Abstract
In foaming processes, the blowing agent has a significant influence on the material behaviour and the necessary processing parameters. Low-density polypropylene foam sheets are usually produced with aliphatic hydrocarbons or alkanes as physical blowing agent. Due to the necessary safety precautions and the environmental impact, there is great interest in using alternative blowing agents such as CO2. The sole use of CO2 often leads to corrugation, open cells or surface defects on the foam sheet and therefore requires modifications to the process technology. For this reason, blowing agent mixtures based on CO2 and organic solvents are used for the production of foam sheets. For developing a process model describing the melt flow in the extrusion die and the formation of cells, specific material data like diffusion coefficients are necessary. For CO2 and N2 as sole blowing agent, experimental data exist in the literature. Since no experimental data are available for co-blowing agents such as ethanol at elevated temperatures as they occur in the foam process, these data were calculated using molecular dynamics (MD) simulations. The benefit of MD simulations lies in their ability to reduce the experimental effort and, in particular, to provide data in cases where this data is not available through experimental measurements. The calculated diffusion coefficient values are compared to experimental data from the literature and presented for CO2, N2 and ethanol in polypropylene. The calculated diffusion coefficients of CO2 and N2 are compared with literature results and agree well with them. For the ethanol molecules, the diffusion coefficient is compared relative to the both aforementioned ones considered the larger size of the ethanol molecule compared to N2 and CO2. The results of the diffusion coefficients for ethanol are reasonable compared to the values found for the other two molecules.
Keywords
Introduction
Foam extrusion with physical blowing agents (PBA) is a key processing technology for thermoplastic polymers like polypropylene (PP). Foamed products can be used in various applications due to their good acoustic and thermal insulation behaviour that can be controlled by the foam structure and density.1–3 In order to govern foam properties, many foam extrusion studies have focused on the optimisation of process parameters (e.g. temperature, pressure gradient, die geometry)4–12 or on material modifications.13–19 Another important influence factor in the foaming process is the choice of blowing agent. Inert gases like carbon dioxide (CO2) or nitrogen (N2) are a safe and environmentally friendly alternative to hydrochlorofluorocarbons (HCFCs) or hydrocarbons as PBA, but have limited solubility in many polymers and can cause cell defects.20,21 Blowing agent mixtures, that are nowadays commonly applied in polystyrene (PS) foam board extrusion, can help to overcome these challenges.22–24 Scientific studies on blowing agent mixtures based on CO2 for PS have been published by
Modelling foam extrusion processes is a challenge, as all mentioned parameters (material, process and blowing agent properties) have to be taken into account and strongly interact with each other, governing the foaming process in the extrusion die.35,36,2 Modelling approaches for foam extrusion processes should therefore include material flow of the molten polymer loaded with blowing agents, cell formation and foaming in the extrusion die with respect to the interaction of material properties and process parameters.28,37,38
For developing a process model describing the melt flow in the extrusion die and the formation of cells, material data like diffusion coefficients at process conditions, that is, elevated temperatures and pressures, are necessary. For CO2 and N2 as sole blowing agent, experimental data exists in the literature.41,42 For co-blowing agents like ethanol or isopropanol, these data are not available and are therefore calculated by molecular dynamics (MD) simulations.
MD simulations are a widely used atomistic modelling approach to describe the dynamic behaviour of a system by solving Newton’s equations of motion. The areas of application for MD simulations are very broad, ranging from mechanistic examination on small organic molecules to the description of mechanical behaviour of solid phase material.43,44 The use of MD is also of great benefit in the field of polymer science, although it is hampered by the enormous size of the polymer systems and the associated high computational cost. Nonetheless, MD simulations are widely used in the field of polymers, with the determination of glass transition temperature and the description of gas penetrant permeation being two of the most important applications.
44
The great advantage of the MD simulations arises from its ability to describe time-dependent properties, such as diffusion or solubility, making it a very valuable tool especially in the field of membranes.
45
In this context, knowledge of the mobility of small penetrant molecules through the matrix of barrier material is of great importance. Some examples of applications are materials for packaging,
45
drug release
46
or CO2 storage.
47
Due to this broad range of applications as barrier materials, great efforts have been made to study the gas penetrant permeation and diffusion of small molecules in polymers. There are a lot of studies concerning the penetrant diffusion of small gas molecules in polymers and many researchers have published results of MD simulations for that purpose.
In our work we use MD simulations to describe the diffusion process of the blowing agent during foam extrusion. Therefore, the diffusion of three different gas penetrant molecules (N2, CO2 and EtOH) in isotactic polypropylene is studied for three different temperatures. The selected temperatures of 453 K, 473 K and 493 K represent relevant processing temperatures, which are all above the melting temperature of polypropylene. A full atomistic approach is applied, since for the selected temperatures the diffusion coefficients are expected to be large enough, so that they can be calculated with a reasonable computational effort. Furthermore, the aforementioned drawbacks of the united atom approach can be avoided. The calculated diffusion coefficient of N2 and CO2 are compared to experimental data from the literature which was measured at temperatures around 450 K. To our best knowledge, there is no such experimental data for ethanol. Furthermore, there are no values calculated by MD simulations for ethanol in polypropylene at elevated temperatures. The elevated temperatures lead to a higher mobility of the penetrant molecules, so that they can cover a greater distance in the simulated time interval compared to simulations at lower temperatures.
Since there are no literature values for the diffusion coefficient of ethanol, the calculated values are compared and the accuracy of the ethanol diffusion coefficients is evaluated in comparison to the other two penetrant molecules, considering the different molecule sizes.
Fundamentals of foaming
Physical foaming and formation of cells
Foam formation in the extrusion process with physical blowing agents can be divided into the phases shown in Figure 1 and described below.
4
The injection of the blowing agent into the melt takes place at a pressure above the saturation pressure. By intensive mixing, solution and diffusion processes, a homogeneous polymer/gas solution is achieved. The blowing agent load has a significant influence on the viscosity of the melt.
21
As the melt pressure in the extrusion die drops below the saturation pressure, the nucleation of the foam cells and their growth begins. Nucleation can be divided in heterogeneous and homogeneous nucleation and is often described by the classical nucleation theory.
55
Homogeneous nucleation applies for uncontaminated polymer melts. In contrast to this, cell nuclei are formed on the surface of particles during heterogeneous nucleation. Since the activation energy required for homogeneous nucleation is comparatively high, finely dispersed nucleation particles are usually used in foam extrusion, which reduce the activation energy.
56
The cell nucleation in the foam then results mainly from heterogeneous nucleation, enhanced by shear and extensional flow.5,9 Process steps of foam extrusion with physical blowing agents.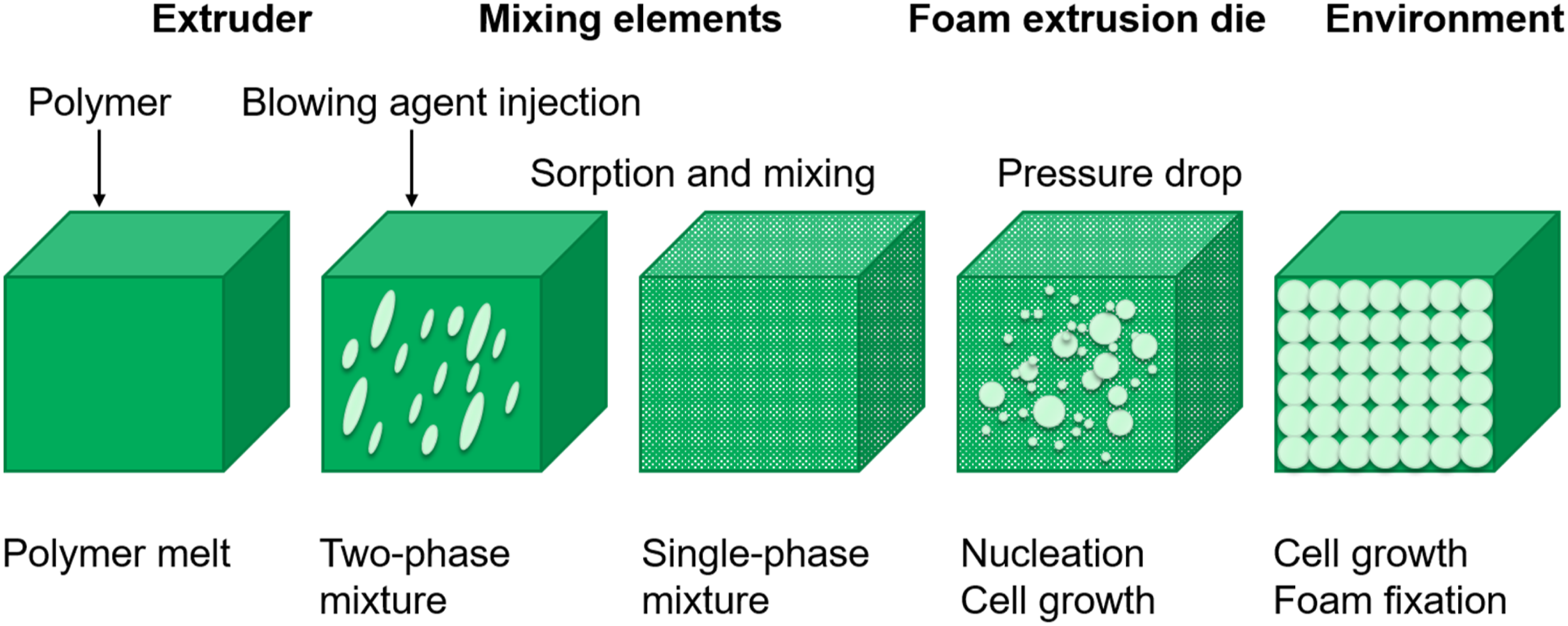
After leaving the extrusion die, foaming of the extrudate continues until the foam structure is solidified. Important parameters for successful formation of cells is the complete dissolution of the blowing agent in the melt, the creation of a high number of nuclei due to the pressure drop as well as the kinematics of cell growth and diffusion of blowing agent into the cells.12,28
Modelling flow and foaming in the extrusion die
When modelling foam extrusion, different phenomena have to be taken into account. Besides material properties, nucleation and cell growth, the flow field in the die gap should be considered. 21 Additionally, further effects like the shear rate can be taken into account.7,57
Based on the works of
Based on these initial parameters, the model calculation can be carried out in steps along the flow channel, respecting the change of material properties like viscosity, surface tension and solubility due to the formation of cells and the change of gas pressure in the cells. The volume flow rate, shear rate and pressure gradient calculated for each segment of the flow channel can be used to calculate the full pressure profile along the flow channel or to calculate dimensionless numbers to describe the foaming process in the extrusion die, as it was done by
The modelling approach is therefore dependent of the material properties and the PBA used, as the blowing agent load influences the material behaviour. To set up foam extrusion models, experimental data, e.g. from rheometer or solubility experiments, are therefore often combined with literature data, for example, specific density data.35,39,21
One of the specific material properties needed for setting up a material model is the diffusion coefficient, which can be used in an Arrhenius approach to consider the temperature dependence
Experimental data are available for the diffusion coefficients of CO2 and N2 in PP and other polymers, mainly from measurements with a sorption cell.
Fundamentals of the diffusion of small penetrant molecules in polymers and their determination by MD simulations
Diffusion process
The diffusion process of a small penetrant molecule in glassy or rubbery polymers is widely described in literature as a so-called ‘hopping’ mechanism.59–64 This mechanism can be divided in to two types of motion. The first one is the oscillation of the penetrant molecule in a microcavity in the polymer matrix. The duration of this quasi-stationary period depends on the polymer/penetrant combination. This first motion is interrupted by a ‘jump’ of the penetrant to another cavity in the polymer matrix. This is done through a ‘tunnel’ that is opened by the thermal motion of the polymer chains. In this manner, the diffusion in glassy or rubbery polymers is a combination of these two motions, while in rubbery state one might find a higher mobility of the chains and hence a higher ‘jump’ frequency. This also applies for the glass transition temperature (
The described ‘hopping’ mechanism undergoes a transition to a so-called ‘liquid-like’ mechanism for elevated temperatures, which leads to faster movement of the penetrant through the polymer.48,65,66 This is due to the fact that the chain mobility in the polymer increases with increasing temperature.
Since the diffusion coefficients obtained in this work are all simulated at elevated temperatures, it is expected for the penetrant to rather follow a ‘liquid-like’ diffusion than a ‘hopping’ mechanism, although ‘hopping’ should not be completely overcome, due to the long chain length of the polymer.
Calculation of diffusion coefficients from molecular dynamics simulations
The diffusion coefficient can be obtained from the well-known Einstein relation,
67
by determining the slope of the mean square displacement (MSD) over time. The MSD is given by the following equation
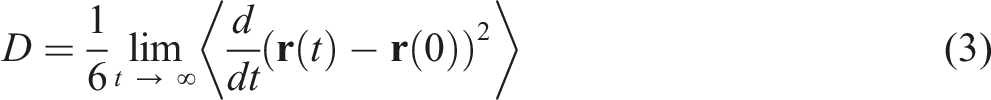
Here
Methodology
All simulations in this work were performed with the BIOVIA
For the production runs, a
Results and discussion
Discussion of the diffusion results
Results of the diffusion coefficients calculated in this work and their corresponding standard deviation, as also experimental results from literature data for comparison. All diffusion coefficients listed, are given in the units of cm2/s ∙ 10−5. Where column five shows experimental results from
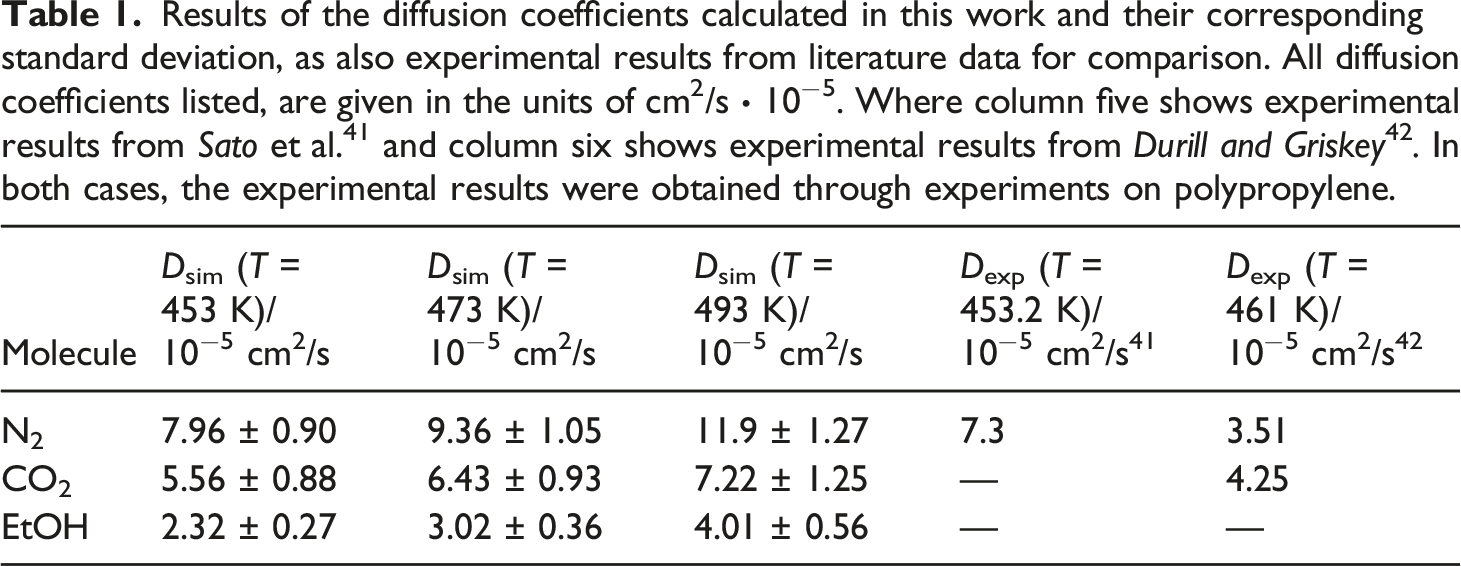
As Table 1 shows, for all three penetrant molecules the diffusion coefficients increase with increasing temperature. The comparison of the diffusion coefficients for the selected temperatures shows that the simulated results are all within the same order of magnitude as the experimental results. The diffusion coefficient for N2 at 453.2 K estimated by
Figure 2 shows three MSD over time diagrams as an example, representing only one out of 10 starting configurations. It shows the Mean-Square-Displacement (equation (2)) of CO2 over time in polypropylene for three different temperatures. The first 100 ps weren’t used for the evaluation, due to a clear non-linear behaviour in this time domain. This non-linear behaviour is due to the free flight of the penetrant in the microcavity at the beginning of the simulation and is of non-diffusive nature.
54
MSD vs. 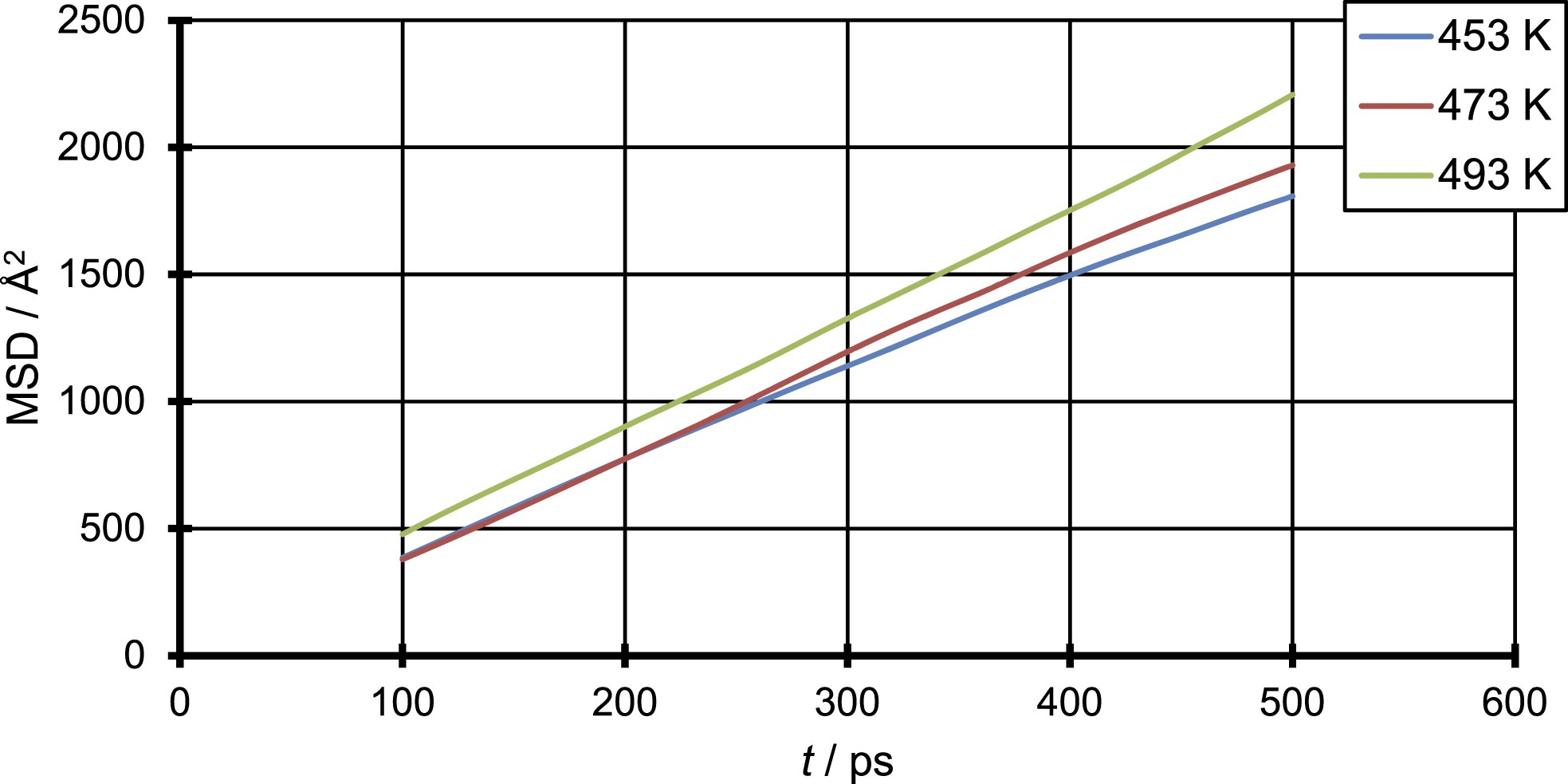
The results of the MSD show a clear linear behaviour. Furthermore, it can be seen that the MSD increases with increasing temperature, as it is expressed in the increasing diffusion coefficients. To ensure the domain of normal diffusion is reached, a double logarithmic scaled plot of MSD vs. MSD vs. 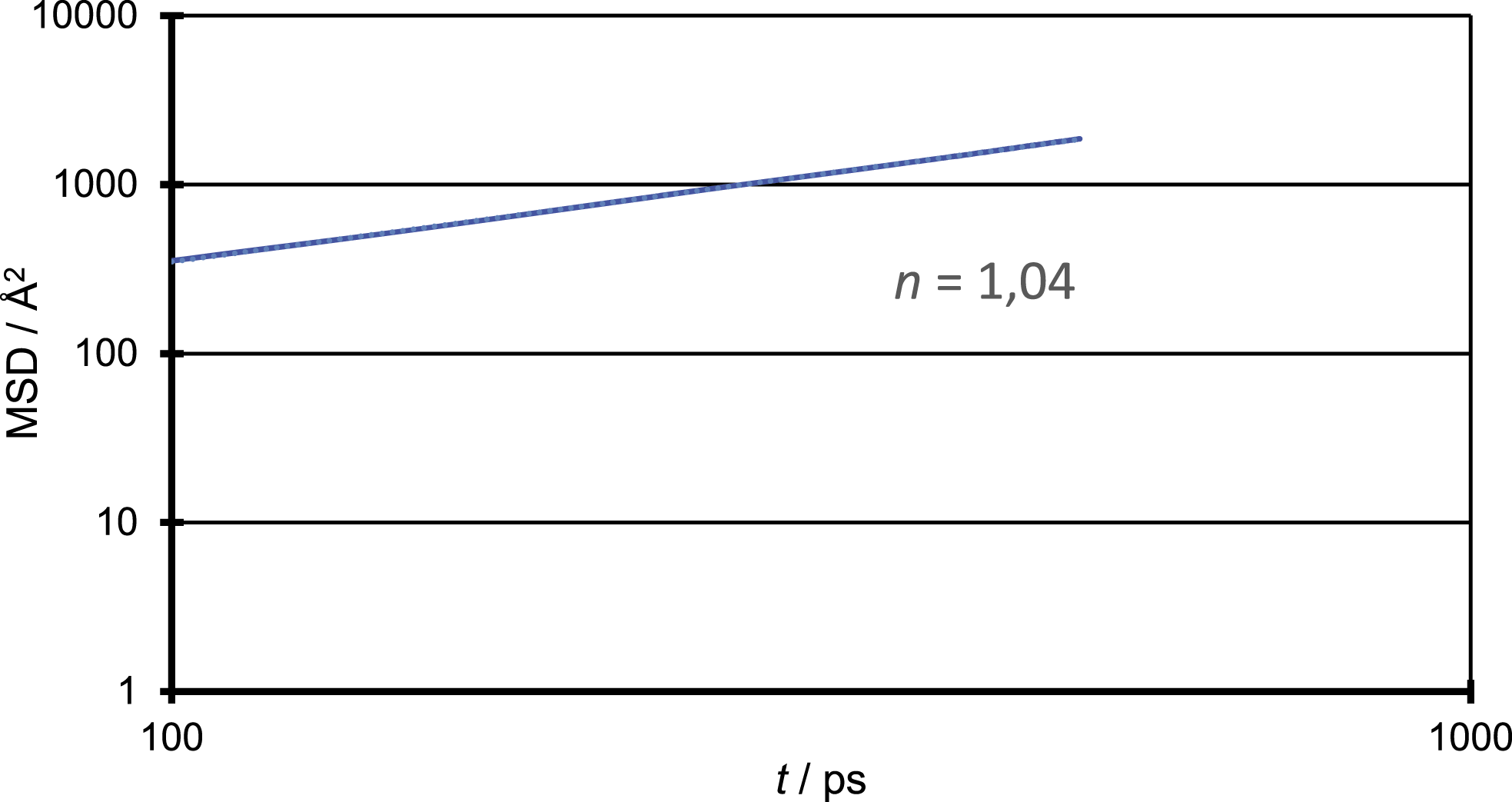
To illustrate the underlying mechanism of the diffusion in the polymer penetrant systems and compare them with findings in literature, in Figures 4 – 6, the total displacement over time is shown for all three penetrant molecules and all three temperatures. The results were generated for a single penetrant molecule, which can be viewed as representative. Total displacement of a N2 molecule for the three different temperatures. Total displacement of a CO2 molecule for the three different temperatures. Total displacement of an EtOH molecule for the three different temperatures.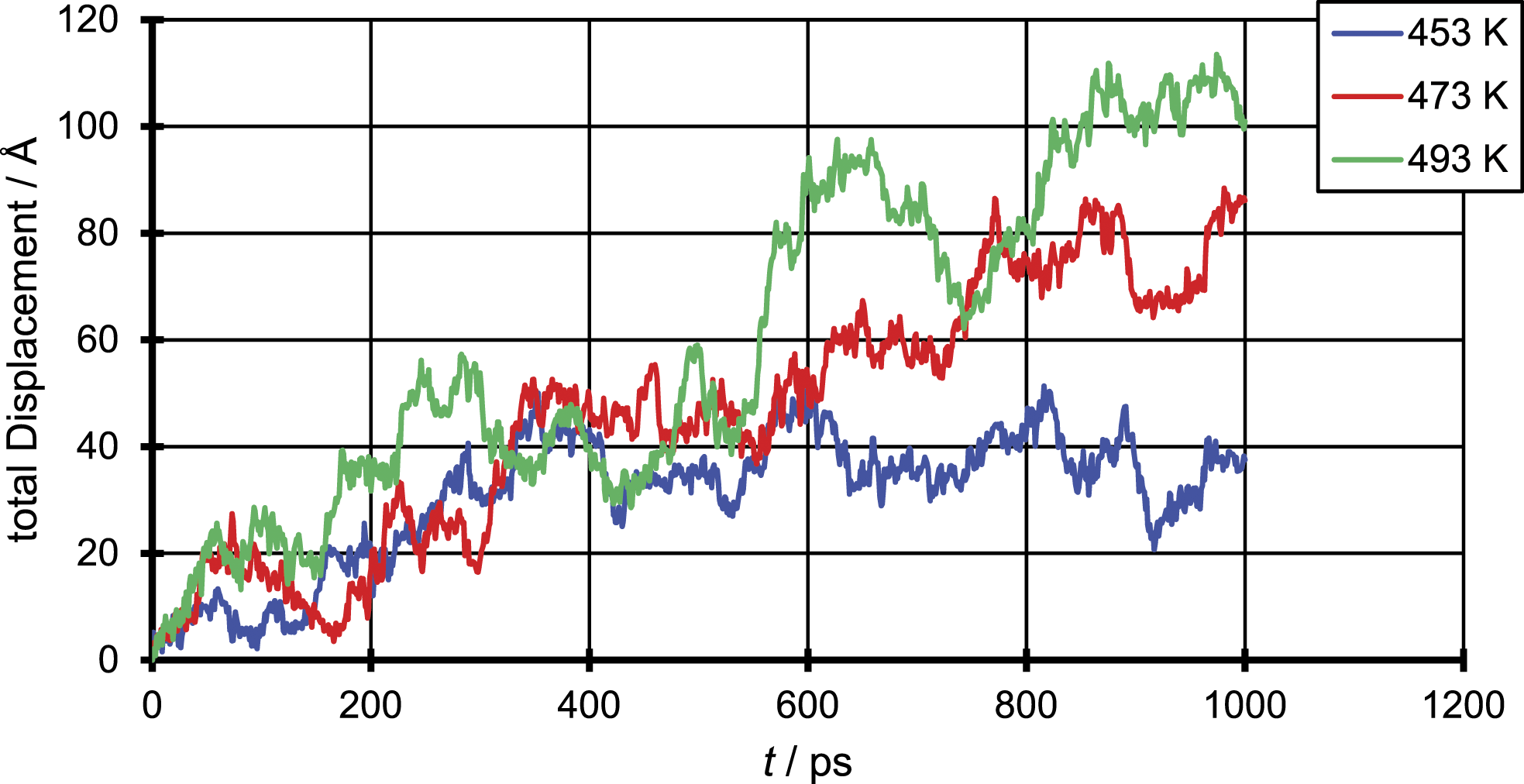
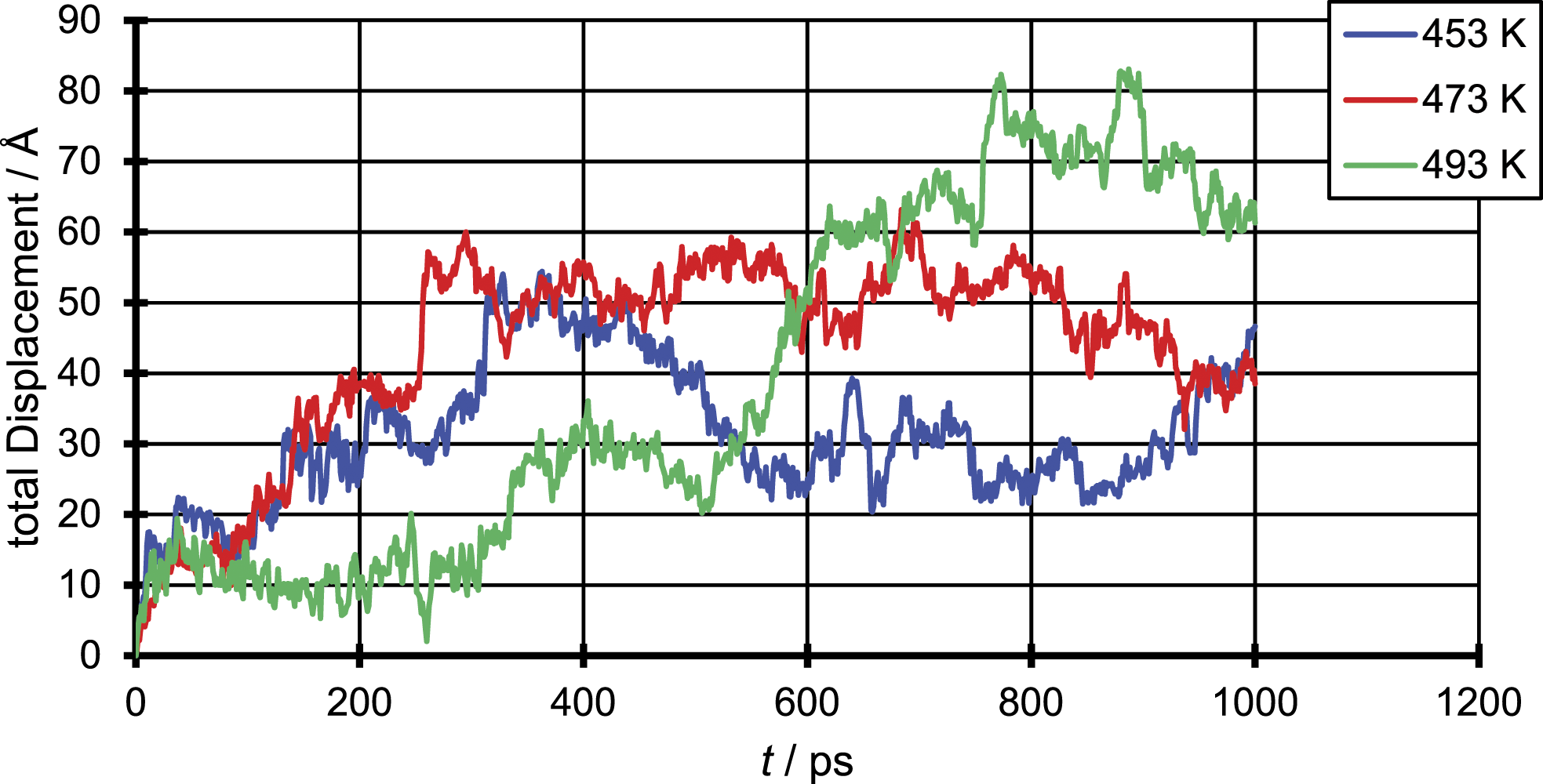
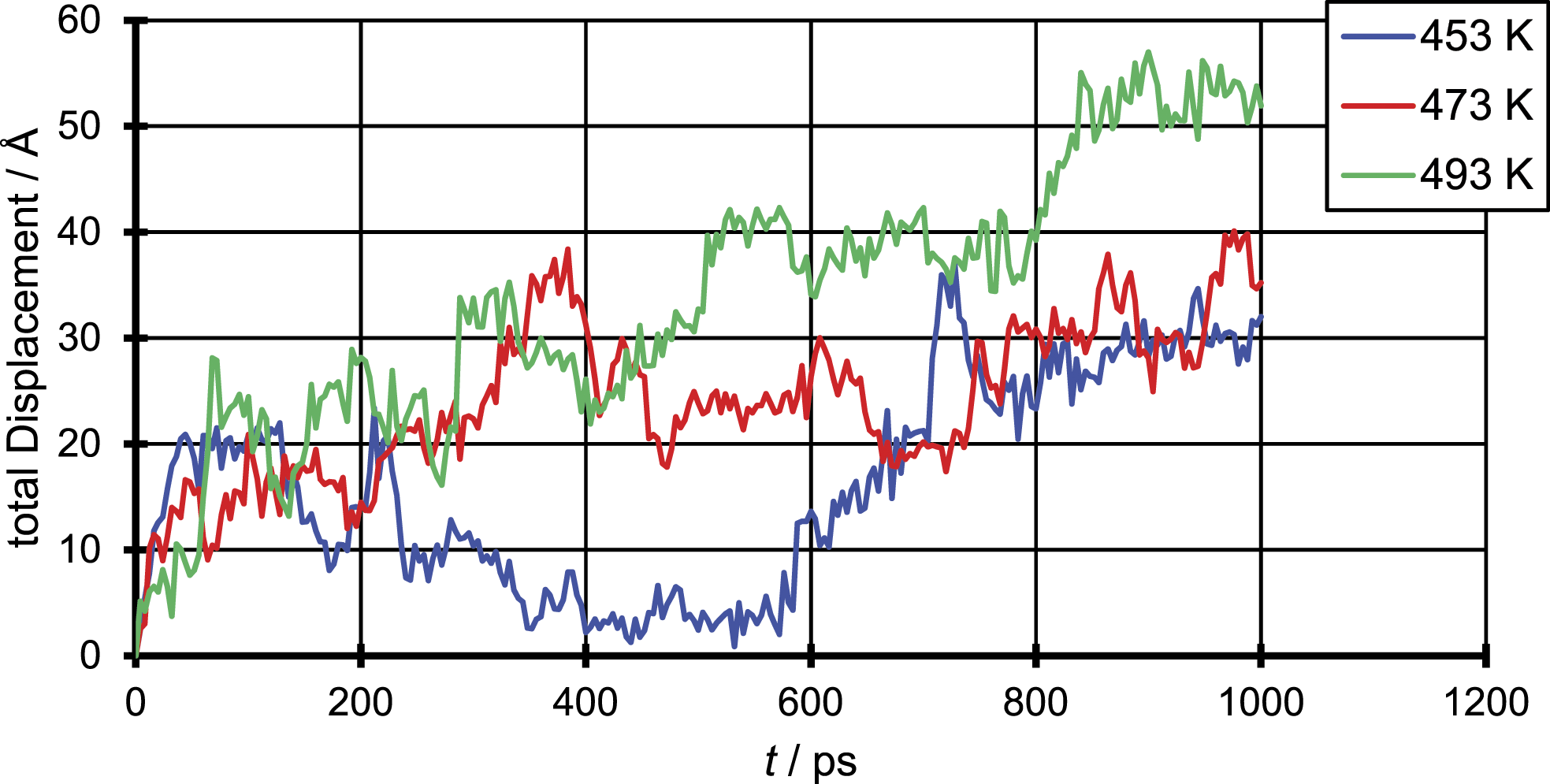
The figure of the total displacement over time shows the aforementioned dependency of the diffusion coefficient from the temperature. It can be seen that the total displacement for the chosen penetrant particle increases with higher temperatures and decreases with increasing penetrant size which in average over all penetrant molecules leads to an increase of the diffusion coefficient with increasing temperature and decreasing particle size. More important is that the total displacement over time shows that the penetrant diffusion follows a ‘hopping’ mechanism. On the one hand one can see areas of high oscillation in a range of just a few Å which correspond to the movement of the penetrant in a cavity. On the other hand, this oscillations are interrupted by bigger movements of about 10 Å which correspond the to a single jump event taken place.
69
This means even at such elevated temperatures the ‘hopping’ mechanism does not completely merge into a ‘liquid-like’ mechanism, but the number of ‘hopping’ events taken place increases and so smoothens the curve compared to simulations at room temperature. This can be due to the fact that polymers even in liquid state are quite immobile, due to the slow motion of the single chains. Instead, like in a low molecular weight fluid were the fluid velocity is of the same magnitude as the velocity of the penetrant, in a polymer system the penetrant diffusion still depends on the slower chain mobility of the polymer.
66
The above describe mechanism for the penetrant diffusion, found in this work compares well to the findings by 2-Dimensional representation of the total displacement.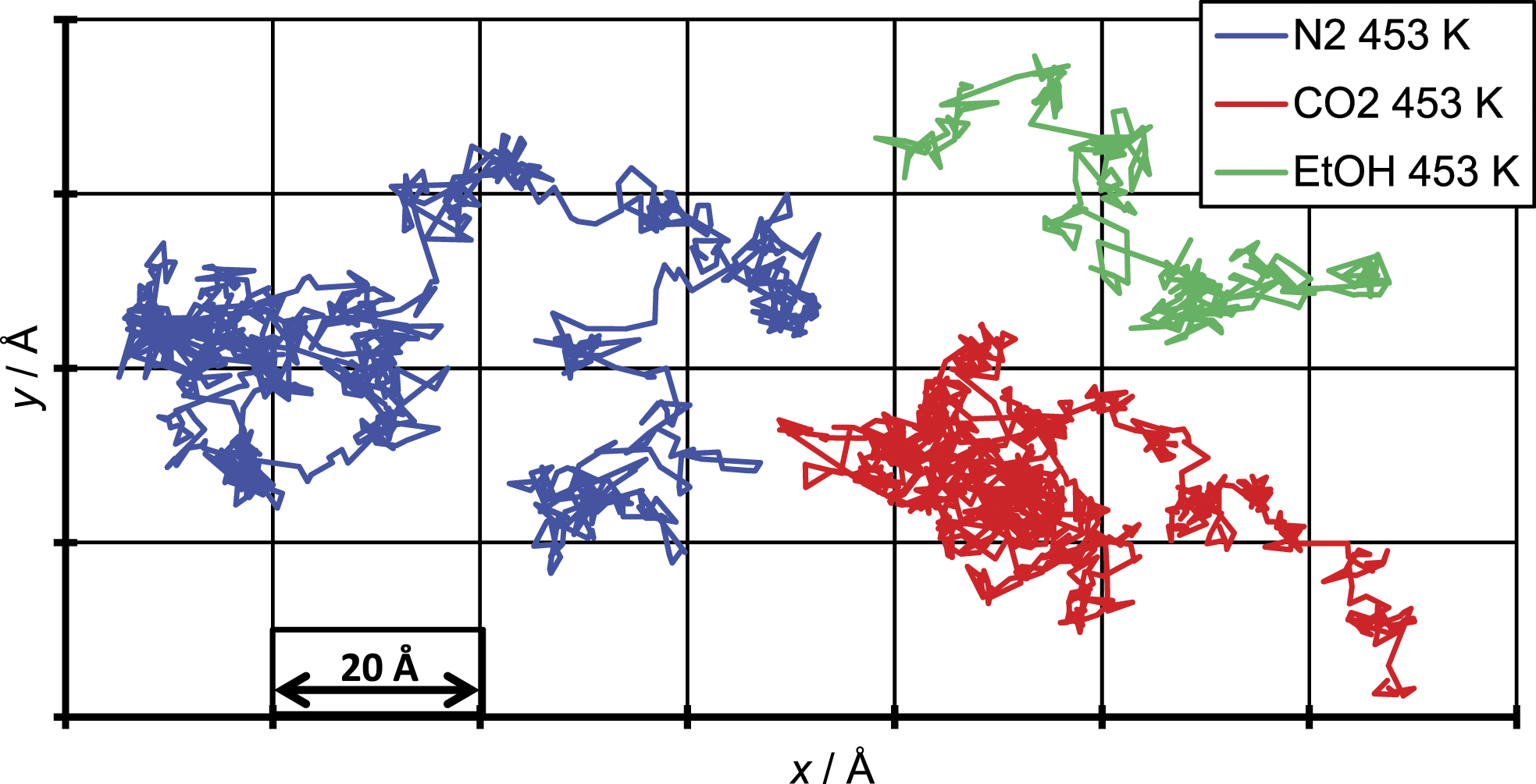
Figure 7 depicts the single jumps which has taken place while the penetrant molecule moves through the polymer matrix. For all three penetrant molecules shown here, one can also see the higher number of jumps for the N2 molecule compared to the CO2 and ethanol molecule, which in average is represented by the higher diffusion coefficient of the N2 molecule.
The temperature dependence of the diffusion coefficient can be described by an Arrhenius plot following equation (1), which is shown in Figure 8. Arrhenius plot of the three penetrant molecules N2, CO2 and EtOH.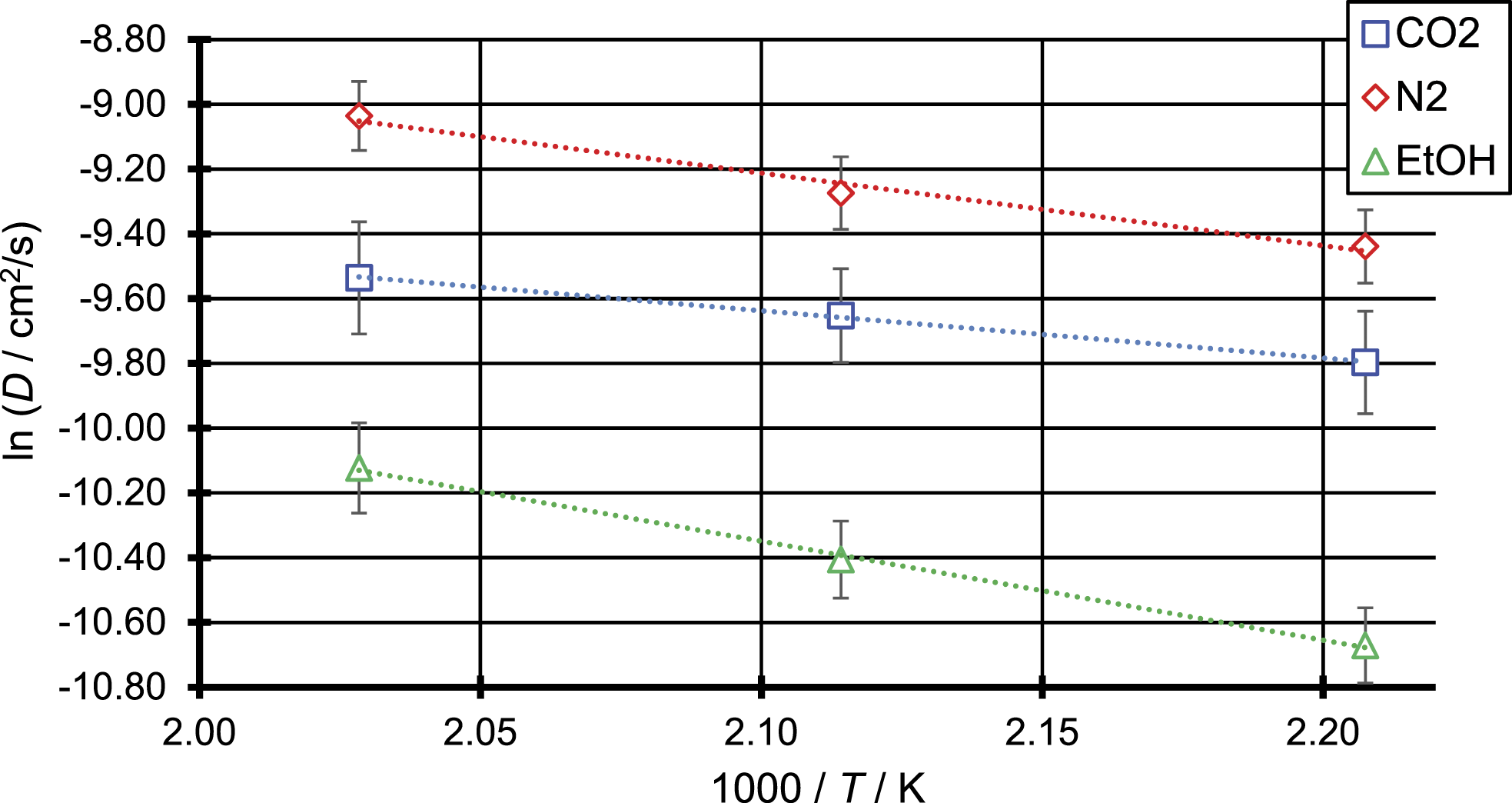
As can be seen in Figure 8 for all three penetrant molecules the diffusion coefficient shows a linear dependence with 1/
For ethanol as the penetrant molecule, an activation energy of 25.4 kJ/mol is found. This is significantly higher than the activation energies of N2 and CO2 which was to be expected, due to the bigger size of the ethanol molecule compared to the other two penetrant molecules. In contrast to this result the activation energy of N2 determined from MD simulations is higher than the activation energy obtained for CO2, although N2 is the smaller molecule. This result is consistent with the activation energies
Conclusion
For modelling foam extrusion processes, knowledge of the diffusion behaviour of the blowing agent in the molten polymer is crucial. As an alternative to experimental methods, molecular dynamics (MD) simulations were used to calculate the diffusion coefficients of the penetrant molecules N2, CO2 and ethanol in polypropylene at elevated temperatures. The calculated diffusion coefficients were found to be in the same order of magnitude as those described in literature obtained from solubility experiments. Only for the relation between penetrant size and diffusion coefficient the experimental results by
Overall, the MD simulations allowed to calculate diffusion coefficients at elevated temperatures, which agreed well with experimentally determined diffusion coefficients, except for the deviations mentioned before. Thus, MD simulations can be a valuable tool in calculating diffusion coefficients for blowing agents and blowing agent mixtures allowing predictions of the solubility for foam extrusion processes. Especially when novel blowing agents or blowing agent mixtures are used, this is helpful because experimentally determined diffusion coefficients cannot be found in literature yet. The next step would be to expand the MD simulations to other blowing agents like acetone or isopropanol and to different combinations of these blowing agents as blowing agent mixtures. For modelling foam extrusion, MD simulations offer promising possibilities to improve the model quality and enhance the precision of calculated material data from these models without the need to run extensive series of experiments. The findings allow to calculate diffusion coefficients independently from experimental for various combinations of blowing agents and polymers at elevated temperatures and pressures, allowing sophisticated models to be set up faster and easier in the future.
Footnotes
Acknowledgments
Computational results were obtained by using Dassault Systèmes BIOVIA software programs. BIOVIA Materials Studio was used to perform the calculations and to generate the graphical results.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funded by the Deutsche Forschungsgemeinschaft (DFG, German Research Foundation) under Germany’s Excellence Strategy - EXC-2023 Internet of Production - 390621612.
