Abstract
Recently, the combination of electronic devices with living insects has been explored to manipulate their crawling or flying, similar to manipulable biological actuators. However, the investigations of controlled flight cessation or flight modulation of the coleopteran characterized by the strong payload capacity have rarely been reported thus far. On the basis of anatomy, this study proposed and validated two separate muscular electrical stimulation protocols to stop and modulate the flight of coleopteran, respectively. The experiment results demonstrated that the suggested cessation protocol realized flight cessation control with a success rate of 83% and a short response time. Meanwhile, the flight modulation stimulation protocol was able to increase the flapping frequency by an average of 7.3%. Finally, we designed a wireless miniature backpack weighing 1 gram to actuate the flight cessation and modulation of coleopteran in free-flying through the remote command transmission. The proposed method has potential applications in the development of the insect-cyborg micro air vehicle.
Introduction
Insects exhibit impressive flight skills over eons of evolution. Engineers have been dedicated to researching those naturally occurring fliers and building biomimetic micro-aircraft vehicles (MAVs) for several decades. 1–5 However, the design of insect-scale bionic aircraft with comparable aerodynamic performance to a natural insect remains a challenge. 6,7 In recent years, a novel approach called insect-machine interface (IMI) that directly manipulates insects' movement behaviors by either neural or neuromuscular stimulus has been reported. 8–12 The actuation of such insect-machine hybrid robots is generated by insect platforms instead of artificial actuators. Therefore, these hybrid designs feature low power consumption and stable locomotion capabilities.
One fundamental challenge in developing the insect-machine hybrid robot is to establish a stable flight cessation protocol. In most insects, such as bees and fruit flies, the flapping stops immediately after touchdown. 13,14 Controllable flight cessation allows researchers to steer the hybrid insect to land at a specific position. For instance, hybrid insects that carry miniature devices, such as the camera or microphone, can be dispatched to the narrow and hazardous environments inaccessible to humans as disaster rescue biobots (e.g., localizing survivors in disaster areas). Researchers have demonstrated several controllable flight cessation in insects through a wide spectrum of neural actuation mechanisms. For example, the flight of Apis mellifera could be immediately terminated by the electrical excitation of the brain center. Similarly, the same cessation behavior could be realized in Manduca sexta through stimulating the antennal lobe. 15 The stimulus on the beetle’s optic lobes 16 or simultaneously on the brain central complex and ventral nerve cords 17 could also stop its flying behavior. Nevertheless, the optic lobe stimulation will inevitably impair the photoreception of the beetles and thereby cause flight discontinuity. 18,19 Furthermore, this kind of neural stimulation requires precise operations to identify target tissues and implant electrodes into tiny neurons with microscopic auxiliary instruments. 9,16
Compared with neural stimulation, stimulating the specific muscles is a more direct method as the sub-centimeter dimensions of muscles facilitate the location and implantation of the electrodes in the absence of auxiliary tools. The muscular stimulation has been applied to control insect biobots (e.g., moth and beetle) to realize guided flight maneuvers, including flight initiation, 19–21 cessation, 22 and directional steering. 20,23,24 Nevertheless, the proposed cessation technique in moth flight 22 relied on medication injections to paralyze the dorsoventral muscles (DVMs) and the dorsal longitudinal muscles (DLMs) as these two muscles are responsible for powering the wing beat. 25,26 Both the complexity of the microfluidic system and the impairment of the chemical drug are the impediments to wider application of the method. For beetle, several groups have reported their work on stimulating the DLM and further exploring the optimum pulse parameters to elicit the flight initiation, 19,27 but the potential of those muscles (DLM and DVM) on flight cessation has not been explored thus far.
Meanwhile, the wingbeat frequency variation is a common flight modifying behavior for insects with asynchronous flight muscles. For instance, the tethered Drosophila exhibits an average 10% increment in wingbeat frequency as a response to the visual stimulus, 28 and the bumblebee adapts to different loadings through either wingbeat frequency or stroke amplitude. 29 For Mecynorhina torquata (M. torquata, beetle), Sato et al. demonstrate that the brain stimulus is able to depress its flight. 30 As the power source and end-effector for wing-flapping activity, 31,32 the beetle’s DLM and DVM (asynchronous flight muscles) probably also play a crucial role in its wingbeat frequency modulation.
Herein, we pose and verify a hypothesis that stimulations exerting on either or both the DVM and DLM will interfere with beetles' (Mecynorhina torquata) flight modality, including the induction of flight ceasing or modulation. The anatomy of beetles was first conducted to observe and locate the flight muscles. Secondly, we made a comparison among three possible stimulation methods (DLM-stimulation, DVM-stimulation, and DLM-DVM combined stimulation) and analyzed the experimental data to propose a generally applicable flight cessation protocol. According to the statistics, the proposed cessation protocol based on DLM-DVM combined stimulation was selected. Thirdly, a miniature wireless electronic backpack equipped with a microphone successfully recorded the acoustic information of the flapping and fulfilled flight cessation control in multiple flight conditions. Fourthly, we demonstrated the modulation of wing oscillations frequency (frequency increase) through a weak DLM-DVM combined stimulation and quantitatively recorded their responses. Finally, we made a possible explanation for this frequency variation.
Materials and methods
Beetles
Mecynorhina beetles were kept in chambered transparent plastic boxes with moist sphagnum moss and fed with beetle jelly or banana every other day. The room was maintained LED light for 13 h every day, similar to the daylight duration in nature. Beetles were all purchased from the same insect breeding company. The species do not belong to the protected insects prescribed by our government and can be traded freely. Our university or institute only carries out the ethical review for experiments related to humans and animals, and insect-related experiments do not require ethical review.
Wireless Communication
The wireless communication system consisted of two customed printed circuit boards (PCB) equipped with NRF52840 microcontrollers (7×7 mm2, 64 MHz clock, 2.4 GHz IEEE 802.15.1 compliant RF transceiver Chipcon, Nordic semiconductor); one served as the beetle-mounted electronic backpack (Bluetooth slave), and the other served as the computer-driven RF transceiver (Bluetooth host) (Figure 1(a)). The computer-driven RF transceiver implemented RF transmission with a signal amplifier (1w) and a whip antenna (10 dB) to achieve stable communication in a long distance. (a) Schematic illustration of the whole wireless communication system. (b) Photo of an untethered beetle with an electronic backpack mounted on the posterior pronotum for muscular stimulation. (c) Details of the assembled backpack. LDO is the abbreviation of low dropout regulator. (d) Device setup for the Bluetooth host side. (e) Weight breakdown of the electrical backpack, including printed circuit board, antenna, battery, and electrodes, as well as each component’s percentage of total weight.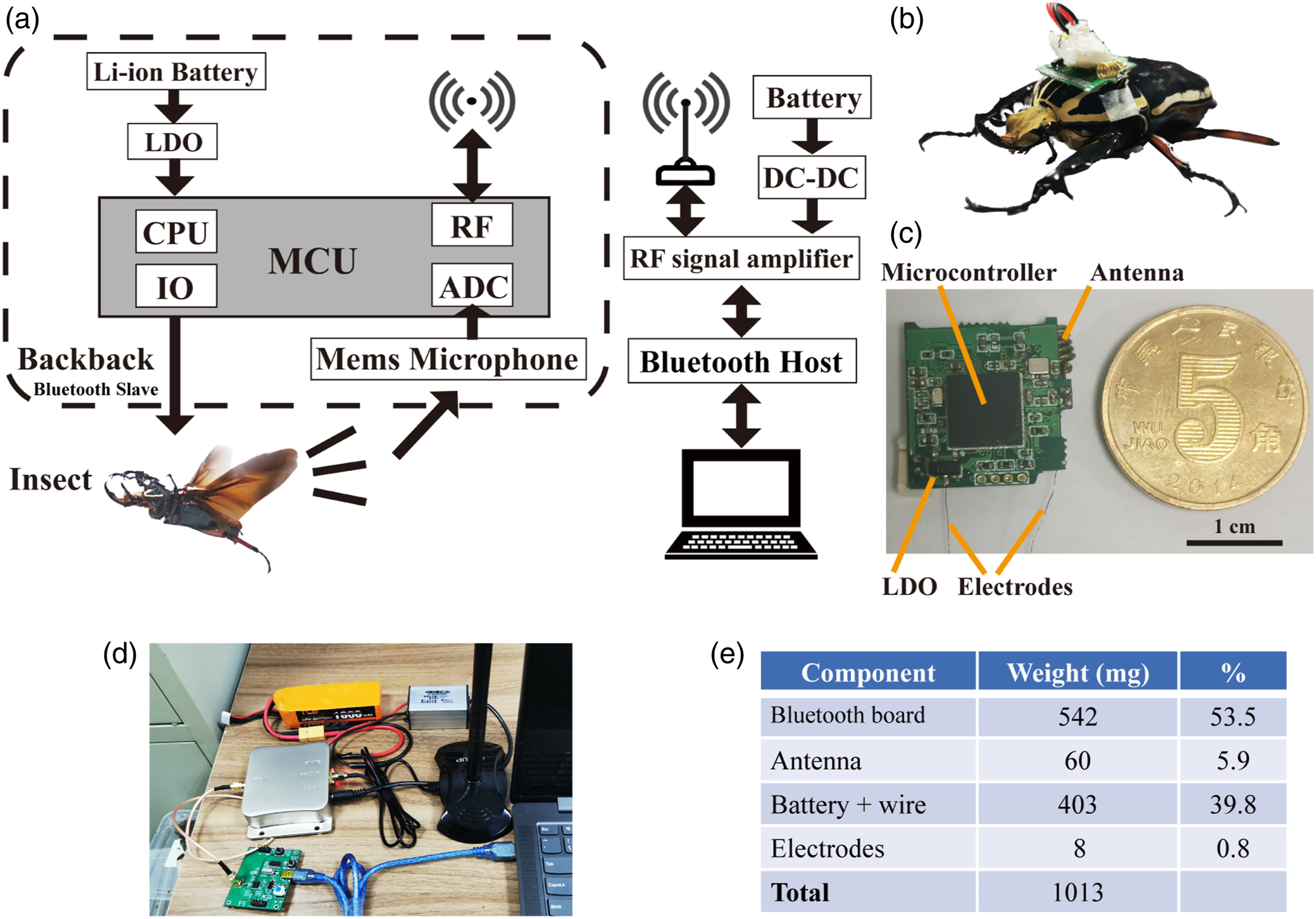
The backpack was powered by a rechargeable li-polymer (Li-Po) battery mounted atop the PCB (3.7 V, 403 mg, 20 mAh). Additionally, the backpack required a 2.4 GHz antenna for transmitting and receiving signals. Although lightweight chip ceramic antennas were available, they needed large ground planes and clearance to other components to achieve better performance. 33 Considering that the required clearance and ground plane exceed the desired PCB size, we designed an alternative helical antenna utilizing 26-AWG wire. The backpack equipped with this helical antenna was capable of transmitting audio data up to 80 m to the RF transceiver (Bluetooth host). A dedicated LABVIEW program was run on the PC to command the backpack through the RF transceiver (Figure 1(d)). The backpack could stimulate flying beetles and transmit data to the computer and weighed 1013 mg in total (Figure 1(e)).
Anatomical observation
The prothorax and abdomen were removed to dissect the pterothorax to which the wings and flight muscles were attached. We carefully cut off the elytra and scutellum (Figure 2(b)), followed by the cuticles (Figure 2(c)), to observe the flying muscles underneath. It was clear that the dorsoventral muscles (DVMs) were located on both sides of the dorsal longitudinal muscles (DLMs), and the high concentration of mitochondria and cytochromes gave the exposed DLMs and DVMs a characteristic pinkish-brown color.
18
Then, we dissected the pterothorax longitudinally to further study those muscles (Figure 2(d)). The DVMs and DLMs were exposed after the scrupulous removal of the sternal apophysis. The DVMs vertically run from the tergum to the sternum, and the DLMs horizontally run from prephragma to postphragma. As shown in Figure 2(e), the DLM-attached postphragma could indicate the horizontal position of DVMs and the vertical position of DLMs in the thorax. We observed that the average horizontal length (Haverage) of the postphragma was 9 mm, and the average vertical distance (Vaverage) from the metasternum to the left or right edge of the postphragma was 10.3 mm (n = 5 beetles). Those measurements assisted us in determining the implantation sites and depth. Anatomical views of the beetle and the implantation sites for electrical stimulation. (a) Dorsal view and ventral view of a beetle with DVM implantation sites. The electrodes were inserted into DVMs via holes on the sternum (site 1, site 2). (b) Close-up views of the cuticle after removal of the elytra and scutellum. (c) Close-up views of the DLM and DVM after removal of the cuticle. (d) The sagittal section of the pterothorax (right half pterothorax) showed the relative positions of DLMs and DVMs. (e) The cross-section of the metathorax indicated the DVMs stimulator sites, the postphragma DVMs attached, and the sternal apophysis. Haverage was the average horizontal length of the postphragma, and Vaverage was the average vertical distance from the metasternum to the left or right edge of the postphragma. DVM = dorsoventral muscles; DLM = dorsal longitudinal muscles.
Electrode implantation
Firstly, the beetle was anesthetized by placing it in a plastic box filled with food-grade CO2 for 90 s. Exposure to CO2 was the usual anesthetic method in entomological research, but the anesthetic effect normally lasted only several minutes.
34,35
Secondly, the exoskeleton around implant sites was polished with abrasive paper to rough the surface. Thirdly, several small holes were made on the exoskeleton using an insect pin (00#). The Polyvinylidene Fluoride (PVDF)-coated thin silver wire (a diameter of 100 μm) was chosen as the electrode. And then, we removed the insulation layer with a wire stripping tweezer to bare the wire core and cut it short to 4 mm with a scissor. Later, a pair of silver wires were inserted into the holes for 9 mm (site 1, site 2) to the left and right DVMs. The DLMs located in the mid-domain of the thorax were sandwiched between the left and right DVMs (Figure 2(c) and (d)). Therefore, a pair of electrodes embedded into the DVMs was anticipated to achieve the simultaneous contract of both kinds of muscles (Figure 3(b)). The selection of stimulation sites was based on pre-comparative experiments provided in supplementary materials. The principle of the different stimulation protocols and corresponding experiment appliances. (a) Photograph of a beetle tethered on a frame to perform flight cessation experiment. (b) The principle of DLM-DVM combined stimulation. Blue represented muscle relaxation, and red represented muscle contraction. The darker red indicated the intensified muscle contraction. In natural flight (left figure), the contraction of the DLM raised the tergum causing the wing to flap down. Then, the contraction of the DVM pulled the tergum down and hence also brought the wing flaps up. Appling stimuli through electrodes implanted into the DVMs on both sides(right figure), the current can flow through the left-DVM, DLM, and right-DVM in turn. The method could achieve the simultaneous contract of both kinds of muscles. (c) Schematic of flight modulation experiment. (d) Photograph of a beetle rotated the inner ring as it flaped the wing. DVM = dorsoventral muscles; DLM = dorsal longitudinal muscles.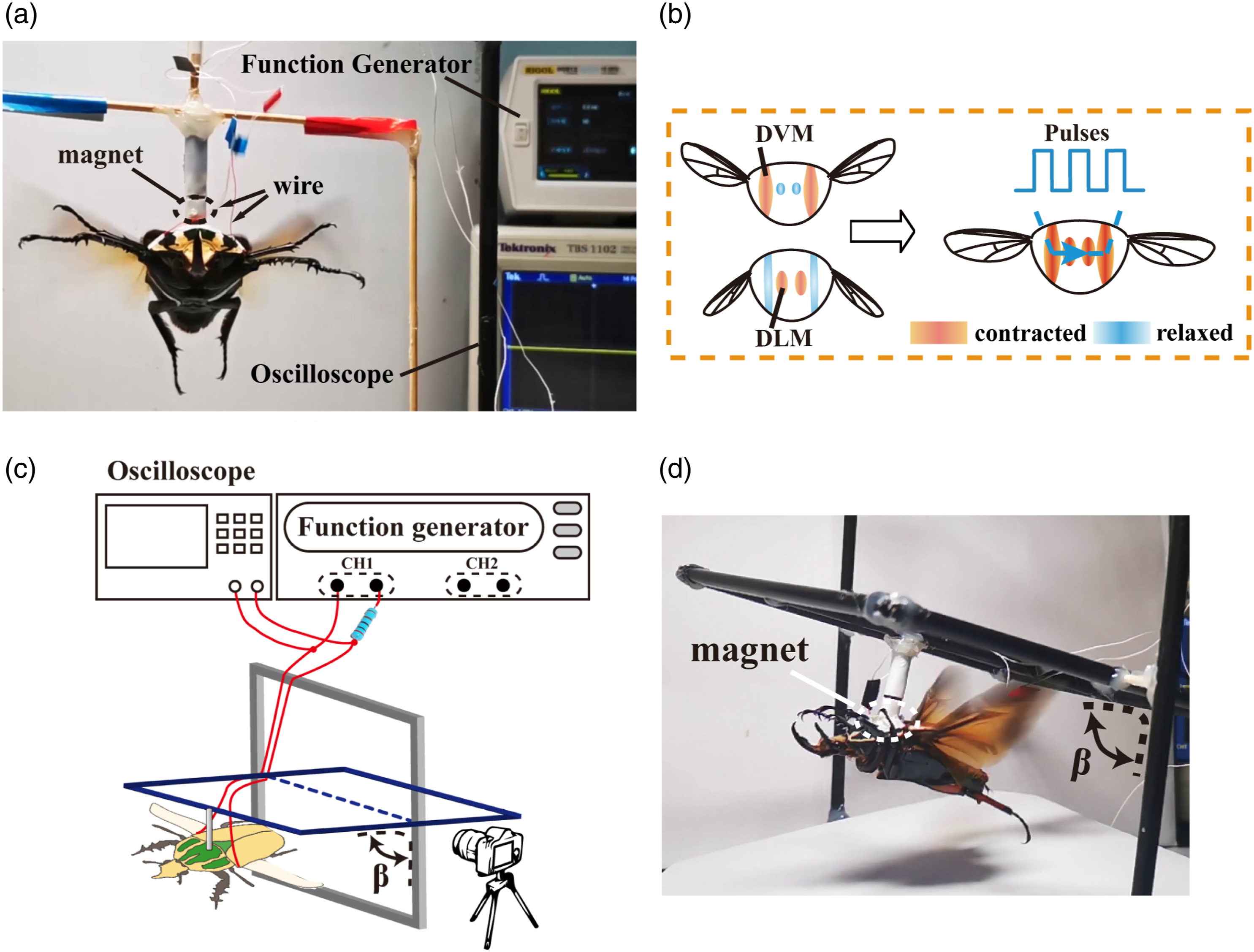
After insertion, several small pieces of paper adhesive tape were attached to immobilize electrodes. The adhesive tape was more convenient than the previous gluing with melt beeswax 19,21 as it did not need heat, and the tape was thin and sticky enough to reduce the inevitable interference from legs. For example, the midlegs would rub the beeswax-caused protrusions on the surface of the metasternum, which probably further lead to the electrode drift.
Flight cessation experiments
The experimental device and preparation
Prior to stimulation, the target beetle was tethered to the frame with a magnet (Figure 3(a)). The stimulus pulses could be generated by the function generator (RIGOL DG812) as well as the electronic backpack. In order to protect the target muscle, the stimulation circuit connected a current-limiting resistor (1kΩ) in series. Simultaneously, the effective voltage drop on the target muscle was monitored by an oscilloscope (Tektronix, TBS 1102). Based on the stimulation signals used in previous insect control studies, 12,15,17,23,27,36 a number of pilot tests were conducted for the purpose of getting proper stimulation parameters. We preliminarily selected the parameters of pulse trains at 50 Hz, 50% duty cycle, 1 s duration, with a voltage range of 1.5 V–5V. The stimulation signal was proved as reliable for inducing muscular activities in beetles. The flight was induced by airflow over the beetle and then was filmed at 30 frames per second during the trials. The cessation response time was calculated by the frame-to-frame playback on open source software Kinovea. 37 We counted the number of frames from the stimulation signal trigger to the apparent flapping cessation behavior (the last wingbeat). Besides, we employed a high-speed camera to observe the beetle’s response after stimulation. The camera sampling rate was 1000 Hz, considered sufficient to capture wing movements.
Statistical analysis of the wingbeat cessation experiment
A pilot experiment was conducted to compare three muscle group stimulation for flight cessation (DLM-stimulation, DVM-stimulation, and DLM-DVM combined stimulation). Details of the comparison experiment were given in supplementary materials. The DLM-DVM combined stimulation was experimentally proved to be satisfactory for the high success rate (Figure S1). To determine the stimulus parameters with universal applicability, we further analyzed the distribution of flight cessation threshold voltage under the determined optimal muscle group stimulation (the DLM-DVM combined stimulation, Figure 2(a), site 1, site 2). In this section, flight cessation experiments started by applying 2V amplitude pulse trains as described in section 2.5.1, and the amplitude was then increased by 0.5 V. This was repeated until the beetle stopped flight or the value reached 5V. We defined the amplitude at which the flight cessation occurred as the cessation voltage. This test was repeated ten times for each beetle, and we tested six beetles in total (n = 60 trials).
Finally, we would move on to evaluate the obtained flight cessation protocol (drawn from section 3.1, DLM-DVM combined stimulation, 3.5 V, 50 Hz, 50% duty cycle, last for 1s). Eight beetles were tethered as described in section 2.5.1 and then stimulated with the proposed protocol. For each target beetle, stimulation and observation were repeated ten times (n = 8 beetles, n = 80 trials). If the beetle stopped flapping within 2 s, we recorded it as a successful trial, otherwise as a failed trial.
Loosely tethered-flight and free-flight control
As an application, we demonstrated the flight cessation with the obtained cessation protocol in loosely tethered flight and free flight conditions. Firstly, we fixed the electronic backpack onto the posterior pronotum of the beetle with double-sides adhesive tape, which featured easy assembly and disassembly. After initializing the LABVIEW program, the RF transceiver and the electronic backpack were powered in turn. The LED light on the RF transceiver indicated the connection status of the Bluetooth network. Steady light meant the network connection, and twinkling represented the disconnection. Finally, in the loosely tethered-flight experiment, the beetle was wired in a limited area for recording and stimulation. Moreover, in the free-flight experiment, the beetle was induced to initiate flight and gently threw forwards into the air. Wirelessly commanded by the LABVIEW software, the backpack applied electrical stimulation to the beetle.
Modulation of wing oscillation
Insects did not always exhibit flight attenuation or cessation behavior under DLM-DVM combined stimulation. As the studies have advanced, we found a mild stimulus (alternating positive and negative pulse at 2V, 100 Hz, and 10% duty cycle) would enhance the wingbeat rather than depress it. For example, the loudness of the flapping would considerably rise.
In this section, the beetles' responses to stimuli were characterized by tethering them to one custom-fabricated gimbal (Figure 3(c) and (d)), which was designed to lower the horizontal axis turning resistance. Therefore, the beetle could only push the gimble to spin around the fixed axis so as to change the pitch angle during flight while constraining other degrees of freedom. A camera (30fps) was simultaneously adopted to capture the beetle from the side view. To faciliate pitch angle analysis, the filmed videos were processed using the angle tracking function of the motion analysis software Kinovea. 37 Moreover, the wireless backpack recorded the audio information of the insects flapping and then transmitted it to the computer. The received audio data were then normalized and plotted as a scatter diagram using the customized MATLAB software. We also employed a high-speed camera to observe the beetle response, such as the change of flapping frequency, under the weak muscular stimulation. The camera sampling rate was 1000 Hz, considered sufficient to capture wing movements.
Results
Statistical analysis of beetle wingbeat cessation
Figure 4(a) depicted the voltage distribution of flight cessation under the DLM-DVM combined stimulation (N = 6 beetles, ten times each beetle, n = 60 trials). The voltage ranged from 2.0 V to 5V in all successful flight cessation trials (nsuccess = 54 trials). More than two-thirds of flights stopped with a 2.0V–3.0 V amplitude, and less than 10% stopped with an amplitude higher than 3.5V. We hypothesized that the pulse trains with a voltage higher than the value, which was able to stop flight in at least 80% trials, could be considered as the stimulation with universal validity. It was apparent that beetles stopped stroking at pulse amplitudes below or equal to 3.5 V in most trials (n = 49, indicated by the blue blocks). Therefore, we concluded that the cessation protocol (3.5 V, 50 Hz, 50% duty cycle, lasting for 1s) was effective in most flight cessation trials. Quantitative analysis of the beetle flight cessation. (a) The distribution of the cessation voltage required to trigger flight cessation (N = 6 beetles, ten times per beetle, n = 60 trials). The voltage values from 2V to 5V were shown with the appropriate colormap. Blue shades characterized cessation threshold voltages below 3.75 V, and red shades characterized those higher than 3.75 V. Black shades indicated that the failed trial (the beetle did not stop flight). (b) The variation of wing flapping angle in the flight cessation trial. As shown in the inset, we defined the angle between the tangent to the costal margin and the horizontal line as the wing-flapping angle. The points c1 to c5 indicated five moments shown in (c). (c) Photos of high-speed filming corresponding to the five points (c1-c5) identified in (b).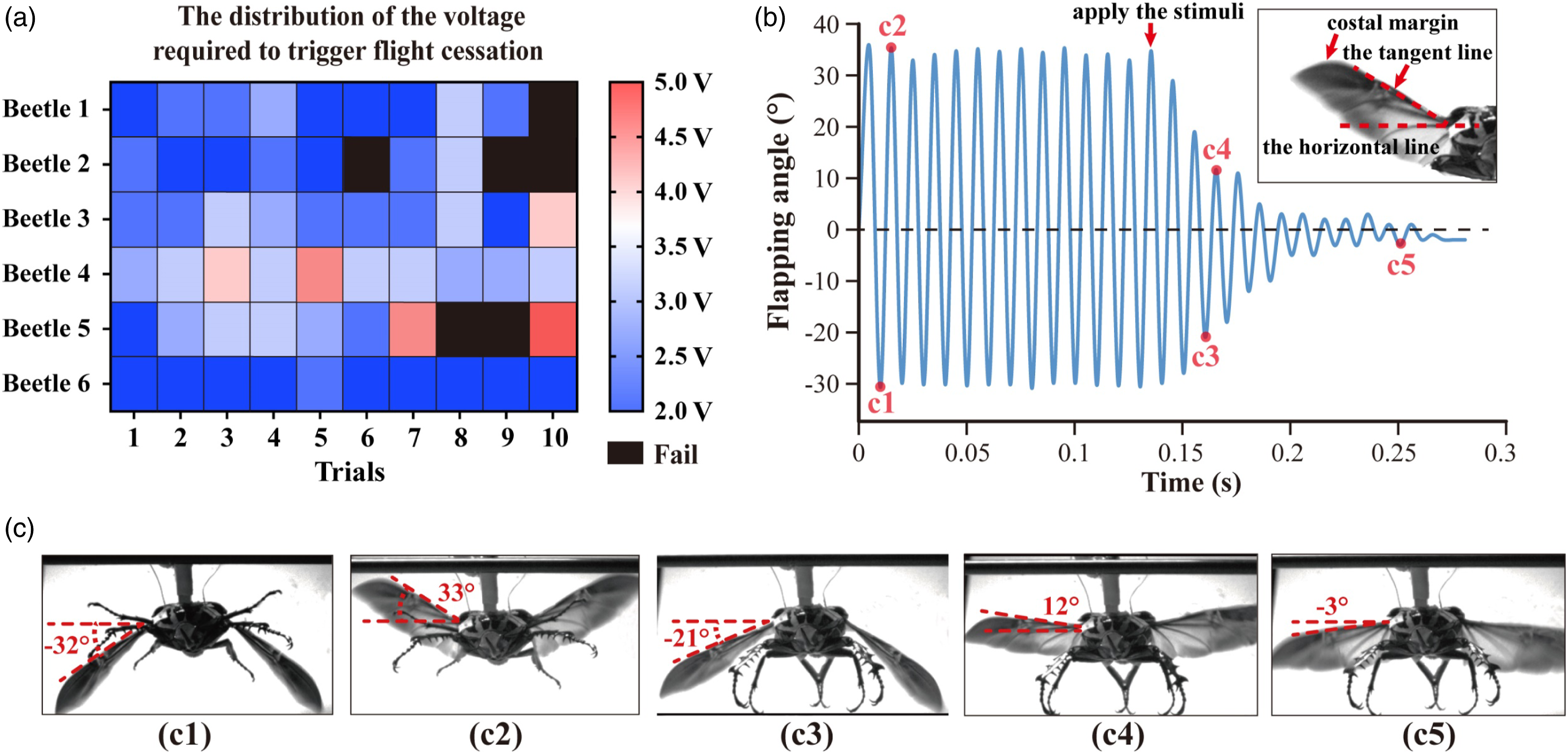
Sequences of high-speed camera images recorded some characteristic states of the beetle flight. Figure 4 (c) provided images of the wingbeat at the maximum and minimum flapping angles of this stroke cycle in natural flight (c1, c2) and DLM-DVM combined stimulated flight (c3, c4, c5). The definition and variation of flapping angle during the flight were given in Figure 4 (b). The observation of multiple flight trials (N = 3 beetles, five times per beetle, n = 15 trials) showed the absolute value of the difference between the maximum and minimum flapping angle in normal flight was 58 ± 5°. Notably, the flapping angle decreased rapidly, and forelegs and middle legs fell underneath the thorax after the DLM-DVM combined stimulation. As the wingbeat stopped gradually, the flapping angle fluctuated until it approached a value close to zero (Figure 4(b)).
For further estimating the reliability of the obtained flight cessation protocol, eight beetles were stimulated with the protocol to evaluate the success rate (Figure 5(a1)). As a result, it was found that in the majority of cases, the stimulated beetles stopped oscillating their wings with a success rate of 83.75% (N = 8 beetles, ten trials per beetle, nsuccess = 67 trials, Figure 5(b)). Moreover, beetles showed a short response time ranging from 0.1s to 1.9 s in all successful flight cessation trials. Although the increase of response time occurred in a minority of stimulation trials (Figure 5(b)), more than three-quarters of flights stopped within 0.5s, and less than 8% of flights stopped with response time longer than 1s. The lengthening time was still sufficiently short for practical application (below 2s). The results implied that the flight cessation protocol proposed in this section was substantiated to be effective under the given criteria. Demonstration of the flight cessation experiment under tethered (a1), loosely tethered (a2), and free-flight conditions (a3). (b) The distribution of response time to flight cessation in all successful trials. (N = 8 beetles, 10 times per beetle, nsuccess = 67 trials).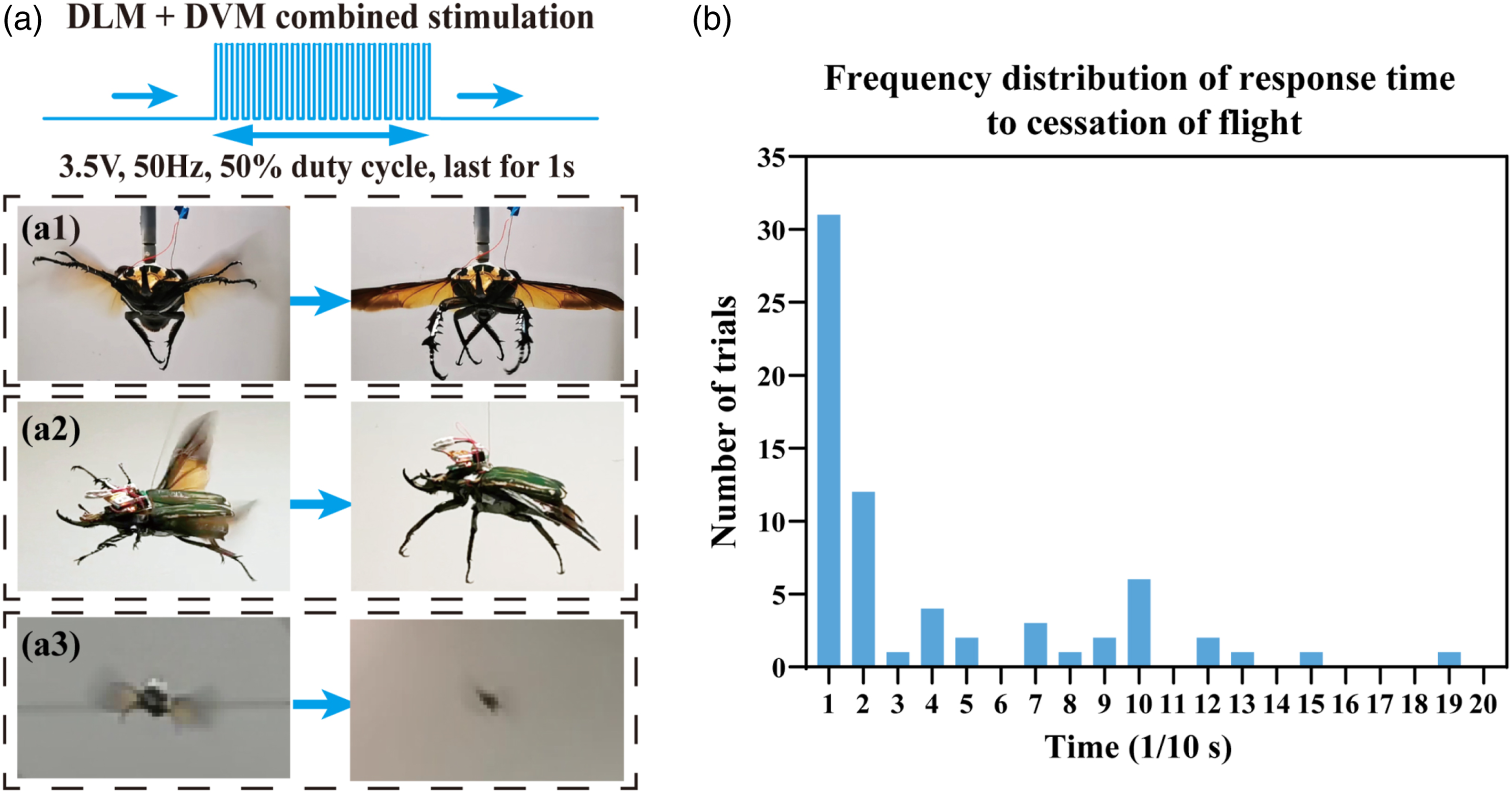
A demonstration of flight cessation was performed in loosely tethered (Figure 5(a2)) and free-flying (Figure 5(a3)) conditions. In loosely tethered flight (Movie S1, 00:00:17-00:00:48), the stimulated beetle stopped flapping its wings and swayed in the air owing to inertia until the next flight. It was noteworthy that the folding of hind wings in those manual intervention cessations was much later than in natural landings, or the folding did not happen. This same behavior was also observed in free-flight experiments. In the free-flying flight (Movie S1, 00:00:48-00:01:05), the electrical stimulation applied by the electronic backpack induced beetles to stop flapping in the air and drop to the ground. Multiple free flight cessation rounds were possible for most tested beetles in the experiment. We did not observe the inevitable loss of steerage under the optic lobe stimulation reported in previous literature 16,19 and did not observe crucial damage to the insects caused by the muscular stimulation. The beetles' behaviors, including eating, mating, and flapping, were as normal as intact beetles and had an average life span of more than 6 weeks in the laboratory.
Modulation of wing oscillation
Through the analysis of the flight video, we noticed that the alternating positive and negative pulse stimulus (2V, 100 Hz, 10% duty cycle) resulted in an increased pitch angle of the gimbal and a higher amplitude (>20%) in the flight audio recordings (Fig. 6(a) and Movie S2). The pitch angle of the un-stimulated flight was between 5° and 10°. The value was determined by the flight thrust as well as the structure and mass of the gimbal. During the stimulated flight, there was a clear increasing trend of the pitch angle (increased by about 15°, marked by the red dashed rectangle). The oscillation of pitch angle was due to the lightweight gimbal and the lack of damping of the horizontal rotation axis. Furthermore, the increase and decrease of the pitch angle were synchronized with the beginning and end of the stimuli, and the time extension of enhanced flight accompanied the progressive extension of the stimuli duration. The behavior showed that the electrical stimulation and the induced wing oscillations modulation occurred simultaneously, without conspicuous time hysteresis. The variation of flight pitch angle and audio signal of the beetle during the flight modulation experiment. (a) Muscular stimulus altered the wing oscillation, hence the pitch angle of the gimbal, during alternating periods of unstimulated and stimulated flight. Red and green arrows indicated the beginning and end of the stimuli. The audio signal was synchronized with the pitch angle in abscissa (Time). (b) The changes in wing flapping frequency before and during stimulation. Each frequency indicated in Figure 6(b) was obtained by calculating the average frequency of fifty consecutive wing strokes in each trial. The frequency was calculated at intervals of 0.1 Hz. (c) Comparison of the wing oscillation speed of unstimulated and stimulated flight in the same beetle. After 163 ms, the stimulated flight (right, stroke frequency: 54.5 Hz) started the 10th pronation (indicated by red cycles), while the unstimulated flight (left, stroke frequency: 52 Hz) ended the 9th pronation (indicated by red cycles). The difference of wingbeat phase (180°) can be observed.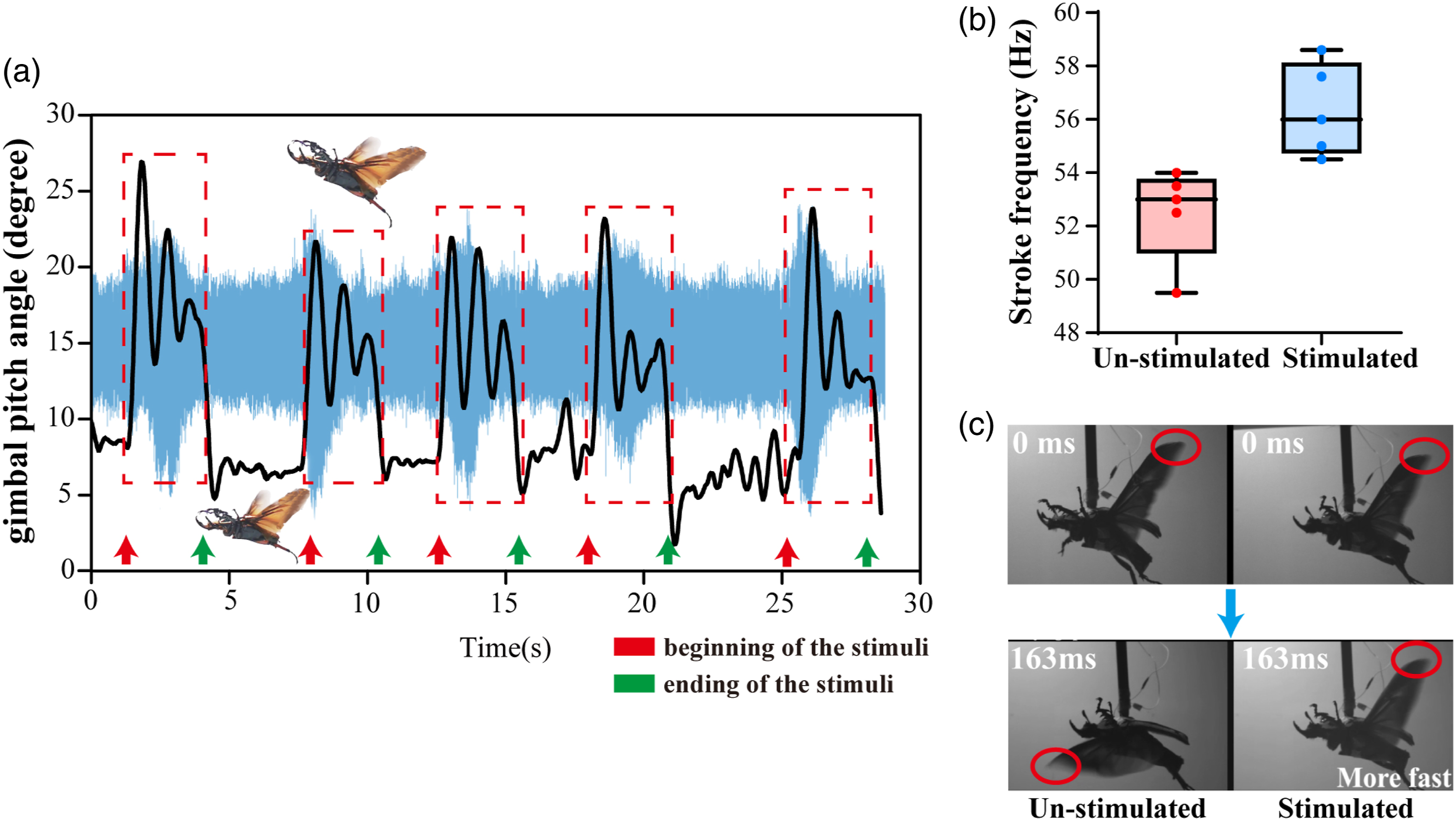
The high-speed camera video demonstrated that, compared with the unstimulated beetle, the average wing-flapping frequency of the stimulated beetle increased from ∼52 Hz to ∼56 Hz (Figure 6(b)). Among the five tested beetles, all beetles showed a similar increasing tendency. Figure 6(c) and Movie S3 demonstrated a representative moment of comparing unstimulated and stimulated flight in the same beetle. Both video clips were synchronized to start at the same wing beat phase (beginning of downstroke). After the same time (163 ms, in 00:21.80 of Movie S3), there was a pronounced wing beat phase difference between the unstimulated and stimulated flights. The stimulated flight started the 10th pronation, while the unstimulated flight ended the 9th pronation. Therefore, stimulated flight (right) resulted in faster wingbeat frequency than un-stimulated flight (left).
Discussion
In general, the flight cessation behavior of beetles proved that the stimulus decoupled the oscillation state established by the alternate contraction of the DVMs and DLMs. The electrical stimulation activated two muscles together, which induced the muscles in a tetanic contractions state. 38 Thus, those muscles were not able to stretch and activate in the next stroke cycle. If this continued for a long duration, the flapping oscillation was broken. We thought the similar threshold cessation behavior could be a generalized decoupling of fibrillar flight muscle oscillation as a response to a long but moderate stimulus. 16
A possible explanation for the elevation of flapping frequency was that mild electrical pulses altered the thorax’s resonance properties by affecting muscle contraction. Pringle et al. had proved that insects with asynchronous muscles stroked wings at the frequency of the wing-thorax mechanical resonant system rather than of the neuromuscular signal. 31,39,40 Previous articles have suggested that insects modulated their wingbeat frequency by tensioning their thorax (increasing the stiffness) via pleurosternal muscle contraction, 41,42 and the stiffness of the resonant system was proportional to the square of oscillation frequency. 42,43 Therefore, we hypothesized that our thoracic muscular stimulation caused or enhanced the contraction of some thoracic muscles, and the generated thorax modifications (stiffness) altered the dynamic response (resonant frequency) of the thorax and finally modulated the flapping frequency.
Meanwhile, insects could utilize wingbeat frequency modulation to overcome flapping-wing instabilities. 28,29,44,45 The direct correlation of beetle wingbeat frequency with the thorax resonant frequency provided a simple mechanism for the insect to adjust wingbeat frequency. It was an important mechanical property for active flight. Therefore, the increase of wingbeat frequency could also be speculated as an instinctive response of insects to eliminate extraneous interference.
Conclusion
In this paper, we reported two muscular electrical stimulation protocols for flight cessation and modulation to transform the beetle into a controllable flight actuator. We compared the beetle flight behavior under three different muscle stimulations (DLM-stimulation, DVM-stimulation, and DLM-DVM combined stimulation) and found that DLM-DVM combined stimulation has the highest cessation success rate in comparison with just stimulating one type of muscle. According to the statistical analysis, flight cessation could be induced with a success rate of over 80% and a relatively short response time under our proposed protocol. The remote flight cessation control was successfully demonstrated in free flight with a miniature wireless stimulation backpack. Moreover, beetles increased the frequency of wing oscillation by 7.3% in response to the alternating positive and negative pulse stimulus (2V, 100 Hz, and 10% duty cycle). The formation of this frequency fluctuation was probably explained by either the passive change in thoracic resonance frequency generated by electrical stimulation or the beetles' active adjustment required to overcome interference.
We noticed that insects with asynchronous flight muscles have similar wingbeat principles and muscle distribution. Therefore, the reliable cessation protocol might have some applications in the future development of insect cyborg actuators or biobots. Our future work will focus on fabricating flexible neural probes with biocompatibility and durability and designing backpacks with multiple sensors that endow the cyborg beetle robot with more practical functions.
Supplemental Material
Supplemental material - Flight cessation and modulation control of coleopteran employing wireless miniature muscular stimulators
Supplemental material for Flight cessation and modulation control of coleopteran employing wireless miniature muscular stimulators by Yongchang Jiang, Bo Yang, Ye Jiang, Wenhao Zhao and Xin Guo in Measurement and Control
Supplemental Material
Supplemental Material
Supplemental Material
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funding by the National Natural Science Foundation of China, grant number 61571126.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
