Abstract
Silicone elastomers filled with silica exhibit a softening effect under cyclic loading, also known as the Mullins effect. There are various theories as to the origin of this effect. One theory cites the breaking of secondary bonds as the reason, while another theory cites molecular slipping and the loosening of entanglements as the reason for the softening. In contrast to previous studies, foamed silicone rubbers are investigated here. In addition to the silica, the samples investigated here contain thermally expandable microspheres, resulting in two types of filler (silica and the microspheres as thermoplastic particles). Two different kinds of these microspheres with different particle sizes and expansion temperatures are used. To study the effect, hysteresis tensile tests (Mullins tests) as well as FTIR analyses, density measurements and microscopy images were carried out. The Mullins tests were evaluated for their curve shape, their hysteresis area of the first cycle and the residual strain. The residual strain shows a correlation with the porosity, as the larger microspheres have a higher porosity and a higher residual strain. The area of the first hysteresis correlates with the number of microspheres, but not with the porosity. The larger microspheres have a smaller hysteresis area but a higher porosity. The FTIR analysis showed that the stabilizer used to produce the microspheres is based on silicon oxide. As a result, there are secondary bonds between the microspheres and the silicone elastomer, which are broken at the beginning.
Introduction
Liquid Silicone Rubber (LSR) is an elastomer that offers excellent properties. It is considered to be very weather-resistant to external influences as well as cold-flexible down to - 50°C and temperature-stable up to 200°C, and even up to 250°C for short periods. LSR is a two-component material, with component A containing the catalyst and component B containing the crosslinker and an inhibitor. The base of LSR consists of vinyl-terminated polydimethylsiloxane, to which silica is added as an active filler. The silica is needed to improve the mechanical properties. For example, the tensile strength increases from 0.4 MPa to up to 10 MPa. 1
Since LSR is a high-priced material, it makes sense to produce foams and thus reduce the density and material costs. These materials could be used for various purposes like insulation (thermal or acoustic), protection (shock absorption or impact damping), resistance (waterproof or moisture) and buoyancy (swimming).2,3 Initial attempts to foam LSR in the injection molding process have been carried out using gas injection. In this process, an inert gas was added to the LSR at the screw, which expanded due to the pressure drop when it entered the cavity, thus producing a foam. 4 However, this process has not been able to establish itself industrially because the machine technology is very expensive and high-maintenance.
Therefore, another method for foaming LSR in the injection molding process was developed at the University of Kassel. Here, water is added as a blowing agent. Since water is liquid at room temperature and only changes to the vapor phase at over 100°C, it is well suited as a blowing agent due to the temperature control in LSR injection molding. In addition, there are fewer problems with the high gas permeability of LSR.5–7 In addition, the water can be added as a liquid via color metering or mixed into the LSR before processing, eliminating the need for special machine technology.
When looking at other silicone rubbers, other processes are commonly used to produce foams. For example, in the area of room temperature crosslinking systems, silicone foams are produced by hydrogen elimination during the crosslinking reaction. Shaping is limited here, as foaming takes place in open molds, which distinguishes it from the injection molding processes used here.8,9 Furthermore, additives such as lignin can be used in silicone rubber, which split off gases during processing and thus produce a foam. 10 The last two processes mentioned are chemical foaming agents.
Physical foaming processes are used for high consistency silicone rubber (HCR), for example. By adding supercritical carbon dioxide, silicone foams are produced in peroxide-curing systems. 11
Another method for foaming LSR in the injection molding process was also developed at the University of Kassel. 12 In this process, thermally expandable microspheres are added to the uncrosslinked LSR as blowing agents. Due to the temperature control, the microspheres remain unexpanded in the cooled screw (20°C) and expand only after injection into the heated cavity (140°C to 200°C). The impact on both the mechanical properties and the cell structure has been previously documented.13–15 Typically, up to 5 wt.-% of microspheres are incorporated to attain density reductions or porosities of up to 50 %. In many instances, the amount used falls between 2 wt.-% and 3 wt.-%, as this yields significant porosity while minimizing the loss of mechanical properties.15–18 Moreover, at 3 wt.-%, expansion during processing reaches saturation, limiting further expansion. 15 This is a physical foaming process.
The material combination of polysiloxanes and thermally expandable microspheres has already been used to produce porous ceramics according to the literature.19–22 The material combination was in the first step either injection-molded, extruded or compression-molded, whereby the studies on this differ in that a significantly higher proportion of microspheres (up to 15 wt.-% 21 ) was used. In the second step the material was pyrolyzed at 1200°C for 1 h to create a ceramic. In addition, a silicone resin with additional components (carbon black, Al2O3 and Y2O3) and no silicone rubber was used.19–22
In the case of filled elastomers, a stress-induced softening effect can often be observed after a load when further loads are applied. This effect was named after its inventor and referred to as the Mullins effect.23,24 The cause of this stress-induced softening varies depending on the material studied and is summarized in detail by Diani et al. 25 Studies on the Mullins effect of silica-filled silicone rubber were carried out by Clément et al. and Hanson et al.26,27 Clément et al. postulated that the stress softening comes due to the detachment of the polymer chains from the filler particles. 26 This hypothesis was first proposed by Bueche for SBR28,29 and was transferred to filled polysiloxanes by Clément. Likewise, Zhang et al. 2021 used this hypothesis for polydimethylsiloxane (PDMS) with glass microspheres as an explanation for the Mullins effect with macroscopic particles. Zhang et al investigated the stress softening of PDMS up to an elongation of 25 % and found that an increase in the proportion of microspheres in the curves leads to a stiffening of PDMS in this low elongation range. 30 As a second influence, the proportion of crosslinker was investigated. Here, a stiffening was also determined with a higher proportion. Since both the increase in chemical crosslinking sites (higher proportion of crosslinker) and the increase in physical crosslinking sites (higher proportion of glass microspheres) resulted in an increase in the hysteresis area and the residual strain. The maximum stress decreased with the number of cycles, but increased with an increasing proportion of glass microspheres or crosslinking sites.
Another hypothesis regarding the Mullins effect of silica-filled silicone rubber was put forward by Hanson et al 2005. 27 By examining the specimens in two dimensions, it was determined that the Mullins effect did not occur with a second strain direction rotated 90° from the first loading direction. Despite the second loading, the curves were at the same height level. Based on this observation, Hanson et al. postulated that the Mullins effect occurs due to molecular slippage and associated dissolution of entanglements.
Nazari et al. highlighted that the mechanical properties of foams primarily rely on the base material since the pores exhibit no significant influence. 31 However, this assertion doesn’t directly apply to the material combination addressed in this paper. Here, the structure isn’t solely composed of pores but comprises a filler comprising a thermoplastic shell and a gas. Additionally, the mechanical properties are influenced by vulcanization and the foam’s structure. 32
Recent publications on the Mullins effect of silicone rubbers deal with the mechanical behavior for the determination of simulation models for numerical applications. This involves the determination of the curve characteristics and the calculation them to simulate industrial applications using FEM.33–35 Other studies have explored how the chemical composition or the use of different silicas affect the Mullins effect.36,37
This publication aims to explain the influence of the addition of thermoplastic microspheres to the LSR on the Mullins effect. This will allow conclusions to be drawn about possible interactions of the microspheres with the LSR matrix. In contrast to Zhang et al., 30 the experiments presented here investigated significantly higher strains up to 300% strain instead of 25% strain by Zhang et al. 30 and thermoplastic microspheres with a flexible shell instead of glass microspheres.
Materials and methods
Materials and injection molding parameters
aunexpanded.
Various proportions (1 wt.-%, 2 wt.-%, and 3 wt.-%) of the unexpanded blowing agent were blended with a hand mixer into the uncrosslinked Liquid Silicone Rubber (LSR) in each component. A homogeneous mixing quality was tested in unpublished preliminary work. Additionally, the compact LSR was examined as a reference. Due to geometry considerations, these low loading quantities result in a density reduction of up to 53 %, 15 which is why higher amounts are not deemed practical.
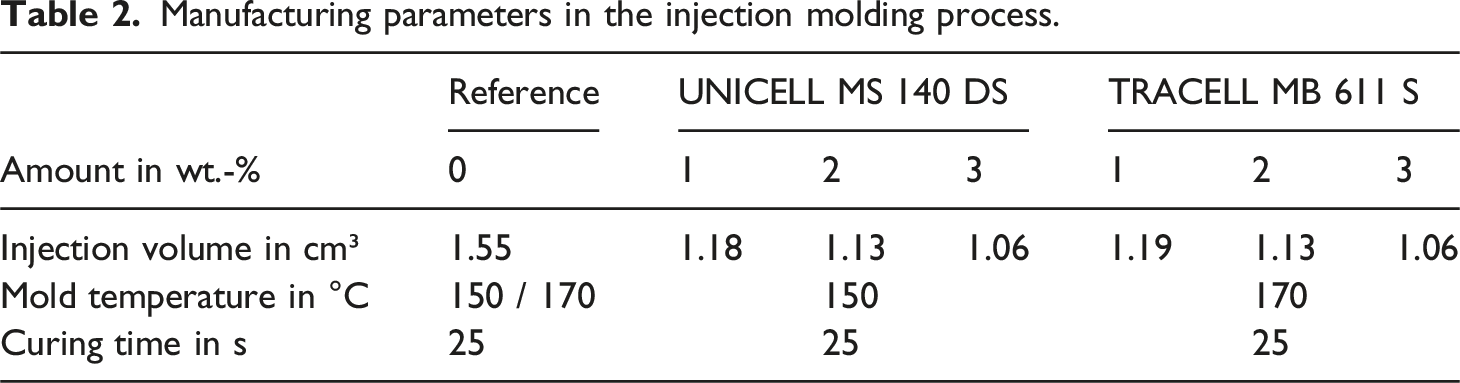
The tensile test bars with geometry S2 following DIN 53504 (cf. Figure 1) were produced by injection molding. A Babyplast 6/610P injection molding machine from Christmann Kunststofftechnik GmbH with a cartridge metering system and a piston injection unit was used for this purpose. Both components of the LSR were mixed directly in the 500 mL cartridge with the blowing agent and thus inserted into the injection molding machine in the uncrosslinked state. There, the material was injected into the heated mold, where it crosslinked in the mold for specimen. Due to the very long cross-linking time for LSR and a dimensionally stable sample when removing the samples, sufficient cross-linking can be guaranteed. In the case of incomplete cross-linking, post-expansion occurs after demolding. Depending on the blowing agent, the mold temperature had to be adjusted to ensure good foam quality. Due to the use of an LTC-LSR, high degrees of cross-linking are achieved at low mold temperatures. For the different blowing agents and their amounts, a filling study was carried out in each case to achieve the lowest density. The manufacturing parameters are shown in the following Table 2. The difference between the mold temperature and the maximum temperature of the microspheres does not pose a problem during processing. The maximum temperature of the microspheres indicates the point at which the shell is too thin and the microspheres start to shrink as the blowing agent diffuses through the shell.48–50 As the silicone rubber acts as an additional shell, leakage of the blowing gas can be prevented. This effect has already been demonstrated in PP foams by Kawaguchi et al.
48
The higher processing temperature is therefore not a problem. Used tensile test bar of geometry S2 with a = 75 mm, b = 12.5 mm and a thickness of 2 mm according to DIN 53504. Manufacturing parameters in the injection molding process.
Test methodes
The tensile test bars S2 produced were analyzed for density on the one hand and in the Mullins experiment on the other. In addition, microscopic images of the foams as well as Fourier transform infrared spectrometric analyses of the microspheres were performed.
Density
Archimedes’ principle was used to determine the density. Five test specimens were weighed first in air and then in demineralized water. From the density of the water and the two weights, the respective density could be calculated by formula (1).
Here
The porosity 
Hysteresis tensile tests
To investigate the Mullins effect, hysteresis curves were recorded on an Inspekt Table 5k universal testing machine from Hegewald and Peschke using a long-travel extensometer. A total of 15 cycles were run, whereby the first 5 cycles went up to 100 % strain, the second 5 cycles up to 200 % strain and the third 5 cycles up to 300% strain (cf. Figure 2). Five cycles were selected for each strain because own, unpublished preliminary work tested that very little change occurred thereafter. In each case, the strain was reduced to the pre-stress of 0.1 MPa. The test speed was 100 mm/min in order to realize a reasonable test time. The clamping length was 55 mm and the distance of the extensometer at the beginning of the preload was 20 mm. The test speed to preload was 50 mm/min. Four specimens were tested for each material and blowing agent content. Exemplary, schematic illustration of the stress-strain diagram of the hysteresis curve with marked hysteresis area and residual strain.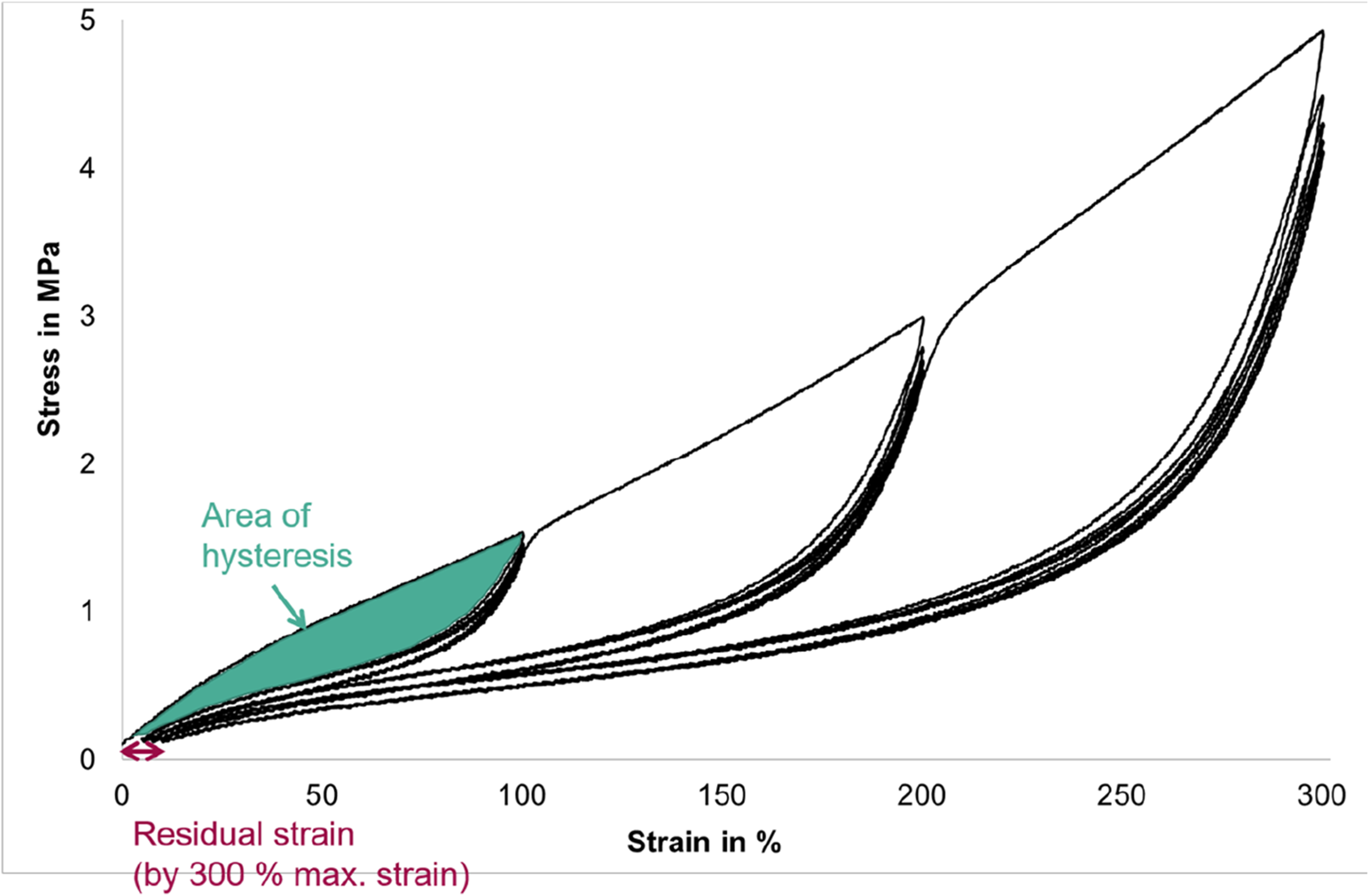
On the basis of the measurement curves, the differences in the curve characteristics, the residual strain as well as the hysteresis areas can be analyzed. The measurement curves provide information about secondary and primary bonds between the microspheres and the LSR matrix. In this context, the curve of the first strain per strain level is particularly important.
The residual strain describes the value of the strain after unloading, when the preload is reached again. The residual strain after the fifth cycle is evaluated here, i.e. before the next load level is approached.
The hysteresis area (Figure 2, in green) between the first load curve and the first unload curve indicates the proportion of irreversible damage caused by the tensile load. It is calculated by calculating the areas of the load curve and the unload curve and then subtracting the area of the unload curve from that of the load curve.
FTIR
To analyze and compare the chemical composition of the microspheres, FTIR analyses were performed. The IR Affinity-1S from Shimadzu was used as ATR-FTIR with the MIRacle and a ZnSe crystal. A wavelength range between 700 cm−1 and 4000 cm−1 was tested. The resolution was 4 cm−1 and 32 scans were performed. No correction of the data was performed.
Microscopy
To compare the foam structure, images were taken in front of cross-sections of the center of the tensile test bar (cf. Figure 3) using a VK-X3000 confocal laser scanning microscope from Keyence. A 20x magnification was used and 35 (5 × 7) individual images were assembled for each sample. The samples were cut with a razor blade. Sample removal position for the microscopy images.
Results
Density
The density of the compact reference components is 1.114 g/cm³. By adding thermally expandable thermoplastic microspheres, the density can be reduced by up to 44% at 3 wt.-% MB 611 S to 0.620 g/cm³. In the case of MS 140 DS, a porosity of 30%, i.e. the density of 0.785 g/cm³, could be achieved with 3 wt.-%. Thus, the degree of foaming of the MS 140 DS is lower than that of the MB 611 S (cf. Figure 4). This can be explained by the smaller particle size of the MS 140 DS (see Table 1). Since, according to the literature,
51
the volume can increase by up to 40 times, the larger microspheres can achieve a greater absolute volume change during expansion. This results in a lower density for the same weight fraction of microspheres. Porosity and density of foamed LSR samples as a function of the content of microspheres. The standard deviation is less than ±0.006 g/cm³, which is why it cannot be shown graphically. The porosity is calculated from the mean values and has no standard deviation.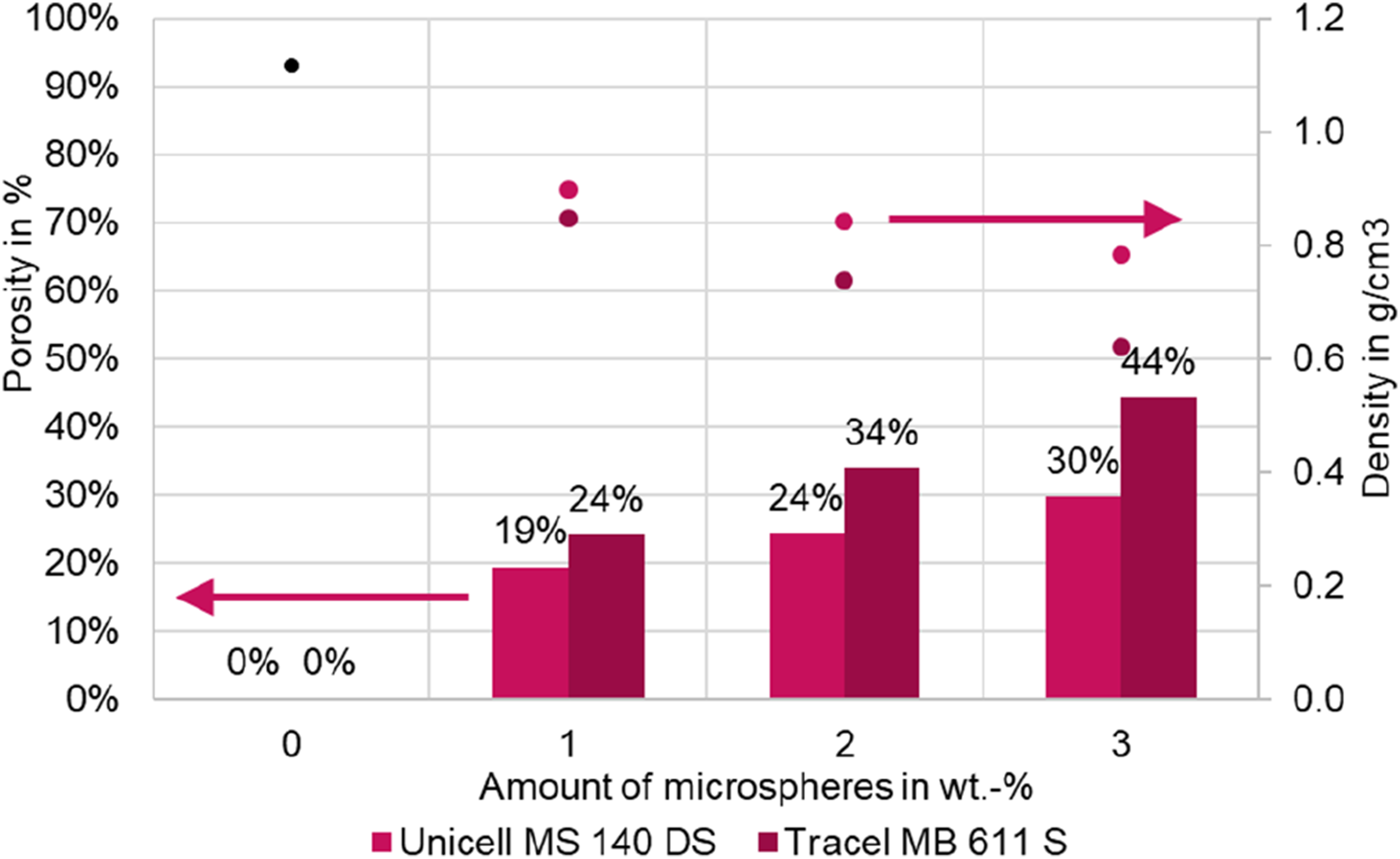
FTIR
The FTIR analysis compares the chemical structures of the thermoplastic microspheres. The spectrum reveals characteristic signals typical of microspheres made from a copolymer of methyl methacrylate, acrylonitrile, and methacrylonitrile.42,43,45 Small variations in the intensity of specific groups are noticeable between the two types (cf. Figure 5). Notably, both materials exhibit siloxanes (Si-O-Si) at around 1110 cm−1 and 1220 cm−1, consistent with Fredlund’s observation that silica particles are used as stabilizers during production.
42
FTIR spectra of the microspheres (TRACELL MB 611 S: black, UNICELL MS 140 DS: red).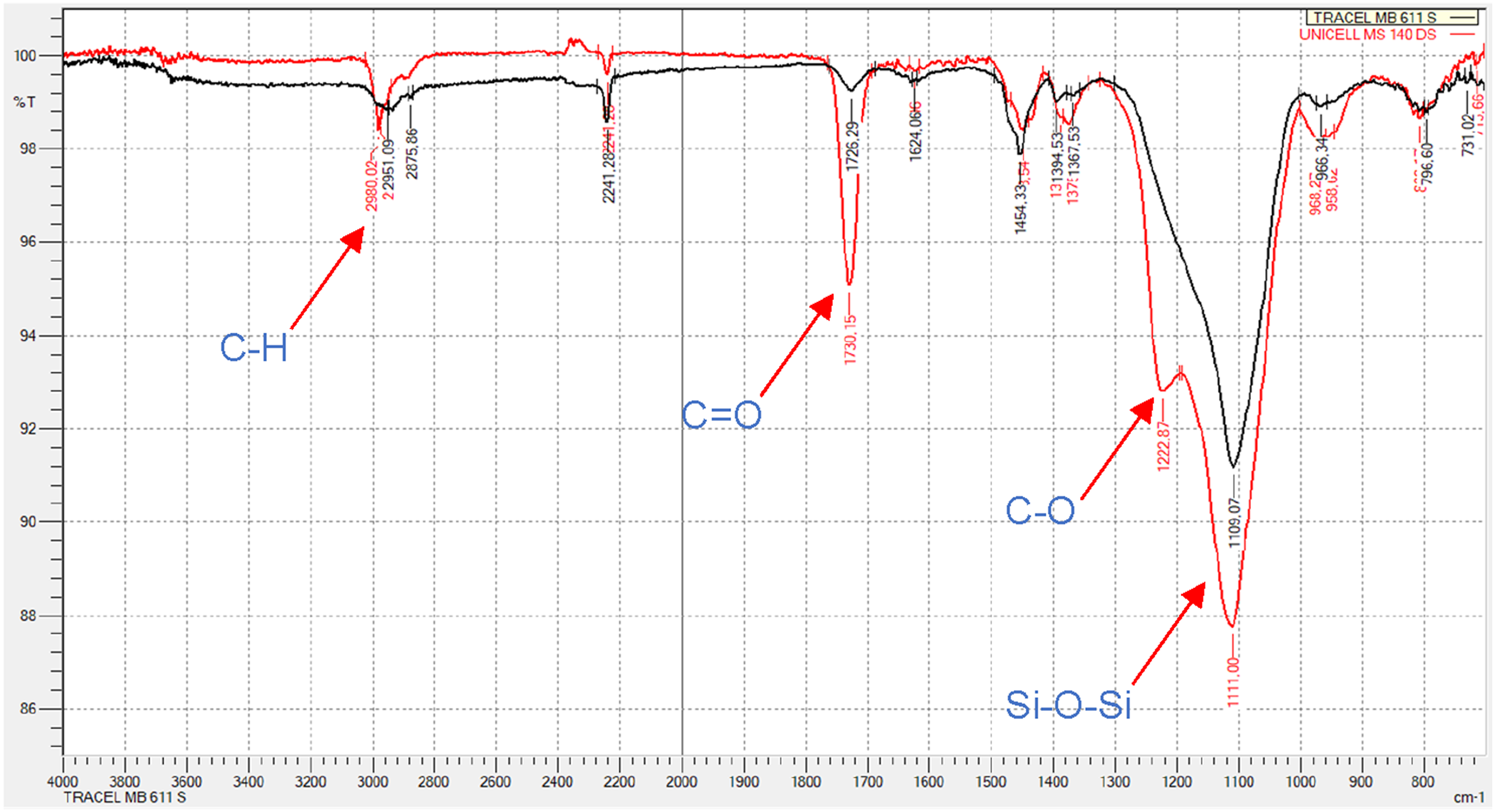
Another discrepancy appears at approximately 1725 cm−1, corresponding to C = O stretching vibrations, 42 indicating a higher proportion of C = O compounds in UNICELL MS 140 DS. Since the shell material typically comprises a copolymer of methyl methacrylate (MMA), acrylonitrile, and methacrylonitrile, the signal at 1725 cm−1 suggests a greater proportion of MMA. This higher MMA content leads to a lower expansion temperature.43,45 The higher intensity of the signal at 1222 cm−1 in the Unicell MS 140 DS can also be explained by the MMA as a monomer, since the stretching vibration of the C-O (ester bond) can be found between 1000 cm−1 and 1260 cm−152 and thus there is a superposition between the signals of the MMA and the Si-O.
The C-H-stretch vibration in the range from 2800 cm−1 to 3000 cm−1 arises from either the blowing agent or the polymer chain.42,45 Despite both microsphere types containing the same blowing agent (isopentane), differences in the polymer chain are discernible here. Further chemical analyses would be necessary to explore this discrepancy in more detail. Although this difference likely explains the distinct properties of the microspheres, it’s not deemed relevant for the interactions between the microspheres and the LSR.
Microscopy
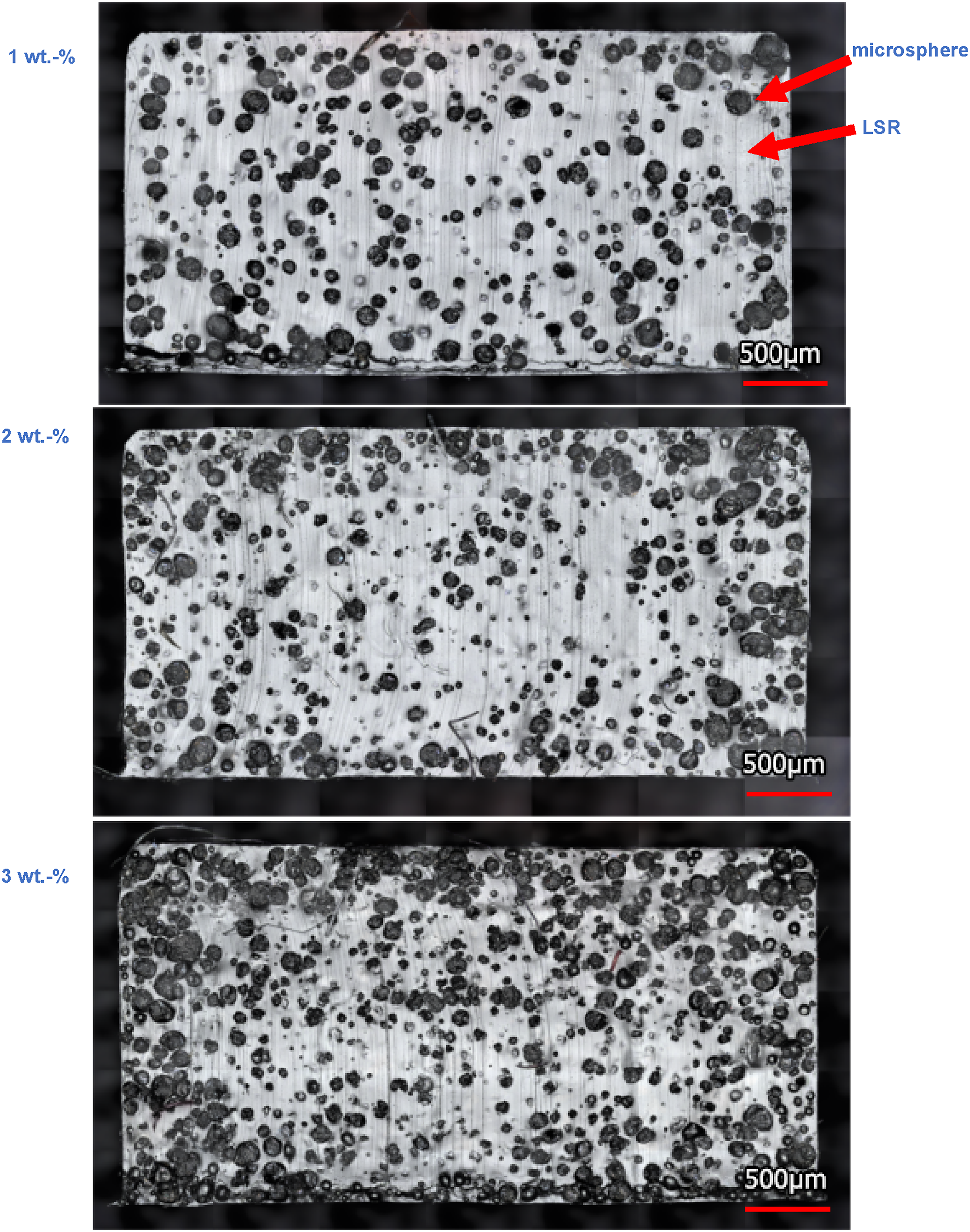
Based on microscopy images (cf. Figures 6 and 7), it was determined that significantly more microspheres are found in the cross-section of the UNICELL MS 140 DS than in the TRACELL MB 611 S. In addition, the smaller microspheres are significantly more homogeneously distributed than the larger ones. With the larger microspheres, more areas of matrix material can be identified, resulting in a smaller contact area between the microspheres and the LSR. Microscopy image of the cross section of the LSR with 1 wt.-% (top); 2 wt.-% (middle) and 3 wt.-% (bottem) UNICELL MS 140 DS; magnification: 20x. Microscopy image of the cross section of the LSR with 1 wt.-% (top); 2 wt.-% (middle) and 3 wt.-% (bottem) TRACELL MB 611 S; magnification: 20x.
Mullins effect
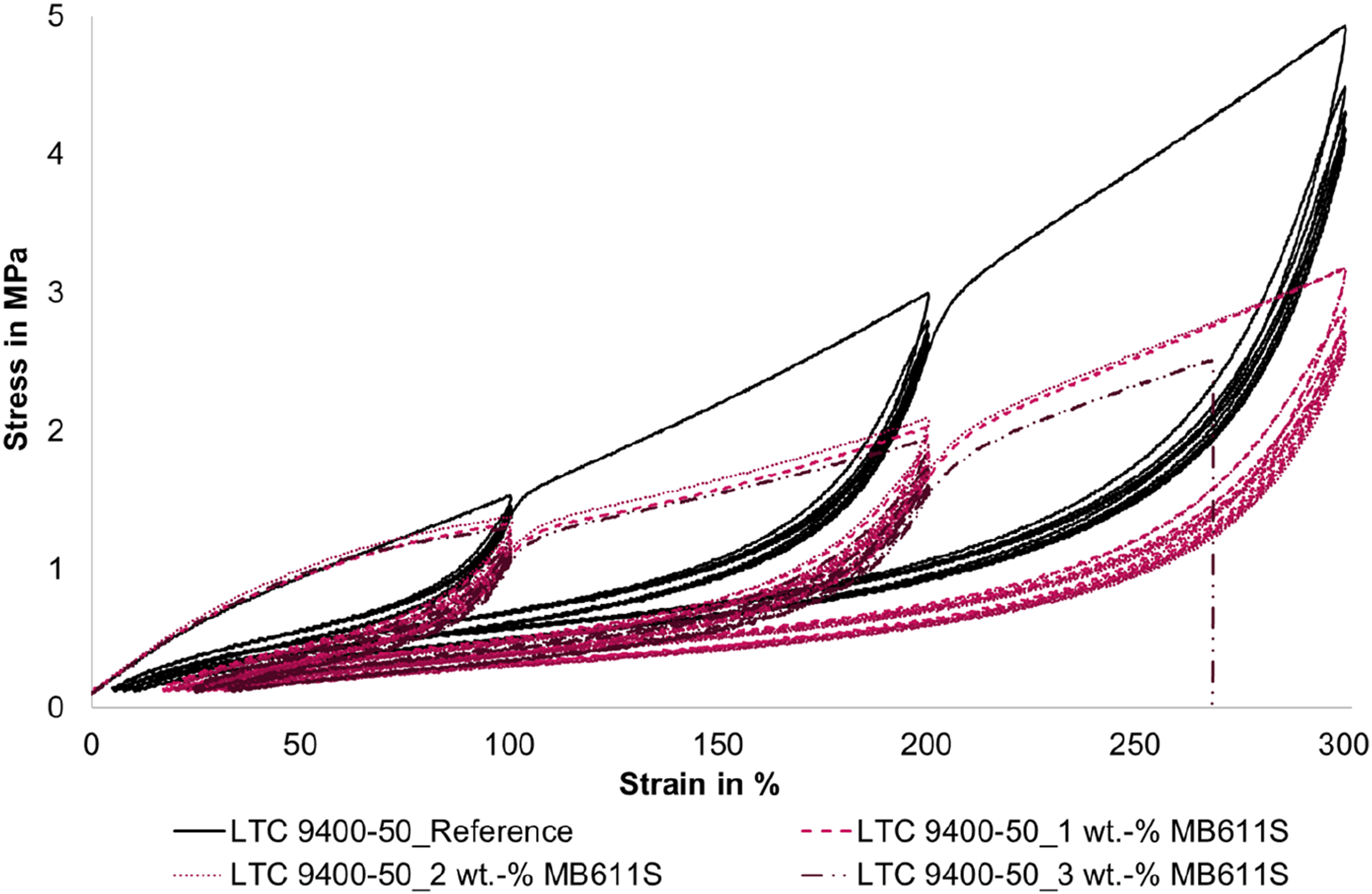
The measurement curves are shown in Figures 8 and 9. In addition, Figure 10 shows the first five cycles in more detail. It is very interesting to note that at the first load up to 100 % strain, the stress of the foamed tensile specimens is higher than that of the compact LSR. However, this behavior changes at the first loads of the higher strains (cycle 6 and cycle 9, respectively). In this range, the slope of the stress-strain curves of the foamed LSR specimens is significantly lower than that of the compact reference and the stress values are below the reference. This behavior is more pronounced for the smaller microspheres (MS 140 DS) than for the larger microspheres (MB 611 S). In addition, the measurement curves of the larger microspheres (MB 611 S) intersect with the measurement curve of the reference much earlier at 60-75 % strain. The curves of the smaller microspheres (MS 140 DS) intersect the reference curve only at about 100 % elongation. An exception is the measurement curve with 1 wt.-% MS 140 DS, which intersects the reference at approx. 60 %. Hysteresis curves of the LSR (LTC 9400-50) as a function of the proportion of blowing agent UNICELL MS 140 DS. Hysteresis curves of LSR (LTC 9400-50) as a function of the amount of blowing agent TRACELL MB 611 S; hysteresis curves stop at 3 wt.-% due to premature failure of the specimens. Section of the first 5 cycles up to maximum 100% elongation for LTC 9400-50 as a function of the proportion of UNICELL MS 140 DS.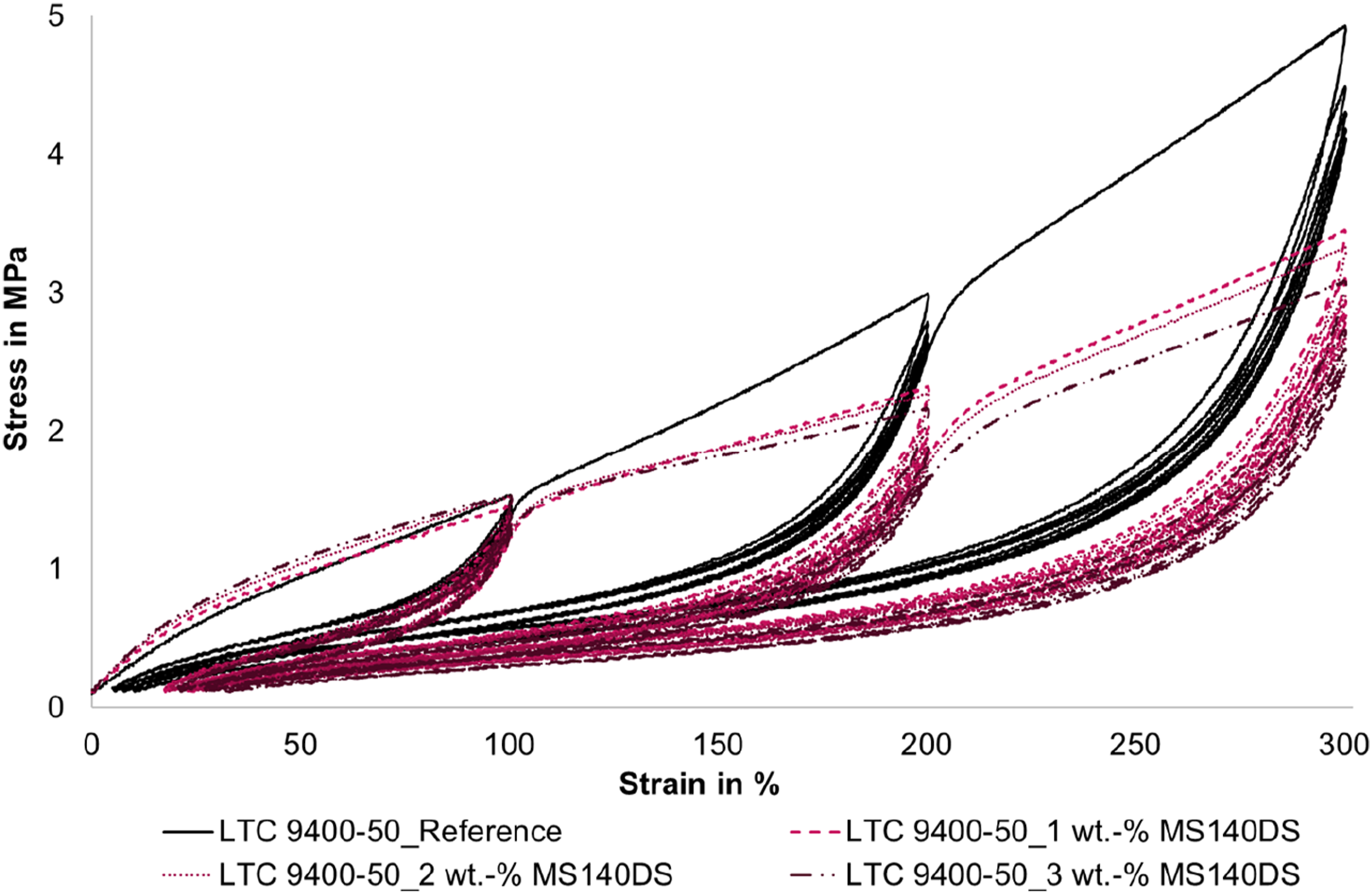
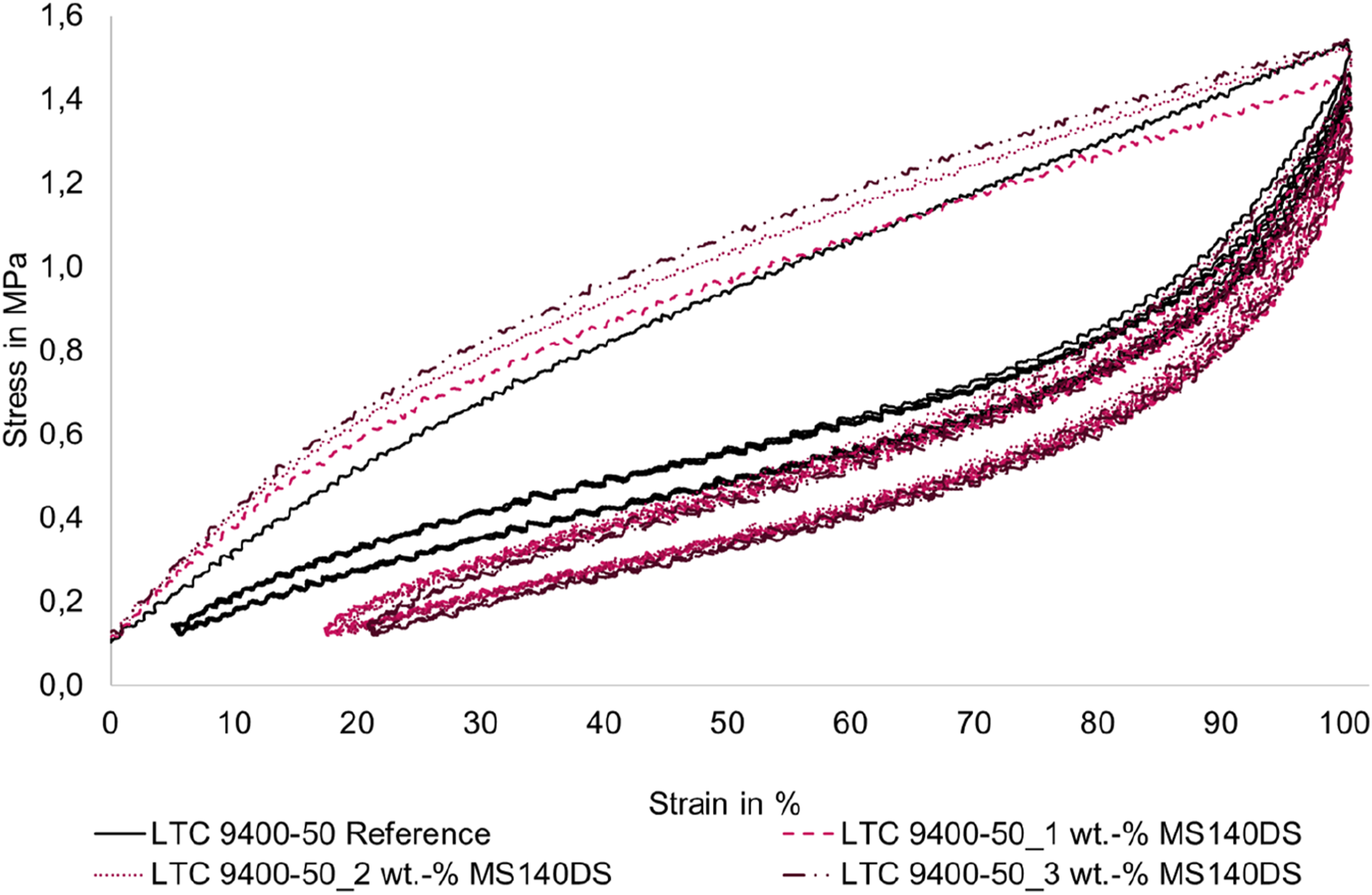
Since the initial loads at each strain level mirror the load curve of a standard tensile test, the explanation for this effect can be sought in the molecular changes of the specimens during the standard tensile test compared to. 53 The curve can divided into three regions. 53 The first area describes the stress increase up to the first inflection point. Here, in elastomers under tensile load, the entanglements of the polymer chains loosen and secondary bonds are broken. After this, the gradient in the second area flattens out, since the micro-Brownian movement causes stretching in conjunction with slippage of the polymer chains from one another. The slippage or stretching of the polymer chains is limited by the crosslinking sites. In the third region, the stress increases again more strongly, since there is now a direct pull on the main bonds. 53
Primary bonds are generally understood to be ionic bonds, atomic bonds and metallic bonds. In the case of polymers, atomic bonds are present within the molecular chains. The term secondary bonds refers to hydrogen bonds, dipole-dipole bonds and van der Waals bonds. The most probable bonds between LSR and thermoplastic microspheres are van der Waals bonds.
The stress at 25% elongation is about 0.6 MPa for the compact LSR in the first loading curve and increases to about 0.74 MPa at 3 wt.-% MS 140 DS. For the MB 611 S, this effect is much less pronounced (increase to 0.65 MPa). Because of the increased stresses experienced by the foamed LSR specimens in the initial region, coupled with the reduced matrix content resulting from foaming, it can be inferred that secondary bonds exist between the polymer chains and the microspheres. Consequently, more secondary bonds are likely broken in the initial region compared to the reference. Based on the chemical structure, it can be assumed that the secondary bonds are van der Waals forces, which increase the tension of the foamed samples. This effect is more pronounced with the smaller microspheres (MS 140 DS) than with the larger microspheres (MB 611 S) (cf. Figure 11). This implies that the smaller microspheres likely contain more secondary bonds. One reason for this could be a higher surface area of the microsphere shells since there are more microspheres at the same weight fraction due to the smaller microspheres (cf. Figures 6 and 7). Comparison between the hysteresis curves of the compact LSR, the sample with 3 wt.-% MS 140 DS and the sample with 3 wt.-% MB 611 S.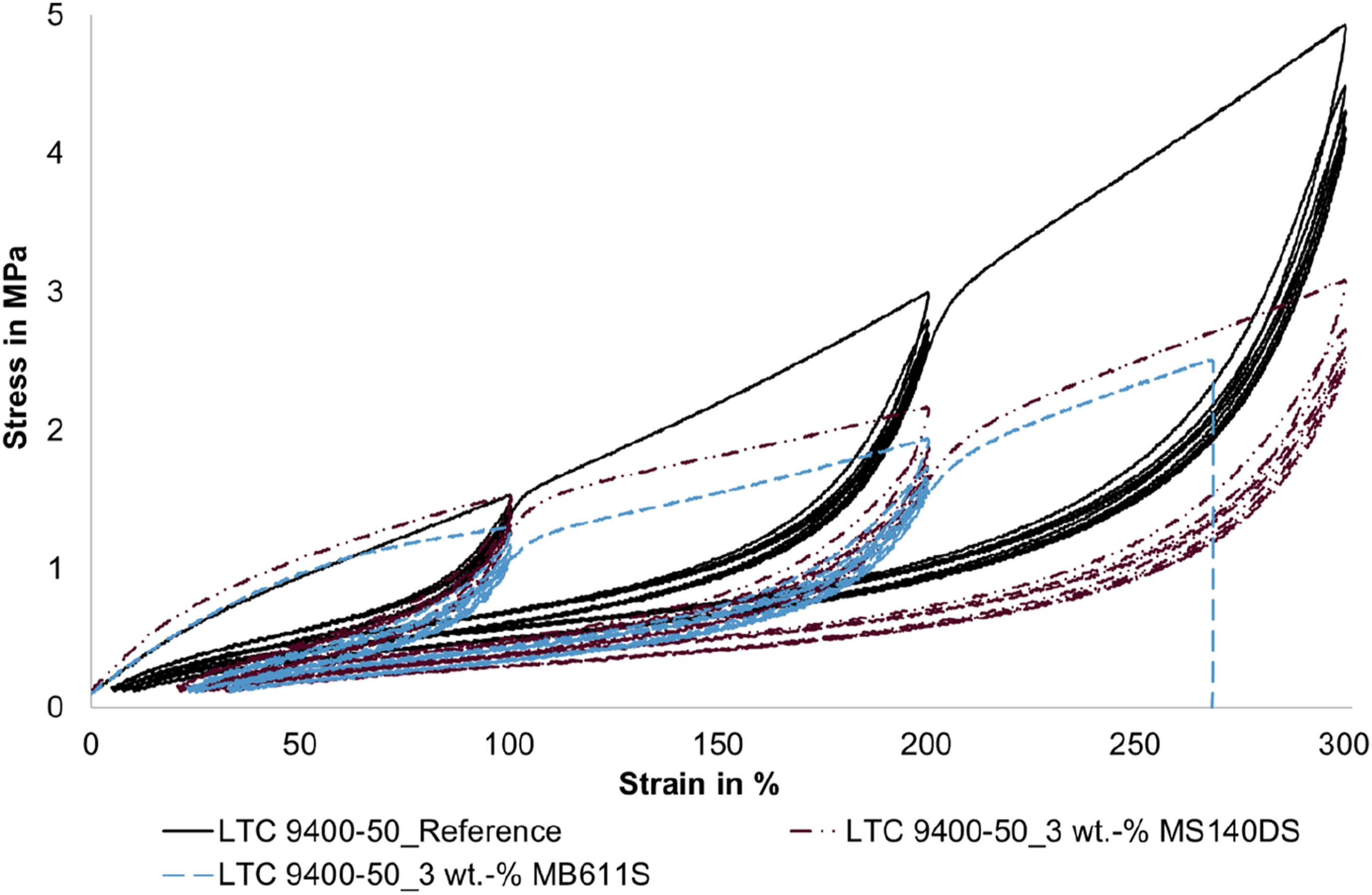
In the second and third regions, the observed changes can primarily be attributed to molecular alterations in the polymer chains. These alterations decrease in absolute number as the proportion of blowing agent increases. Consequently, the reduction in stress, leading to the flattening of the curve compared to the compact sample, can also be elucidated.
Area of hysteresis – dissipated work
In addition to the effect described above that the foamed specimens have slightly higher stresses at the first loading, the stress of the unloading curves of the foamed specimens is lower compared to the reference. To describe the fraction of irreversible damage or work loss, the hysteresis area between the first load and first unload curves is calculated. The results are shown in Figure 12. The hysteresis area is computed solely during the first cycle, reaching up to 100% strain. Within this range, secondary bonds are broken. Therefore, the determined area corresponds to the energy needed to break these secondary bonds. Area of hysteresis depending on the amount of blowing agent in comparison to the density. The standard deviation is below ± 0.015 MJ/m³, making it impractical to depict graphically.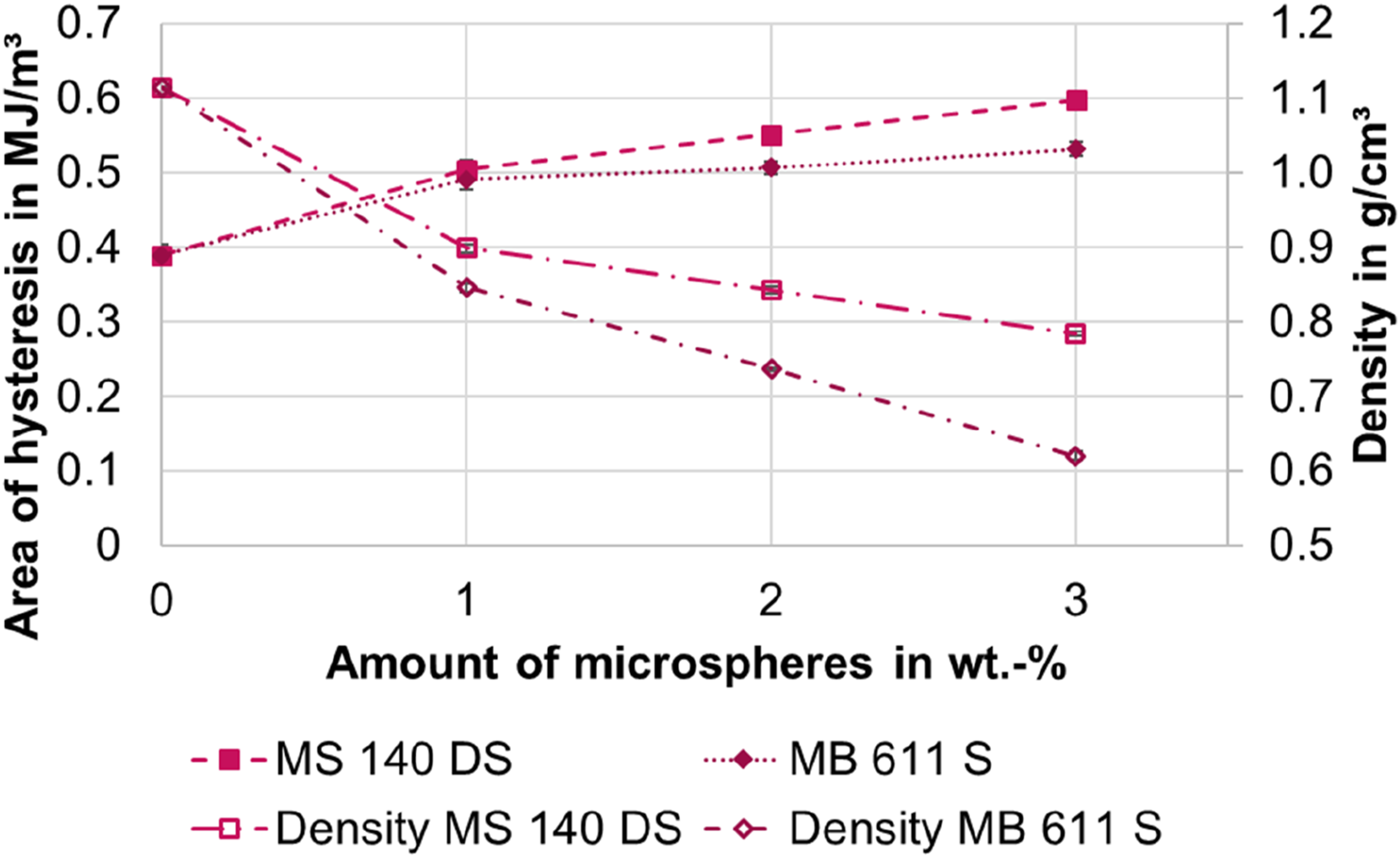
The hysteresis area increases as the amount of microspheres increases. However, it is noticeable that the hysteresis area of the smaller microspheres MS 140 DS is larger than the hysteresis area of the MB 611 S. This effect is opposite to the density reduction (cf. Figure 12). With the addition of the microspheres, the density decreases and the hysteresis area increases. With higher proportions of microspheres, the hysteresis area continues to increase and the density reduction also increases. However, if the samples with 3 wt.-% are compared with each other, the hysteresis area is larger for the MS 140 DS than for the MB 611 S, but the density is reduced to a greater extent for the MB 611 S. Thus, no direct correlation between the density reduction and the hysteresis area can be determined. Rather, there seems to be a correlation between the total surface area of the microspheres or the number of microspheres and the hysteresis area. For this, further more complex investigations of the cell structures are necessary. However, the microscopy images already show that there are significantly more particles in the smaller microspheres than in the larger microspheres for the same weight fraction. The number of cells was counted manually for 1 wt% with ImageJ as an example. Approx. 370 cells were counted for the MS 140 DS and approx. 1260 cells for the MB 611 S.
In summary, smaller microspheres demonstrate stronger secondary forces.
Residual strain
The residual strain describes the deformation at the end of the unloading of a cycle when the preload is reached again. Figure 13 shows how the residual strain increases as the proportion of microspheres increases. Increasing the maximum load also increases the residual strain. A stronger increase of the larger microspheres (MB 611 S) compared to the smaller microspheres (MS 140 DS) can be observed both with the increasing proportion of microspheres and with the increase of the maximum load. Residual strain depending on the amount of microspheres, left: UNICELL MS 140 DS, right: TRACELL MB 611 S; the standard deviation is below ±0.75% and cannot be mapped, value of 300% strain of MB 611 S is missing because the specimen break before 300% strain.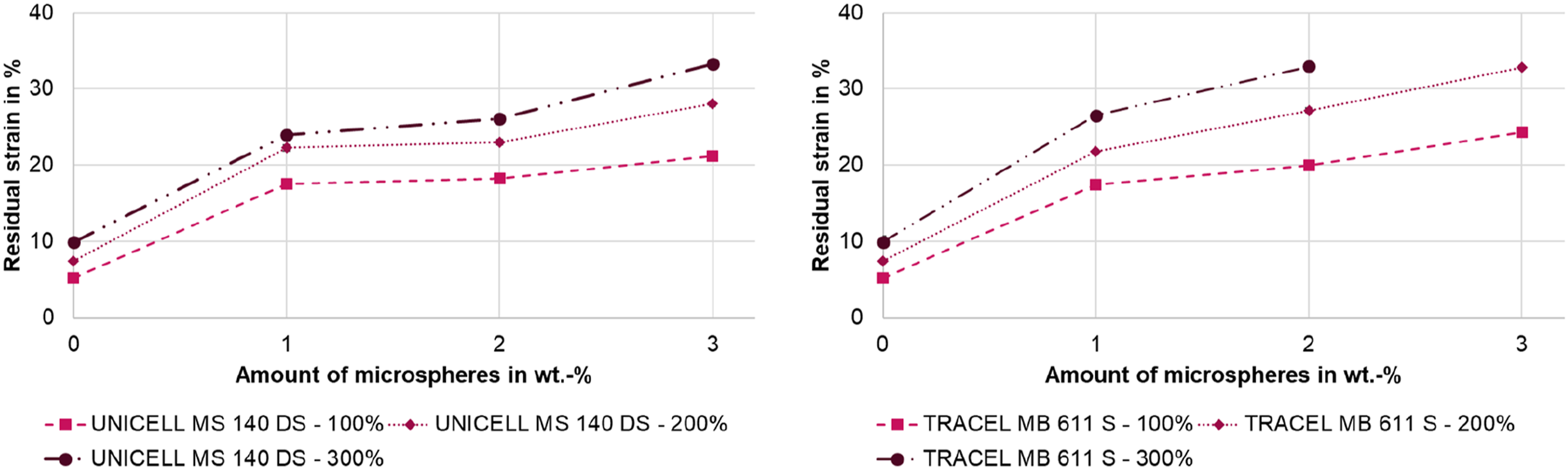
At 100% maximum strain, both microspheres exhibit similar residual strain at 1 wt.-%. However, the residual elongation for the larger microspheres (MB 611 S) increases more strongly with increasing amount of microspheres. This is an opposite trend compared to the hysteresis area from the first loading (see Figure 12) where the smaller microspheres have a larger hysteresis area with increasing proportion.
However, when the residual strain is compared to the density reduction, it is noticeable that the residual strain is larger at a higher density reduction. The larger microspheres (MB 611 S) exhibit greater density reduction than the smaller microspheres (MS 140 DS). Therefore, the percentage of the LSR matrix is lower for the samples with the larger microspheres. Due to the lower proportion of the LSR matrix, there are fewer crosslinking sites when considering the entire volume including the pores, which means that the recovery behavior is lower. Thus, the residual elongation is higher due to the lower recovery.
Discussion
The changes in mechanical properties due to foaming LSR with thermally expandable microspheres is explained in detail by Giesen et al. and Marl et al.13,14 The decrease in elongation at break, which is particularly noticeable in this publication at 3 wt.-% MB 611 S since it is below the third loading level of 300% elongation, can be attributed to the lower matrix content.
The residual strain does not provide any additional information on the mechanism of the Mullins effect in foamed LSR in these tests compared to the compact LSR. Since porosity is proportional to residual strain, the larger residual strain can be explained by the lower percentage of matrix material. A lower proportion of LSR means a lower number of crosslinking sites and thus a lower restoring force when considered over the entire volume.
Hanson et al concluded from his experiments for compact silicone rubber that the Mullins effect is caused by the dissolution of entanglements. 27 Clemént et al. previously postulated the conclusion of detachment of polymer chains from filler particles for PDMS. 26 However, compared to these experiments, since both sources dealt only with compact silicone rubber and not with foamed ones, both theories can be adopted for the experiments of compact LSR as well as for effects in the matrix material. For the changes in the curves of the foamed LSR specimens, on the other hand, other literature sources must be consulted.
Zhang et al carried out experiments with PDMS and glass microspheres. 30 The glass microspheres, which have a higher density than PDMS, were introduced as an additional filler. Since the tests were limited to strains up to a maximum of 25%, only the initial curves can be compared with the results presented here. By adding the glass microspheres, the stress at 25% elongation increases from approx. 0.2 MPa with the pure silicone rubber by 0.6 MPa to approx. 0.8 MPa with the highest proportion of microspheres (weight ratio crosslinking agent and glassmicrosphere: 1:2.7). This has thus increased approximately by a factor of four. As described in section 3.4, the effect is less pronounced for the thermoplastic microspheres than for the glass microspheres. Zhang et al. postulate that the Mullins effect is caused by the breaking of physical bonds between the polymer chains and the microspheres and the sliding of the chains off the microspheres. 30 Since glass is made of silicon oxide and thus has the same base as polydimethylsiloxane (PDMS), stronger bonding between the microspheres and PDMS is more likely than for thermoplastic microspheres. FTIR analysis has revealed the presence of silicon oxide in the expandable microspheres, which helps explain the secondary bonding between the blowing agent and the LSR. However, compared to glass microspheres, the content of silicon oxide in the thermoplastic microspheres studied here is lower, resulting in a lesser increase. These secondary bonds form between the LSR matrix and the microspheres, and they are broken at the onset of the initial loading curve. Due to the different materials, there are fewer secondary bonds in the thermoplastic microspheres than in the glass microspheres studied by Zhang.
The greater abundance of secondary bonds in the MS 140 DS, evidenced by the higher work lost in the hysteresis area and the greater increase in the initial loading curve, can be attributed to several factors. On the one hand, microscopy has shown that the smaller microspheres contain significantly more particles, which are also more homogeneously distributed. As a consequence, a larger interface between the microspheres and the LSR is established, facilitating the formation of a greater number of secondary bonds in absolute terms. Additionally, FTIR analysis reveals a more prominent signal in the Si-O-Si twisting, suggesting a higher fraction of silicon oxide and consequently more functional groups available for secondary bonds. Similar trends are observed with the C = O bonds, particularly pronounced in the MS 140 DS. Given that oxygen, with its free electron pairs, readily engages in physical interactions, this further supports the higher proportion of secondary bonds in the MS 140 DS.
Conclusions
The aim of this publication was to investigate the stress softening of foamed LSR with microspheres as blowing agents. The tests showed that secondary bonds can exist between the microspheres and the LSR. This could be determined from the curve shape and the increase in stress at the beginning of the curve, as well as the larger work lost (hysteresis area). The residual strain is largely determined by the reduced amount of LSR matrix, as it correlates with the porosity.
The secondary bonds arise from interactions between the LSR and the functional groups present on the microspheres. FTIR analysis revealed that silicon oxide was employed as a stabilizer during the microsphere production process and remains present on the microspheres. This allows for interactions between the LSR and the microspheres to occur.
The strength of the secondary bonds and thus the shape of the hysteresis curve is influenced by the composition of the shell and the size of the surface. This was demonstrated by using the two types of microspheres and the different shift of the first load to higher values. In addition, the smaller microspheres have a higher hysteresis area than the larger microspheres despite their lower density. Further experiments are planned to precisely analyze the contact surface between the LSR and the microspheres.
Footnotes
Acknowledgements
We thank Tramaco for providing the microspheres and Dow Silicone for providing the LSR. In addition, we thank Mr Morris Brückner, in whose bachelor thesis the Mullins experiments were carried out.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

