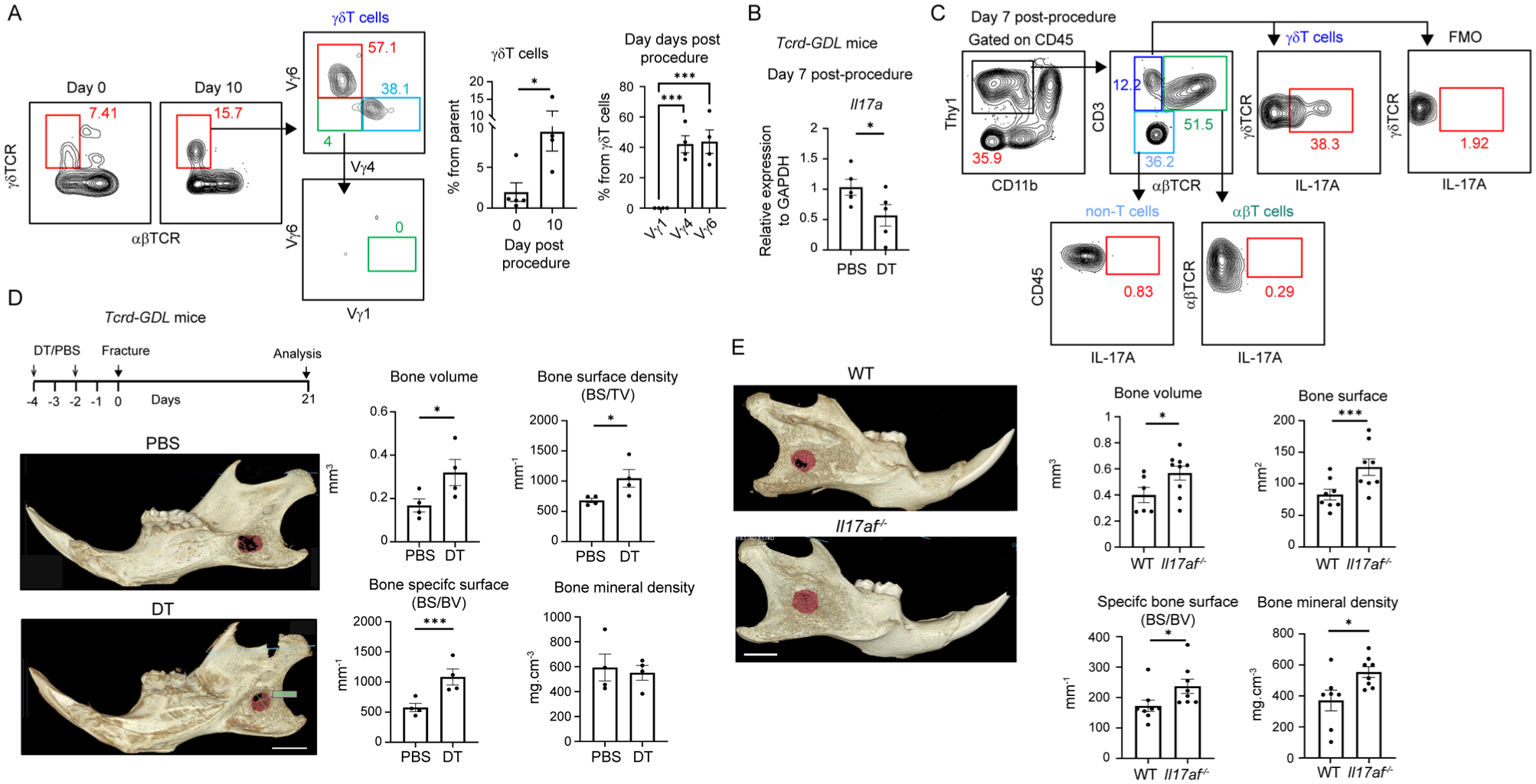
The absence of γδT cells or interleukin 17 (IL-17) accelerates the healing of the mandibular drilled hole defect. (
RNA Sequencing Analysis Reveals Reduced Inflammation and Altered Metabolism in the Bone Defect Tissue of Il17af-/- Mice
To unbiasedly investigate the impact of IL-17 on mandibular bone defect healing, soft tissues from the vicinity of the drill hole were collected on the fifth day from Il17af-/- mice and their littermates for RNA sequencing. Hierarchical clustering and principal component analysis revealed a significant difference between tissues collected from both groups (Fig. 4A, B). As depicted in Figure 4C, gene set enrichment analysis revealed in the Il17af-/- group an upregulation in oxidative phosphorylation, myogenesis, adipogenesis, fatty and bile acid, and heme metabolic pathways. This could reflect improved cellular energy availability and muscle fiber formation/repair, as well as a shift toward lipid utilization, fat storage, and oxidative detoxification associated with the repair processes. These alterations are likely related to the response to the bone defect, as examination of various metabolism-related genes at steady-state mandible revealed no differences between Il17af-/- mice and their controls (Appendix Fig. 5). The downregulated pathways include immune and inflammatory pathways such as interferon responses (IFN-α/γ), TNF-α via NF-κB, IL-6/JAK/STAT3 signaling, and complement. Such dampened inflammatory signals may prevent excessive inflammation and improve healing. In addition, a reduction in cell cycle and proliferation pathways was detected (E2F targets, G2M checkpoint, and mitotic spindle), potentially shifting the balance toward differentiation and repair. In agreement, a reduction in epithelial–mesenchymal transition and apical junctions suggests a stabilization of the tissue microenvironment in Il17af-/- mice, promoting organized repair. The reduction in other metabolic pathways, such as glycolysis and mTORC1 signaling, correlates with a reduced reliance on anaerobic metabolism, favoring more efficient oxidative pathways. We further performed an ingenuity pathway analysis, revealing an upregulation in MSP-RON signaling in the macrophage pathway. Activation of MSP-RON promotes the differentiation of macrophages into the M2 phenotype associated with tissue repair (Huang et al 2020). IL-10 signaling was also upregulated, which, as an anti-inflammatory cytokine, favors tissue repair. Downregulated pathways include a neuroimmune signaling pathway and various immunologic pathways. In agreement with these findings, qPCR analysis identified reduced expression in Il17af-/- mice of inflammatory cytokines and chemokines (Il6, Ifng, Ifna, Il1ba, and Cxcl2 but not Ccl2; Fig. 4E). A reduction was also observed in the expression of Foxp3, a gene involved in immunoregulation, as well as Rankl, which is associated with osteoclastogenesis. Of note, the gene amphiregulin (Areg), which has been proposed to affect alveolar bone remodeling by γδT cells (Krishnan et al 2018), was unaffected. Collectively, these findings suggest that the lack of IL-17 results in reduced inflammation and alteration in metabolic and physiologic pathways associated with accelerated tissue repair in the mandibular defect site.
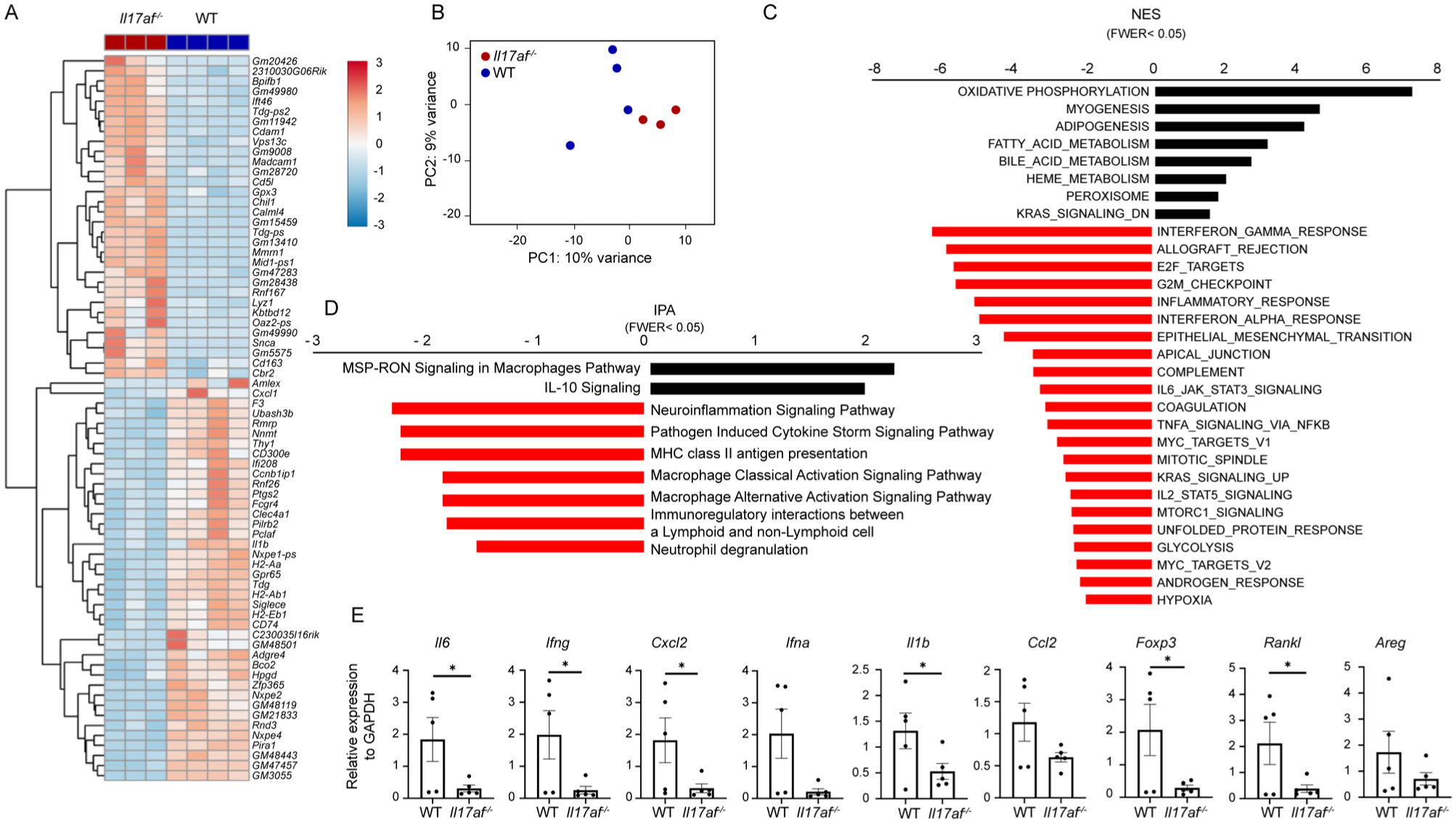
Reduced inflammatory and alerted metabolic pathways in bone defect tissue of Il17af-/- mice. Mandibular soft tissues were harvested from the drilled hole site in Il17af-/- mice (n = 3) and littermate controls (n = 4) 7 d postprocedure for global gene expression analysis. (
Decreased Bone Defect Immune Responses and Osteoclastogenic Activity in Il17af-/- Mice
Given the role of γδ17T cells as key initiators of inflammation (Papotto et al 2017), we examined the early immune response at the defect site. As shown in Figure 5A, flow cytometry analysis conducted 3 d postprocedure revealed a reduction in the frequency of total CD45+ leukocytes in Il17af-/- mice as compared with their littermate controls. However, the relative frequencies and numbers of neutrophils and monocytes, which represent innate immunity, were not reduced (Fig. 5B–D). In contrast, the relative frequencies and numbers of adaptive immune cells, including CD4+ and CD8+ T cells, as well as B cells, were significantly lower in Il17af-/- mice. This observation suggested that the absence of IL-17 specifically dampens adaptive immunity. Reduced frequencies of leukocytes at the defect site were also detected in Il17af-/- mice 7 d after the defect, when adaptive immunity takes place (Fig. 5E). Analysis of T regulatory cells at this time revealed that whereas their frequencies among the CD4 T cells (CD4+FOXP3+) was unchanged, the overall frequencies among the total leukocytes were reduced (Fig. 5E). Interestingly, on day 7 postprocedure, reduced frequencies of CD11b+MHCII+ APCs were found in the Il17af-/- mice, while the relative fraction of monocyte-derived dendritic cells (DCs; CD11c+CD64+) outnumbered those of monocyte-derived macrophages (CD11c-CD64+; Fig. 5F). Moreover, Il17af-/- mice showed reduced expression of Nos2 and Il6 in the injured mandible, with increased Cd206 but not Arg1, indicating a shift toward an M2-like phenotype (Fig. 5G). We next investigated whether the observed reduction in immunity influenced bone remodeling parameters. Notably, systemic CTX-1 levels were significantly reduced in Il17af-/- mice, whereas P1NP levels remained unchanged (Fig. 5H). Staining for osteoclasts by tartrate-resistant acid phosphatase (TRAP) further revealed a reduced number of TRAP+ cells in Il17af-/- mice (Fig. 5I). These findings suggest that the absence of IL-17 diminishes local immune responses at the bone defect site, leading to decreased osteoclastogenic activity.
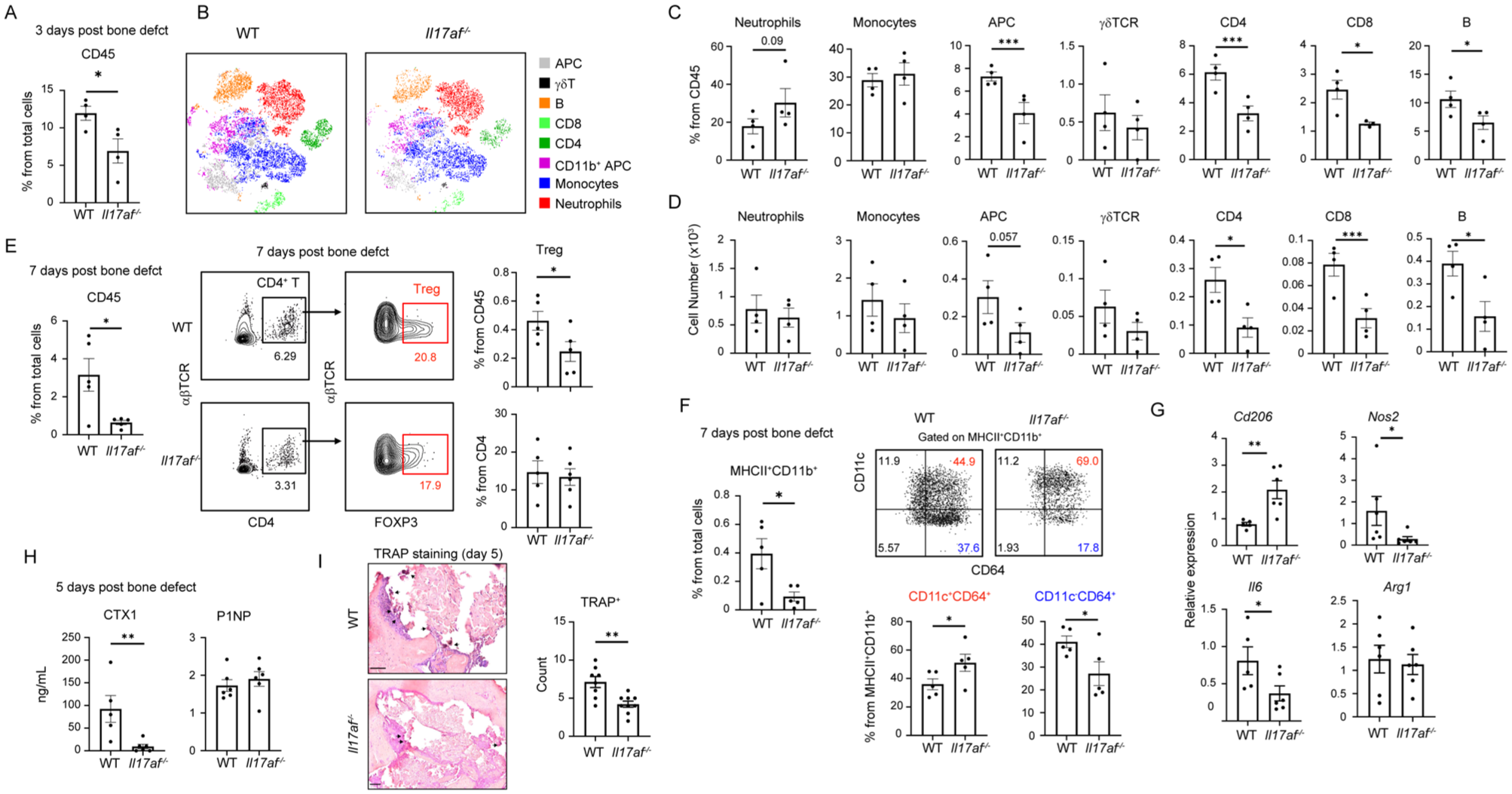
Decreased immunity and systemic CTX-1 levels in drilled hole tissues of Il17af-/- mice. A drilled hole model was induced in Il17af-/- mice and littermate controls. Three days postprocedure, tissues were collected for flow cytometry analysis. (
Discussion
This study demonstrates that γδ17T cells impede the spontaneous healing of mandibular drill hole defects by delaying the resolution of inflammation and repair. While studies on long bones, such as the femur, suggest a beneficial role for γδT cells (Ono et al 2016; Dar et al 2023), a tibia fracture model reported the opposite (Colburn et al 2009), indicating that bone type alone may not explain this discrepancy. IL-17 has also shown beneficial and detrimental effects in long and flat bones (Kim et al 2014, 2020; Ono et al 2016; Dixit et al 2017; Dar et al 2023), suggesting that the role of γδ17T cells is complex and controlled by other factors. One such factor is the microbiota, which regulates fracture healing (Dar et al 2023) and shapes γδT-cell development and function (Papotto et al 2021). Differences in microbial composition could thus influence the role of γδT cells in bone repair, especially in the mandible, where oral γδT cells are strongly affected by microbial cues (Wilharm et al 2019; Koren et al 2021), and the microbiota also affects bone mass and matrix properties (Vahidi et al 2023). Although the mandibular defect is created externally, it lies close to the buccal mucosa and is likely influenced by the oral immune system and microbiota. Microbial components, immune cells, and cytokines from the adjacent mucosa can modulate inflammation and bone remodeling, affecting the healing process. Given the abundance of γδ17T cells in the oral mucosa, their interaction with the microbiota likely plays a role in mandibular bone healing (Wilharm et al 2019).
The presence of Vγ6+ and Vγ4+ subsets in the defect site suggests that γδT cells populate the tissue via local proliferation of the embryonically derived Vγ6+ and from circulating Vγ4+ cells (Wilharm et al 2019). Vγ4+ can be induced to produce IL-17 and thus might support the Vγ6+ subset, which has a natural capacity to secrete this cytokine (Chien et al 2013). To eliminate γδT cells, the present study utilized Tcrd-GDL mice instead of the Tcrd-/- mice employed in previous studies investigating their role in fracture healing (Colburn et al 2009; Ono et al 2016; Dar et al 2023). This represents a significant difference, as Tcrd-GDL mice undergo normal postnatal development, and γδT cells are depleted only during the experiment (Sandrock et al 2018). In contrast, Tcrd-/- mice lack γδT cells throughout development, which may lead to compensatory mechanisms that obscure the genuine physiologic role of γδT cells. This distinction is particularly relevant in the oral mucosa, where γδT cells play a critical role in postnatal development (Koren et al 2021). Furthermore, studies have shown that Tcrd-GDL and Tcrd-/- mice exhibit distinct outcomes in conditions such as natural or ligature-induced bone loss during adulthood (Krishnan et al 2018; Wilharm et al 2019; Barel et al 2022). It would be intriguing to investigate whether the use of Tcrd-GDL mice in various long bone fracture models would yield different healing outcomes as compared with Tcrd-/- mice, providing further insight into the role of γδT cells in fracture repair.
In the absence of IL-17, inflammatory responses at the defect site were reduced, facilitating a more rapid transition to the reparative phase (Baht et al 2018). Interestingly, the early recruitment of neutrophils and monocytes remained unaffected, indicating that γδ17T cells may not directly regulate their recruitment. Instead, other mechanisms, such as those mediated by damage-associated molecular patterns (Pittman and Kubes 2013), might play a role. Our analysis also demonstrates that adaptive immunity is predominantly suppressed, consistent with the ability of γδT cells to activate DCs (Wang et al 2021). Among cytokines downregulated in the absence of IL-17 was IL-1β, whose expression was persistently elevated. As high or prolonged IL-1β can impair repair (Lange et al 2010; Hengartner et al 2013; Wang et al 2020), its reduction in IL-17af -/- mice suggests an additional pathway by which IL-17 delays healing.
Macrophages play a pivotal role in fracture healing, transitioning from a proinflammatory (M1) phenotype to an anti-inflammatory (M2) phenotype that promotes repair during later stages (Frade et al 2023). Our analysis revealed a reduction in CD64-expressing CD11b+CD11c−MHCII+ APCs, suggesting impaired differentiation of macrophages from monocytes in the absence of IL-17. Despite their reduced frequencies, RNA sequencing data and cytokine profiles indicate that the remaining macrophages predominantly exhibit an M2 phenotype (Yunna et al 2020). Interestingly, ingenuity pathway analysis highlighted a reduction in classical (M1) and alternative (M2) macrophage activation pathways. IL-17 has been reported to directly promote M1 macrophage polarization (Zhang et al 2013). The reduction in Nos2 and Il6 expression, typically associated with the M1 phenotype, with the increased expression of Cd206 (M2), substantiates this view. Notably, although IL-17 deficiency reduces monocyte-derived CD11b+MHCII+ APCs, more of these cells adopt a CD64+ DC phenotype (Langlet et al 2012), which may support repair and compensate for reduced macrophages, as monocyte-derived DCs can contribute to regeneration (Stobener et al 2023).
In conclusion, this study demonstrates that γδ17T cells delay mandibular defect healing by promoting local inflammation. Future research should investigate the factors regulating γδ17T-cell function to understand their complex role and harness their potential to improve fracture healing strategies.
Author Contributions
R. Naamneh, F.L. Shoukair, M. Gera, Y. Jaber, Y. Netanely, Y. Saba, K. Zubeidat, contributed to conception and design, data acquisition, analysis, and interpretation, critically revised the manuscript; S. Yacoub, contributed to conception and design, data analysis and interpretation, critically revised the manuscript; A. Wilensky, I. Prinz, N. Casap, contributed to conception and design, critically revised the manuscript; A.H. Hovav, contributed to conception and design, drafted and critically revised the manuscript. All authors gave final approval and agreed to be accountable for all aspects of the work.
Supplemental Material
sj-docx-1-jdr-10.1177_00220345251391194 – Supplemental material for γδ17T Cells Hinder Mandibular Bone Defect Healing
Supplemental material, sj-docx-1-jdr-10.1177_00220345251391194 for γδ17T Cells Hinder Mandibular Bone Defect Healing by R. Naamneh, F.L. Shoukair, M. Gera, Y. Jaber, S. Yacoub, Y. Netanely, Y. Saba, K. Zubeidat, A. Wilensky, I. Prinz, N. Casap and A.H. Hovav in Journal of Dental Research
Supplemental Material
sj-pdf-2-jdr-10.1177_00220345251391194 – Supplemental material for γδ17T Cells Hinder Mandibular Bone Defect Healing
Supplemental material, sj-pdf-2-jdr-10.1177_00220345251391194 for γδ17T Cells Hinder Mandibular Bone Defect Healing by R. Naamneh, F.L. Shoukair, M. Gera, Y. Jaber, S. Yacoub, Y. Netanely, Y. Saba, K. Zubeidat, A. Wilensky, I. Prinz, N. Casap and A.H. Hovav in Journal of Dental Research
Footnotes
Acknowledgements
We thank Yuval Nevo, Inbar Plaschkes, and Hadar Benyamini for the bioinformatic analysis.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Israel Science Foundation grant 2272/20 and the Samuel Badihi Foundation for A.H.H.
Data Availability
The RNA sequencing data generated in this study will be deposited in the NCBI Gene Expression Omnibus GEO database (GSE312365). All data are available in the main text and the supplementary materials.
A supplemental appendix to this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
