Abstract
Oral diseases remain a pressing global health challenge, affecting billions and disproportionately affecting underserved and vulnerable populations. Despite strong evidence supporting preventive measures, this evidence often fails to translate into real-world practice due to fragmented health systems, a treatment-dominated model of dental care, and a lack of focus on upstream, population-wide prevention strategies. Implementation research helps to bridge the effectiveness-implementation gap by systematically studying how interventions can be integrated into real-world settings and sustained effectively. Implementation research requires a multidisciplinary approach that often goes beyond traditional research settings, prioritizing stakeholder engagement, local context, and iterative testing and aligning interventions with community needs. To support the design and evaluation of these implementation efforts, logic models are practical tools that can be used to help map determinants, strategies, and outcomes using underlying causal reasoning. Once determinants have been identified, trials can be developed to incorporate implementation outcomes such as acceptability (how agreeable it is among stakeholders), adoption (the uptake), and fidelity (how closely implemented as intended), providing insights into the real-world performance of interventions. Implementation strategies should be clearly defined and documented by detailing their rationale, specific actions, and outcomes they are intended to affect. Specifying and contextualizing strategies in this way ensures they can be appropriately evaluated. This article identifies 3 examples that show the importance of embedding interventions within existing health systems and policies and how bottom-up approaches, such as engaging stakeholders from the outset, are critical to the success of the implementation. Integrating implementation considerations early in the research process and fostering strategic partnerships between research organizations, policymakers, and communities are essential to translating evidence into meaningful oral health improvements and bridging the gap between research and real-world impact.
Keywords
Introduction
Oral diseases remain one of the most pressing global health challenges, affecting nearly half of the world’s population (Bernabe et al 2025). Despite well-established evidence that preventive measures at individual and community levels, such as fluoride use, sugar reduction, and tobacco elimination, can reduce dental caries, periodontal diseases, and tooth loss, the global burden of oral diseases is projected to increase, particularly among underserved and vulnerable populations (NHS Improvement 2021; Bernabe et al 2025). It has been estimated that more than 660 million people will be edentulous by 2050, with low- and low-middle-income countries being disproportionately affected (Nascimento et al 2024). However, translating evidence into practice to effectively promote and maintain oral health at the population level remains a major challenge. Implementation research addresses gaps that traditional clinical research often overlooks by providing frameworks that assess stakeholder engagement, integrate innovations into existing health systems, and promote sustainability. Implementation research applies not only to novel therapeutic innovations but also to existing procedures that remain underutilized in routine practice.
The 2023–2030 Global Strategy on Oral Health affirmed oral health as a critical area within the World Health Organization’s (WHO’s) noncommunicable disease agenda, emphasizing accessibility and affordability as essential components of health equity (WHO 2024). The WHO agenda sets ambitious targets: ensuring 80% of the global population has access to essential oral health care services and achieving a 10% reduction in the global prevalence of major oral diseases and conditions across the life course. To support countries in adapting these targets to their specific contexts, key actions include strengthening oral health governance, promoting prevention, building the health workforce, and embedding oral health into information systems and research agendas (WHO 2024). In 2024, the WHO held its first-ever Global Oral Health Meeting in Bangkok, Thailand, where more than 100 member states endorsed the Bangkok Declaration—“No Health Without Oral Health”—affirming their commitment to achieving universal oral health coverage by 2030 (WHO 2025). To realize these ambitions, the WHO is recognizing implementation research as one of the key workstreams in its Global Coalition on Oral Health, highlighting its essential role in achieving equitable improvements in oral health outcomes. Integrating implementation thinking early in the development of interventions ensures that they are contextually relevant, accessible, and sustainable.
Despite a substantial evidence base for effective clinical and public health oral interventions, a recent review revealed that implementation research in support of oral and craniofacial health remains almost negligible (Randall 2023). The disconnect between research and real-world implementation remains a formidable challenge. The implementation gap is driven by multiple factors, including the hegemony of a treatment-dominated, interventionist model of dental care with an emphasis on clinical efficacy over real-world application; inadequate integration of dental services within primary health care systems; a lack of focus on upstream, population-wide prevention strategies; and a lack of engagement of key partners when implementing health policies (Watt et al 2019; Benzian et al 2021; Burgette et al 2023). In addition to these structural issues, practical barriers also impede implementation. These include workload burdens on already overstretched staff, funding strategies that do not support sustainability or undervalue implementation outcomes over publications, and fragmented data systems that hinder workflow integration (Glasgow et al 2012; Bauer and Kirchner 2020). Implementation research can navigate these barriers by identifying these operational constraints early.
Rather than relying on the traditional linear biomedical pipeline, where research moves through preintervention studies, clinical trials, pragmatic trials, and only then implementation research, research should consider a more integrated approach (Fig). Such an approach involves co-developing research and implementation from the outset and embedding research within real-world contexts. With continuous feedback loops between clinicians, researchers, policymakers, and communities, this framework increases the likelihood that research is relevant and adopted into practice. This article offers a novel contribution by demonstrating how implementation research can be integrated into routine oral health care through the use of logic models (ie, graphic depictions that map out resources, outputs, and desired outcomes), hybrid study designs, and tailored implementation strategies.
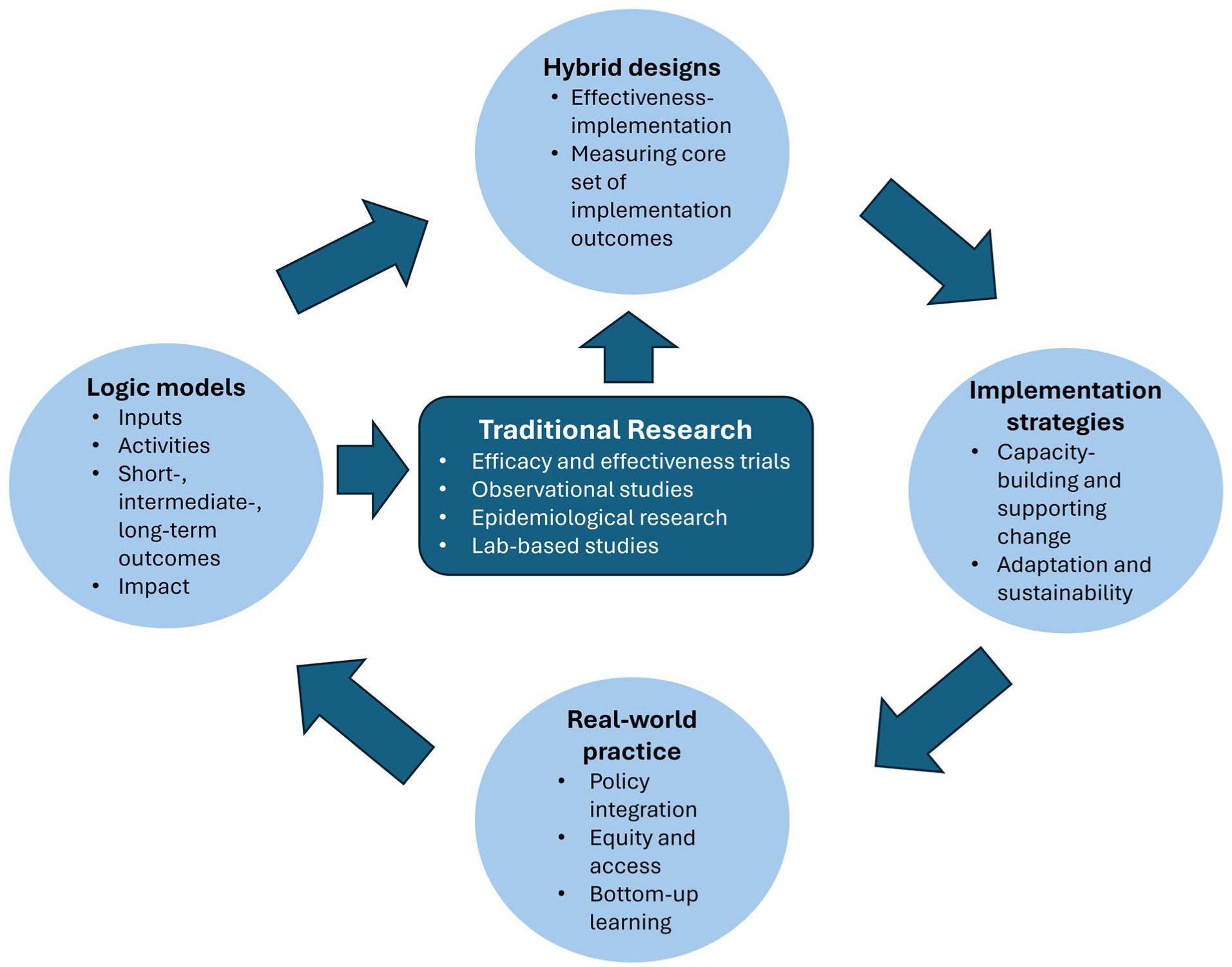
Integrating implementation research by emphasizing the dynamic integration of interventions in real-world settings, as opposed to a traditional, linear biomedical pipeline from preintervention to real-world evaluation. This figure illustrates an iterative approach in which traditional research (central box) interacts with 4 complementary domains (peripheral boxes) to facilitate the implementation of evidence-based interventions. A key component is the logic model, which maps the pathway from input (eg, resources, personnel) to activities (eg, training) and ultimately to outcomes. By making these causal linkages, logic models serve as a foundation for implementation planning. Hybrid designs (type 1, 2, or 3) evaluate both clinical and implementation outcomes, with the emphasis varying by type; for example, type 3 places greater focus on implementation outcomes. Based on the logic model, implementation strategies are selected to achieve desired outcomes. These strategies can be iteratively adapted using early feedback from real-time data to optimize delivery. As interventions are integrated in real-world practice, ongoing field observations inform revisions to the logic model, adaptation of implementation strategies, and the design of subsequent studies that consider diverse contexts.
What Is Implementation Research?
At its core, implementation research is about putting policies, programs, or practices into routine use within real-world settings (Peters et al 2013). In the context of oral health, implementation research means not only communicating evidence-based interventions to target audiences (dissemination) but also embedding them into practice within communities and health care systems to ensure they are adopted, scaled, and sustained effectively.
Unlike efficacy or effectiveness studies, which aim to control the context to test whether an intervention works under ideal conditions, implementation research takes a step forward and explicitly addresses the complexity of health systems (Bauer and Kirchner 2020). To understand how interventions work in practice, implementation research often uses mixed methods, combining system-level data with qualitative methods such as interviews and focus groups to capture the perspectives of patients, providers, and communities.
Implementation research is inherently collaborative and often extends beyond traditional academic boundaries. Patients, caregivers, and community leaders provide insights into acceptability and equity; policymakers and insurers influence system-level feasibility and sustainability; and industry partners contribute technical expertise and integration with existing platforms. Co-design that actively involves these groups can help ensure interventions are contextually relevant and adaptable across settings.
It is important to distinguish implementation research from quality improvement (QI), although the boundaries between the two have increasingly become blurred in practice. QI typically begins with an operational problem within a specific health care setting and seeks to iteratively test solutions aimed at improving quality dimensions such as safety, timeliness, efficiency, effectiveness, patient centeredness, and equity, while implementation research generally starts with the recognition that an evidence-based intervention or practice is underutilized and aims to generate generalizable knowledge to bridge the evidence–practice gaps across different health care contexts.
Although there is overlap, patient-centered outcomes research (PCOR) and implementation research are not interchangeable concepts. PCOR prioritizes outcomes that are important to patients and frequently involves designing effectiveness studies with active patient and caregiver engagement. In many instances, PCOR can serve as a foundation for implementation research by generating patient-centered evidence that can then be translated into practice. For example, the United States Patient-Centered Outcomes Research Institute not only funds comparative effectiveness studies with strong patient involvement but also supports efforts to translate patient-centered findings into real-world practice.
Formulating a Logic Model
To guide the process of implementation research systematically, a logic model that links determinants, strategies, and outcomes with underlying causal reasoning can be helpful. One such tool is the Implementation Research Logic Model (IRLM), which provides an outline of how determinants inform the selection of implementation strategies and how these strategies influence proximal implementation outcomes through specific mechanisms of action (Smith et al 2020). Utilizing the IRLM begins with identifying determinants, which can be guided by frameworks such as the widely used Consolidated Framework for Implementation Research (CFIR) (Damschroder et al 2022).
CFIR specifies domains that capture barriers and facilitators to implementation: the characteristics of the innovation, the outer setting (eg, patient needs and resources), the inner setting (eg, organizational culture and infrastructure), the individuals involved, and the implementation process. To address these determinants, strategies can be drawn from the Expert Recommendations for Implementing Change (ERIC) taxonomy, which catalogues and standardizes implementation strategies across health systems (Powell et al 2015). These strategies can then be linked to implementation outcomes such as acceptability, feasibility, adoption, or sustainability (Proctor et al 2011). The Table provides an illustration in which the row headings correspond to CFIR domains while the column headings reflect the IRLM’s core elements—the determinants, the strategies used to address them, and the outcomes against which progress can be measured.
Framework Mapping for Implementation Research Using a Mobile Oral Health Screening Tool as an Illustrative Example.
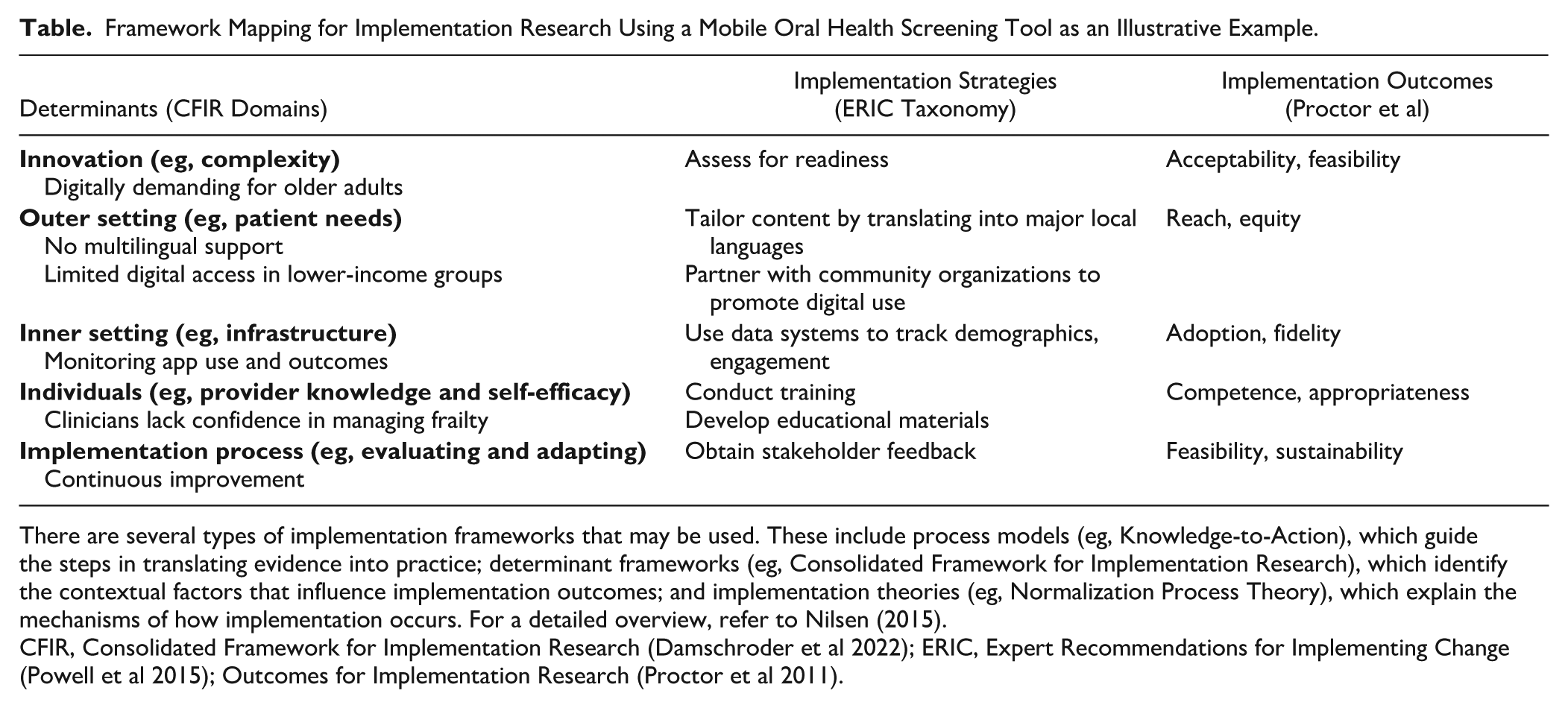
There are several types of implementation frameworks that may be used. These include process models (eg, Knowledge-to-Action), which guide the steps in translating evidence into practice; determinant frameworks (eg, Consolidated Framework for Implementation Research), which identify the contextual factors that influence implementation outcomes; and implementation theories (eg, Normalization Process Theory), which explain the mechanisms of how implementation occurs. For a detailed overview, refer to Nilsen (2015).
CFIR, Consolidated Framework for Implementation Research (Damschroder et al 2022); ERIC, Expert Recommendations for Implementing Change (Powell et al 2015); Outcomes for Implementation Research (Proctor et al 2011).
An important part of assessing the feasibility of implementation is considering equity and access to care to ensure that innovations do not inadvertently reproduce existing inequities. Assessing feasibility through an equity lens includes (1) making health equity explicit during the design process, for example, through leadership commitment, designated equity funding, and accountability structures, and (2) aligning strategies across system levels so that macro-level policy and local delivery are complementary—two “cardinal conditions” for equitable implementation (Bouckley et al 2025). Frameworks such as the Health Equity Implementation Framework can guide the process of designing and implementing with equity in mind (Woodward et al 2019). The Health Equity Implementation Framework makes explicit how recipient factors (eg, health literacy, cultural beliefs, competing demands), provider-level influences (eg, bias, attitudes, training), and structural determinants (eg, policy and regulatory environments, organizational resources and culture, and broader societal inequities such as stigma or discrimination) interact, with the clinical encounter serving as the point where the innovation is delivered and where these determinants converge. The framework can be applied by pairing disaggregated quantitative monitoring (eg, reach, adoption, fidelity) with qualitative interviews that capture the perspectives of underserved populations.
Implementation-Effectiveness Hybrid Designs in Oral Health Research
Determining implementation outcomes is crucial for assessing the effectiveness of oral health interventions in real-world settings. Implementation outcomes may include user acceptability of an intervention, the extent to which it reaches its intended target population, or whether it drives subsequent positive actions, such as seeking dental care or adopting recommended behaviors. In the context of research, one approach is to use an implementation-effectiveness hybrid study to evaluate the clinical effectiveness of an oral health screening tool while gathering implementation data (Curran et al 2012). If there is strong face validity regarding the intervention’s effectiveness, minimal risk, and organizational support (eg, a novel fluoride varnish protocol for a school-based dental program), a study design that places greater emphasis on testing the implementation may be more appropriate than testing only the efficacy while still gathering data on clinical outcomes (Curran et al 2012). Integrating implementation research early in the research pipeline allows for flexibility, including bypassing efficacy trials for low-risk interventions or supporting bottom-up interventions that are developed directly within the real-world settings, as seen during the COVID-19 pandemic (Appleton et al 2021).
Use of Implementation Strategies and Documentation
Once contextual barriers and facilitators have been identified, the next step is to select and tailor implementation strategies that address these determinants (Powell et al 2015). These strategies aim to improve the adoption, acceptability, and sustainability of the innovation (Powell et al 2015). For example, if a mobile technology is being developed to support public health practices, a local needs assessment is essential to understand barriers such as literacy, access, and user preferences. The local assessment will help determine if the intervention is feasible and sustainable. Identifying a clinical champion within the care setting can help drive stakeholder buy-in, while partnering with community organizations can promote digital literacy (Ngiam et al 2022). In parallel, engaging data experts is necessary to establish systems for tracking usage patterns, user demographics, and engagement metrics. Finally, the health care system should be prepared to support the use of the mobile health tool, such as training dentists to interpret the results and ensuring the infrastructure is in place to respond to changes in patient demand and care pathways.
The selection of these strategies can be further informed by implementation theories such as the COM-B model, which conceptualizes behavioral change through 3 necessary components: capabilities (eg, knowledge, skills), opportunities (eg, physical and environmental), and motivation (eg, beliefs, habits) (Michie et al 2011). For example, in the case of a mobile oral health app, capability can be addressed through training providers to use the app confidently, opportunity can be increased by ensuring multilingual access, and motivation can be enhanced through clinicians to highlight the value of digital screening to their patients. Each strategy should be clearly documented, including its rationale, specific actions, responsible individuals, timelines, and intended implementation outcomes. For example, a tracking system within the mobile health app could monitor indicators such as patients using periodontal risk assessments, patient demographics, usage patterns, and technical issues. Regular evaluations (eg, quarterly) would help refine the implementation process and provide a more comprehensive assessment at 12 months to inform its long-term sustainability.
Real-World Practice: Lessons from Different Contexts
While some interventions may be rapidly scaled up through top-down initiatives led by enthusiastic funders, hospitals, or health systems, these efforts can overlook the complex realities of embedding interventions within existing health systems and policies. This urgency to scale an intervention is understandable—translation of clinical innovations into routine practice is widely recognized to take many years (Rubin 2023), slowed by the need for validation studies, regulatory hurdles, fragmented health systems, remuneration challenges, and resistance to change among clinicians and within the community. This inefficiency is part of a broader challenge in research, where approximately 80% of research investments fail to yield meaningful public benefit (Chalmers and Glasziou 2009). Implementation research balances speed with evidence, local stakeholder engagement, and effective strategies. Three examples illustrate the real-world complexities of putting research into practice and, when these challenges are addressed, the successes that can be achieved, along with the transferable lessons from each case.
Example 1: Childsmile Program in Scotland
The Childsmile program (https://www.childsmile.nhs.scot/) exemplifies how a context-sensitive approach can achieve improvements in oral health. It was developed in response to the high prevalence of caries among children in Scotland—about 60% of 5-y-olds in the early 2000s—with the greatest burden in lower socioeconomic groups. To address barriers to care, Childsmile aimed at delivering both universal and targeted preventive measures across dental, community, and education settings (Ross et al 2023). This included supervised toothbrushing, targeted fluoride varnish, and family and community support, with greater provision for children in deprived areas. Evaluation of the program has been ongoing since its inception in the mid-2000s, using logic models, process evaluation, and data linkage to track clinical and implementation outcomes. A key part of Childsmile is the supervised nursery and kindergarten toothbrushing program, supported by accredited training for dental nurses to extend their role in providing interventions, such as applying fluoride varnish alongside toothbrushing. Implementation evaluations have documented high adoption across Scotland and are independently associated with reduced odds of caries (Macpherson et al 2019; Ross et al 2023). By 2020, fewer than one-third of 5-y-olds had caries experience, although children in the most deprived areas were still about 3 times more likely to have decay than their more affluent peers. Evidence is also limited on whether the program influences home-based behaviors such as daily toothbrushing, with additional concerns about implementation fidelity, such as the quality of preventive advice delivered and the consistency of children’s participation in supervised nursery toothbrushing.
Example 2: Health Buddy App in Singapore
SingHealth is Singapore’s largest health care cluster, comprising public hospitals, community hospitals, national specialty centers, and 8 polyclinics, which account for more than 5 million patient visits per year. In 2011, it launched the Health Buddy app to improve patient access to health services. In 2024, the app integrated specialty care modules that include personalized assessments for conditions such as sleep apnea and anxiety in older adults, alongside an oral health screening tool featuring a questionnaire-based nomogram developed using local data (Sim et al 2022) for self-assessed periodontitis risk and the Oral Frailty Index-8 (Tanaka et al 2021) to assess oral frailty. While the integration reflects a systems-level effort to provide a seamless, patient-facing platform and to increase the oral health literacy of patients as part of nationwide preventive care (Foo et al 2023), key implementation questions remain unclear, such as the app’s reach, user engagement, equitable access, feedback mechanisms, outcome monitoring, and long-term sustainability. Those most at risk of oral frailty and periodontitis are also less likely to possess the digital literacy necessary to benefit from these tools (Dibello et al 2021; Ngiam et al 2022). In addition, the app’s design raises questions about how personalized recommendations integrate into existing pathways, with uniform recommendations to seek a dental consultation regardless of risk score. Given that more than half of adults in Singapore already visit the dentist at least annually (Lee et al 2024), the app may offer value through early detection and patient engagement between visits. However, the system’s readiness to respond to these needs remains unaddressed, with many local dental professionals lacking the training and confidence to manage frailty and periodontitis, including related “personalized” care issues such as dysphagia, xerostomia, or diabetes (Chuang et al 2024; Tay et al 2025).
Example 3: MeTree in the United States
In primary care, collecting a family health history detailed enough to guide risk stratification is often hindered by competing clinical demands and limited patient knowledge. To address this barrier, MeTree was developed as a co-designed, patient-facing Web-based platform in the United States to collect family health history and calculate the risk of cancer or cancer syndromes, including hereditary syndromes (eg, familial adenomatous polyposis, Cowden syndrome), where early signs may present in the oral cavity, such as papillomas, jaw osteomas, and supernumerary teeth. The development process actively engaged patients and primary care clinicians, with pilot testing conducted to ensure usability and workflow integration. The platform provides prevention-guided strategies, and using an implementation-effectiveness hybrid design, the proportion of patients meeting criteria for recommended services rose from 10.5% preimplementation to 80.7% postimplementation, while overall agreement with risk-stratified recommendations improved from 1.1% to 16.1% (Orlando et al 2013; Orlando et al 2016). MeTree’s success in patient uptake, acceptability, and equity engagement may be attributed to its continuous adaptation based on contextual needs to improve uptake, alignment with clinical workflows, and its ease of use (Wu et al 2013; Wu et al 2019).
Conclusion: Lessons Learned and the Path to Wider Impact
By using logic models and hybrid study designs, researchers can bridge the gap between research and the communities who stand to benefit from these interventions, helping to reduce inequalities in oral health care while generating learning that can be shared to support implementation elsewhere.
An important mechanism for ensuring that evidence translates to everyday care is the development and use of clinical practice guidelines. Produced by professional bodies or public health agencies, guidelines synthesize evidence into actionable recommendations, and when adapted to the local workforce, financing, and infrastructure, they can accelerate uptake and inform financing and monitoring arrangements (Panteli et al 2019). Furthermore, the WHO Global Strategy on Oral Health calls for country-specific, evidence-based practice guidelines as a foundation for implementation (World Health Organization 2024). When well-adapted and disseminated, guidelines act as powerful enablers of implementation, but when absent or misaligned, they become barriers that research must address.
Sustainability also requires structural commitments across health systems, including workforce and financing. From a financial perspective, integrating oral health into primary care funding models can safeguard against reliance on short-term project-based funding. Workforce development through continuous training, upskilling, and task shifting can enable not just dentists but other health professionals to play an expanded role in prevention and early management. At the systems level, incorporating oral health within national noncommunicable disease strategies, strengthening referral and care pathways, and incorporating implementation monitoring into routine electronic health record systems are critical steps.
Achieving equitable improvements in oral health requires strategic partnerships between the WHO and key stakeholders such as the International Association for Dental, Oral, and Craniofacial Research and the FDI World Dental Federation as well as other international and national dental organizations; these are essential for advancing intersectoral collaboration. These alliances can assist leaders, researchers, and practitioners on the ground in translating global strategies into locally relevant actions by fostering knowledge exchange, aligning research agendas with public health priorities, and supporting the development and implementation of evidence-based policies and interventions. Implementation research must also be applied at the local level when new programs, policies, or procedures are introduced, to ensure that context-specific barriers and facilitators are addressed and that innovations are adapted, evaluated, and sustained.
Author Contributions
J.R.H. Tay, contributed to conception and design, data analysis and interpretation, drafted and critically revised the manuscript; H.B. Bosworth, G.G. Nascimento, contributed to conception and design, data analysis and interpretation, critically revised the manuscript; C.H. Fox, J.E. Gallagher, N.S. Jakubovics, F. Schwendicke, contributed to design, data analysis and interpretation, critically revised the manuscript. All authors gave final approval and agreed to be accountable for all aspects of the work.
Footnotes
Acknowledgements
J.R.H. Tay received PhD funding from the Singapore National Medical Research Council (NMRC) Research Training Fellowship (RTF24jan-0007).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Registration Number
Not applicable.
Availability of Data
Data sharing is not applicable to this article as no datasets were generated or analyzed during the current study.
