Abstract
Background:
Smoking cigarettes is highly prevalent in psychosis. However, the relationship between smoking and psychosis is still unclear.
Aims:
This paper aims to investigate the probable association in a 6-year follow up of community-based population.
Methods:
A general population sample were contacted at baseline (n = 4,011) and at 6-year follow-up (n = 2,185). Both at baseline and follow-up (newly developing/persistent), smoking cigarettes and psychotic experiences were assessed using the relevant sections of Composite International Diagnostic Interview 2.1. The associations between baseline smoking features and follow-up psychotic experiences were analysed using logistic regression models. To accurately assess the impact of smoking, participants were screened and controlled for alcohol and substance use. Interactions between baseline smoking features and existence of baseline psychotic experiences on the persistence of baseline psychotic experiences and its progression to psychotic disorder were analysed using interaction contrast ratios.
Results:
Follow-up psychotic experiences were 1.8 (OR = 1.8; CI [1.3, 2.6]) and follow-up psychotic disorders were 5.9 times (OR = 5.9; CI [2.9, 12.0]; p < .001) more common in individuals whose first smoking age were ⩽15 years old at T1 and who were an active regular smoker. The risk of persistence (OR = 1.5, 95% CI [1.1, 2.2]) and progression to psychotic disorder (OR = 5.3, 95% CI [1.6, 17.8]) was higher in smokers compared to non-smokers. There was a significant additive interaction between baseline psychotic experiences and smoking on persistence of psychotic experiences at follow-up (ICR = 1.8; 95% CI [0.2, 3.6]).
Conclusions:
These findings suggest that early and regular cigarette smoking is associated with the persistence of psychotic experiences and increased risk of progression to psychotic disorder.
Introduction
On a global scale, the estimated prevalence of smoking among adults in 2020 was approximately 32.6% in men and 6.5% in women (Dai et al., 2022). Cigarettes are consumed by almost one third of the adult population in Türkiye (Özer et al., 2018). In many countries, individuals affected by mental illnesses are also more prone to smoking cigarettes (Fagerström & Aubin, 2009; Garey et al., 2020; Yargic & Baykan, 2013). There is growing debate whether smoking cigarettes may contribute to the development of mental disorders, including psychotic disorders (PDs) (Jackson et al., 2015; Taylor et al., 2014; Treur et al., 2021). Neurochemical effects of nicotine may be related to mental distress (Jobson et al., 2019). Toxic compounds released from inhaled cigarettes trigger inflammatory and oxidative stress processes which may lead to neural changes and eventually to mental disorders (Berk et al., 2011). Some evidence has implied desensitisation of nicotinic acetylcholine receptors following recurrent nicotine exposures (Mineur & Picciotto, 2009).
Psychotic experiences (PEs) refer mostly to subclinical positive psychotic symptoms, which may be temporary or persistent. PEs may also progress into PDs in interaction with genetic and environmental risk factors (Staines et al., 2022; van Nierop et al., 2012). Schizophrenia is considered the most severe form of PDs. The fact that the lifetime prevalence of smoking cigarettes in schizophrenia is over 60% indicates that smoking is two to five times more common in schizophrenia than in the general population (de Leon & Diaz, 2005). Additionally, schizophrenia patients had been smoking cigarettes for years before the first psychotic episode (Hickling et al., 2018). Smoking is both prevalent in clinical forms of psychosis and in individuals with subclinical PEs (Bhavsar et al., 2018).
The association between smoking cigarettes and psychosis is complicated and has been the subject of various studies for a long time (Coustals et al., 2020; Scott et al., 2018). Several hypotheses on this relationship have been proposed so far (Quigley & MacCabe, 2019): (i) Reverse-causation hypothesis; (ii) Shared liability hypothesis; (iii) Confounding hypothesis; and (iv) The hypothesis proposing smoking as an independent causal factor.
First, the reverse causation hypothesis proposes that individuals with PDs smoke for improvement of clinical symptoms as well as side effects of the antipsychotic medication (Kumari & Postma, 2005). Nicotine reduces antipsychotic related extrapyramidal side effects such as parkinsonism (Goff et al., 1992) and akathisia (Barnes et al., 2006), and improves negative symptoms in schizophrenia (Smith et al., 2002). Furthermore, a recent meta-analysis has reported positive effects of nicotine on cognitive functioning in PDs (Coustals et al., 2020). However, this interpretation has been challenged by recent large-scale prospective findings. It was shown that both individuals with PDs and their unaffected siblings who smoked experienced more frequent positive, negative and depressive symptoms, as well as poorer quality of life, compared with non-smokers. Importantly, participants who initiated smoking during follow-up subsequently showed an increase in symptom severity. These findings argue against the self-medication hypothesis and suggest that smoking may contribute to the persistence or worsening of PEs rather than merely serving as a coping mechanism (Vermeulen et al., 2019). Second, the shared liability hypothesis proposes that both psychosis and nicotine addiction share a similar genetic background. Some RNA and RNA-binding protein genes, protein regulatory genes and energy production genes have been associated with both PDs and smoking-related variables such as nicotine addiction levels, number of cigarettes smoked per day and plasma cotinine (predominant metabolite of nicotine) concentration (J. Chen et al., 2016). Third, the confounding hypothesis suggests that smoking is associated with some conditions such as migration, victimisation, urban living, and cannabis use, which are also risk factors for psychosis (Stilo & Murray, 2019). Finally, recent studies suggest that smoking may be an independent causal risk factor for psychosis (Gage et al., 2017; Kendler et al., 2015; Wium-Andersen et al., 2015). Despite the emerging evidence, this area of research is far from being conclusive.
The aims of this paper are:
I. To investigate the associations between various characteristics of smoking cigarettes at baseline and presence of PEs/PDs at the 6-year follow-up of a community-based population.
II. To investigate whether PEs and smoking cigarettes interact additively on the persistence of PEs and progression into PDs during the follow-up period.
Methods
This study is a prospective longitudinal study designed mainly to screen and follow up phenotypes within the psychosis spectrum and associated individual, familial, and neighbourhood-level variables. The sample included individuals from the general population of a metropolitan area. Our study consisted of baseline (T1: 2008) and follow-up (T2: 2014) assessments with an approximately 6-year interval (mean follow-up time: 6.11 ± 0.94 years). A summary of the types of data collected at T1 and T2 is presented in Supplementary Table 1.
Sampling and Participants
This study is a prospective longitudinal study designed mainly to screen and follow up phenotypes within the psychosis spectrum. The baseline assessment (T1: 2008) was applied in a sample which was randomly selected by the Turkish Institute of Statistics (TurkStat) to represent the Izmir metropolitan area. A multistage clustered probability sampling frame was used, covering 11 districts and 302 neighbourhoods. From 6,000 initially selected households, 5,242 were eligible. Within each household, one individual aged 15 to 64 years was randomly selected using the Kish method. Inclusion criteria were being aged 15 to 64 years and residing at the selected address. There were no specific exclusion criteria to ensure a representative community sample. Trained interviewers visited addresses in person, informed potential participants about the study’s goals, procedures, and confidentiality, and obtained written informed consent. Participants did not receive direct financial compensation for their participation at either wave. They were provided with a brochure detailing the study and, upon request, general feedback and guidance on accessing mental health services following the assessment. In T1, 4,011 individuals were interviewed for affective and psychotic symptoms, as well as smoking, drinking, and help-seeking behaviours. In the follow-up assessment (T2: 2014), all baseline participants were revisited at their previously recorded addresses. During these visits, 2,185 individuals from the original sample were successfully re-screened for affective and psychotic symptoms, as well as smoking, drinking, and help-seeking behaviours.
Ethical approval for the study was obtained from the Ege University Clinical Research Ethics Committee on 10 May 2007 (Decision No: 12-3/60) and on 4 June 2012 (Decision No: 07-5.1/10). Written informed consent was obtained from all participants.
Psychotic Experiences and Psychotic Disorders
The procedure used in both baseline (T1) and follow-up (T2) assessments to identify PEs and PDs has been previously described in detail (Binbay et al., 2012; Kırlı et al., 2019). The time frame was lifetime in T1, whereas it was the last 6 years in T2. Hallucinations, delusions, and associated impairment/help-seeking were assessed according to the relevant sections of the Turkish version of the Composite International Diagnostic Interview (CIDI) 2.1 (Kılıç & Göğüş, 1997). The CIDI was selected as the primary screening instrument because it is a fully structured interview designed for large-scale epidemiological studies in the general population, allowing for administration by trained lay interviewers. While clinician-rated scales like the Positive and Negative Syndrome Scale (PANSS) and Brief Psychiatric Rating Scale (BPRS) offer granular symptom detail, they are less feasible for community-based screening of thousands of participants. To enhance the validity of CIDI-based psychosis assessments, a rigorous two-stage procedure was employed: all endorsed PEs were reviewed by a team clinician, and participants with probable PDs were subsequently re-interviewed by a psychiatrist using the Structured Clinical Interview for DSM-IV (SCID-I), a gold-standard diagnostic tool. This methodology balances epidemiological feasibility with clinical diagnostic accuracy (Robins et al., 1988). CIDI was designed by the World Health Organisation to be implemented in epidemiological studies of mental disorders and it provides information about frequency, duration, and severity of symptoms, psychosocial impairment, and help-seeking due to symptoms (Andrews & Peters, 1998). Furthermore, CIDI evaluates whether the symptoms are due to a somatic illness, medication, use of alcohol, or other substances.
PEs were screened using the relevant section of the CIDI 2.1 (module G: Schizophrenia and other psychotic disorders). Assessment of PEs was based on 14 CIDI delusions items (G1, G2, G3, G4, G5, G7, G8, G9, G10, G11, G12, G13, G13b, and G14) and five CIDI hallucinations items (G17, G18, G20, G20C, and G21). All items were rated dichotomously, indicating presence or absence. A measure of impairment associated with PEs was defined using CIDI items G16, G23 (told interviewer about psychotic beliefs), G25 (duration of the PE: between 1 day and 6 months or more), G26, G28, G29, and G29A (level of dysfunction). Participants with probable PDs were re-interviewed with the SCID-I.
At both T1 and T2, sections were devoted to defining patterns of help-seeking for mental health problems. Questions included any self-report mental problem, help-seeking for a mental problem, the route of help-seeking, the probable outcome of the help-seeking (diagnosis) and prescribed medicines and any hospitalisation during the time frame (lifetime and last 12 months at T1 and last 6 years at T2). If this was the case, the person was asked for permission to contact the clinician involved in the diagnosis or the treatment of the participant to verify the diagnosis and review case material.
If a participant was deemed to have a diagnosis of probable PD according to the algorithm mentioned, the participant was recontacted by the team psychiatrist and invited to the hospital for a clinical evaluation with the Structured Clinical Interview for DSM-IV (SCID). When the participant did not attend the hospital, clinical interviews were conducted at the participant’s residence by the psychiatrist. Thus, 225 participants at T1 and 263 participants at T2 were clinically re-interviewed to identify participants with PD.
The PDs group included all individuals diagnosed with any DSM-IV disorder with psychotic features. The PEs group included individuals who had at least one CIDI PE.
Smoking and Related Features
Smoking was assessed using the relevant module of the CIDI 2.1 (section B, disorders caused by tobacco). Smoking status in baseline (T1) was categorised as follows: (a) non-smokers: respondents who have never smoked in their life; (b) active irregular smoker: respondents who define themselves as occasional smokers but who have not smoked on a daily basis for a month or more; (c) active regular smoker: respondents who smoke frequently and on a daily basis; and (d) quitted smoking: respondents who used to smoke before but quitted smoking and were not smoking during the baseline assessment. Second, a variable was created combining the age of first smoking (⩽15 or >15 years old) and status of active regular smoking (present or not). Third, a variable “chain-smoking” was created and defined as starting to smoke as soon as the latest one finishes. Fourth, a variable assessing the intention to quit smoking was created and coded as follows: (a) currently non-smoker; (b) intend to cease smoking is absent; and (c) intend to cease smoking is present.
Covariates
The baseline assessment included a sociodemographic questionnaire. At the follow-up assessment, the same questionnaire was used to determine changes over time. Covariates used in this paper included age, sex, education, marital status, socioeconomic status, family history of mental disorders, life events for the last 6 years and childhood adversity.
Socioeconomic status depended on the participant’s occupation and was divided into three ordinal categories: professional and non-manual employees, owners of small businesses, manual workers (Noble & Goldthorpe, 1981). Childhood adversity was assessed based on self-reported history of major disruptive events before the age of 15 years, including the death of any parent, divorce of parents, and separation from parents for at least 3 months. It was coded dichotomously (none versus at least one event). While this method captures significant early-life stressors, it does not constitute a comprehensive assessment of childhood maltreatment, such as that provided by dedicated instruments like the Childhood Trauma Questionnaire (CTQ). Using questions derived from the Family Interview for Genetic Studies (National Institute of Mental Health, 1992), history of mental disorders in the father, mother, siblings, and offspring was assessed. Thus, a “family history of mental disorders” variable was defined and recoded into four categories guided by previous literature (none, unknown mental problem, common mental disorder, and severe mental disorder) (Mortensen et al., 2010). Family history of common mental disorders include any diagnosis of depression, anxiety, conversion, or somatisation in absence of severe mental disorders among parents or siblings of the participant. Severe mental disorder in the family was coded positive if the participant’s parents or siblings had been diagnosed with PD or bipolar disorder, had died because of suicide or had been admitted to a psychiatric inpatient unit.
Alcohol, cannabis, and other substance use was investigated using module J (alcohol) and module L (substances) of the CIDI 2.1. Alcohol use was recoded into four categories: never, none in last year, non-heavy drinking and heavy drinking. The upper limit of 14 standard drinks per week (males) and 7 standard drinks (females) was used for the definition of heavy drinking, as previously suggested for our country (Öztürk & Uluşahin, 2015). Furthermore, substance use was assessed using the criterion of “at least five times” based on previous research and recoded into three categories: none, cannabis only, cannabis, and other substances (Kuepper et al., 2011).
Statistical Analyses
Data was analysed using STATA v13.1. First, sociodemographic characteristics of the participants at the baseline assessment and proxy measures of exposures (family history of mental disorders, childhood adversity, recent stressful life events, alcohol use, cannabis, and other substance use) were presented. In addition, the associations between these variables and smoking status (in three categories: active regular smokers/active irregular smokers/current non-smokers) were assessed using the chi-squared tests. These analyses were presented with the relevant effect size measure (Cramer’s V). Cramer’s V may have values in the range of 0 to 1 and greater values represent stronger associations between the variables (Cramer, 1946). Second, the associations between various smoking features at baseline (e.g. age of starting smoking and current smoking status) and follow-up PEs/PDs were analysed using logistic regression models. Third, to assess a possible synergism between baseline PEs and smoking on persistence of PEs and effects of smoking on progression to PDs, logistic regression models were used with the dichotomous dependent variables “follow-up PEs” and “follow-up incident PDs.” The independent variable combined presence of baseline PEs and active regular smoking at baseline (0: no PEs no Smoking, 1: no PEs Smoking +, 2: PEs + no Smoking, and 3: Both PEs and Smoking +). Using the odds ratios (OR) derived from these logistic regression models, additive interactions between PEs and smoking on the outcome variables were tested using interaction contrast ratios (ICRs). ICRs were calculated using the following formula (ICR = OR both PE and smoking − OR PEs (+), no smoking − OR no PE, smoking (+) + 1) (Knol et al., 2007). In all logistic regression models, estimates for OR were a priori adjusted for sociodemographic variables (age, sex, educational level, and socioeconomic status) and proxy measures of other exposures (family history of mental disorders, childhood adversity, recent stressful life events, alcohol use, cannabis, and other substance use). In order to nullify the multiple comparison problem, Bonferroni correction was performed. Multicollinearity was checked using the VIF command in STATA (variance inflation factor). Furthermore, other assumptions of logistic regression were also checked and none of the assumptions were violated. In all analyses, alpha was set to 0.05.
Results
Characteristics of the Sample and Associations With the Smoking Status
The sociodemographic characteristics of the participants and the proxy measures of exposures stratified by smoking status (active regular smokers/active irregular smokers/current non-smokers) are presented in Table 1. In T1, 45.1% (n = 985) of the participants were non-smokers, 15.1% (n = 330) quitted smoking, 33.4% (n = 729) were active regular smokers, and 6.4% (n = 141) reported active irregular smoking behaviour. Smoking was significantly more common in males, age group of 31 to 45 years, high school graduated participants and participants with alcohol use and substance use. Nevertheless, effect sizes were generally very small (Cramer’s V ⩽ 0.05) and only the associations with alcohol use and male sex had relevant, but still small effect sizes (Cramer’s V > 0.1).
Characteristics of Participants at Baseline (T1) and Associations With Smoking Status.
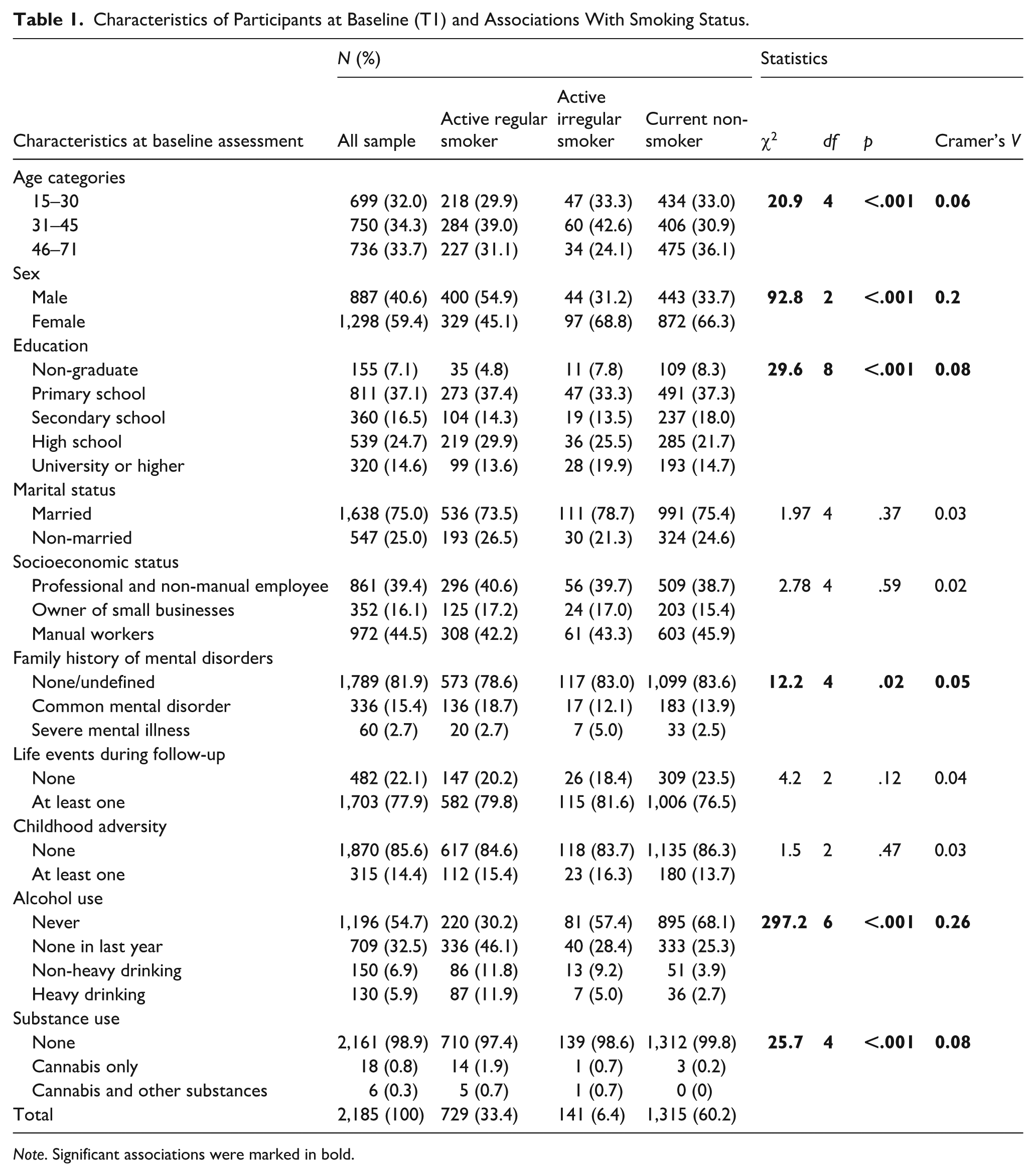
Note. Significant associations were marked in bold.
Associations Between Baseline Smoking Features and Follow-Up PEs/PDs
The associations between baseline smoking features and follow-up PEs/PDs are presented in Table 2. First, follow-up PEs were 1.8 times (OR = 1.8; CI [1.3, 2.6]; p < .01) and follow-up PDs were 5.9 times (OR = 5.9; CI [2.9, 12.0]; p < .001) more common in individuals whose first smoking age were ⩽15 years old at T1 and who were an active regular smoker, compared to non-smokers, adjusting for proxy measures of other exposures and after performed Bonferroni correction. Second, the odds of follow-up PEs (OR = 1.6; CI [1.2, 2.3]; p < .001) and follow-up PDs (OR = 4.9; CI [2.6, 9.2]; p < .001) were higher in chain-smokers than non-smokers and smokers without chain-smoking. Finally, the odds of follow-up PEs (OR = 1.7; CI [1.1, 2.5]; p < .01) and PDs (OR = 3.8; CI [1.7, 8.4]; p < .001) were highest in participants who had no intention to cease smoking at baseline, in comparison with participants who had intention to cease smoking and with non-smokers.
The associations Between Baseline Smoking Features and Follow-Up Psychotic Experiences/Disorders.
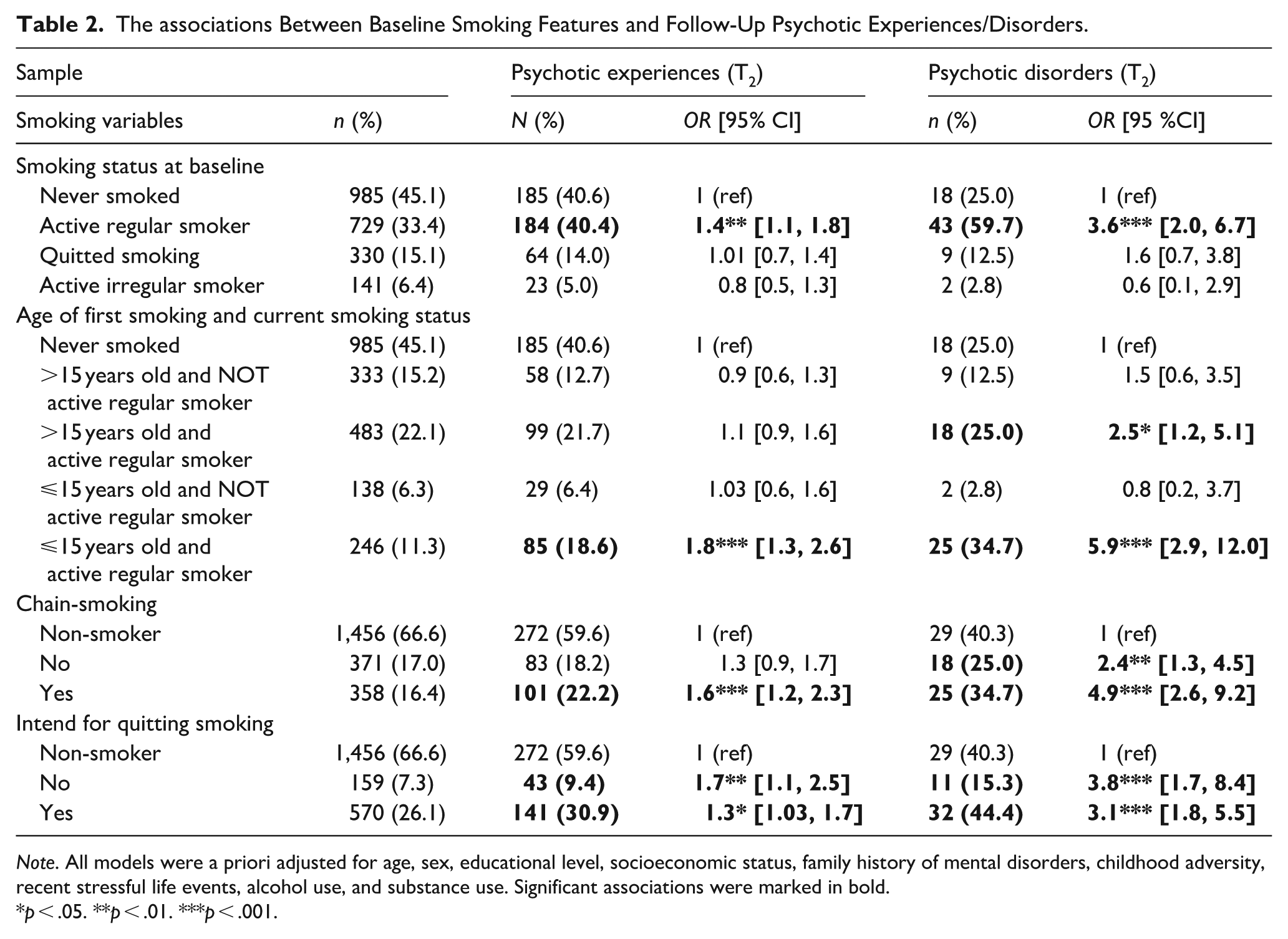
Note. All models were a priori adjusted for age, sex, educational level, socioeconomic status, family history of mental disorders, childhood adversity, recent stressful life events, alcohol use, and substance use. Significant associations were marked in bold.
p < .05. **p < .01. ***p < .001.
Additive Interactions Between Smoking and Baseline PEs on Persistence of PEs and Progression to PD
The additive interactions between smoking and baseline PEs on persistence of PEs and progression to PDs during follow-up are presented in Table 3. Persistence of PEs (OR = 1.5; CI [1.1, 2.2]; p < .01) and transition from PEs to PDs during follow-up period (OR = 5.3; CI [1.6, 17.8]; p < .01) was significantly associated with regular smoking behaviour at baseline, adjusting for proxy measures of other exposures and after performed Bonferroni correction. Test for additive interactions showed significant synergism between baseline regular smoking and PEs for persistence of PEs during the follow-up period (ICR = 1.8; CI [0.2, 3.6]; p < .05).
Additive Interactions Between Smoking and Baseline Psychotic Experiences on Persistence of Psychotic Experiences and Progression to Psychotic Disorders During Follow-Up.
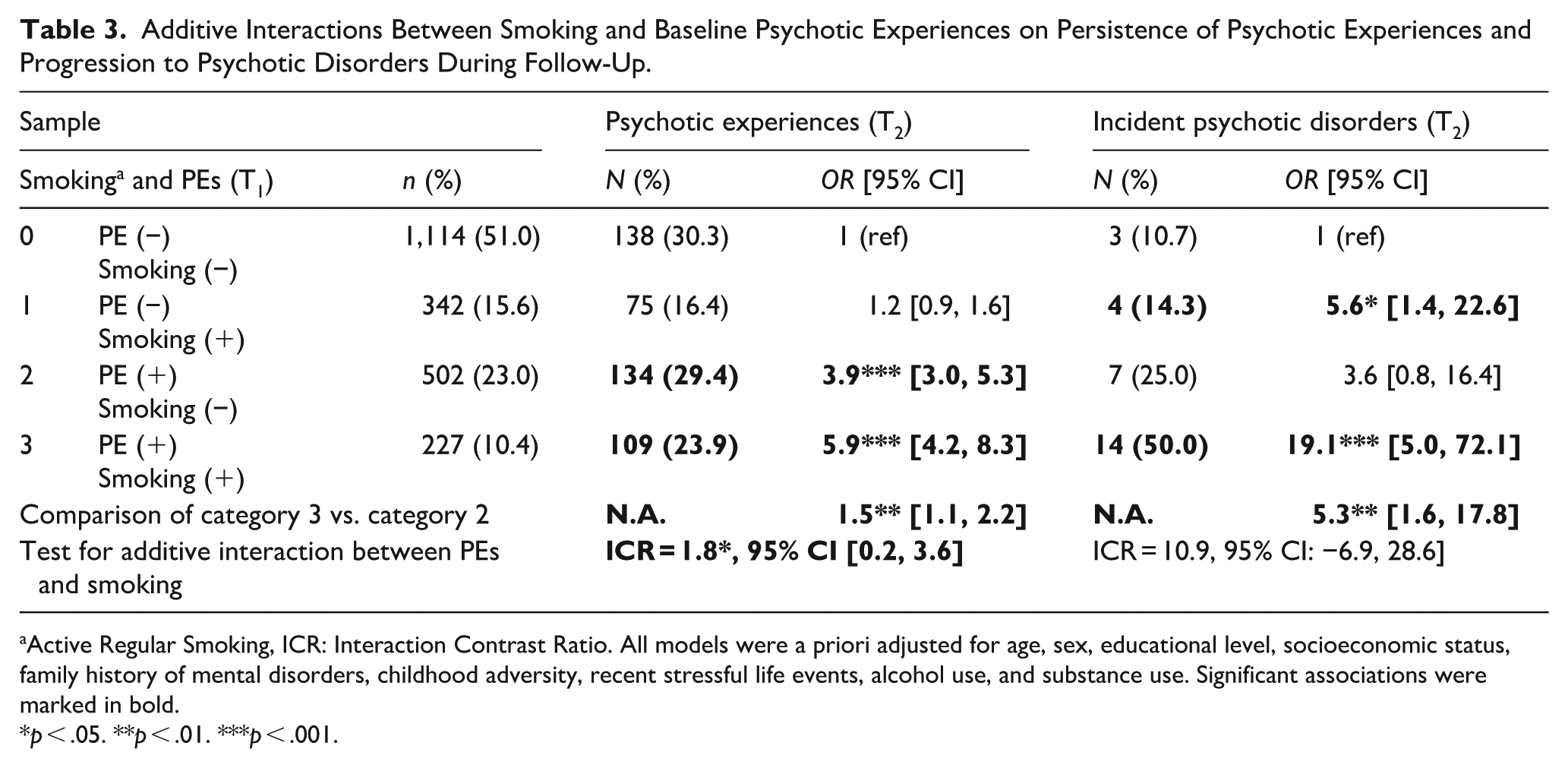
Active Regular Smoking, ICR: Interaction Contrast Ratio. All models were a priori adjusted for age, sex, educational level, socioeconomic status, family history of mental disorders, childhood adversity, recent stressful life events, alcohol use, and substance use. Significant associations were marked in bold.
p < .05. **p < .01. ***p < .001.
Discussion
In this paper, we evaluated the longitudinal associations between baseline smoking features and follow-up PEs and PDs in a 6-year interval of a community-based population. Furthermore, interactions between PEs and smoking cigarettes on the pathway to persistence of PEs, and progression to PDs were investigated. Our results indicated an increased risk of follow-up PDs in individuals who started smoking before the age of 15 years and have been smoking regularly. The increase in risk for follow-up PDs was not significant in individuals who ceased smoking cigarettes. In addition, smoking features such as no intention to cease and chain-smoking were associated with increased risk of follow-up PEs and PDs. Finally, our results showed significant evidence that baseline PEs and smoking cigarettes combined synergistically (on an additive scale) on the pathway to persistence of PEs.
Both incidence and prevalence of PDs are higher in tobacco smokers (de Leon & Diaz, 2005). It may be related to underlying neurodevelopmental problems in sensory gating and stimulus filtering in individuals who are prone to psychosis. Sensory gating mechanism is modulated by hippocampal nicotinic cholinergic neurons (Moxon et al., 2003). Prepulse inhibition, which reflects sensorimotor gating was reported to be reduced in high risk for psychosis subjects and untreated early psychosis patients (Quednow et al., 2008). Some evidence showed smoking cigarettes has positive effects on sensory gating deficits (X. Chen et al., 2011). Furthermore, individuals with early psychosis might have more tendency to smoke for the momentary self-medication of other prodromal symptoms such as decreased motivation, anergy, anxiety, and sleep problems (Gogos et al., 2019; van der Heijden et al., 2021). Our results also showed that the risk for follow-up PEs and PDs were higher in individuals with no intention to cease smoking cigarettes. Previous studies suggested that patients with PDs had lower motivation to give up smoking (Zeng et al., 2020). This may be related to poor insight about the hazards of smoking due to the momentary positive influence of smoking.
In our analysis, the risk for PEs and PDs at follow-up was not significantly increased in individuals who ceased smoking. Successful cessation in these individuals may partly be associated with the lack of underlying neurodevelopmental predisposition to PDs. In other words, these individuals might have used much more useful coping strategies with daily stressors and do not need such compensatory effects of smoking (J. Chen et al., 2016). Relevant associations were found between chain-smoking and follow-up PEs/PDs. This finding can also be related to the increased need for self-medication demand in individuals with relevant predisposition to psychosis (Coustals et al., 2020). Individuals with psychosis may only obtain a similar of effect with non-psychotic individuals by chain-smoking because of neurobiological dysfunctions (Ward et al., 2022).
The risk for follow-up PEs and PDs was significant in individuals who started smoking cigarettes before the age of 15 years old and have been smoking regularly so far. Association between the PDs and regular smoking in which participants started smoking after 15 years old, was not significant. This result suggests that smoking cigarettes during a critical developmental period may be associated with significantly increased risk for psychosis, which may be interpreted in two ways. First, smoking may be more common in individuals who have underlying neurodevelopmental predisposition to psychosis (Goff et al., 1992; Gogos et al., 2019; Lucatch et al., 2018). Second, smoking cigarettes itself may also be triggering some changes leading to psychosis in critical neurodevelopmental period (Scott et al., 2018). From the perspective of this results, it may be asserted that in the first 15 years of life is the most vulnerable period for psychosis, due to harmful and irretrievable effects of smoking. After the first 15 years of life, seems that smoking has not devastating effects on the neurodevelopmental process, which may trigger PDs.
Our results showed that smoking had an association with the poorer longitudinal outcomes of PEs than its association with the emergence of PEs. The association between smoking and incidence of PEs was below the conventional significance level. However, smoking was significantly associated with persistence of PEs and incidence of PDs during follow-up, adjusting for proxy measures of other exposures. Finally, test for additive interactions between PEs and smoking showed significant synergism on the pathway to persistence. The psychosis proneness-persistence-impairment model suggest that factors impacting during earlier period in lifespan (e.g. genetic factors) has an impact on emergence of PEs (Linscott & van Os, 2013; van Os & Reininghaus, 2016). Greater proportion of PEs are temporary during development. However, depending on the degree of subsequently exposed additional risk factors, the risk for poorer outcomes (e.g. persistence and transition to PDs) gradually increase. Our results may be consistent with this model. However, it is important to emphasise that our findings do not provide definitive evidence that smoking is a direct causal factor in the development of psychosis. Although several smoking-related variables were associated with the persistence of PEs and the transition to PD within the psychosis spectrum, our study design did not allow for the examination of the neurobiological mechanisms through which smoking may influence these outcomes. Therefore, the higher likelihood of persistent PEs or subsequent transition to PD among individuals who began smoking before the age of 15 years or who were regular smokers may also be explained by alternative hypotheses. Establishing a definitive causal pathway between smoking and the persistence or progression of psychotic symptoms requires more advanced research designs, such as Mendelian Randomisation studies. For instance, some individuals who later develop PD or schizophrenia may already exhibit subtle prodromal symptoms, and they may smoke more frequently in an attempt to alleviate these early manifestations. In addition, individuals who show both higher smoking levels and a greater likelihood of developing persistent PEs may share underlying genetic or neurodevelopmental vulnerabilities that predispose them to early initiation, more intense or consecutive smoking patterns, and lower motivation to quit. Thus, the association between smoking and poorer longitudinal outcomes in the psychosis spectrum may reflect a non-causal correlation rather than a direct causal effect.
Finally, although several sociodemographic and environmental variables were statistically controlled in our analyses, the influence of residual confounding cannot be excluded. For example, factors known to increase psychosis risk—such as migration status (Stilo & Murray, 2019) or winter birth season (Coury et al., 2023)—were not included in the present analyses and should be considered when interpreting the results. Further studies are needed to elucidate the mechanisms underlying these longitudinal associations between smoking cigarettes and development of psychosis.
The relatively large representative community-based sample, longitudinal design, comprehensive assessment including various types of exposures besides smoking cigarettes, inclusion of psychosis phenotypes in non-help seeking individuals, and construction of the dependent variables (PEs and PDs) via clinical interviews performed by physicians are important strengths of the study. However, our results must be interpreted with some limitations. The low number of individuals diagnosed with PDs at the follow-up assessment (n = 72) resulted in limited number of participants in some subgroups in the analyses with this dependent variable. Therefore, results of these analyses need replication, particularly for the clinical end of the phenotype. The use of the CIDI for symptom screening, rather than clinician-rated instruments like the PANSS or BPRS, may have limited the depth of psychopathological assessment. However, this was mitigated by clinical re-interviews for probable cases using the SCID-I. Childhood adversity was measured using a limited set of major life events and not a detailed, validated scale for maltreatment, which may have led to an underestimation of its true prevalence and impact. Furthermore, although we collected data on help-seeking and treatment, we lacked detailed, continuous information on antipsychotic medication use from health records throughout the follow-up period, which is a potential confounding factor we could not fully adjust for. Due to the longitudinal design with an epidemiological population, the sample was reduced through attrition. This could lead to potential sampling bias. However, analysis of attrition showed very small effect sizes (Cramer’s V value lower than 0.09) as a function of a series of baseline sociodemographic and clinical variables indicating evidence of weak attrition. Furthermore, the dropout rate is comparable with similar designed studies (Dominguez et al., 2011; Zammit et al., 2013). Therefore, it is unlikely that participants and non-participants at the follow-up assessment have been differentially distributed per smoking cigarettes. The time frame between the baseline and follow-up assessments might have led to recall bias and missing of some details in the association between smoking cigarettes and the extended psychosis phenotype. Unlike the data on psychosis phenotype and the covariates, data for smoking cigarettes was only available for the baseline assessment. Therefore, analysing the longitudinal associations from the opposite direction (i.e. from psychosis to smoking cigarettes) was not possible. Analyses were based on self-report information on smoking cigarettes and no biochemical measurement was present. In the literature, exhaled carbon monoxide (CO) is considered an objective biochemical indicator of smoking status, providing a sensitive measure of recent tobacco exposure as well as smoking behaviour (e.g. inhalation depth, puff number, and speed) (Bittoun, 2008). For this reason, exhaled CO measurement is widely regarded as the gold standard in the assessment of smoking status, and the absence of such an objective marker in our dataset should be acknowledged as a limitation. On the other hand, self-report measures may also be sufficiently informative on cigarettes use (Zhang et al., 2018). Finally, although data on a wide range of exposures was collected, it cannot be claimed that the associations with smoking cigarettes were adjusted for any possible exposures outside smoking.
Conclusion
This prospective study with a community-based sample showed that both the incidence and the prevalence of PDs are higher among cigarettes smokers. Furthermore, baseline PEs and smoking cigarettes combine synergistically on the pathway to persistence. Smoking cigarettes seems to be associated with poorer longitudinal outcomes of PEs. Further studies are needed to elucidate the mechanisms underlying the associations between smoking cigarettes and the extended psychosis phenotype.
The findings of this study have important implications for both clinical practice and mental health policy. The observed association between early-onset and regular cigarette smoking and the increased likelihood of persistence of PEs and transition to PD underscores the need to integrate structured smoking prevention and cessation programmes into youth-focussed and community-based mental health services. Restricting tobacco points of sale in school environments has been shown to reduce adolescents’ access and exposure to tobacco products. Similarly, increasing the distance between vendors and limiting the number of licensed retailers within a given area may contribute to a decrease in smoking among young people. Individuals presenting with PEs should be regarded as a high-risk group who may benefit from proactive screening for tobacco use and tailored cessation support. Mental health professionals should also be trained to address clinical misconceptions surrounding the self-medication hypothesis and to encourage smoking cessation rather than viewing tobacco use as an adaptive coping strategy. At the broader policy level, enhancing the availability and funding of smoking cessation interventions within mental health services, strengthening early prevention efforts aimed at reducing smoking initiation before age 15 years, and incorporating routine assessment of tobacco use into community mental health protocols may help reduce the long-term psychiatric burden associated with tobacco exposure. Public education and awareness initiatives alone may not be sufficient; they must be accompanied by raising the minimum legal age for tobacco sales and intensive regulatory enforcement strategies, such as routine identification checks, regular retailer inspections, and consistent application of license revocation or financial penalties for violations, to achieve meaningful population-level impact.
Supplemental Material
sj-docx-1-isp-10.1177_00207640251409953 – Supplemental material for The Effect of Smoking Cigarettes on the Progression of Psychotic Experiences to Psychotic Disorder: Evidence From a 6-Year Follow-Up Study
Supplemental material, sj-docx-1-isp-10.1177_00207640251409953 for The Effect of Smoking Cigarettes on the Progression of Psychotic Experiences to Psychotic Disorder: Evidence From a 6-Year Follow-Up Study by Eldem Güvercin, Tolga Binbay, Umut Kırlı, Bilge Targıtay Öztürk, Hayriye Elbi and Köksal Alptekin in International Journal of Social Psychiatry
Footnotes
Acknowledgements
We extend our gratitude to all participants involved in this study.
Ethical Considerations
This study was approved by the Research Ethics Committee of Ege University, both in 2007 (reference number: 07-5.1/10) and 2012 (reference number: 12-3/60).
Consent to Participate
Written informed consent was obtained from all participants.
Consent for Publication
Not applicable.
Author Contributions
Eldem Güvercin: Formal analysis, writing original draft, and review & editing. Tolga Binbay: Investigation, methodology, formal analysis, writing original draft, supervision, and review & editing. Umut Kırlı: Investigation, methodology, formal analysis, writing original draft, and review & editing. Bilge Targıtay Öztürk: Formal analysis, writing original draft, and review & editing. Hayriye Elbi: Investigation, methodology, funding acquisition, formal analysis, writing original draft, and review & editing. Köksal Alptekin: Investigation, methodology, formal analysis, writing original draft, supervision, and review & editing.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is a part of the TürkSch Project, funded by the Scientific and Technological Council of Turkey (TUBITAK) 1001 programme, under project no: 107S053 and 112S476.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Köksal Alptekin has received grants and honoraria for consulting work, lecturing and research from Boehringer Ingelheim, Abdi İbrahim, Abdi İbrahim Otsuka, Janssen, ARİS, Nobel, Santa Farma, EU, and TUBITAK.
Data Availability Statement
The datasets analysed during the current study are available from the corresponding author on reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
