Abstract
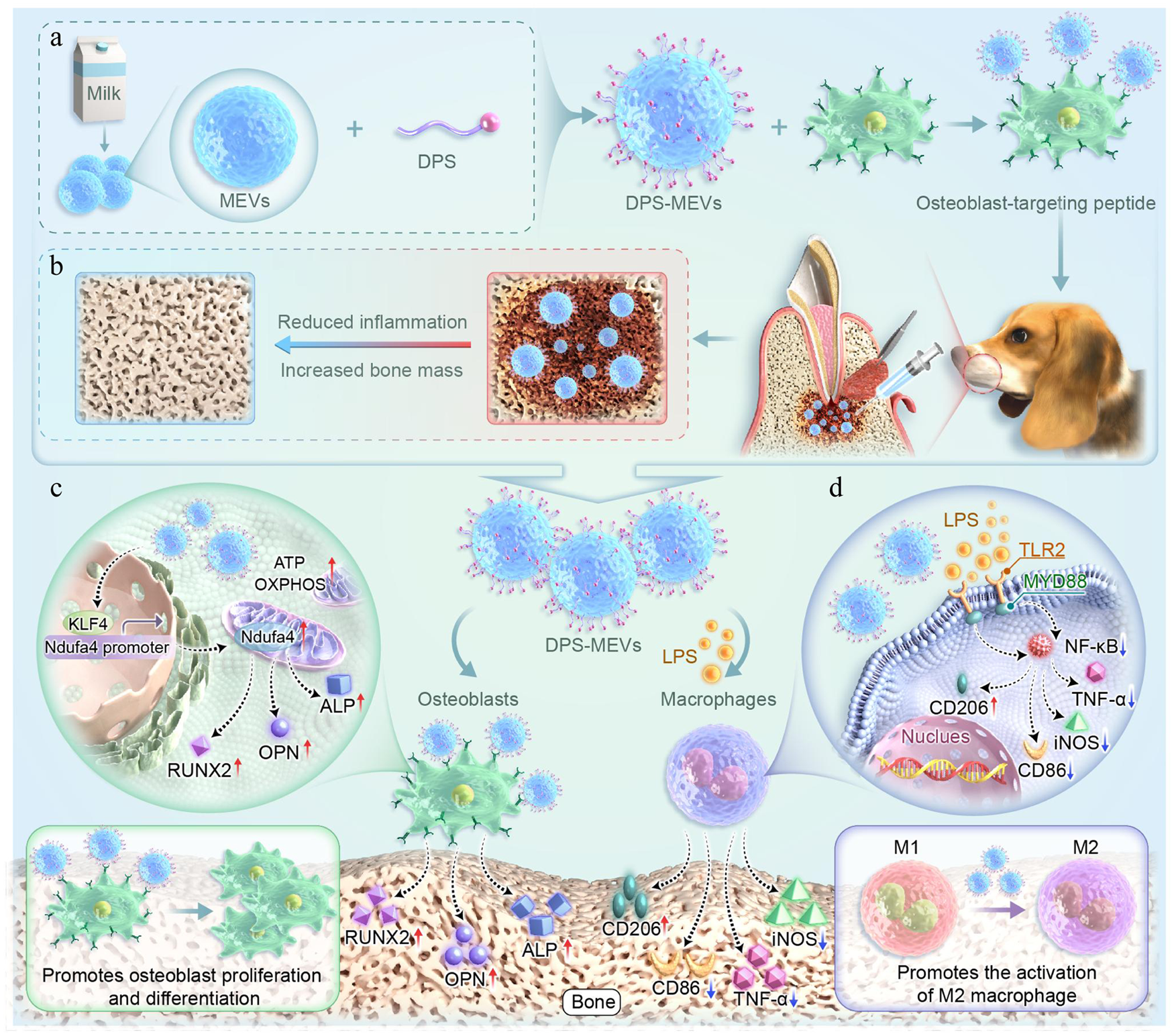
Chronic apical periodontitis (CAP), an inflammatory disease of periapical tissues, leads to alveolar bone destruction. Current therapies lack osteoblast-targeting specificity and fail to effectively promote bone repair. Extracellular vesicles (EVs), particularly milk-derived EVs (MEVs), show potential for bone regeneration and have anti-inflammatory effects. We investigated the therapeutic potential of engineered MEVs modified with the osteoblast-targeting peptide DSPE-PEG-Mal-Cys-SDSSD (DPS) in CAP. DPS-MEVs enhanced osteogenic capacity and exhibited greater osteoblast targeting compared with unmodified MEVs through oxidative phosphorylation (OXPHOS) activation, driven by Kruppel-like factor 4 (KLF4)–mediated upregulation of NADH dehydrogenase 1 alpha subcomplex 4 (Ndufa4) in vivo and in vitro. We also transplanted DPS-MEVs into a CAP model of dogs through apical microsurgery and found that DPS-MEVs enhanced bone repair and reduced inflammation by promoting polarization of M2 macrophages. These findings highlighted the potential of engineered DPS-MEVs as a dual-functional therapy for CAP, combining immunomodulation and tissue repair to advance precision treatment for inflammatory bone diseases.
Get full access to this article
View all access options for this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
