Abstract
This study compares the effectiveness of pharmacological treatments to develop guidelines for the management of acute pain after tooth extraction. We searched Medline, EMBASE, CENTRAL, and US Clinical Trials registry on November 21, 2020. We included randomized clinical trials (RCTs) of participants undergoing dental extractions comparing 10 interventions, including acetaminophen, nonsteroidal anti-inflammatory drugs (NSAIDs), opioids, and combinations to placebo. After duplicate screening and data abstraction, we conducted a frequentist network meta-analysis for each outcome at 6 h (i.e., pain relief, total pain relief [TOTPAR], summed pain intensity difference [SPID], global efficacy rating, rescue analgesia, and adverse effects). We assessed the risk of bias using a modified Cochrane RoB 2.0 tool and the certainty of evidence using the Grading of Recommendations, Assessment, Development, and Evaluation approach. We implemented the analyses in RStudio version 3.5.3 and classified interventions from most to least beneficial or harmful. We included 82 RCTs. Fifty-six RCTs enrolling 9,095 participants found moderate- and high-certainty evidence that ibuprofen 200 to 400 mg plus acetaminophen 500 to 1,000 mg (mean difference compared to placebo [MDp], 1.68; 95% confidence interval [CI], 1.06–2.31), acetaminophen 650 mg plus oxycodone 10 mg (MDp, 1.19; 95% CI, 0.85–1.54), ibuprofen 400 mg (MDp, 1.31; 95% CI, 1.17–1.45), and naproxen 400–440 mg (MDp, 1.44; 95% CI, 1.07–1.80) were most effective for pain relief on a 0 to 4 scale. Oxycodone 5 mg, codeine 60 mg, and tramadol 37.5 mg plus acetaminophen 325 mg were no better than placebo. The results for TOTPAR, SPID, global efficacy rating, and rescue analgesia were similar. Based on low- and very low-certainty evidence, most interventions were classified as no more harmful than placebo for most adverse effects. Based on moderate- and high-certainty evidence, NSAIDs with or without acetaminophen result in better pain-related outcomes than opioids with or without acetaminophen (except acetaminophen 650 mg plus oxycodone 10 mg) or placebo.
Keywords
Background
North America is amid an opioid crisis, which is a leading public health and safety concern. In dentistry, many patients are prescribed opioids for the first time to manage acute postoperative pain after dental impaction surgery, which often results in prescription of an excess number of opioid pills, thereby increasing the risk of misuse, abuse, and addiction (Denisco et al. 2011; Maughan et al. 2016; Moore et al. 2016).
Acute dental pain includes pain from both surgical and nonsurgical dental conditions. Of over half a million dental patient visits between 2011 and 2015, 29% of prescribed opioids exceeded the recommended morphine equivalent for appropriate management of acute pain, and over half exceeded the recommended days of supply (Suda et al. 2020).
The current clinical practice guidelines lack evidence-based guidance on effective management of acute dental pain (Herzig et al. 2018). The National Academies’ report, titled Framing Opioid Prescribing Guidelines for Acute Pain, highlighted the need to formalize evidence-based alternatives to opioid analgesics in a clinical practice guideline (National Academies of Sciences, Engineering, and Medicine et al. 2019).
The objective of this systematic review (SR) and network meta-analysis (NMA) was to assess the comparative effectiveness of pharmacological treatments for the management of pain subsequent to simple and surgical tooth extraction, as well as pain associated with pulpitis or its complications. This SR was conducted to inform the 2022 evidence-based clinical practice guidelines produced by the American Dental Association (ADA) Council on Scientific Affairs, the ADA Science & Research Institute (ADASRI), and the University of Pittsburgh’s and the University of Pennsylvania’s Schools of Dental Medicine in partnership with the US Food and Drug Administration (FDA) for the management of acute dental pain.
Methods
We report this SR following the guidelines of the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) checklist for SR and NMAs (see Appendix Table 1) (Hutton et al. 2015). We did not register this SR but followed preestablished methodology outlined in the plan for guideline development and used eligibility criteria determined by the recommendation questions addressed by the guideline panel.
Eligibility Criteria
We included randomized clinical trials (RCTs) including individuals ages 12 and above undergoing simple or surgical tooth extraction or affected by symptomatic pulpitis or its complications that compared 10 interventions, including acetaminophen, nonsteroidal anti-inflammatory drugs (NSAIDs), opioids, and combinations against one another or placebo. We limited the literature to peer-reviewed articles and English language.
The interventions and dosages were selected by the guideline panel as the most commonly prescribed and relevant to acute dental pain management after a prioritization exercise that considered more than 30 different drugs as monotherapies or combinations (Moore et al. 2006, 2018). Similar to Cochrane Library overviews of multiple analgesics, a follow-up time point of 6 h was selected to eliminate distortions of findings (i.e., summed pain intensity difference [SPID], total pain relief [TOTPAR], proportion of rescue medications) when comparing short-duration (4–6 h) and long-duration (8–12 h) agents (Moore et al. 2011). The outcomes included pain relief at 6 h, TOTPAR at 6 h, SPID at 6 h, global efficacy rating at 6 h, proportion of participants receiving rescue analgesia at 6 h, and adverse effects (central nervous system and gastrointestinal) at the longest reported follow-up time point. See Appendix Table 2 for a list of included interventions and outcomes.
To establish the aforementioned eligibility criteria, the guideline panel relied on peer-reviewed literature regarding dental pharmacology, professional experience, a national survey of US oral surgeons of drug selection and prescribing behaviors, and selective randomized clinical trials of analgesic efficacy following third molar extractions (Moore and Hersh 2013; Hersh et al. 2020; American Dental Association 2021). All decisions regarding the eligibility criteria for drug/dose selection and time points were made with the consensus of the panel.
Information Sources
We performed searches in Medline, EMBASE, CENTRAL, and US Clinical Trials registry from inception through November 21, 2020. See Appendix Table 3 for a sample search strategy.
Study Selection
Using a systematic review software (Covidence), pairs of reviewers, following training and calibration exercises, independently screened all titles and abstracts, followed by full texts of trials that were identified as potentially eligible. A third reviewer adjudicated conflicts.
Data Collection
For each eligible trial, pairs of reviewers, following training and calibration exercises, extracted data independently using a standardized, pilot-tested data extraction form. Reviewers collected information on trial characteristics (i.e., design), patient characteristics (i.e., age, gender, country), and outcomes of interest. Reviewers resolved discrepancies by discussion and, when necessary, with adjudication by a third party.
Risk of Bias within Individual Studies
For each eligible trial and outcome, pairs of reviewers, following training and calibration exercises, independently used a modification of the Cochrane tool to assess risk of bias in randomized trials (RoB 2.0). Reviewers resolved discrepancies by discussion and, when necessary, with adjudication by a third party.
Data Synthesis
Treatments were grouped into nodes that could include more than 1 dose for a monotherapy or a combination, according to the eligibility criteria listed above. The comparator/reference for all networks was selected to be the intervention/placebo reported in the highest number of studies.
We summarized the effect of interventions on dichotomous outcomes (i.e., proportion of participants receiving rescue analgesia and adverse effects) using odds ratios (ORs) and risk differences (RDs), as well as their corresponding 95% confidence interval (CI). For continuous outcomes (i.e., pain relief, TOTPAR, SPID, global efficacy rating), we used the mean difference (MD) (between pre- and postoperative scores) and corresponding 95% CI. When studies reported the same outcome using a scale with a different range, we converted data to the scale range most commonly reported before conducting analyses (Thorlund et al. 2011). When standard deviation (SD) was not reported, we calculated SD using standard error, confidence intervals, means, and sample sizes. In rare instances, when neither of the beforementioned statistics were reported, we imputed SD by choosing a median SD of 3 studies with similar means (Higgins and Green 2011).
We performed frequentist NMAs for outcomes with sufficient data. If data were insufficient for an NMA but adequate for a pairwise meta-analysis (i.e., at least 2 studies), we conducted pairwise meta-analyses comparing specific interventions. All analyses were completed using a random-effects model and weighting studies according to the inverse of their variance.
We implemented the analyses in RStudio version 3.5.3 (R Studio). We used the package netmeta (Rucker 2021) to conduct NMAs and the packages meta (Schwarzer 2015) and metafor (Viechtbauer 2021) to conduct pairwise meta-analyses.
Certainty of the Evidence
We assessed the certainty of the evidence using the Grading of Recommendations, Assessment, Development, and Evaluation (GRADE) approach for NMAs (Puhan et al. 2014; Brignardello-Petersen et al. 2018; Brignardello-Petersen et al. 2019). The GRADE domains included risk of bias, inconsistency, indirectness, publication bias, imprecision, transitivity, and incoherence (i.e., agreement between direct and indirect evidence). Two methodologists rated each domain for each outcome and comparison independently, resolving discrepancies by discussion. We used a minimally contextualized approach to rate our certainty that there was an important effect (Zeng et al. 2021), using a threshold of 10% of the length of the scale as the minimally important difference for continuous outcomes and baseline risk for dichotomous outcomes.
Presentation of Results
To facilitate interpretation of results of dichotomous outcomes, we calculated absolute effects (95% CI) per 100 participants. To draw conclusions, we classified interventions in groups from the most to the least effective by considering the estimates of effect and the certainty of the evidence (Brignardello-Petersen et al. 2020).
Subgroup and Sensitivity Analyses
Subgroup and sensitivity analyses were not planned.
Results
After screening 4,716 titles and abstracts, we included 85 unique RCTs reported in 82 publications (Fig.). Reasons for exclusion at the full-text screening stage (n = 776) are presented the Figure.
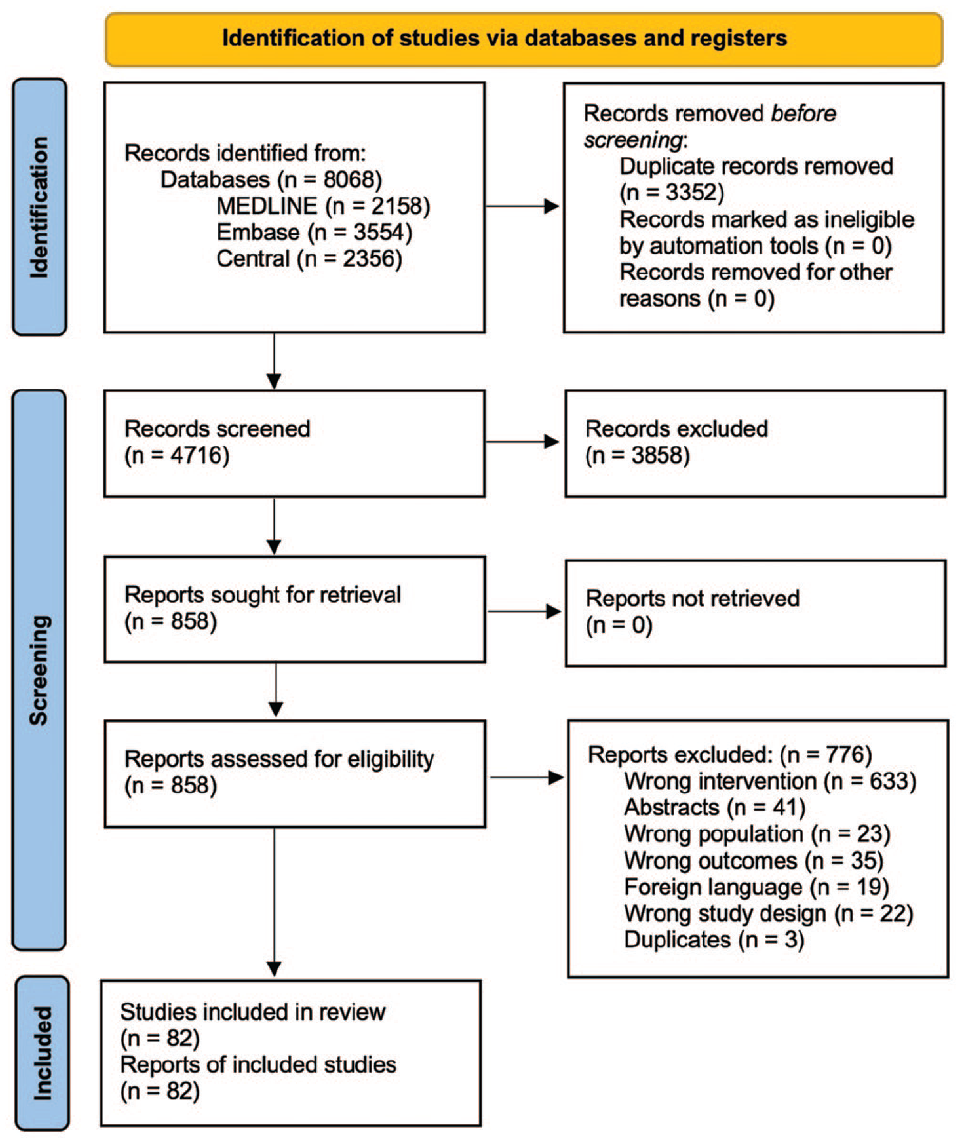
Study identification and selection flowchart.
Characteristics of Included Studies
Characteristics of the included studies are reported in Table 1. All studies were parallel group RCTs. Most studies were conducted in the United States (75%). Number of participants ranged from 31 to 540. Mean age of participants across studies varied from 13.5 (SD = 2.64) to 40.7 (SD not reported). Interventions were administered orally. Surgical tooth extraction was the type of extraction performed in all included studies. Studies assessing the interventions of interest in patients with symptomatic pulpitis or its complications were not found.
Characteristics of Included Studies.
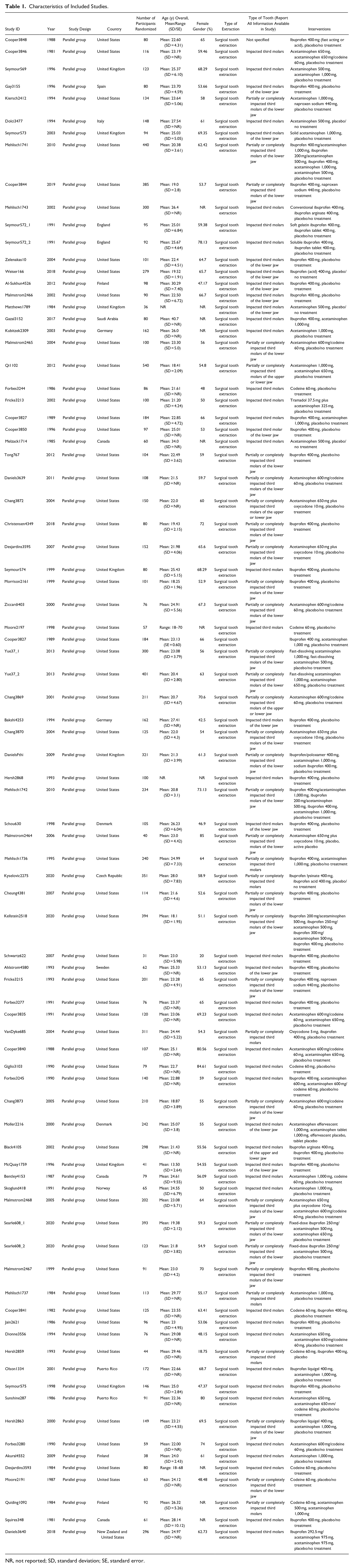
NR, not reported; SD, standard deviation; SE, standard error.
Risk of Bias in Included Studies
Appendix Tables 4 to 9 present the assessment of risk of bias of the included RCTs for each outcome. The domains in which most of the RCTs were judged at high risk of bias were missing outcome data and selection of reported results. Overall, 9 of 85 RCTs were judged at low or probably low risk of bias.
Effects of the Interventions
Table 2 presents a summary of the effects of interventions on pain outcomes. Appendix Tables 10 and 11 summarize the effect of the interventions on adverse effects. Appendix Figures 1 to 21.6 and Appendix Tables 12 to 28 include network plots and forest plots of pairwise meta-analyses for all outcomes, as well as detailed relative and absolute effect estimates from the NMAs and the certainty of evidence for all comparisons and outcomes. All 85 RCTs were included in at least 1 of the 5 NMAs.
Summary of Benefit Outcomes Compared with Placebo (No Treatment).
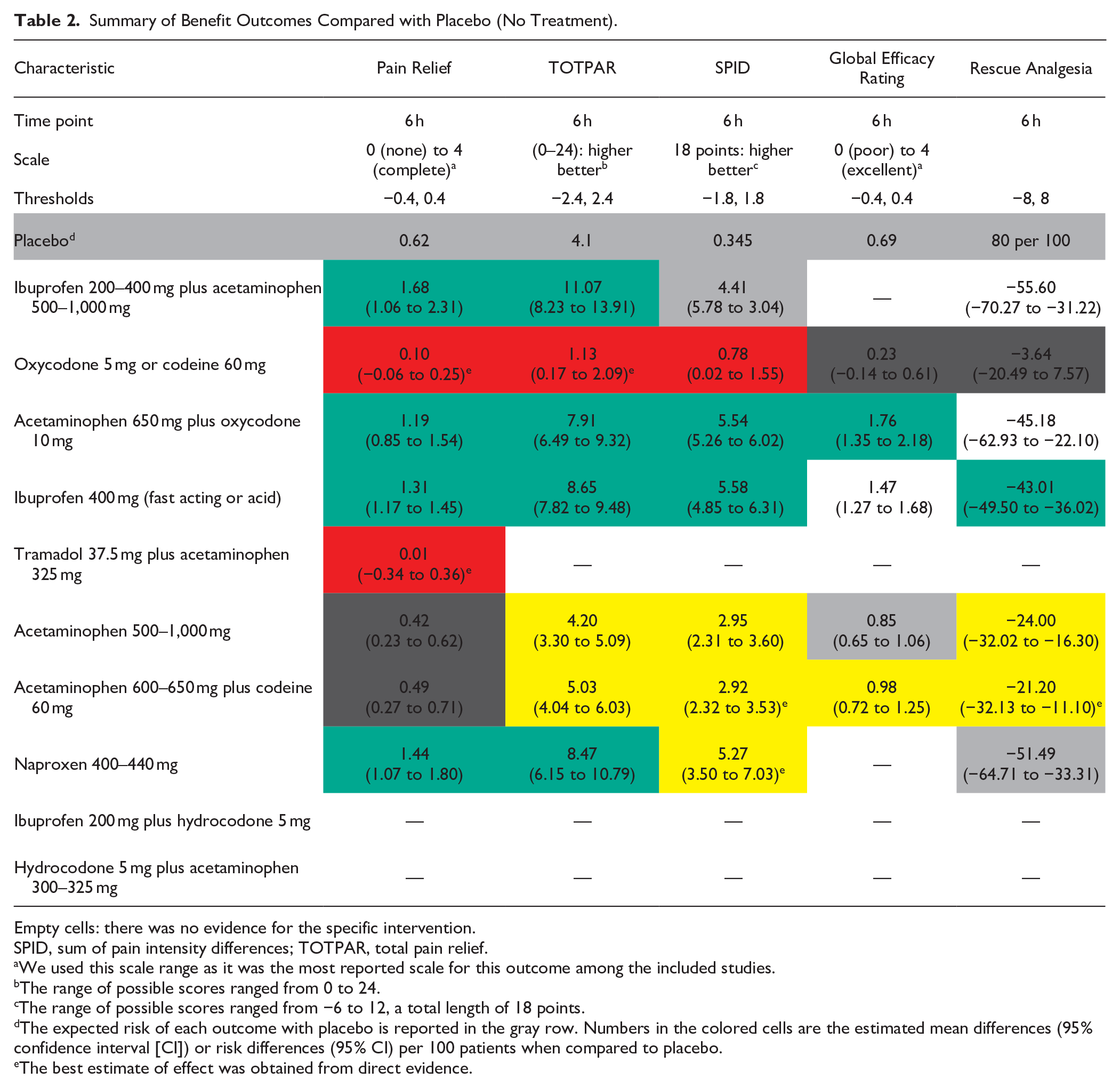
Empty cells: there was no evidence for the specific intervention.
SPID, sum of pain intensity differences; TOTPAR, total pain relief.
We used this scale range as it was the most reported scale for this outcome among the included studies.
The range of possible scores ranged from 0 to 24.
The range of possible scores ranged from −6 to 12, a total length of 18 points.
The expected risk of each outcome with placebo is reported in the gray row. Numbers in the colored cells are the estimated mean differences (95% confidence interval [CI]) or risk differences (95% CI) per 100 patients when compared to placebo.
The best estimate of effect was obtained from direct evidence.

Pain relief
Fifty-six studies including 9,095 participants were included in the NMA for pain relief. Using a scale from 0 to 4, where higher scores represent more pain relief, the interventions classified as among the most effective for this outcome were (effect estimates in reference to placebo) as follows: ibuprofen 200 to 400 mg plus acetaminophen 500 to 1,000 mg (MD, 1.68; 95% 95% CI, 1.06−2.31; moderate certainty), acetaminophen 650 mg plus oxycodone 10 mg (MD, 1.19; 95% CI, 0.85−1.54; moderate certainty), and ibuprofen 400 mg (fast acting or acid) (MD, 1.31; 95% CI, 1.17−1.45; moderate certainty). There was no convincing evidence that any of the other interventions were different from placebo for this outcome.
Totpar
Forty-four studies including 7,282 participants were included in the NMA for TOTPAR. Using a scale from 0 to 24, where higher scores represent more total pain relief, the interventions classified as among the most effective for this outcome were (effect estimates in reference to placebo) as follows: ibuprofen 200 to 400 mg plus acetaminophen 500 to 1,000 mg (MD, 11.07; 95% CI, 8.23−13.9; moderate certainty), acetaminophen 650 mg plus oxycodone 10 mg (MD, 7.91; 95% CI, 6.49−9.32; moderate certainty), ibuprofen 400 mg (fast acting or acid) (MD, 8.65; 95% CI, 7.82−9.48; moderate certainty), and naproxen 400 to 440 mg (MD, 8.47; 95% CI, 6.15−10.79; moderate certainty). Interventions that were more effective than placebo but less effective than the interventions above were acetaminophen 500 to 1,000 mg (MD, 4.20; 95% CI, 3.30−5.09; moderate certainty) and acetaminophen 600 to 650 mg plus codeine 60 mg (MD, 5.03; 95% CI, 4.04−6.03; moderate certainty). Oxycodone 5 mg and codeine 60 mg were not more effective than placebo.
Spid
Thirty-one studies including 6,721 participants reported on SPID. Using an 18-point scale, where higher scores represent better outcomes, the interventions classified as the most effective were (effect estimates in reference to placebo) as follows: acetaminophen 650 mg plus oxycodone 10 mg (MD, 5.54; 95% CI, 5.26−6.02; moderate certainty) and ibuprofen 400 mg (fast acting or acid) (MD, 5.58; 95% CI, 4.85−6.31; moderate certainty). Acetaminophen 500 to 1,000 mg (MD, 2.95; 95% CI, 2.31−3.60; moderate certainty), acetaminophen 600 to 650 mg plus codeine 60 mg (MD, 2.92; 95% CI, 2.32−3.53; moderate certainty), and naproxen 400 to 440 mg (MD, 5.27; 95% CI, 3.50−7.03; moderate certainty) were better than placebo but less effective than the interventions above. Ibuprofen 200 to 400 mg plus acetaminophen 500 to 1,000 mg (MD, 4.41; 95% CI, 5.78−3.04; low certainty) were classified as possibly better than placebo. Oxycodone 5 mg and codeine 60 mg were not more effective than placebo.
Global efficacy rating, rescue analgesia, and adverse effects are reported in the Appendix.
Discussion
This SR and NMA summarizes the comparative effects of the analgesic drugs considered by the guideline panel when making recommendations for treating dental acute pain. Based on moderate- and high-certainty evidence, in individuals undergoing surgical tooth extractions, the interventions classified as the most effective for pain relief were ibuprofen 200 to 400 mg plus acetaminophen 500 to 1,000 mg, acetaminophen 650 mg plus oxycodone 10 mg, ibuprofen 400 mg, and naproxen 400 to 440 mg. Oxycodone 5 mg or codeine 60 mg and tramadol 37.5 mg plus acetaminophen 325 mg were no better than placebo. The results for TOTPAR, SPID, global efficacy rating, and rescue analgesia were similar to pain relief. Based on low- and very low-certainty evidence, most interventions were classified as no more harmful than placebo for most adverse effects.
Evidence for ibuprofen 200 mg plus hydrocodone 5 mg and hydrocodone 5 mg plus acetaminophen 300 to 325 mg (i.e., Vicodin) was not available. A comprehensive national survey conducted in the United States showed that hydrocodone and acetaminophen combinations (i.e., Vicodin, Vicodin ES, Vicodin HP) are the most preferred combination of analgesics prescribed by oral and maxillofacial surgeons for the management of pain following third molar extractions (Levy et al. 2015). Future research should focus on exploring the comparative effect of hydrocodone and acetaminophen formulations. Furthermore, evidence regarding the effect of the 10 selected analgesics on the temporary management of symptomatic pulpitis or its complications prior to dental treatment was not found.
A recent overview of SRs summarizing benefits and harms of analgesic agents for the management of acute dental pain concluded that “relief of postoperative pain in dental practice with the use of nonsteroidal anti-inflammatory drugs, with or without acetaminophen, is equal or superior to that provided by opioid-containing medications” (Moore et al. 2018). This finding is similar to the finding of this SR and NMA that, except for acetaminophen 650 mg plus oxycodone 10 mg, NSAIDs with or without acetaminophen were superior to placebo as opposed to opioid-containing interventions that were not. Furthermore, similar to this NMA, the overview of SRs suggested that opioid analgesics and their combinations are associated with higher rates of acute adverse events (Moore et al. 2018).
Another overview of SRs aiming to summarize the efficacy of analgesics for acute dental pain in adults investigated 41 single-dose analgesics or analgesic combinations for acute postoperative pain and showed that the best interventions in terms of number needed to treat (NNT) for at least 50% maximum pain relief over 4 to 6 h compared with placebo were ibuprofen 200 mg plus acetaminophen 500 mg, ibuprofen 200 mg, ibuprofen 200 mg plus caffeine 100 mg, diclofenac potassium 50 mg, and etoricoxib 120 mg (Moore et al. 2015b). The worst intervention was codeine 60 mg (Moore et al. 2015b). These results were comparable to the results of this SR and NMA, specifically the superiority of ibuprofen plus acetaminophen and ibuprofen alone and inferiority of codeine 60 mg compared to placebo.
An overview of SRs summarizing adverse event rates associated with 41 single-dose oral analgesics or analgesic combinations compared with placebo for acute postoperative pain in adults showed that there were few instances of participants experiencing significantly more or fewer adverse events than with placebo for most NSAIDs, acetaminophen, and combinations not containing opioids (Moore et al. 2015a). However, for aspirin 1,000 mg, diflunisal 1,000 mg, opioids, or fixed-dose combination drugs containing opioids, participants often experienced significantly more adverse effects than with placebo. In this SR and NMA, most included opioid-containing interventions (oxycodone 5 mg and codeine 60 mg, acetaminophen 650 mg plus oxycodone 10 mg, and acetaminophen 600–650 mg plus codeine 60 mg) were worse than placebo in terms of at least 1 adverse effect. Ibuprofen 400 and acetaminophen 500 to 1,000 mg were worse than placebo in terms of drowsiness.
Several differences between this SR and NMA and review of reviews by P.A. Moore et al. (Moore et al. 2018) and R.A. Moore et al. (Moore et al. 2015a, 2015b; Moore, Wiffen, et al. 2015) may have contributed to the differences in conclusions between these reviews. In this SR and NMA, data from individual RCTs were included in a single NMA that comprised the results, whereas in the overviews of SRs, results from multiple analyses were aggregated to constitute the results. In terms of pain relief, P.A. Moore et al. (Moore et al. 2018) and R.A. Moore et al. (Moore et al. 2015b; Moore, Wiffen, et al. 2015) reported NNT for at least 50% pain relief, whereas this SR and NMA reported mean pain relief measured on a scale from 0 (none) to 4 (complete) at 6 h. The variability in inclusion of interventions may have contributed to the differences in the conclusions between these publications.
Furthermore, in this SR and NMA, a modification of the Cochrane tool for assessing risk of bias (RoB 2.0) was used to examine risk of bias in individual RCTs, whereas the overviews of SRs by P.A. Moore et al. (Moore et al. 2018) and R.A. Moore et al. (Moore et al. 2015a, 2015b; Moore, Wiffen, et al. 2015) did not mention risk of bias assessment. The quality of the evidence using the GRADE approach was assessed in this SR, but it was not addressed in the overviews by P.A. Moore et al. (Moore et al. 2018) and R.A. Moore et al. (Moore et al. 2015a, 2015b; Moore, Wiffen, et al. 2015).
Strengths of the Review Process
This review is the first to synthesize NMAs for comparison of analgesics for treatment of acute dental pain associated with postsurgical extractions, allowing for streamlined clinical decision-making. Each of the review process stages was conducted in duplicate, with adjudication of conflicts by a third reviewer. The risk of bias of each individual study, as well as the certainty of the evidence for each outcome of interest, was assessed. We performed analyses and interpreted the results using the latest methodological guidance from the GRADE Working Group. In order to make the results easier to interpret, instead of using standardized mean difference, we reported continuous outcomes using mean difference by converting all scale scores to the most reported scale (Thorlund et al. 2011).
Limitations of the Review Process
This SR and NMA was limited to inclusion of only 10 interventions, which may limit its applicability. These 10 interventions, however, were chosen by a panel of clinical experts who determined that they were the most relevant to practice. Furthermore, this SR and NMA included only peer-reviewed studies published in English, but this likely did not affect the study conclusions.
Conclusion
Based on moderate- and high-certainty evidence, in individuals undergoing surgical tooth extractions, the interventions classified as the most effective for pain relief were ibuprofen 200 to 400 mg plus acetaminophen 500 to 1,000 mg, acetaminophen 650 mg plus oxycodone 10 mg, ibuprofen 400 mg, and naproxen 400–440 mg. Oxycodone 5 mg or codeine 60 mg and tramadol 37.5 mg plus acetaminophen 325 mg were no better than placebo. The results for TOTPAR, SPID, global efficacy rating, and rescue analgesia were similar to pain relief. Based on low- and very low-certainty evidence, most interventions were classified as no more harmful than placebo for most adverse effects. Future research should focus on the assessment of ibuprofen 200 mg plus hydrocodone 5 mg, hydrocodone 5 mg plus acetaminophen 300 to 325 mg, and tramadol 37.5 mg plus acetaminophen 325 mg through RCTs.
Author Contributions
A. Miroshnychenko, contributed to conception and design, data acquisition, analysis, and interpretation, drafted and critically revised the manuscript; S. Ibrahim, M. Azab, contributed to conception and design, data acquisition, analysis, and interpretation, critically revised the manuscript; Y. Roldan, D. Tamilselvan, L. He, J.W. Little, contributed to conception and design, data acquisition, critically revised the manuscript; J.P.D. Martinez, contributed to conception and design, data analysis, critically revised the manuscript; O. Urquhart, M. Tampi, D.E. Polk, P.A. Moore, E.V. Hersh, B. Claytor, A. Carrasco-Labra, contributed to conception and design, data interpretation, critically revised the manuscript; R. Brignardello-Petersen, contributed to conception and design, data analysis and interpretation, critically revised the manuscript. All authors gave their final approval and agree to be accountable for all aspects of the work.
Supplemental Material
sj-docx-1-jdr-10.1177_00220345221139230 – Supplemental material for Acute Postoperative Pain Due to Dental Extraction in the Adult Population: A Systematic Review and Network Meta-analysis
Supplemental material, sj-docx-1-jdr-10.1177_00220345221139230 for Acute Postoperative Pain Due to Dental Extraction in the Adult Population: A Systematic Review and Network Meta-analysis by A. Miroshnychenko, S. Ibrahim, M. Azab, Y. Roldan, J.P.D. Martinez, D. Tamilselvan, L. He, J.W. Little, O. Urquhart, M. Tampi, D.E. Polk, P.A. Moore, E.V. Hersh, B. Claytor, A. Carrasco-Labra and R. Brignardello-Petersen in Journal of Dental Research
Footnotes
A supplemental appendix to this article is available online.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: E.V. Hersh and the Trustees of the University of Pennsylvania have received grant funding from Pfizer Consumer Healthcare and Bayer Healthcare. In addition, within the past 5 y, E.V. Hersh received consulting funds for reviewing data and sharing his expertise with the National Advertising Division of the Better Business Bureau concerning pain studies that compared Aleve and Extra Strength Tylenol.
This project was financially supported by the FDA of the US Department of Health and Human Services (HHS). The contents are those of the author(s) and do not necessarily represent the official views of, nor an endorsement by, FDA/HHS or the US government.
The findings and conclusions in this presentation are those of the author(s) and do not necessarily reflect the official views, position, or policy of the American Dental Association, nor does this presentation constitute an endorsement by the American Dental Association.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
