Abstract
Biomimetic mineralization of dentin collagen appears to be a promising strategy to optimize dentin bonding durability. However, traditional postbonding mineralization strategies based on Ca/P ion release still have some drawbacks, such as being time-consuming, having a spatiotemporal mismatch, and having limited intrafibrillar minerals. To tackle these problems, a prebonding rapid intrafibrillar mineralization strategy was developed in the present study. Specifically, polyacrylic acid–stabilized amorphous calcium fluoride (PAA-ACF) was found to induce rapid intrafibrillar mineralization of the single-layer collagen model and dentin collagen at just 1 min and 10 min, as identified by transmission electron microscopy, scanning electron microscopy, and atomic force microscopy. This strategy has also been identified to strengthen the mechanical properties of demineralized dentin within a clinically acceptable timeframe. Significantly, the bonding strength of the PAA-ACF–treated groups outperformed the control group irrespective of aging modes. In addition, the endogenous matrix metalloproteinases as well as exogenous bacterial erosion were inhibited, thus reducing the degradation of dentin collagen. High-quality integration of the hybrid layer and the underlying dentin was also demonstrated. On the basis of the present results, the concept of “prebonding rapid intrafibrillar mineralization” was proposed. This user-friendly scheme introduced PAA-ACF–based intrafibrillar mineralization into dentin bonding for the first time. As multifunctional primers, PAA-ACF precursors have the potential to shed new light on prolonging the service life of adhesive restorations, with promising significance.
Introduction
Due to the great progress of dental materials and growing esthetic demands of the public, adhesive restorations are becoming increasingly popular. The stability of adhesive–dentin interface determines the service life of restorations to a large extent. The hybrid layer based on the micromechanical interlocking between adhesive monomers and dentin collagen forms the structural foundation of the bonding interface (Breschi et al. 2018). Nevertheless, current etch-and-rinse (ER) adhesive systems cannot present a well-integrated hybrid layer. Specifically, insufficient infiltration of solvated adhesive monomers always leads to the appearance of exposed collagen, which is susceptible to hydrolytic degradation, matrix metalloproteinase (MMP) attack, and bacterial invasion (Stoyanov et al. 2010). As a result, the exploration of appropriate tactics to strengthen dentin collagen has gained prominence.
Currently, researchers have proposed various collagen enhancement strategies, such as cross-linking agents, plant extracts, enzyme inhibitors, and antibacterial agents (Moussa et al. 2019; Peng et al. 2020; Cai et al. 2021). Among these, biomimetic mineralization of dentin collagen seems to be promising because of its biomimetic repair concept. The most common method is “postbonding mineralization.” This means that the dental adhesive is usually modified with amorphous calcium phosphate (ACP) nanoparticles, and then the slowly released Ca/P ions supposedly act as seed crystals to facilitate remineralization at the hybrid layer, leading to acceptable bonding durability (Tao et al. 2020; Cai and Wang 2022; Wu et al. 2022). Although the results are exciting, the disadvantages of postbonding mineralization for the adhesive–dentin interface cannot be ignored. First of all, the required mineralization time is an enormous challenge, since enzymatic or bacterial attacks may occur immediately, and the exposed dentin collagen cannot receive timely protection before time-consuming remineralization (Gungormus and Tulumbaci 2021). Second, the appearance of remineralization is always accompanied by the dissolution of ACP nanoparticles, and whether this would create new weak areas in the hybrid layer is yet to be answered (Yang et al. 2017). To overcome these problems, a rapid intrafibrillar mineralization strategy employed before the application of adhesives (determined as “prebonding mineralization”) would be highly desirable to obtain a more structurally intact hybrid layer.
Based on the nonclassical crystallization theory, polymer-induced liquid precursors (PILPs) have been developed to explain intrafibrillar mineralization of collagen, in which amorphous minerals could be stabilized by biomimetic analogues of noncollagenous proteins (NCPs), such as polyaspartic acid (PAsp), polyacrylic acid (PAA), and poly(allylamine hydrochloride) (PAH) (Niu et al. 2017; Yao et al. 2019; Ping et al. 2022). According to previous studies, ACP with fluidic characteristics may penetrate into the gap regions of collagen first, then transform into needle-like hydroxyapatite (HAp) and later develop the 2-dimensional shape (Niu et al. 2017; Xu et al. 2020). However, in classical PILP process, intrafibrillar mineralization generally takes approximately 6 h to 3 d (Shao et al. 2018; He et al. 2020; Qu et al. 2020), but dentists only have an operation window of no more than 10 min in clinical practice. That is, only by developing a faster mineralization system can the prebonding mineralization strategy be feasible in real clinical application scenarios. Recently, Fang et al.’s studies on the rapid mineralization of collagen with amorphous calcium fluoride (ACF) shortened the mineralization time to 2 h (Fang, Ping, Huang, et al. 2021; Fang, Ping, Wagermaier, et al. 2021). Considering fluoride preparations as effective antibacterial agents, ACF potentially allows for rapid biological tissue mineralization as well as interfacial antibiosis. However, no related reports are available about rapid intrafibrillar mineralization mediated by ACF in dentin bonding.
Therefore, we put forward the concept of “prebonding rapid intrafibrillar mineralization” and adapted polyacrylic acid–stabilized amorphous calcium fluoride (PAA-ACF) as a dentin bonding primer in the present study. This study specifically verified the rapid intrafibrillar mineralization ability of PAA-ACF on demineralized dentin in a short time (within 1 min and 10 min) and further evaluated the impacts of this pretreatment on the physicochemical properties of the dentin surface, as well as the adhesive–dentin bonding stability. The null hypotheses of the study are as follows: pretreatment with PAA-ACF for 1 min and 10 min would have no effects on 1) rapid mineralization of the single-layer collagen and dentin collagen, 2) chemical and mechanical behaviors of dentin, 3) the immediate and durable bond strength, 4) MMP activity at the adhesive–dentin interface, and 5) the antibacterial activity of dentin.
Materials and Methods
Details of materials and methods are provided in the Appendix.
Results
Characteristics of PAA-ACF Precursors and Mineralization of the Single-Layer Collagen
Cryogenic transmission electron microscopy (Cryo-TEM) results showed that PAA-ACF precursors with a granular appearance and diameters of approximately 50 to 70 nm immediately aggregated into clusters (Fig. 1A). The amorphous phase was confirmed by selected area electron diffraction (SAED, inset in Fig. 1A). After 7 d, the nanoparticles aggregated into larger structures, and the SAED pattern revealed that the ACF precursors could be stabilized by PAA for a relatively longer time, although some crystalline CaF2 formed.
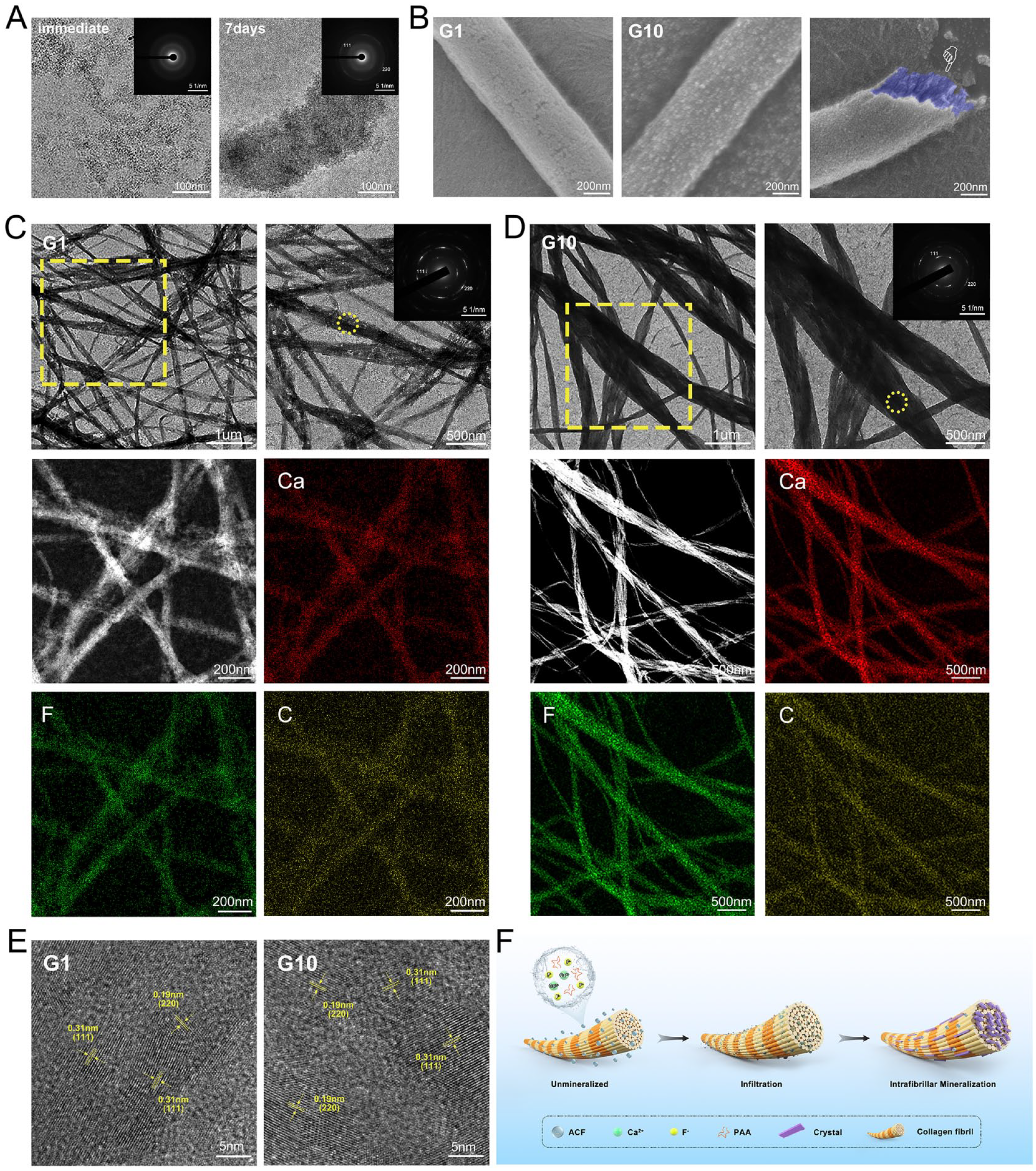
Rapid mineralization of reconstituted single-layer collagen fibrils by polyacrylic acid–stabilized amorphous calcium fluoride (PAA-ACF) for 1 min and 10 min (G1 and G10). (
Reconstituted single-layer collagen was used to achieve intrafibrillar mineralization at 1 min and 10 min, as observed by scanning electron microscopy (SEM) (Fig. 1B). Mineralized collagen fibrils at 1 min were broken to create the cross-section area, painted in blue, displaying the internal nanocrystals oriented along the long axis of collagen fibrils. Dense nanoparticles deposited on the surface, and the interior of collagen was filled and reinforced by regularly shaped crystals.
Transmission electron microscopy (TEM) images illustrated similar results (Fig. 1C, D). The (111) and (220) crystal planes in the SAED patterns indicated that the internal minerals were aligned CaF2. Fully mineralized collagen fibrils were observed at 10 min since no further mineralization appeared from then on, as shown in Appendix Figure 1. Energy-dispersive X-ray spectroscopy (EDS) mapping demonstrated the integration and uniform distribution of calcium and fluoride elements within collagen fibrils (Fig. 1C, D). The lattice fringe spacings of 0.19 nm and 0.31 nm in the high-resolution transmission electron microscopy (HRTEM) images were aligned with the (111) and (220) crystal planes of CaF2 (Fig. 1E). The schematic in Figure 1F reveals the possible intrafibrillar mineralization mechanisms mediated by PAA-ACF, as reported by previous studies (Gower 2008; Xu, Tijssen, et al. 2018; Xu et al. 2020).
Mineralization of Dentin
The SEM images in Figure 2A depict that demineralized dentin from the phosphate acid–etched group (PA group) was devoid of intrafibrillar and extrafibrillar minerals, with patent dentinal tubules and a porous collagen network. The typical periodic structures of exposed collagen fibrils could be observed at high magnification. EDS mapping illustrated scarce calcium content and nearly no fluoride in peritubular dentin. After mineralization by PAA-ACF for 1 min, CaF2 crystals grew and distributed densely on the dentin collagen network, resulting in a rugged surface morphology. For Group 10 min (G10), more minerals could be observed on the collagen matrix. At high magnification, the collagen fibrils were completely wrapped and supported by crystals, separated by finer extrafibrillar spaces. The enhanced calcium and fluoride signal in EDS mapping also confirmed crystal growth in the peritubular dentin collagen network. Similarly, the intrafibrillar mineralization of demineralized dentin is also shown in TEM images (Appendix Fig. 2).
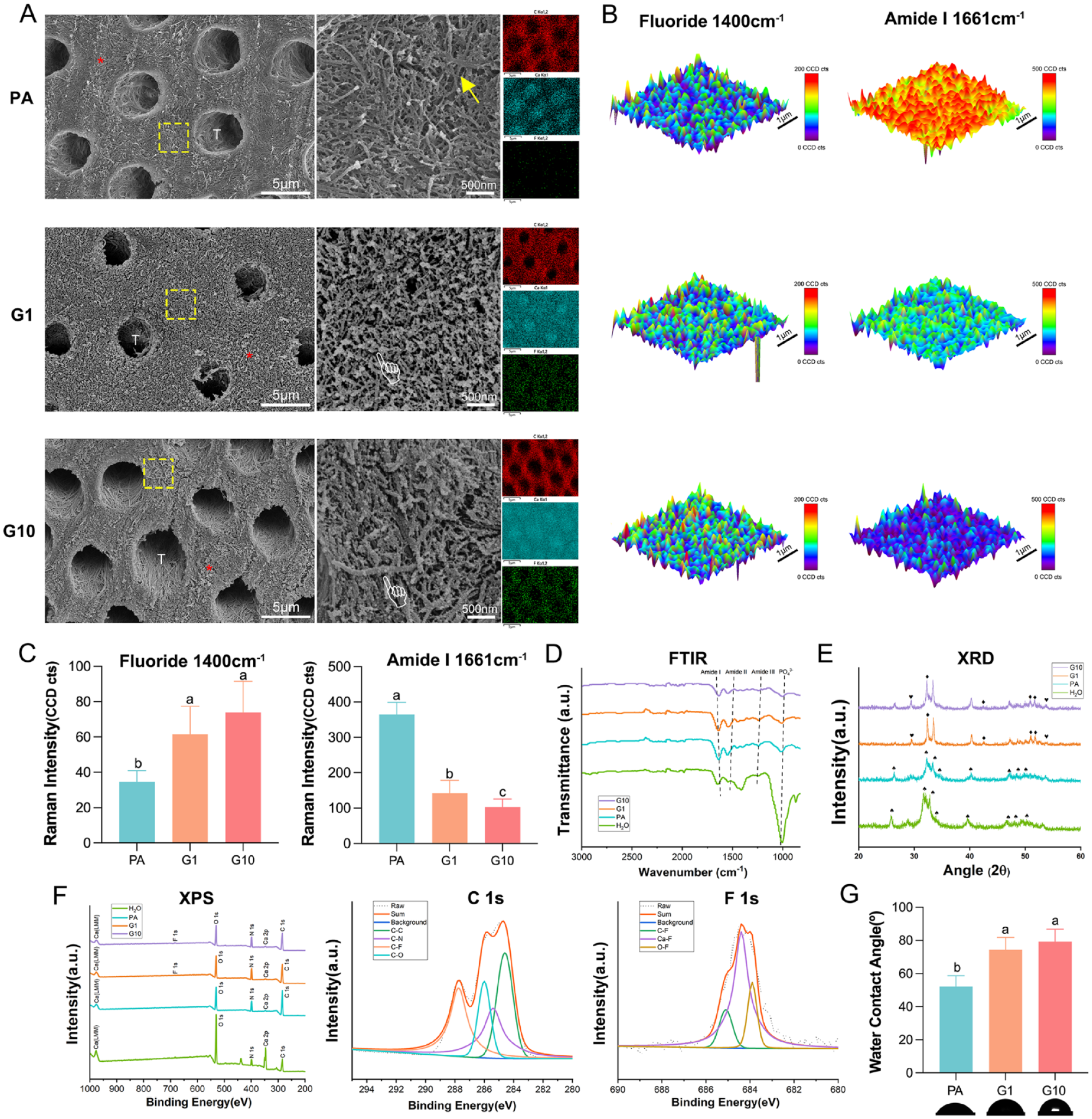
Characterization of the differently conditioned dentin. (
Three-dimensional Raman mapping of fluoride and amide I is illustrated in Figure 2B. The evidence of CaF2 crystals deposited into dentin collagen and the organic groups of protein were represented by the fluoride peak at 1,400 cm−1 and the amide I peak at 1,661 cm−1, respectively. Compared with demineralized dentin, fluoride mapping of Group 1 min (G1)and G10 displayed a larger area of high intensity, while the intensity of amide I mapping was generally weak, which was particularly evident in G10. The quantitative calculation is shown in Figure 2C (
For Fourier transform infrared (FTIR) spectra, the phosphate peak at 1,010 cm−1 and amide I, II, and III peaks at 1,643 cm−1, 1,550 cm−1, and 1,238 cm−1 corresponded to inorganic minerals and organic proteins of dentin, respectively (Fig. 2D). Compared with natural dentin, the PA group showed a significant decrease in the phosphate peak intensity but an increase in the amide peak intensity, ascribed to the exposure of organic groups on the surface of demineralized dentin collagen. However, the intensity of the amide peaks of G10 and G1 decreased, attributed to the integration of collagen and CaF2 minerals. As shown in Figure 2E, the (002), (211) and (300) peaks of natural dentin (group H2O) in X-ray diffraction (XRD) spectra indicated a strong signal of HAp, which decreased sharply after acid treatment, related to mineral loss. For remineralized dentin, peaks (111) and (220) represented the CaF2 nanocrystals. Peaks (211), (310) and (213) of fluorapatite were also found.
In the full spectrum of X-ray photoelectron spectroscopy (XPS), peaks of calcium and fluoride elements were observed in G1 and G10, with G10 increasing more obviously (Fig. 2F). In the PA group, the peaks of calcium and fluoride were extremely low. The main peak of the high-resolution deconvoluted spectra of F 1s from G1 and G10 represented the ionic bond between fluoride and calcium; the 2 secondary peaks were attributed to the covalent bonds O–F and C–F. Thus, the existence of chemical bonds between CaF2 crystals and dentin collagen molecules was speculated. The 4 deconvoluted peaks in the C 1s spectrum corresponded to C–C, C–N, C–F, and C–O bonds.
Figure 2G shows the contact angles of water on dentin surfaces and the corresponding views. Water contact angles significantly increased in G1 and G10 compared to PA, suggesting improved hydrophobicity (
Adhesive–Dentin Interface
mTBS test
Two-way analysis of variance (ANOVA) illustrated that the bond strength was affected by both dentin pretreatment (
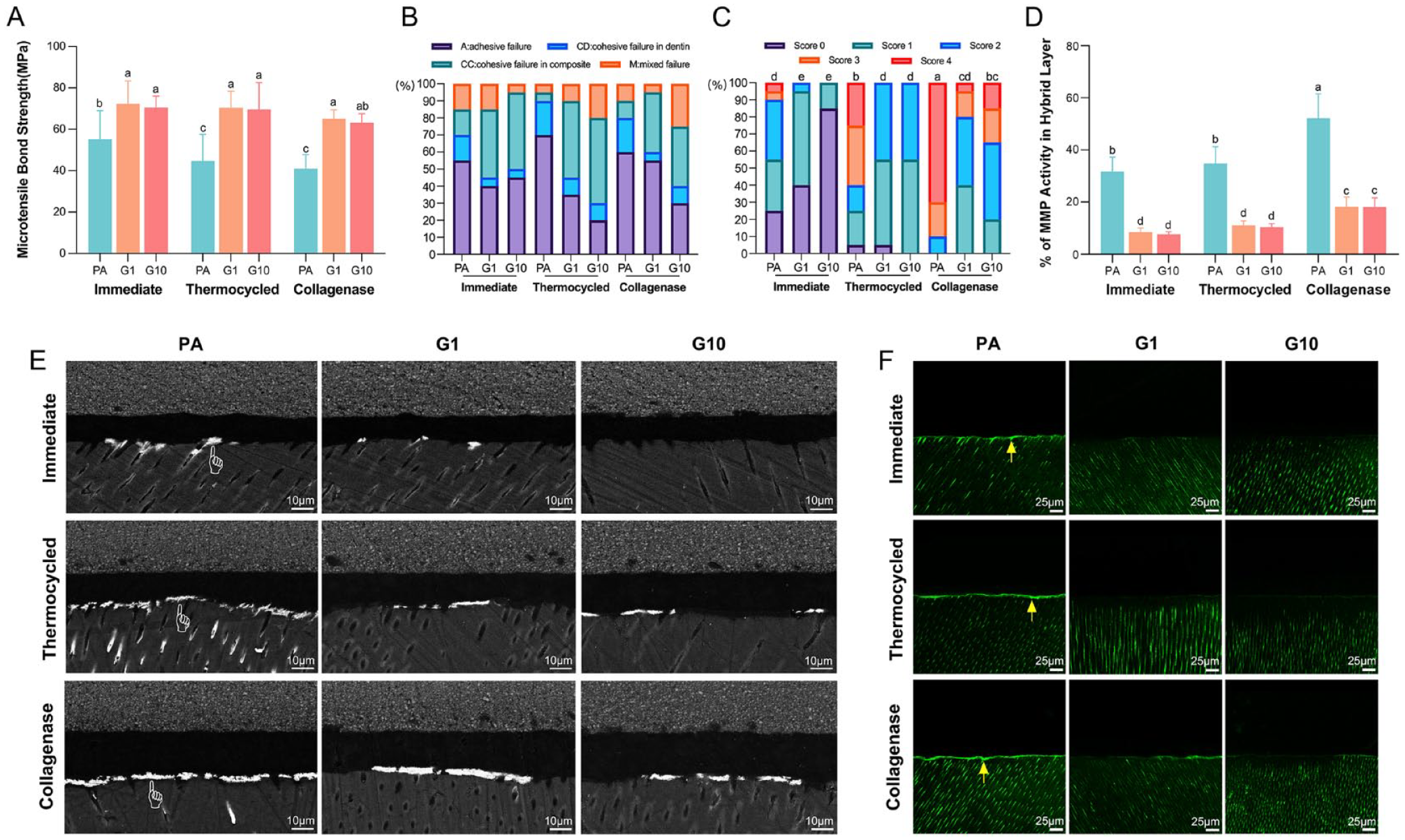
Bonding performance of adhesive–dentin interface. (
Failure mode analysis
The frequency distribution and representative images of fracture patterns are shown in Figure 3B and Appendix Figure 3, respectively. Regardless of aging modes, cohesive failure in dentin (CD) in G1 and G10 exhibited a further downward trend compared with the PA group. By contrast, cohesive failure in the composite (CC) accounted for a particularly higher percentage in G1 and G10.
Nanoleakage evaluation
The score distribution of interface nanoleakage is presented in Figure 3C. Both the conditioner and aging method obviously influenced the statistical results of nanoleakage evaluation (
In situ zymography
Stronger fluorescein-conjugated gelatin activity was observed in the PA groups than in G1 and G10, regardless of aging challenges. Continuous green fluorescence was shown along the hybrid layer in the PA groups after aging, while almost no increase was detected in G1 and G10 (Fig. 3F). In addition, quantitative calculation in Figure 3D demonstrates the evidently stronger signal of gelatinolytic activity in the PA group, especially after a collagenase challenge (
Modulus Evaluation
The 2-dimensional and 3-dimensional atomic force microscopy (AFM) images in Figure 4A reveal a granular, uneven morphology with nanoparticles in G1 and G10, consistent with the SEM images. Open dentin tubules and smooth dentin surfaces were observed in the PA group, the roughness of which was relatively lower than that of G1 and G10 (
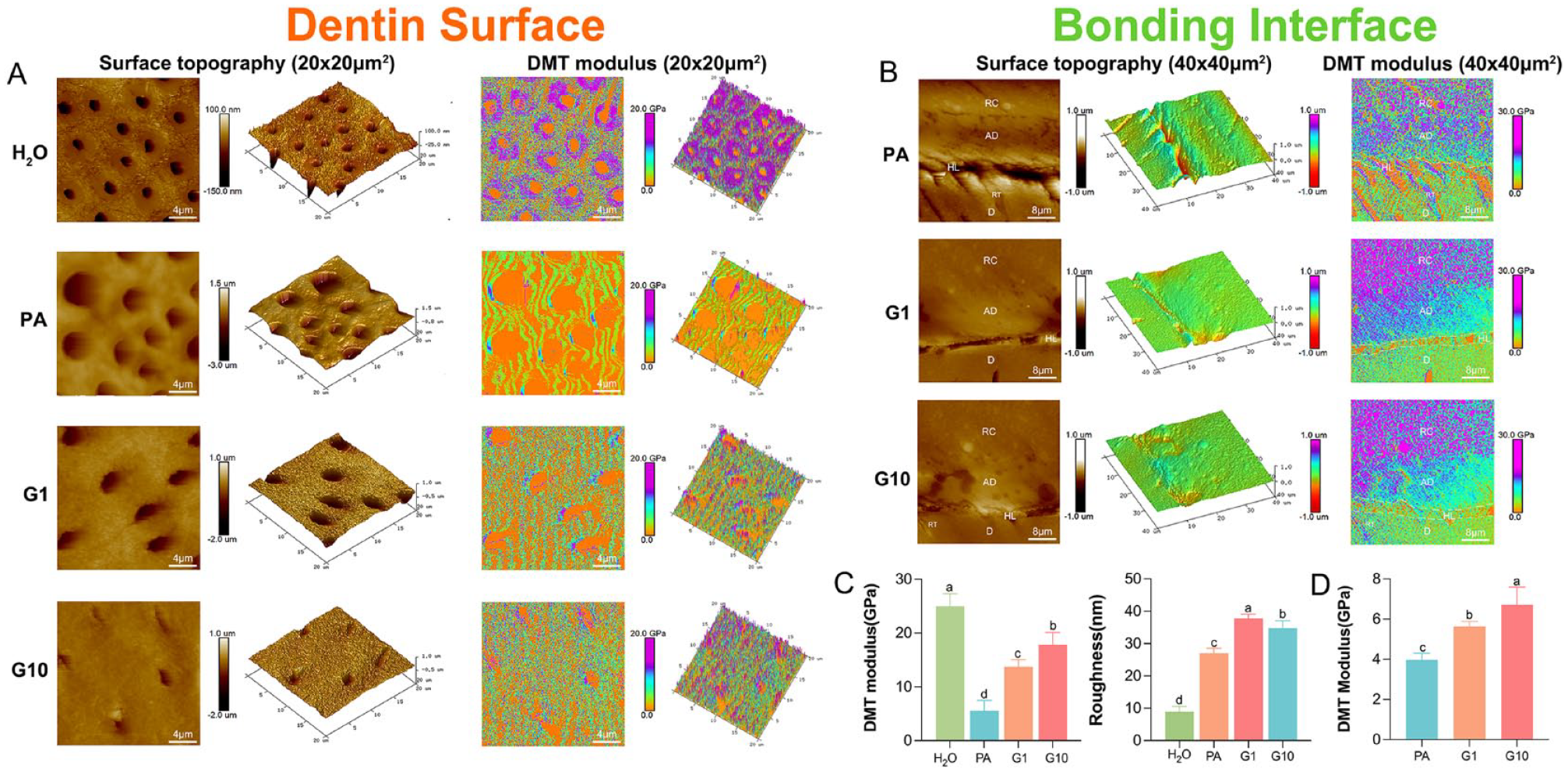
Atomic force microscopy (AFM) characterization of dentin surface and adhesive–dentin interface. (
Figure 4B shows typical AFM images of the adhesive–dentin interface. For superficial topography, G1 and G10 exhibited a more continuous and smooth transition, indicating their tight interlocked structure. By comparison, there was an obvious gap between the adhesive layer and the underlying dentin of the PA group, denoting the collapse of the collagen network that may cause microleakage and low bonding quality. Correspondingly, a stable mechanical transition was illustrated by a constant color gradient in the DMT modulus mapping of G1 and G10. The quantitative mechanical performance also revealed an increasing trend in G1 and G10 (
Antibacterial and Biocompatible Activities
Antibacterial properties
Figure 5A demonstrates the results of OD 570 values of the MTT test and colony-forming unit (CFU)/disk of
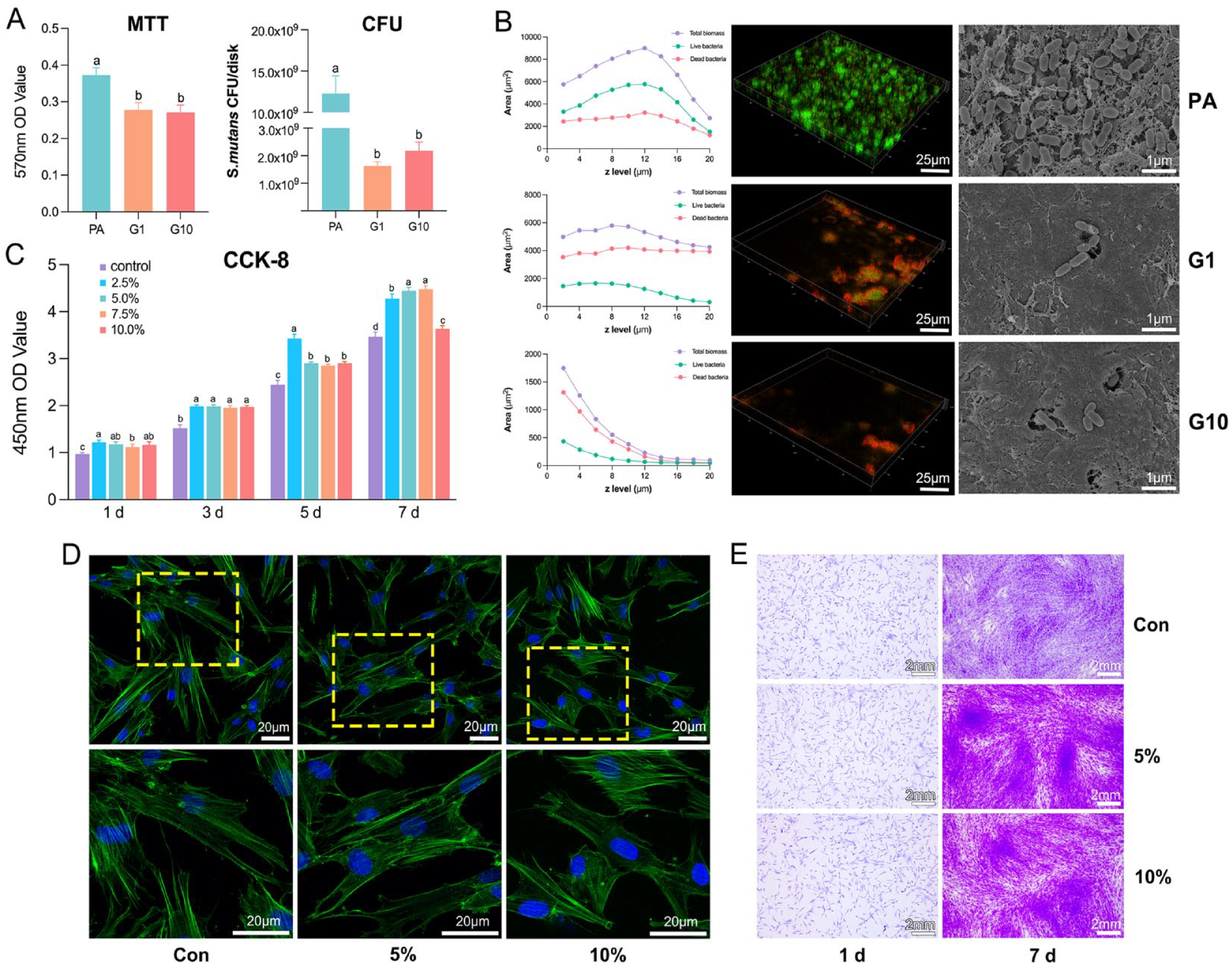
Antibacterial properties of dentin surfaces and biocompatible activities of different concentrations of polyacrylic acid–stabilized amorphous calcium fluoride (PAA-ACF). (
Biocompatible activities
The proliferation of human dental pulp stem cells (hDPSCs) cultured in medium containing mineral solutions at concentrations of 2.5%, 5%, 7.5%, and 10% was obviously promoted compared with the blank control group at 1, 3, 5, and 7 d (
Discussion
Unlike the demineralized dentin collagen that collapses immediately after acid etching in ER modes, the 3-dimensional collagen scaffold with intrafibrillar minerals facilitates the infiltration of adhesive resin, thereby reducing nanoleakage (Liu et al. 2011). Beyond that, the water phase within the fibril compartments would be replaced with crystals after intrafibrillar mineralization. Therefore, recovering intrafibrillar minerals could enhance the bonding interface by a physiologic dehydration mechanism (Ihli et al. 2014; Xu, Zhao, et al. 2018). In this study, we proposed a “prebonding rapid intrafibrillar mineralization strategy,” and the results showed that the physicochemical properties of dentin were improved, as well as the bond strength and bond durability.
As shown by SEM and TEM, the earliest intrafibrillar mineralization occurred at 1 min, when aligned internal minerals appeared. Full mineralization was reached at 10 min. This minimized pretreatment time (from 1 to 10 min) is more user-friendly, bringing the possibility of clinical application in dentin bonding. Therefore, the first null hypothesis should be rejected.
As for the detailed process illustrated in Fig 1F, the collagen fibrils are immersed in a liquid environment surrounded by PAA-ACF precursors, which continuously infiltrate into collagen from gap zones first. Then, ACF gradually transforms from amorphous precursors to crystalline CaF2 and fills up the internal area of collagen (Fang, Ping, Wagermaier, et al. 2021). The mineralization process enlarges the diameter of collagen fibrils, with arranged CaF2 minerals along the central axis of collagen. However, it still needs to be further verified.
Considering that the 3-dimensional collagen matrix is more complex than the 2-dimensional reconstructed single-layer collagen model, it is necessary to further verify the clinical feasibility of the rapid mineralization strategy in demineralized dentin. Dentin surfaces treated by PAA-ACF were morphologically rougher than those of the PA group under SEM observation. AFM images also presented a granular appearance and higher roughness values of G1 and G10.
The distribution of organic and inorganic components on dentin surfaces was also fully investigated. The signal enhancement of fluoride in Raman mapping confirmed the appearance of CaF2 crystals on dentin after pretreatment with PAA-ACF. XRD involving the alignment of the crystal structure indicated a mixture of CaF2 and underlying HAP. For the organic matrix, the existence of amide I in Raman mapping illustrated the well-preserved structure of collagen. The intensity of amide I in G1 and G10 was lower than that of the PA group, indicating that the binding of minerals onto carboxyl groups attenuated the stretching vibration of the C═O bonds.
AFM with the function of mechanical property characterization was applied in the present study. Apparently, the elastic modulus of PAA-ACF–conditioned dentin was significantly improved by the reincorporation of minerals. The results showed positive support for the relationship between the intrafibrillar minerals and mechanical behaviors of dentin, in line with our previous study (Zhao et al. 2022). Hence, the second null hypothesis was rejected.
Inadequate stability of dentin bonding is mainly ascribed to the degradation of fragile dentin collagen, while the enhanced mechanical properties of dentin contribute to better bonding performance (Vidal et al. 2021). Therefore, we speculated the high feasibility of PAA-ACF mineralization agents as primers for dentin bonding. Thermocycling and collagenase aging were utilized to evaluate wet bonding durability in the present research (Ho et al. 2005; Deng et al. 2014). Bond strength testing showed that G1 and G10 displayed remarkably improved mTBS values, irrespective of immediate or aging samples. Infiltration of adhesive monomers was assessed by a nanoleakage test, in which continuous silver deposition was observed in PA group, while only sparse deposition appeared in G1 and G10. The third null hypothesis was therefore rejected. As shown by AFM, the remineralized dentin served as a 3-dimensional scaffold to integrate a hybrid network of hydrophobic adhesive monomers and hydrophilic collagen, thus forming a smooth interface transition.
MMPs are incapable of being inactivated by a faintly acidic environment during the application of dentin etching (De Munck et al. 2009; Mazzoni et al. 2012). The attenuated fluorescence signal in G1 and G10 suggested the correlation between intrafibrillar minerals and inhibition of endogenous enzyme activity, similar to previous studies (Gu et al. 2019; Guo et al. 2019). In addition, fluoride ions themselves have direct inhibitory effects on MMPs due to their strong electronegativity (Kato et al. 2014). Based on the above 2 explanations, the fourth null hypothesis was rejected.
In addition to endogenous enzymes, the fragile bonding interface could also be undermined by exogenous enzymes from microorganisms. It is widely believed that the application of fluoride preparations could release fluoride ions, acting as an antimicrobial (Fei et al. 2020; Lu et al. 2020). Likewise, our results revealed that PAA-ACF–conditioned dentin tended to restrain the survivability of biofilms. This indicated that intrafibrillar minerals generated by topical application of PAA-ACF may act as a reservoir of fluoride ions. It was reported that CaF2 nanoparticles may suppress microbial biofilms by inhibiting the enzyme activity related to glucan synthesis and the gene involved in bacterial toxicity (Kulshrestha et al. 2016; Fei et al. 2020). Consequently, the fifth null hypothesis was rejected.
Conclusion
In conclusion, the concept of “prebonding rapid intrafibrillar mineralization” was proposed for dentin bonding in the present study. This strategy enhanced bonding stability and reduced the degradation of dentin collagen by inhibiting endogenous MMPs and exogenous bacterial erosion. The findings have promising significance to foster the longevity of adhesive restorations. The mechanisms of rapid mineralization of PAA-ACF will be further elaborated in our future studies, while in vivo tests should be carried out to provide more convincing evidence.
Author Contributions
X. Gao, contributed to conception, design, data acquisition, analysis and interpretation, drafted the manuscript; Z. Wang, contributed to data acquisition, critically revised the manuscript; H. Yang, C. Huang, contributed to conception, design, data analysis and interpretation, critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of the work.
Supplemental Material
sj-docx-1-jdr-10.1177_00220345231205492 – Supplemental material for Rapid Intrafibrillar Mineralization Strategy Enhances Adhesive–Dentin Interface
Supplemental material, sj-docx-1-jdr-10.1177_00220345231205492 for Rapid Intrafibrillar Mineralization Strategy Enhances Adhesive–Dentin Interface by X. Gao, Z. Wang, H. Yang and C. Huang in Journal of Dental Research
Footnotes
Acknowledgements
The authors thank the Core Facility of Wuhan University for the use of Cryo-TEM and the Tescan MIRA. The authors thank Dr. Yin Liu from Wuhan University of Technology for the assistance with SEM analysis of the single-layer collagen model, as well as XPS analysis.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by National Natural Science Foundation of China (82371004,82271010) and the Natural Science Foundation of Hubei Province (2022CFB068). The funding agencies have no role in the submitted work.
A supplemental appendix to this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
