Abstract
Oral squamous cell carcinoma (OSCC) is a highly aggressive malignancy, with a low 5-y survival rate and frequent local recurrence or metastasis. This study explores the role of γδT cells in the development and progression of OSCC. γδT cells, which exhibit innate and adaptive immune characteristics, are known for their dual role in cancer, acting as anti- and protumor agents depending on the context. Using a murine model of OSCC induced by the carcinogen 4-nitroquinoline-1-oxide (4NQO), which adequately mimics the progression of human OSCC, we investigated the impact of γδT cells on tumor growth and the tumor microenvironment. We first characterized the γδT cells of the tongue epithelium, the primary site for cancer development in this model. The results indicate that γδT cells are predominantly of the Vγ6+ subset, expanding postnatally in a microbiota-dependent manner. Upon 4NQO administration, depletion of γδT cells did not significantly alter the kinetics of OSCC progression but did result in a reduction in tumor size and number, suggesting a role in promoting tumor growth. Interestingly, the absence of IL-17, a key cytokine produced by the Vγ6+ subset, also resulted in reduced tumor volume without affecting disease progression, corroborating the protumor role of these cells in OSCC. Further analysis revealed that IL-17–producing γδT cells facilitate angiogenesis within the tumor microenvironment by promoting the expression of angiogenic factors. Of note, while 4NQO treatment increased the oral microbial load and altered its composition, IL-17 deficiency did not affect the oral microbiota, indicating that the effects of IL-17–producing γδT cells on OSCC are independent of microbial changes. This study highlights the pathologic role of IL-17–producing γδT cells in OSCC, particularly in promoting tumor growth through angiogenesis. This underscores the importance of γδT cells in OSCC and the need for further research into therapeutic strategies targeting these cells.
Introduction
Oral squamous cell carcinoma (OSCC) is a highly prevalent and invasive malignancy (Rivera 2015). Since OSCC originates in the epithelium, epithelial leukocytes are the first to detect and respond to early carcinogenic events. Indeed, Langerhans cells have been shown to play a protective role during murine OSCC (Saba et al. 2022). Alongside Langerhans cells, the oral epithelium harbors γδT cells, which possess adaptive and innate immune characteristics (Chien et al. 2014). Murine γδT cells consist of 6 subsets: Vγ1+, Vγ2+, Vγ4+, Vγ5+, Vγ6+, and Vγ7+ cells. In adult mice, oral γδT cells are mainly composed of the Vγ6+, Vγ1+, and Vγ4+ subsets (Krishnan et al. 2018; Wilharm et al. 2019), while their relative frequencies might vary in different oral niches. Whereas Vγ6+ and Vγ4+ cells are the primary sources of IL-17 (γδ17T cells), the Vγ1+, Vγ5+, and Vγ7+ subsets are associated with IFN-γ secretion (Papotto et al. 2017). γδT cells play a role in maintaining oral homeostasis and are involved in oral pathologies (Hovav et al. 2020). Nevertheless, despite increasing knowledge about oral γδT cells, their role in OSCC is ill-defined.
γδT cells can function independently of the major histocompatibility complex (Willcox et al. 2020). They exhibit natural killer cell–like cytotoxicity and express various receptors that detect and eliminate cells showing stress signals (Ribot et al. 2021). Consequently, γδT cells are recognized as cytotoxic and antitumor lymphocytes (Gentles et al. 2015). However, certain subsets of γδT cells, particularly γδ17T cells, have the potential to function as tumor-promoting entities (Silva-Santos et al. 2019). The tumor-promoting effects of IL-17 can manifest through impacts on macrophage polarization, angiogenesis, or neutrophil recruitment (Silva-Santos et al. 2019). Conversely, γδ17T cells have also demonstrated anticancer activity (Ma et al. 2011). Therefore, the precise role of γδT cells is contingent upon the specific type of cancer and the interactions with the tumor microenvironment.
Human studies have demonstrated that γδT cells exhibit anti- and protumor activities during head and neck squamous cell carcinoma (HNSCC; Bas et al. 2006; Sureshbabu et al. 2020; Arias-Badia et al. 2024). Additionally, while a bioinformatic study linked high intratumoral γδT-cell abundance with a favorable prognosis in HNSCC (Lu et al. 2020), recent findings showed these cells as negative predictors of survival (Parikh et al. 2023). In murine models, typically utilizing transplantation of OSCC cell lines, γδ17T cells responded to periodontitis-induced dysbiosis, facilitating OSCC via M2 macrophage recruitment (Wei et al. 2022). These reports underscore the imperative need for understanding the precise role of γδT cells in OSCC.
Materials and Methods
Experimental OSCC
One gram of 4-nitroquinoline-1-oxide (4NQO; N8141-5G [Sigma]) was dissolved in 200 mL of propylene glycol (P4347-500ML; Sigma) over 3 to 5 h, ensuring homogeneity. Aliquots of the stock solution (5 mg/mL) were stored at 4 °C. For experimental use, the stock solution was diluted in 250 mL of drinking water, reaching a final concentration of 60 μg/mL. The water, housed in a dark bottle, was replenished twice a week. Following 16 wk of treatment, the mice were transitioned to regular water.
Extensive experimental details are presented in the Appendix.
Animal protocols were approved by the Hebrew University Institutional Animal Care and Use Committee. The study conforms to the ARRIVE guidelines.
Results
Vγ6+ T Cells Are the Main γδT-Cell Subset in the Tongue Epithelium That Expands Postnatally in a Microbiota-Dependent Manner
Since the tongue is a primary site for tumor growth in experimental OSCC, we initially focused on profiling the γδT cells in the tongue epithelium. As illustrated in Appendix Figure 1A, γδT cells were identified in the epithelium, with the majority expressing the Vγ6+ chain and the rest Vγ1+ and Vγ4+. Further examination revealed that the number of γδT cells in the tongue increased gradually postnatally, peaking during adulthood (Appendix Fig. 1B). The frequencies of γδT cells, however, remained stable among total leukocytes until weaning, subsequently declining in adults. We next explored the influence of the microbiota on the development of tongue γδT cells. As shown in Appendix Figure 1C, reduced frequencies of γδT cells were observed in germ-free mice as compared with specific pathogen–free mice. These findings indicate that the microbiota regulates the expansion of γδT cells in the tongue epithelium until adulthood, predominantly comprising the Vγ6+ subset.
Ablation of γδT Cells Increases the Size and Number of Oral Tumors
To investigate the role of γδT cells in experimental OSCC, we used the carcinogen 4NQO, which adequately mimics the gradual progression observed in patients with OSCC. Additionally, we used Tcrd-GDL knock-in mice, which express 3 reporter genes (GDL: Gfp-Dtr-Luc) specifically in γδT cells under the control of successfully rearranged TCR δ genes, enabling the ablation of γδT cells upon administration of diphtheria toxin (DT). The Tcrd-GDL mice were exposed to the chemical carcinogen 4NQO for 16 wk in their drinking water, concomitantly with the depletion of γδT cells via DT injections (Fig. 1A, B). Assessment of the tongue revealed that all 4NQO-treated mice eventually developed carcinogenesis, with no significant differences in carcinogenesis kinetics observed between mice with γδT-cell depletion and controls (Fig. 1C). However, pathologic evaluation classified all tumors in the 4NQO + DT group as benign papillomas or high-grade dysplasia, whereas in the 4NQO + phosphate-buffered saline (PBS) group, 3 out of 10 mice developed invasive tumors, with the remainder presenting benign papillomas (Fig. 1D). No spontaneous tumors were observed in the control groups receiving DT injections or PBS (Fig. 1E). Consistent with these results, the 4NQO-treated groups exhibited weight loss, which was accelerated by the coadministration of DT (Fig. 1F). Notably, injection of DT in Tcrd-GDL mice, either alone or in combination with 4NQO, resulted in a sharp and temporary weight loss. Subsequently, we examined the tumors on the tongues of 4NQO-treated mice. As depicted in Figure 1G, the average tumor volume in the 4NQO + DT group (1.4 mm3) was significantly smaller than in the control 4NQO + PBS group (4.2 mm3). Additionally, the number of tumors (papilloma and invasive) per mouse was lower in the mice depleted of γδT cells, with an average of 1 or 4 tumors per tongue in the DT or PBS group, respectively. These findings suggest that while γδT cells do not affect the kinetics of OSCC progression, they influence the number and volume of the established tumors.
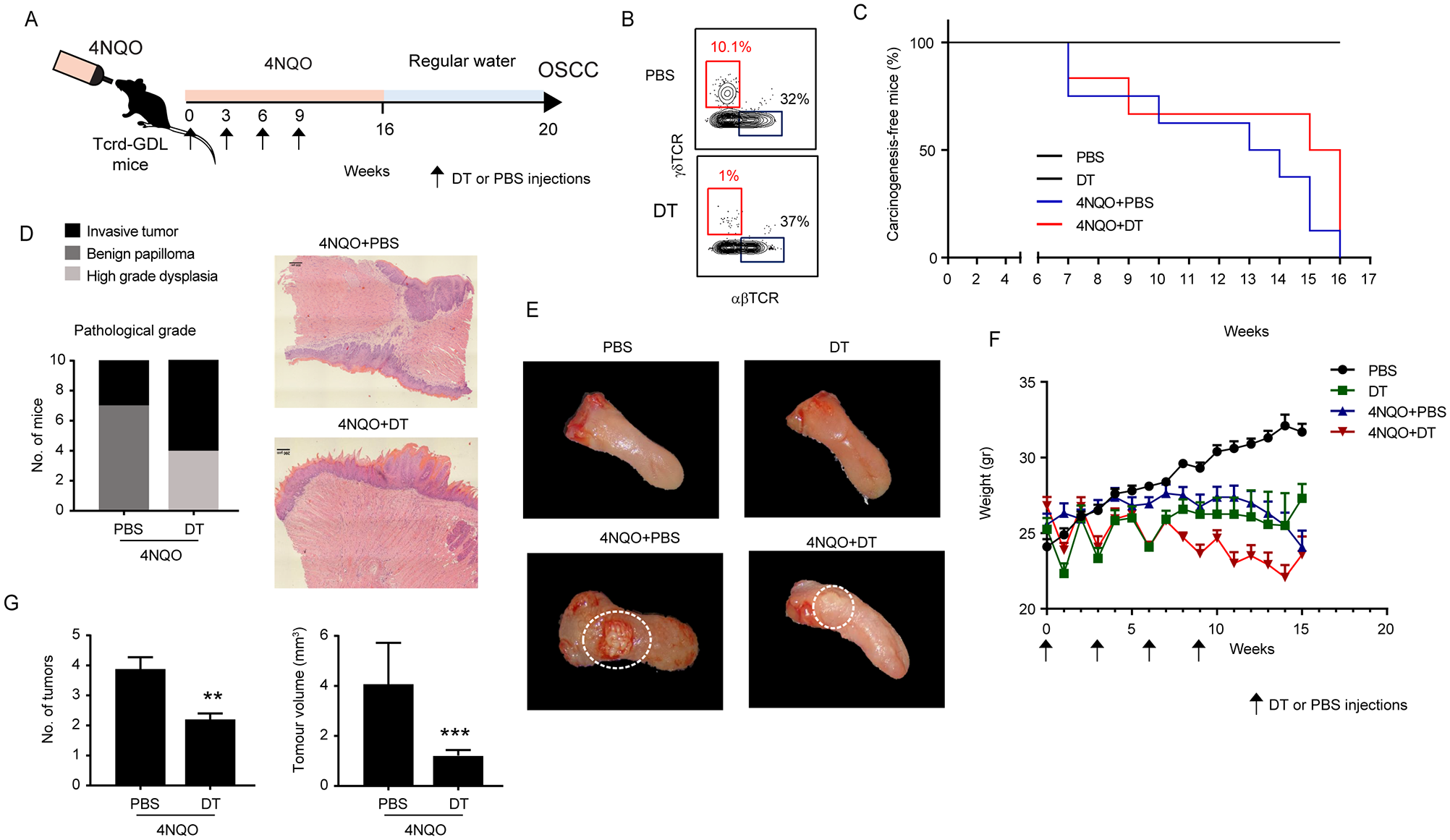
Impact of conditional ablation of γδT cells on 4NQO-induced carcinogenesis. (
Depletion of γδT Cells Has a Limited Impact on the Development of Tumor-Promoting Conditions in the Epithelium
It has been demonstrated that 3 to 5 wk after exposure to 4NQO before visible tumors emerge, the tongue epithelium shifts to a protumor state (Saba et al. 2022). This transition is characterized by reduced Langerhans cell frequencies and increased T-regulatory (Treg) cells, plasmacytoid dendritic cells (pDCs), and macrophages (Saba et al. 2022). We aimed to determine whether γδT-cell depletion affects this pathologic progression. As depicted in Appendix Figure 2A, 3 wk after 4NQO treatment—a stage in which no significant epithelial pathology was observed—the percentage of Langerhans cells in the tongue epithelium decreased, with no significant impact from γδT-cell ablation. Although the frequencies of epithelial Treg cells and pDCs increased after 4NQO treatment, γδT-cell depletion limited the expansion of pDCs but not Treg cells. Nevertheless, after 5 wk of treatment, a time when hyperplastic and dysplastic lesions were detected, pDCs and Treg cells were comparably elevated irrespective of the presence of γδT cells (Appendix Fig. 2B). Furthermore, the differentiation of CD64+ antigen-presenting cells in the epithelium, identified as macrophages with immunosuppressive capacities in a prior study (Saba et al. 2022), remained unaffected by the presence of γδT cells. These results suggest that γδT cells have a limited influence on the transition of the epithelium into tumor-permissive conditions.
Induction of OSCC in Il17af-/- Mice Results in Reduced Tumor Size without Affecting Carcinogenesis Progression
Given that the Vγ6+ subset is the primary producer of IL-17 (Wilharm et al. 2019), we investigated the role of this cytokine in OSCC. To achieve this, Il17af-/- mice and littermate wild type (WT) controls were exposed to 4NQO in the drinking water, following the previously described protocol. As depicted in Figure 2A, the development kinetics of carcinogenesis in Il17af-/- mice were similar to those observed in WT control mice. However, while WT mice started to lose weight 8 wk after exposure to 4NQO, Il17af-/- mice maintained their body weight, with a slight reduction observed only after 16 wk (Fig. 2B). Pathologic characterization classified the tumors in the Il17af-/- mice as a benign papilloma or high-grade dysplasia, while in the littermate WT controls, 2 out of 8 mice developed invasive tumors and the rest benign papillomas (Fig. 1C). Quantification of tumors in the mice (papilloma and invasive) showed similar numbers of tumors per mouse in both groups (Fig. 2D). Nevertheless, tumor volumes, were approximately 10 times higher in littermate control mice as compared with Il17af-/- mice (Fig. 2E). Representative clinical (Fig. 2F) images underscored the substantial difference in tumor volume between Il17af-/- mice and littermate WT controls. This implies that the absence of IL-17 recapitulates most of the effects of γδT cells during OSCC.
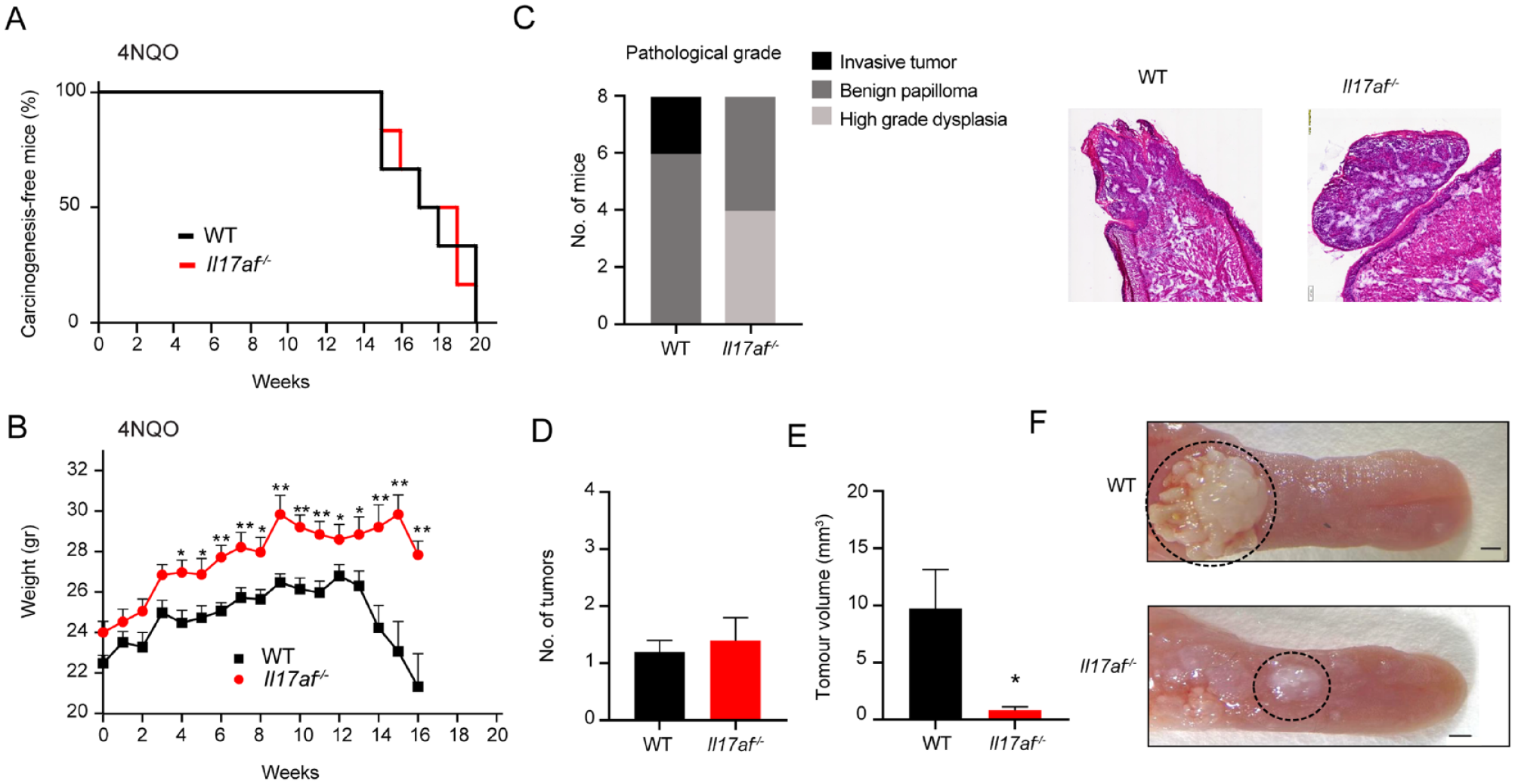
Increased tumor volume in Il17a/f-/- mice. Il17a/f-/- mice were exposed to 4NQO in the drinking water for 16 wk. (
Absence of IL-17 Modulates the Expression of Intratumoral Pathways Linked to Tumor Growth, Metabolism, Immune Responses, and Angiogenesis
To gain insights into the influence of IL-17 on OSCC, we conducted a comprehensive analysis of global gene expression in tumor tissues isolated from Il17af-/- and WT littermate controls classified as either papilloma or early invasive stage. Hierarchical clustering of the 2 cell populations, Silhouette score analysis, and principal component analysis underscored a significant divergence between the tumors of Il17af-/- and WT littermates (Fig. 3A–C). Subsequently, we performed gene set enrichment analysis to elucidate hallmark pathways differentially expressed in the tumors from both groups of mice (Fig. 3D). The most upregulated pathway in Il17af-/- mouse tumors was myogenesis, aligning with the known capacity of IL-17 to inhibit myogenic differentiation (Kocic et al. 2012) and a report indicating myogenesis as the most downregulated pathway in human OSCC (Mumtaz et al. 2022). Notably, K-ras and hedgehog signaling pathways, implicated in the pathology and progression of human OSCC, were also upregulated (Srinath et al. 2016; El Hanbuli and Abou Sarie 2022). Additional upregulated pathways were associated with metabolic dysregulation linked to cancer, including glycolysis, heme metabolism, peroxisome function, oxidative phosphorylation, fatty acid, and bile acid metabolism (Kim 2020). An upregulation of adipogenesis was also observed, as a process supporting tumor growth and known to be inhibited by IL-17 (Ahmed and Gaffen 2013). The analysis also uncovered several downregulated pathways in tumors isolated from Il17af-/- mice (Fig. 3D). The allograft rejection pathway was reduced, which strongly reflects anticancer immune activity and infiltration of immune cells (Oshi et al. 2022). Other key immunologic pathways were downregulated, encompassing IL6/JAK/STAT3 signaling, inflammatory response, IFN-γ and IFN-α responses, and IL2/STAT5 signaling, all of which have been correlated with better prognosis in HNSCC (She et al. 2020). The reduced pathways also included signaling involved in cell proliferation, such as MYC targets V2 and V, E2F targets, G2M checkpoint, DNA repair, and mitotic spindle. Pathways such as TNF-α via NF-κB signaling and unfolded protein response were reduced, representing responses associated with cancer aggressiveness (Oshi et al. 2022), invasion, and metastasis of OSCC (Tang et al. 2017). Recognizing the reported role of IL-17 in regulating angiogenesis in various cancers, we employed ingenuity pathway analysis to investigate this aspect. The analysis indeed revealed a substantial reduction in several pathways associated with angiogenesis (Fig. 3E). Importantly, gene set enrichment analysis of the impact of IL-17A expression in human HNSCC revealed overall similar pathway changes to those observed in our murine analysis, although the angiogenesis pathway was not significantly altered (Appendix Fig. 3A). However, in contrast to the detrimental effect observed in mice, higher levels of IL-17A were associated with an improved probability of overall survival (Appendix Fig. 3B). In conclusion, our analysis sheds light on the intricate impact of IL-17 on experimental OSCC, unveiling significant alterations in key pathways related to tumor growth, metabolism, immunologic responses, and angiogenesis. In humans, although IL-17 similarly influences biological pathways in HNSCC, it is associated with a protective effect on survival.
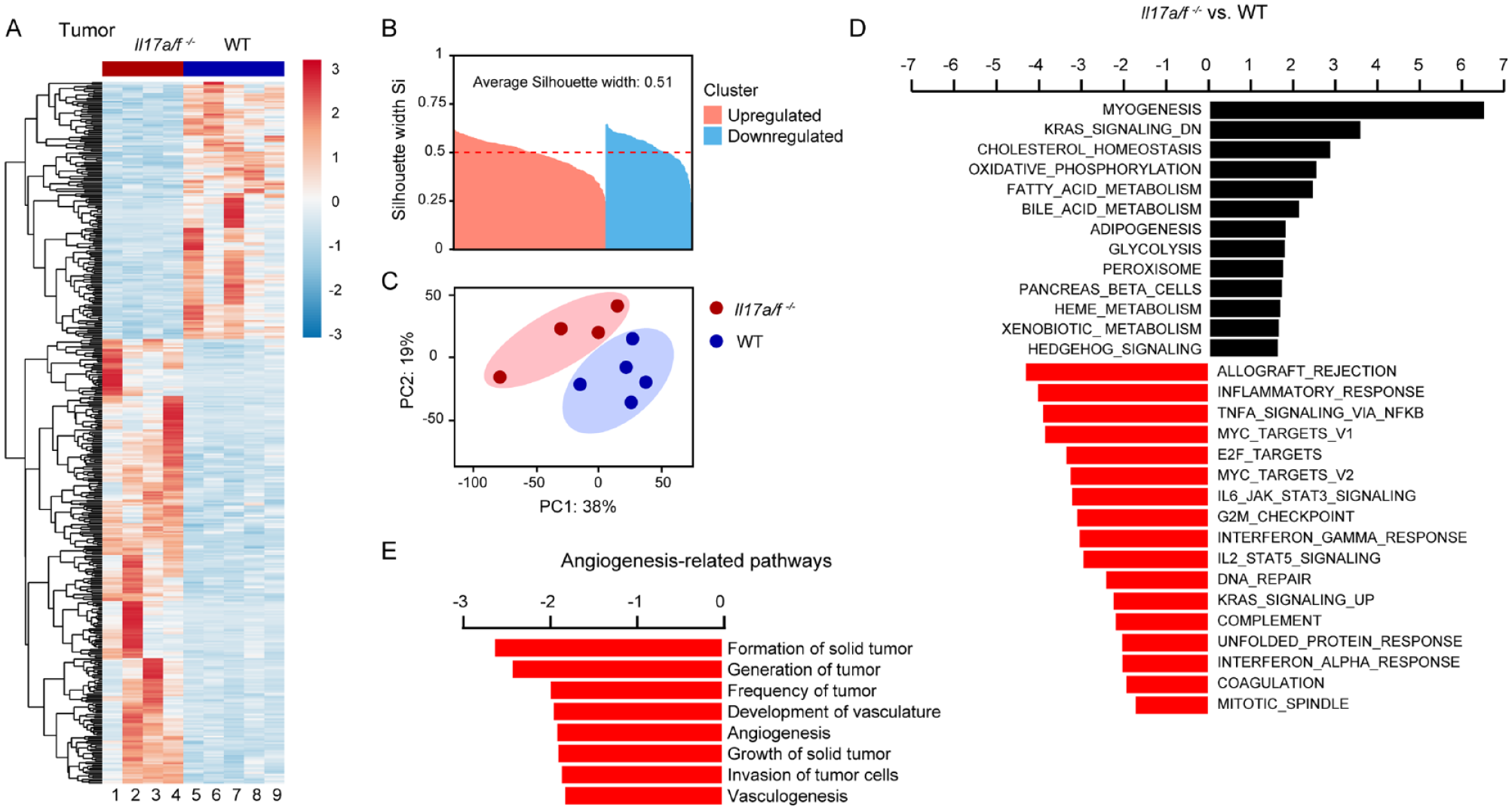
IL-17 induces transcriptomic changes in OSCC tumors. OSCC tumor cells were collected from Il17a/f-/- mice and littermate controls for global gene expression analysis. (
Intratumoral γδ17T Cells Promote Angiogenesis in Experimental OSCC
To assess the influence of IL-17 on tumor development and local angiogenesis, we initially examined the leukocyte composition within the tumor, which graded from papilloma to early invasive stages. As illustrated in Figure 4A and B, tumors developed in Il17af-/- mice exhibited decreased frequencies of neutrophils, which constitute the predominant leukocyte subset among the total intratumoral leukocytes in the WT group. The frequency of tumor macrophages remained unchanged in the absence of IL-17. However, given the role of IL-17 in M1/2 macrophage polarization, we measured the expression of representative cytokines. As depicted in Figure 4C, Il6, associated with M1 macrophages, was elevated in Il17af-/- tumors, while Tgfb1, associated with the M2 phenotype, decreased. Intriguingly, although no significant alterations were noted in αβT-cell frequencies between WT and Il17af-/- tumors, higher frequencies of γδT cells were observed in the latter. Next, as IL-17 can be generated by various lymphocytes, we aimed to identify the specific cell types responsible for its production within the tumors. Tumors were collected from WT mice, and by focusing on Thy1+ lymphocytes and αβ or γδ TCR expression, we identified the αβT cells, γδT cells, and a double-negative population likely representing innate lymphoid cells (Fig. 4D). Despite the diverse lymphocyte populations, the predominant IL-17–producing cells were identified among the γδT-cell subset in terms of percentages and absolute numbers. Subsequently, we utilized anti-CD31 antibodies to stain tumor cross sections and visualize intratumoral blood vessels. As illustrated in Figure 4E, blood vessels were evident in the tumor. Yet, depletion of γδT cells reduces fluorescence intensity, indicating a decrease in blood vessel formation. A similar effect was observed in mice lacking IL-17 (Fig. 4F). Furthermore, analysis of tumors isolated from Il17af-/- mice revealed a diminished expression of Vegf and Tgfb1 (as demonstrated in Fig. 4C), known facilitators of angiogenesis, as compared with control counterparts (Fig. 4G). These findings underscore the crucial role of γδ17T cells in regulating intratumoral neutrophils, γδT cells, and macrophage M1 polarization, as well as in promoting angiogenesis within the OSCC microenvironment.
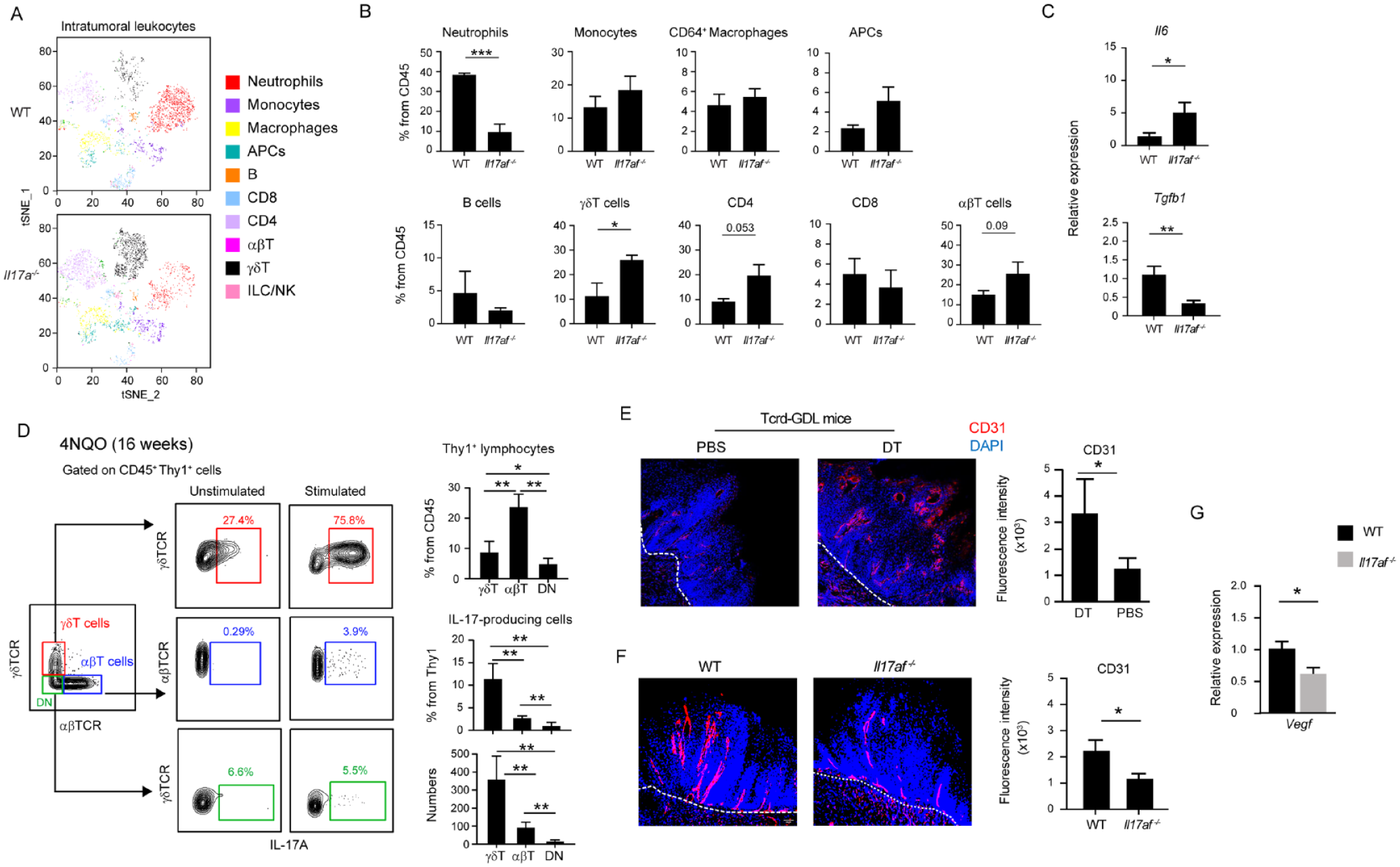
Intratumoral IL-17–producing γδT cells promote angiogenesis in experimental OSCC. (
4NQO Treatment, Rather Than IL-17, Controls the Oral Microbiome
Since oral microorganisms have been linked to the progression of oral cancer (Lan et al. 2023), we analyzed the oral microbiota in our system. Samples were collected from Il17af-/- and littermate WT mice at steady state and 16 wk after the initiation of 4NQO treatment, when tumors had developed. As depicted in Figure 5A, there were no differences in the load of cultivated aerobic and anaerobic bacteria between WT and Il17af-/- mice under either condition. However, the 4NQO treatment itself led to an increase in the oral microbiota load. Alpha diversity analysis revealed no differences in taxa richness between WT and Il17af-/- mice, either at steady state or following 4NQO treatment (Fig. 5B). The diversity of the microbiota, though, was influenced by the 4NQO treatment. Given that 4NQO, rather than IL-17, affects the load and diversity of the oral microbiota, we combined the data from WT and Il17af-/- mice to further examine the impact of 4NQO treatment on the oral microbiota. Principal component analysis showed that the composition of oral taxa differed significantly due to 4NQO treatment (Fig. 5C). Heat tree analysis demonstrated an abundance of Proteobacteria over Firmicutes in 4NQO-treated mice (Fig. 5D), and a single-factor analysis identified a significant expansion of Pasteurellales alongside a reduction of Bacillales in these mice (Fig. 5E). These findings suggest that the smaller tumors developed in the absence of IL-17 cannot be attributed to changes in the oral microbiota. In addition, exposure to 4NQO has a significant impact on the load and diversity of the oral microbiota.
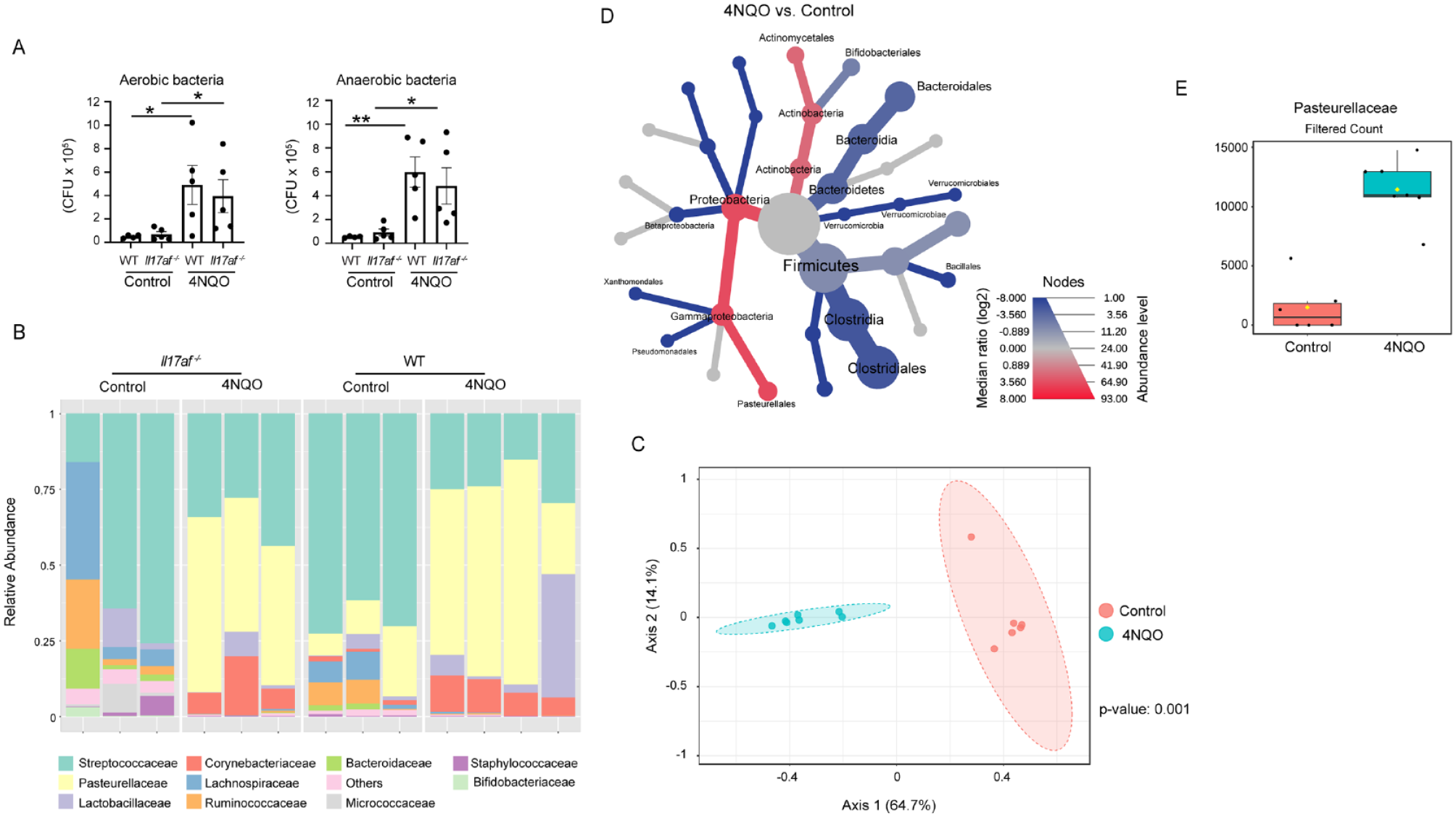
4NQO treatment, rather than IL-17, controls the oral microbiome. (
Discussion
This study underscores the pathologic role of γδ17T cells in carcinogen-induced OSCC. Although these cells did not influence disease progression, they regulate angiogenesis within the tumor, thereby promoting its growth. These findings align with previous reports indicating that γδT cells are unable to eliminate γH2AX+ cells early after 4NQO exposure, a process mediated by αβT cells (Saba et al. 2022). In contrast to their dispensable role in the early stages, γδT cells play a pathologic function in established tumors. Interestingly, bioinformatic studies have reported conflicting correlations between intratumoral γδT cells and HNSCC prognosis (Lu et al. 2020; Parikh et al. 2023). Moreover, while IL-17 affects HNSCC similarly to murine OSCC, its presence appears beneficial in humans, which contrasts with its negative role in mice. These contradictions are challenging to explain, particularly as human oral γδT cells have not yet been well characterized. However, unlike mice, humans have high levels of circulating γδT cells (Urban et al. 2010), which expand in patients with HNSCC (Bas et al. 2006). These circulating γδT cells may enter the tumor and exert protective or pathologic effects depending on the microenvironment. Supporting this idea, human γδT cells exhibit functional plasticity, influencing their anti- or protumor activities. In mice, although circulating γδT-cell subsets such as Vγ4 and Vγ1 are present in the oral mucosa (Wilharm et al. 2019), their low frequencies make it unlikely for them to be heavily recruited into the tumor. Moreover, the Vγ6 subset has shown a strong proliferative capacity (Rei et al. 2014), suggesting that it might be the predominant subset in OSCC tumors.
The lack of influence by IL-17 on the oral microbiome under normal conditions corresponds with its minimal role in OSCC progression. The oral microbiome is essential for developing OSCC by 4NQO (Stashenko et al. 2019), and the presence of periodontal pathogens exacerbates tumor progression (Binder Gallimidi et al. 2015). It is intriguing to consider whether inducing periodontitis in mice deficient in γδT17 cells would affect 4NQO-induced OSCC. Previous research has shown that periodontitis worsens 4NQO-induced OSCC in WT rats (Spuldaro et al. 2022). While γδT cells have shown a limited effect on ligature-induced periodontitis, they do promote periodontitis caused by Porphyromonas gingivalis infection (Barel et al. 2022). Since P. gingivalis is a key pathogen known to influence the oral microbiome (Hajishengallis et al. 2011), the detrimental effect of γδT cells on the infection might involve changes in the microbiota, which could in turn affect 4NQO-induced OSCC. Regardless, the expansion of Pasteurellales (Gammaproteobacteria class) and reduction of Bacillales (Bacilli class) in the presence of a tumor are consistent with findings involving human OSCC (Radaic et al. 2023), underscoring the significant impact that OSCC has on the local microbiota.
In addition to the absence of IL-17A, the Il17af-/- mice utilized in this study lack the IL-17F cytokines. Notably, serum levels of IL-17F have been proposed as a potential diagnostic biomarker for OSCC (Ding et al. 2015) and its salivary concentration linked to disease progression (Zielinska et al. 2020). Furthermore, polymorphisms in IL-17A and IL-17F are associated with an increased risk of OSCC and are correlated with tumor stage (Li et al. 2015). Conversely, at the tumor invasion front, IL-17F exhibits a protective effect in patients with tongue OSCC (Almahmoudi et al. 2018). It is thus challenging to speculate on the precise role of IL-17F in our setting.
In conclusion, this study highlights the role of γδT cells in promoting murine OSCC through angiogenesis. Further research is needed to understand how γδT cells influence tumor dynamics, particularly in the context of periodontitis and microbial dysbiosis, potentially leading to novel therapies targeting immune and microbial components of OSCC.
Author Contributions
Y. Saba, S. Yacoub, Y. Netanely, Y. Jaber, K. Zubeidat, A. Meyer, Y.E. Shlomovitz, L. Eli-Berchoer, contributed to conception, design, data acquisition, analysis, and interpretation, critically revised the manuscript; R. Naamneh, contributed to conception, design, data analysis and interpretation, critically revised the manuscript; A. Wilensky, I. Prinz, contributed to conception, design, critically revised the manuscript; A. Hovav, contributed to conception, design, data and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of the work.
Supplemental Material
sj-docx-1-jdr-10.1177_00220345241305564 – Supplemental material for γδ17T Cells Aggravate Carcinogen-Induced Oral Squamous Cell Carcinoma
Supplemental material, sj-docx-1-jdr-10.1177_00220345241305564 for γδ17T Cells Aggravate Carcinogen-Induced Oral Squamous Cell Carcinoma by Y. Saba, S. Yacoub, Y. Netanely, Y. Jaber, R. Naamneh, K. Zubeidat, A. Meyer, Y.E. Shlomovitz, L. Eli-Berchoer, A. Wilensky, I. Prinz and A. Hovav in Journal of Dental Research
Footnotes
Correction (August 2025):
Article updated online to correct the affiliations cited for A. Wilensky and I. Prinz.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Israel Cancer Research Foundation, the Israel Cancer Association through a donation by Billi and Bernie Marcus, the Samuel Badihi Foundation (for H.A.-H.), and the Deutsche Forschungsgemeinschaft (project 431861465 for I.P.).
Data Availability
The RNA sequencing data generated in this study will be deposited in the NCBI Gene Expression Omnibus GEO database. All data are available in the main text and the supplementary materials.
A supplemental appendix to this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
