Abstract
We performed synchrotron-based micro-X-ray absorption near-edge spectroscopy (µ-XANES) coupled with micro-X-ray fluorescence (µ-XRF) for the identification of elements that included biometal zinc (Zn) and nonmetal sulfur (S) (and its species) in the condylar zones of a rat temporomandibular joint (TMJ). Zone-specific spatial localization of biometal Zn and nonmetal S from a materials viewpoint when correlated with hypoxia inducible factor-1α (HIF-1α) (a surrogate for tissue oxygenation) can provide insights into Zn-specific redox pathways at the vulnerable subchondral interface. Histologic localization of Zn, HIF-1α, and sulfur-rich proteoglycans (PGs) were mapped using an optical microscope. The µ-XRF maps coupled with site-specific micro-X-ray diffraction (µ-XRD) patterns were used to underline Zn-incorporated biological apatite in the subchondral bone and the bone of a rat TMJ condyle. Results demonstrated an association between Zn, PG, and HIF-1α histologic maps with µ-XRF, µ-XANES, and µ-XRD data and provided insights into plausible biological S species in Zn-enriched zones of a rat TMJ condyle. Spatially localized Zn and S underscore their roles in cell and tissue functions in a zone-specific manner. Elemental Zn with organic and inorganic S species at the cartilage–bone interface and the biomineral phase of Zn-enriched biological apatite from subchondral bone to condylar bone were ascertained using µ-XRF-XANES and µ-XRF-XRD. The coupled µ-XRF-XANES in situ complemented with µ-XRF-XRD in situ and immunohistochemistry provided valuable biological insights into zone-specific biological pathways in rat TMJ condyles. Based on these data, we present a workflow to reliably map and correlate S species within Zn-enriched regions of cartilage, bone, and their interface. We suggest the use of this correlative and complementary microspectroscopic spatial information for zone-specific localization of biometal Zn and nonmetal S to gain insights into plausible microanatomy-specific oxidative stress in the TMJ.
Introduction
Articular condyles of synovial joints contain multiple zones with different biochemical compositions (Bergholt et al. 2016; Camarero-Espinosa et al. 2016; Golebiowska and Nukavarapu 2022; Yu et al. 2023). The fibrous, proliferative, and hypertrophic zones within cartilage are delineated by morphologically distinct cells within an environment that contains sulfur (S)-rich macromolecules such as proteoglycans (PGs) and biometals such as zinc (Zn) complexed with other biomolecules (Kim et al. 2014; Malfait 2016; Mukherjee and Das 2024). The gradual changes in mineral densities from the softer unmineralized hypertrophic zone in cartilage through the mineralizing subchondral interface and into the harder mineralized bone is also a characteristic of the condyles. Spatial and temporal changes in the above-mentioned characteristics using preclinical animal models are constantly explored to investigate zone-specific tissue degeneration in condyles with mechanically induced osteoarthritis. The mutual affinity between Zn and S is well recognized in osteoarthritis (Maret 2004). However, there exists no information on S species in Zn-rich condylar zones that could provide valuable biological insights into plausible inception and progression of matrix degeneration in a condyle.
Disruption of the oxygen balance at the subchondral interface due to mechanical forces is known to encourage oxidative stress (Zhang et al. 2023), a hallmark of osteoarthritis. Zone-specific spatial associations between biometal Zn and nonmetal S from a materials viewpoint when correlated with hypoxia inducible factor-1α (HIF-1α) (a surrogate for tissue oxygenation) can provide insights into Zn-specific redox pathways (Perez Castejon et al. 1991; Lee et al. 2016; Huang et al. 2018) at the vulnerable subchondral interface. This is because redox pathways at the subchondral interface are regulated by the shifts in the partial pressure of oxygen between bone and the HIF-1α maintained cartilage (Zhang et al. 2023). However, no studies have addressed Zn colocalization with S-molecules/species in tissues under oxidative stress. For this reason, as a first step, the objective of this study is to demonstrate the use of correlative and complementary microspectroscopic techniques that leverage synchrotron radiation as a tool to identify zone-specific changes in S species in Zn-enriched condylar zones of a temporomandibular joint (TMJ).
Herein, we demonstrate a unique workflow that includes the use of microspectroscopy techniques on rat condylar tissues to identify the predominant organic and inorganic S species within Zn-enriched hypertrophic and subchondral interfacial zones and bone. The histology of Zn colocalized with HIF-1α is performed using optical microscopy. Micro-X-ray fluorescence (µ-XRF) imaging of Zn and S and micro-X-ray absorption near-edge structure (µ-XANES) spectroscopy, along with micro-X-ray diffraction (µ-XRD), enable the identification of zone-specific Zn and S species in the cartilage, subchondral interface, and bone. The S species identified using µ-XANES spectroscopy provide valuable biological insights into zone-specific reduced and oxidized species (Rompel et al. 1998; Hackett et al. 2016) in TMJ condylar zones. In addition, elemental Zn associations with inorganic species (e.g., phosphates and sulfates) at the cartilage-bone interface and condylar bone are ascertained by correlative µ-XRD and µ-XRF analysis in situ.
Materials and Methods
Correlative microspectroscopy and the complementary imaging-related workflow are illustrated in Figure 1. Using these correlative and complementary techniques with microspectroscopic spatial information will allow us to build associations between biometal Zn and nonmetal S.
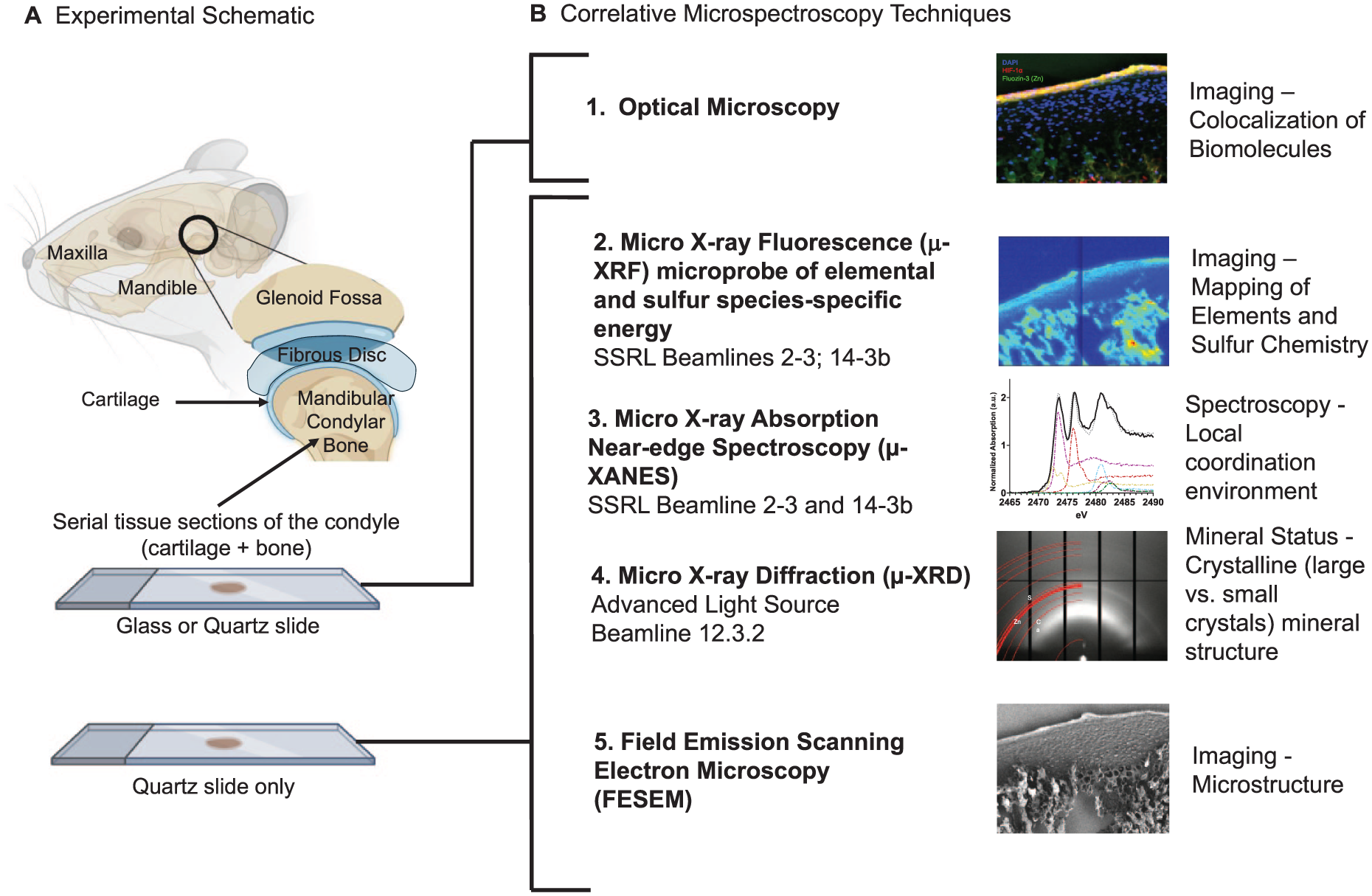
Experimental workflow. (
Specimens and Tissue Sections
Following euthanasia of 4-week-old Sprague-Dawley rats, the TMJ condyles were surgically extracted (IACUC protocol AN183106-03, UCSF; ARRIVE-Animal Research: Reporting of In Vivo Experiments). Fresh condyles (n = 3) were cryopreserved (−180 °C) and cryosectioned before the experiments. Another set of condyles (n = 3) was chemically fixed in 10% neutral-buffered formalin and paraffinized for histology.
Histology
The neutral buffered formalin–fixed specimens were embedded in paraffin, and 5-µm-thick sections were mounted on glass slides. Sections were deparaffinized and rehydrated, before staining with 0.1% Safranin-O for 5 min to examine the region specificity of PGs. Safranin O is a cationic dye that is sensitive to the polyanionic sulfated PGs (Rosenberg 1971). Counterstaining was achieved with 0.001% Fast Green for 5 min (Mino-Oka et al. 2017; He et al. 2022). Images were acquired using a brightfield microscope (AxioObserver2, Carl Zeiss Microscopy).
Immunohistology
Enzymatic epitope retrieval for immunohistochemistry was achieved by treating tissue sections with HistoReveal (ab103720, Abcam) at room temperature for 10 min. Blocking was performed with a commercial hydrogen peroxidase block (ab236469, Abcam) for 10 min and 1% bovine serum albumin (BSA)/phosphate-buffered saline for 5 min. The primary antibody for HIF-1α (100:1, Abcam) was diluted in 1% BSA/Tris-buffered saline and incubated under static conditions overnight at 4 °C. Further incubation with horseradish peroxidase conjugate occurred at room temperature for 15 min (ab236469, Abcam). The section was treated with a secondary antibody (Alexa Fluor 555, Abcam) at room temperature for 1 h, colocalized with Zn using 5 µm FluoZin™-3-tetrapotassium salt (F24194, ThermoFisher) stained at room temperature for 1 h, and counterstained with DAPI (D1306, ThermoFisher). Images were acquired using fluorescence microscopy (AxioObserver2, Carl Zeiss Microscopy). Zone-specific spatial associations between HIF-1α (a surrogate for tissue oxygenation) when correlated with biometal Zn and nonmetal S from a materials viewpoint can provide insights into Zn-specific redox pathways (Perez Castejon et al. 1991; Lee et al. 2016; Huang et al. 2018) at the vulnerable subchondral interface. For this reason, we continued to perform µ-XRF-XANES in situ on a serial tissue section but mounted on a quartz slide (Fig. 1).
µ-XRF-XANES In Situ
µ-XRF imaging
The cryopreserved specimens were cut to 10-µm-thick sections and mounted immediately on quartz slides and cryopreserved until ready for microspectroscopy. Imaging was performed within 24 h after cryosectioning. It should be noted that there is a known effect of time after tissue sectioning particularly on sulfur species (see Appendix). Elemental maps specific to Zn and Ca and to S and phosphorus (P) were collected using µ-XRF at the Stanford Synchrotron Radiation Lightsource (SSRL) at beamlines (BL) 2-3 and 14-3b in a helium environment. Both beamlines are equipped with SIGRAY axially symmetric microfocusing optics that provide beam diameters of 5 and 1 µm.
Spatial maps of Zn and Ca were generated at BL 2-3 using an incident energy beam of 10 keV and a step size of 5 × 5 µm. The incident beam energy was selected using a Si(111) monochromator. The fluorescence signal was detected using a 1-element Hitachi Vortex silicon drift detector. High-resolution elemental maps were acquired at regions of interest with a step size of 1 × 1 µm.
Spatial maps of elements P and S were generated at BL14-3b using an incident beam energy of 2,488 eV and a step size of 5 × 5 µm (incident beam energy selected using an Si(111) monochromator). Multienergy maps of S were acquired at 2,472.68, 2,473.27, 2,476.2, 2,482.4, and 2,482.7 eV to map glutathione disulfide, glutathione/methionine, sulfoxides, organic sulfates, and inorganic sulfates, respectively, using a 7-element Hitachi Vortex silicon drift detector. The monochromator was calibrated by setting the maximum of the first peak of a Na2S2O3 spectrum to 2,472.02 eV. Each pixel within the set of multienergy maps was fit as a linear combination of S XANES spectra for the S species listed above, providing spatially resolved maps of the S species. This procedure is described in Farfan et al., and data were analyzed using Sam’s Microprobe Analysis Kit (SMAK) (Webb 2011; Farfan et al. 2018).
µ-XANES
Principal component analysis and simplex volume maximization were applied to multienergy S µ-XRF maps to identify chemically distinct regions using SMAK (Webb 2011; Farfan et al. 2018), which helps to guide the selection of points of interest for µ-XANES spectroscopy (Appendix Fig. 2). Points of interest for S-spot µ-XANES were also selected by anatomical locations and regions with higher/lower S counts (Appendix Fig. 2). Sulfur spectra were acquired from 2,465 eV to 2,490 eV with a 0.2-eV step and a beam size of 5 × 5 µm at BL 14-3. Zn K-edge XANES spectroscopy was performed at points within regions of interest in tissues. Zn spectra were acquired from higher/lower Zn regions from energy levels 9,640 to 9,760 eV with a spot size of ~1 × 1 µm at BL 2-3. Data were background corrected and fit to in-house Zn and S standards using Sam’s Interface for XAS Package (SIXPack) (Webb 2005). The acquired XANES spectra were fitted to spectra in the European Synchrotron ID21 Database (Appendix Table 1). Finally, principal component analysis was performed to identify clustering of region-specific Zn and S species.
µ-XRF-XRD In Situ
Synchrotron 2-dimensional (2D) µ-XRD patterns were collected from Zn-enriched µ-XRF maps collected using BL12.3.2 of the Advanced Light Source (ALS) at Lawrence Berkeley National Laboratory. Lower-resolution Zn and Ca µ-XRF maps were used to locate regions of interest. X-ray diffraction patterns were measured using a monochromator with an energy of 10 keV, wavelength λ = 1.2398Å, and reflective geometry. The area detector (DECTRIS Pilatus 1M) was placed at an angle of 40° at 158 mm from the sample surface with an X-ray beam spot of 10 × 3 µm. Data were analyzed using in-house software XRDSol and fit to hydroxylapatite (HA) standards acquired from the MINCRYST database (Chichagov et al. 2001).
Field Emission Scanning Electron Microscopy
Following beamline spectroscopy and µ-XRD, the structure of the quartz-mounted section was mapped using field emission scanning electron microscopy (FESEM) (SIGMA VP500, Carl Zeiss Microscopy) at an incident electron energy of 1 keV.
Results
The biomolecular maps from the histology section were visually correlated with biochemical and elemental maps of the serial section from the same condyle (Fig. 1). The red asterisk (Fig. 2) illustrates a fiducial marker across all tissue sections used in this study.
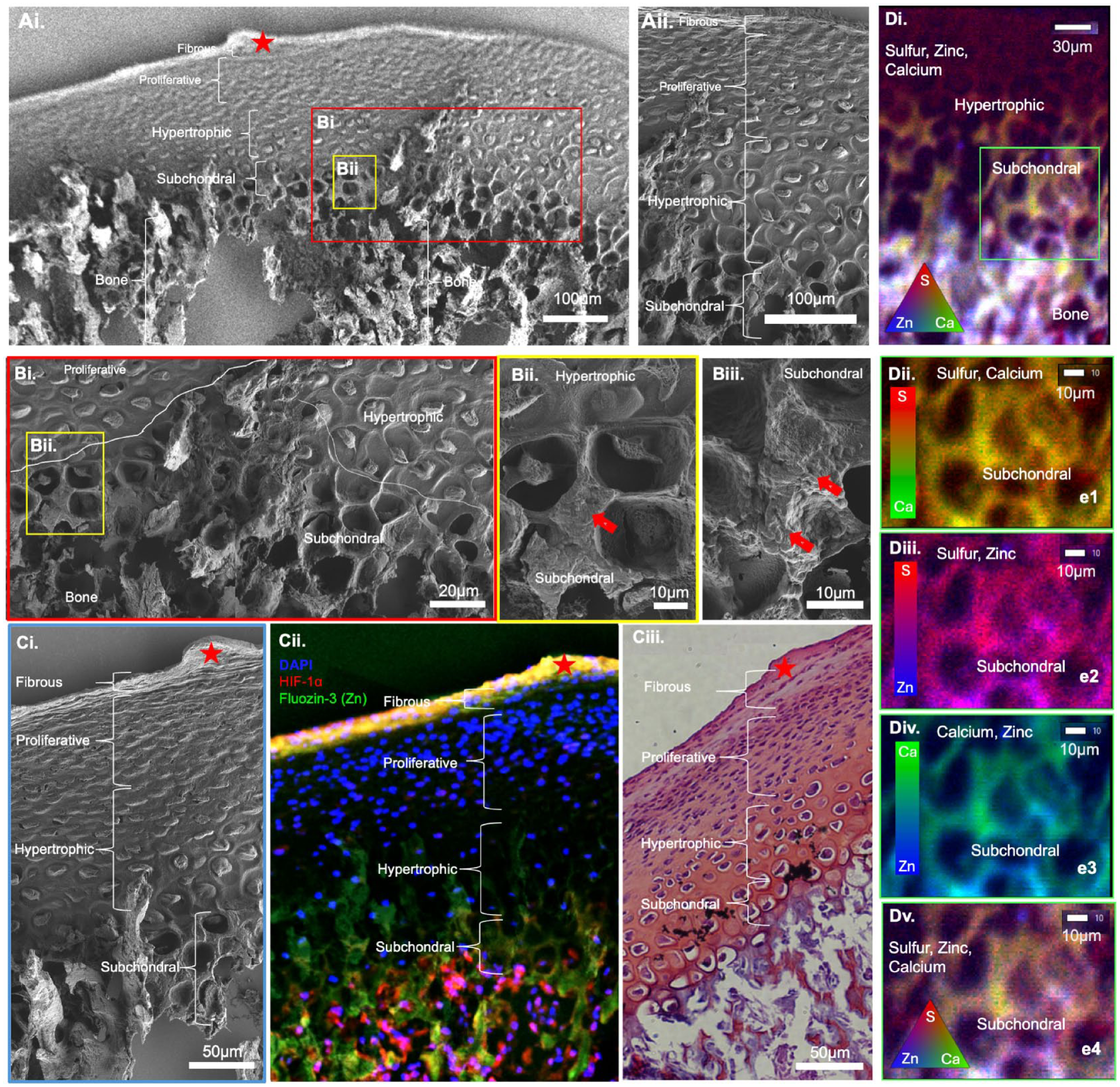
Zonal classification of cartilage, cartilage–bone interface, and bone. (
Structural, Histological, and Elemental Compartmentalization of Condylar Zones; Fibrous, Proliferative, Hypertrophic, Subchondral, and Bone
As mentioned, spatial associations between biometal Zn and nonmetal S from a materials viewpoint when correlated with HIF-1α can provide insights into zone-specific redox pathways (Perez Castejon et al. 1991; Lee et al. 2016; Huang et al. 2018). FESEM of the condylar zones (Fig. 2A) at higher magnification illustrated morphologically distinct cells at the HIF-1α–rich hypertrophic (Figs. 2Aii, 2Bi–Bii) and subchondral regions (Fig. 2Biii). Subchondral regions at a higher magnification were Fluozin-3 positive and exhibited measurable counts of Zn from the spherical nodules in the perilacunar walls (red block arrows in Fig. 2Bii, Biii). Zone-specific correlation between structure (Fig. 2Ci), colocalized Zn and HIF1-α (Fig. 2Cii), and visual correlation with Safranin-O (Fig. 2Ciii) indicated an association between Zn, HIF1-α, and PGs in the fibrous and subchondral zones. The biometal Zn and PGs in the hypertrophic zone were observed. No visual spatial association between Zn and PGs was apparent in the proliferative zone (Fig. 2Cii, Ciii). X-ray fluorescence signals representative of S, Zn, P, and Ca elements were colocalized in the bone and to a lesser extent in the subchondral bone (Fig. 2D; Appendix Fig. S1). Often, bone is characterized by the presence of Ca and P. Visual correlation between FluoZin™-3 identified Zn and immunolocalized HIF-1α with Zn, S, and Ca µ-XRF maps guided further examination of S species in the Zn-enriched hypertrophic and subchondral zones of cartilage and bone.
Sulfur Species in Condylar Zones
Individual spot S µ-XANES from cartilage, the subchondral–bone interface, and bone, as well as average sulfur XANES for cartilage (ii), bone (iii), interface (iv), and sulfur standards (v), are shown (Fig. 3; Appendix Fig. S2). The least-squares fitting of the multienergy µ-XRF maps (Farfan et al. 2018) (a1, Fig. 3A) revealed reduced and oxidized zone-specific sulfur species (glutathione [GSH], glutathione disulfide [GSSG], methionine, sulfoxides, organic sulfates, and inorganic sulfates) in a rat condyle (a3, Fig. 3A). Overlays of the S and P µ-XRF maps on the SEM micrographs illustrated the prevalence of nometal element S and its potential role in cartilage as well as P-rich mineralized regions in bone (b1, Fig. 3B). Results illustrated sulfur species and zone-specific colocalization, such as methionine (or GSH; mostly fibrous and in the proliferative zones) and its oxidized form methionine sulfoxide 2,475.9 eV (Rompel et al. 1998) (fibrous, proliferative, and in the hypertrophic zones) as well as organic sulfate (sulfate ester–chondroitin sulfated–PG at 2,482.7 eV) (Farfan et al. 2018) (includes subchondral zone) (a3. Fig. 3A).
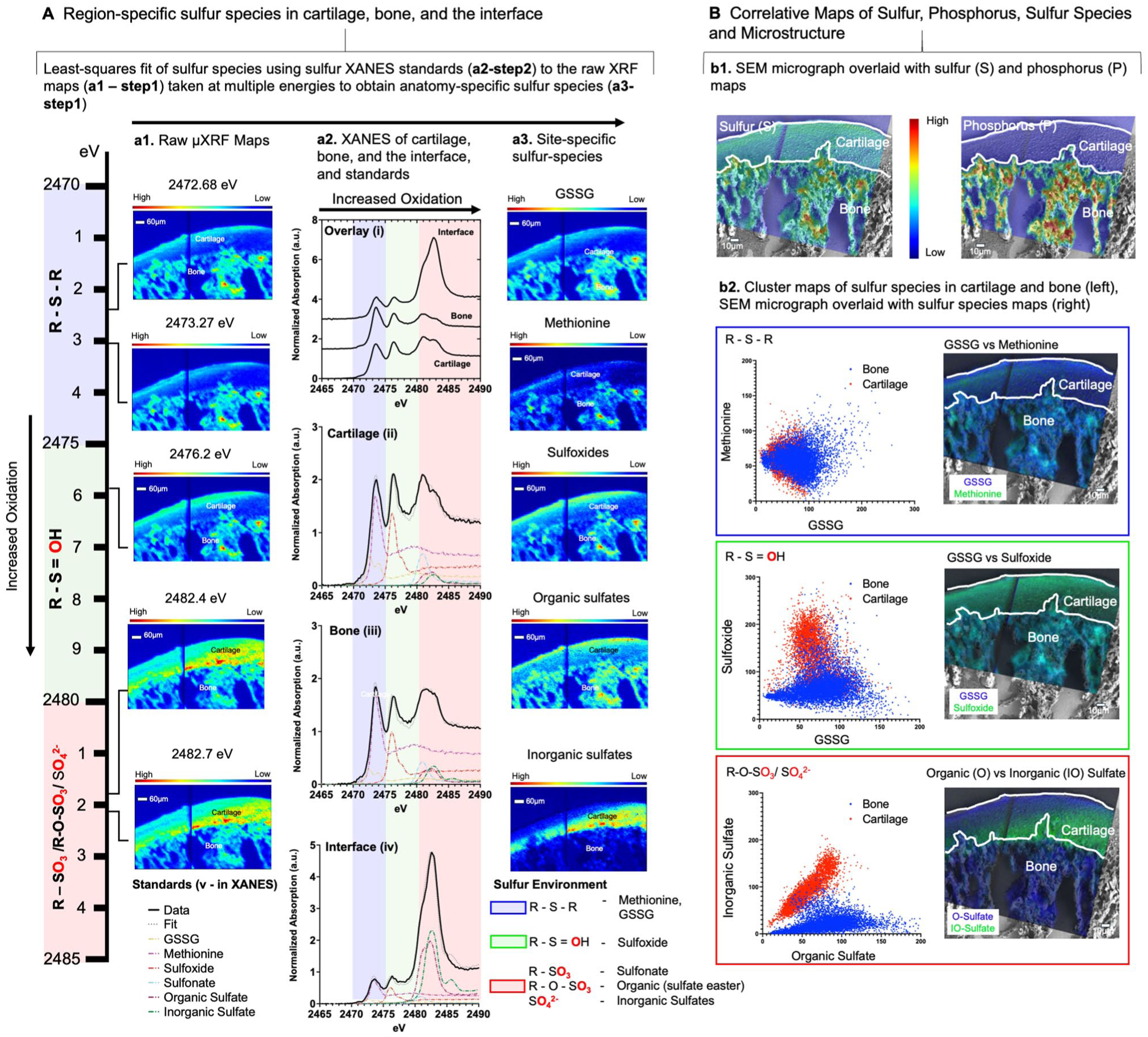
Overview of the methodology used to identify tissue-specific localization of sulfur species in cartilage and bone: (
An increased oxidation signal (left to right – blue to pink in the XANES spectra) was observed at the interface (iv) between bone (iii) and cartilage (ii) (a2, Fig. 3A). The organic sulfate identified by XANES (pink zone) could represent sulfated PGs in cartilage and bone (Farfan et al. 2018). From cluster maps, it is likely that the colocalization of GSSG with methionine and sulfoxide, respectively (b2 in Fig. 3B; Appendix Fig. S2), could indicate methionine sulfoxide as an additional potential species within the cartilage, bone, and the cartilage–bone interface. Based on the µ-XANES analysis, GSSG is a minor species compared with methionine (a2, Fig. 3A). As such, the ratio of methionine-to-methionine sulfoxide (2,473.1 and 2,476.4 eV) could act as a proxy of the GSH to GSSG (2,473.4 and 2,472.2/2,473.3 eV) ratio as a biomarker for oxidative stress (Denno et al. 1995; Gould et al. 2021).
Zn- and S-Species in Cartilage, Subchondral Bone, and Bone
Zn-positive regions in cartilage, bone, and the interface using the µ-XRF-XRD in situ at ALS 12.3.2 (Fig. 4A) were confirmed using the higher-resolution µ-XRF at SSRL, BL2.3 (Fig. 4A, ai–aii). Representative spectra of scan lines 1–4 (Fig. 4; Appendix Fig. S3) are plotted with integrated 1-dimensional (1D) µ-XRD pattern of HA (Ca5[PO4]3OH) (red line) standard. An integrated 1D µ-XRD pattern 2θ from 20° to 40° is shown (Fig. 4B, bii). The corresponding 2D µ-XRD pattern with HA is overlayed, and hkl:002 representative of HA is marked in the 2D X-ray diffraction pattern (Fig. 4B, bi). Lower Zn with Ca in subchondral region, and higher Zn with Ca in bone were observed (biii, Fig. 4B). It was interesting to note that the d-spacing and its variance were less for higher counts of Zn (high Zn; Fig. 4B, biv) compared with an increased d-spacing and its variance for lower counts of Zn (low Zn; Fig. 4B, biv), which may suggest the presence of Zn in bone. The Zn-enriched subchondral interface contained inorganic sulfates (Figs. 4 and 5). Cluster analysis illustrated that zinc sulfate in cartilage closer to the subchondral bone clustered with R-S-R and R-S=O-R (GSH, GSSG, methionine, sulfoxide), zinc phosphate in the subchondral interface clustered with R-SO3/O4, whereas zinc sulfate and HA in bone clustered with R-S-R and R-S=O-R sulfur species (Fig. 5).
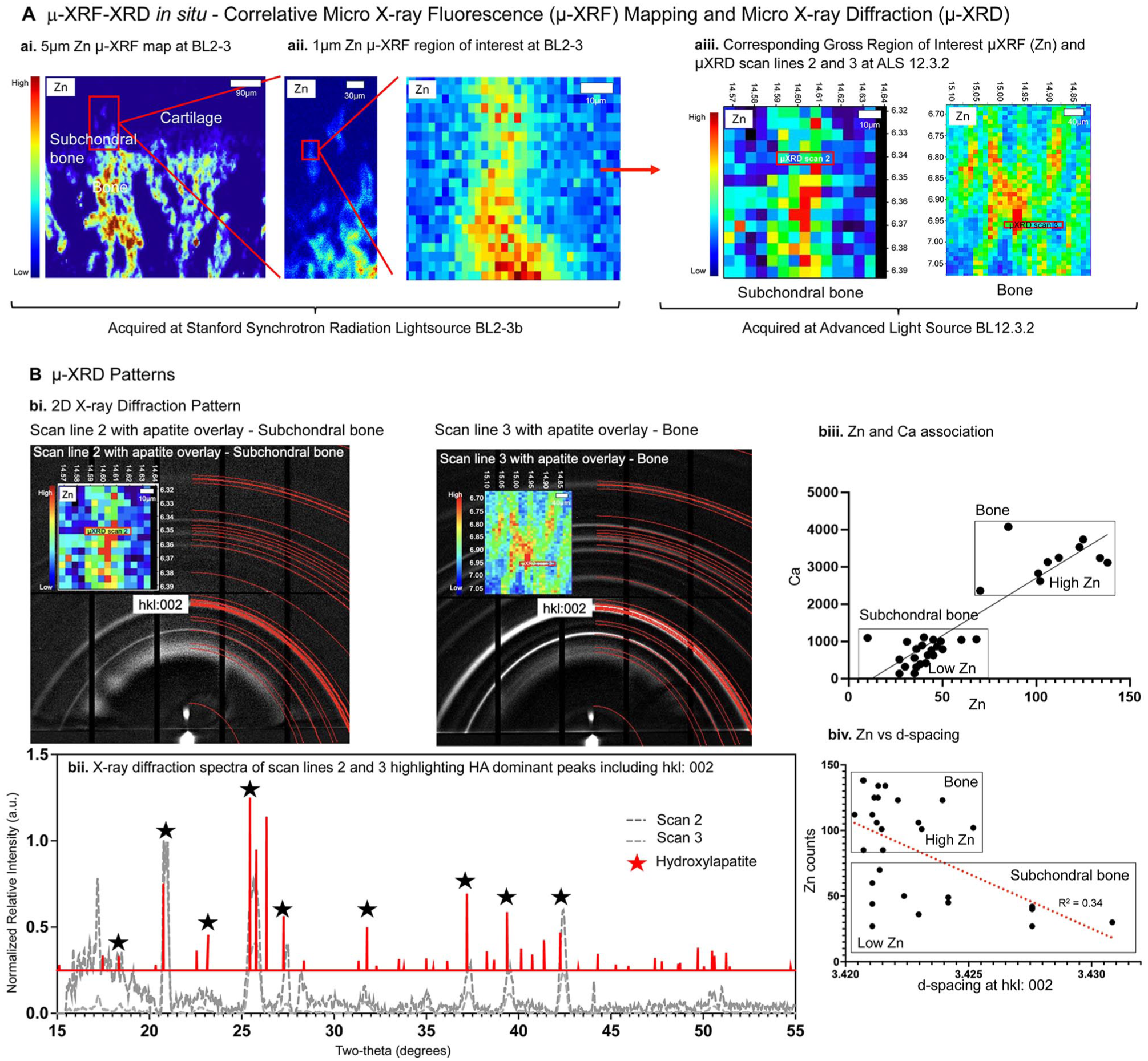
Characterization of mineral at the subchondral interface reveals differences in crystallinity and crystal size that are dependent on zinc counts. (
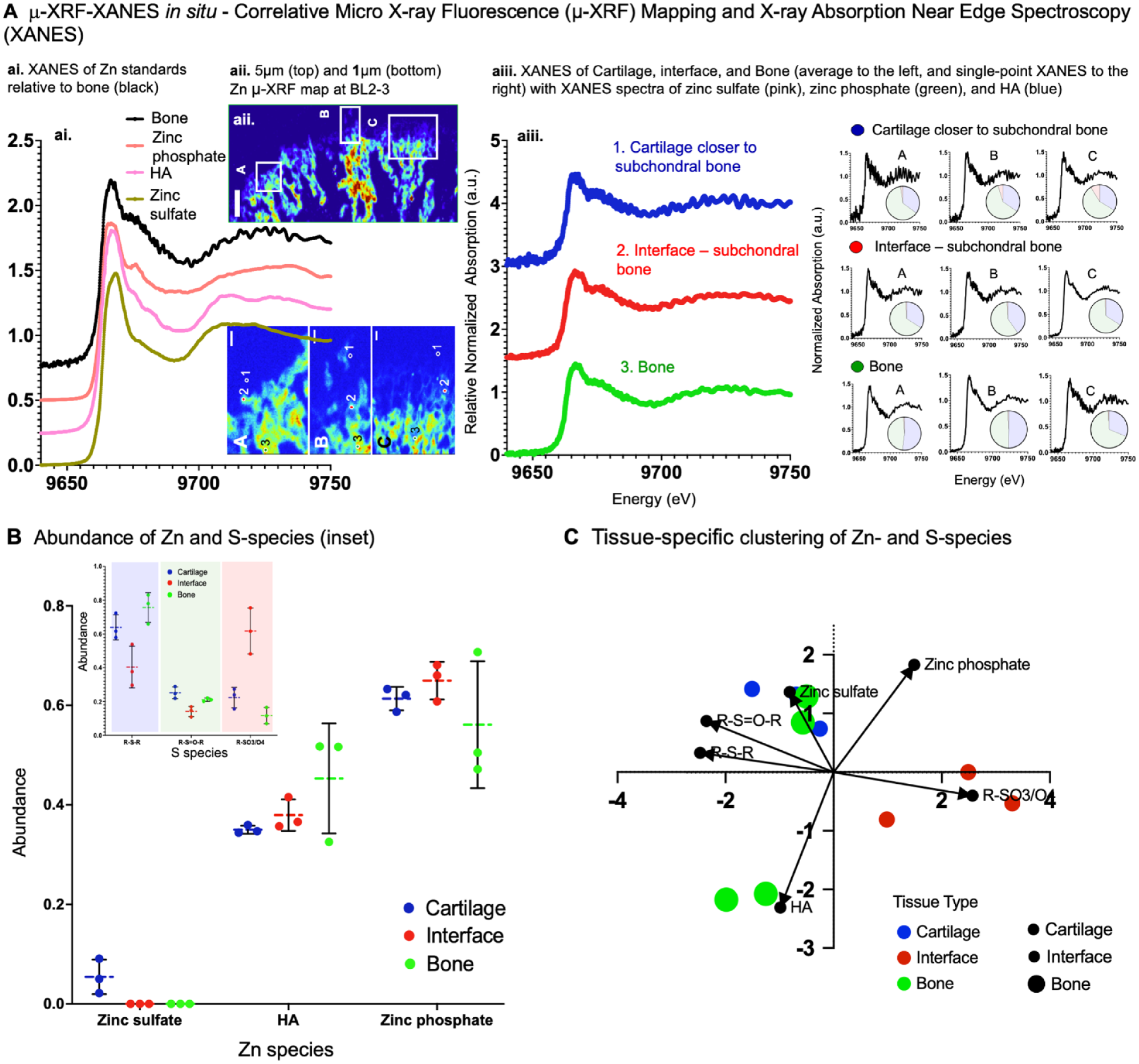
Zinc and sulfur species in cartilage closer to the subchondral interface and bone. (
Discussion
The individual roles of biometal zinc (Zn) and nonmetal sulfur (S) in tissues and their collective partnership in multiple biological pathways continue to be discussed (Maret 2004; Maret 2019; Krezel and Maret 2021). This study approach (Fig. 1) demonstrated the need to correlate and complement microspectroscopic techniques to identify region-specific tissue chemistry in situ that otherwise would be missed using a single technique.
Microspectroscopy in situ using SSRL BLs 14-3 and 2-3 is able to distinguish S species in lower and higher Zn regions of the same condyle tissue section followed by visual association of the same regions with zone-specific HIF-1α expressions. Visual correlation of spatially immunolocalized biomolecules on the first tissue section to site-specific µ-XANES, µ-XRF, and µ-XRD data on a serial tissue section confirmed Zn with hypoxia factor (HIF-1α) in (1) a fibrous matrix in the articular zone with cells (Figs. 2, 3) and organic sulfates, including reduced (GSH/methionine) and oxidized (GSSG/sulfoxides) forms (Mathews and Lozaityte 1958; Gross et al. 1960; Mathews 1962) and (2) hypertrophic and subchondral cells with organic and inorganic sulfates and their potential influence on mineralization kinetics (Tao et al. 2023) (Figs. 2, 3).
The function of most biomolecules is governed by their metal centers, and noting these metals and their associated species will provide insights into biomolecules that could play a role in inciting redox pathways. More than 3,000 biomolecules need Zn to function (Frangos and Maret 2020). The Fluozin-3 fluorophore and Safranin-O chromophore permitted the localization of available biometal Zn in a matrix rich in sulfated polyanionic macromolecules, the PGs. The noted Zn and HIF-1α in the fibrous and the hypertrophic and subchondral regions are positive regulators of collagen and sulfated PGs (Fig. 2C). The biometal Zn and HIF-1α are known to promote chondrocyte differentiation and matrix synthesis necessary for health and function of the cells in the fibrous region and hypertrophic cells in proximity with osteoblasts and osteoclasts in subchondral bone (Rest 1976; Rossi et al. 2001; Lee et al. 2016; Fan et al. 2023). This implies that an acute or chronic disruption to HIF-1α through mechanical stress/injury can incite a biochemical cascade and could result in degeneration of cartilage, bone, and the interface and not necessarily in that order (Zhang et al. 2023). The ensuing tissue chemistry can then be explored to help explain heterogenous biomineralization in the subchondral region between cartilage and bone (see Appendix).
The PG function in cartilage is generally regulated by sulfation. Standard histologic stained regions by Safranin O, a cationic dye that is sensitive to the polyanionic sulfated PGs (Rosenberg 1971), are comparable to the sulfur signal in condylar zones using µ-XRF (Fig. 2). Spectroscopically observed higher sulfate esters possibly from sulfated PGs and methionine from the extracellular matrix of cartilage (Figs. 2 and 3 could be the contributors to the observed organic sulfate µ-XRF signal in the Zn and HIF-1α–rich articular cartilage zone (Figs. 2 and 3). Spectroscopically observed lower sulfation (sulfate esters) and methionine and higher levels of inorganic sulfates (Fig. 3A, B; Appendix Fig. S2; TS2) could be S species in the Zn and HIF-rich hypertrophic and subchondral zones (Fig. 2Cii) (see Appendix).
From a biomechanics standpoint, studies to map the direct influence of mechanical stimuli on Zn-related redox chemistry independent of oxygen tension are warranted. Regardless, sulfation in the cartilage matrix is directly proportional to bound water and determines the compression resistance (Hackett et al. 2016) (see Appendix). The proliferative cells in the mid-zone (Fig. 2Ci-iii) regulate chondrocyte hypertrophy (Wang et al. 2002). Hypertrophic chondrocytes promote changes in the surrounding tissue, undergo apoptosis, and increase Zn-enriched minerals and bone replacement. The observed nodular aggregation reinforcing the organic perilacunar walls within the Zn-enriched region (Fig. 2Bii, 2Biii) was akin to matrix vesicle-mediated endochondral ossification observed in our previous studies (Wang et al. 2015). An aggregate of inorganic sulfates in the subchondral region (Fig. 2A, B) could act as a chelator of metal ions and, in this study, possibly of Zn (Krezel and Maret 2016). Multimodal microspectroscopy illustrated the presence of organic and inorganic sulfates in the presence of Zn in cartilage and the subchondral bone. The observed gradient in the colocalization of S, Zn, and Ca from subchondral bone to bone (Fig. 2D; Appendix Fig. S1), provided insights into the Zn-enriched species (Fig. 5) including the commonly known apatite.
We identified a nuanced signal highlighting the multiple roles of biometal Zn in the fibrous and subchondral interface with sulfur species for the first time by performing microspectroscopy in situ. Following cluster analysis, the Zn-enriched subchondral interface contained inorganic sulfates (Figs. 4, 5). In bone, the observed organic sulfates included sulfate ester, methionine, and inorganic sulfates. It is postulated that these Zn-associated S species could be the zone-specific precursors and tightly regulate mineral precipitation. Although the diffraction pattern was dominated by an hkl index of the 002-lattice plane of calcium-based apatite, it is likely that other crystalline minerals at lower concentrations could also exist. With different anionic/cationic substitutions and vacancies generally expressed in biological apatite, a variance in d-lattice spacing with varying Zn counts (Fig. 4) should be no surprise (Procopio et al. 2019a, 2019b; Dornelas et al. 2024).
In summary, the coupled µ-XRF-XANES and µ-XRF-XRD in situ informed us of the potential roles of biometal Zn in an S-rich environment of cartilage zones in situ (Fig. 5). The sulfur species in the Zn-enriched regions of the cartilage, subchondral bone interface, and bone can be identified to underpin the oxidative risk in a zone-specific manner in a biomechanically active TMJ condyle using a high-resolution microspectroscopy technique.
Author Contributions
B. Lee, contributed to design, data acquisition, analysis, and interpretation, critically revised the manuscript; Z. Yang, Y. Wang, contributed to data acquisition, critically revised the manuscript; J. Levy, contributed to data analysis, critically revised the manuscript; N. Tamura, S. Webb, S. Bone, contributed to design, data acquisition, analysis, and interpretation, critically revised the manuscript; S.P. Ho, contributed to conception, design, data analysis and interpretation, drafted and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of the work.
Supplemental Material
sj-docx-1-jdr-10.1177_00220345251361124 – Supplemental material for Sulfur Species in Zinc-Rich Condylar Zones of a Rat Temporomandibular Joint
Supplemental material, sj-docx-1-jdr-10.1177_00220345251361124 for Sulfur Species in Zinc-Rich Condylar Zones of a Rat Temporomandibular Joint by B.H. Lee, Z. Yang, Y. Wang, J. Levy, N. Tamura, S. Webb, S. Bone and S.P. Ho in Journal of Dental Research
Footnotes
Acknowledgements
This research used beamline 12.3.2 of the Advanced Light Source, a DOE Office of Science User Facility under contract No. DE-AC02-05CH11231. The use of the Stanford Synchrotron Radiation Lightsource (SSRL), SLAC National Accelerator Laboratory, is supported by the U.S. Department of Energy, Office of Science, Office of Basic Energy Sciences under contract No. DE-AC02-76SF00515. The SSRL Structural Molecular Biology Program is supported by the DOE Office of Biological and Environmental Research and by the National Institutes of Health, National Institute of General Medical Sciences (P30GM133894). Beamline 12.3.2 of the Advanced Light Source, a DOE Office of Science User Facility under contract No. DE-AC02-05CH11231 and is supported in part by the ALS-ENABLE program funded by the National Institutes of Health, National Institute of General Medical Sciences, grant P30 GM124169-01. The contents of this publication are solely the responsibility of the authors and do not necessarily represent the official views of the National Institute of General Medical Sciences or the National Institutes of Health. The image of the rat TMJ was created with BioRender.com.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by NIH/NIDCR R01 DE022032 (SPH).
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
A supplemental appendix to this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
