Abstract
The incidence of stroke in India is one of the largest worldwide. With this scoping review, we assessed the evidence on the costs of stroke, which is essential to evaluate whether stroke care interventions are cost-effective. We adopted a healthcare sector and broader societal perspective and searched electronic databases for records including stroke cost estimates (up to 2020). Following deduplication and screening of 2,510 records, we extracted the data (converted into 2020 Indian rupees [INR]) and assessed the quality of eight eligible studies. These studies, published between 2011 and 2020, covered the whole of India and specific localities and were: cost-of-illness studies (n = 3); economic evaluations (n = 2); cross-sectional costing study (n = 1); simulated costing study (n = 1); and policy/clinical review (n = 1). Among the extracted estimates, the mean total costs of stroke care per patient for the period up to six months post admission ranged from ₹19,428.86 in a government hospital in Punjab to ₹118,040.15 in a private tertiary care centre in Ludhiana. The median total out-of-pocket (OOP) payments for cardiovascular disease-related expenditures ranged from ₹18,148.88 to ₹68,464.39 across different income groups. Despite methodological limitations, the data from our scoping review will help in designing economic evaluations of stroke care interventions in India.
Introduction
In India, approximately 1.44–1.64 million new cases of stroke are diagnosed every year (Marfatia et al., 2014), placing a burden on the health sector, economy and the wider society. It was projected that India’s GDP will lose $54 billion in 2015 due to stroke, coronary heart disease and diabetes (Taylor & Suresh Kumar, 2012), mainly through personal expenditures (Downey et al., 2018; Marfatia et al., 2014; Srivastava et al., 2020). The pressure on the health sector places an emphasis on the need for ensuring that high-quality and cost-effective stroke care is provided to optimise the use of limited resources (Srivastava et al., 2020), in addition to the necessity of reducing preventable deaths and disability. This is particularly important in India, where the burden of stroke affects relatively young people (mean age of onset is 63 years, with 20% people aged 40 years or less; Pandian & Sudhan, 2013). Given the incidence of stroke among the working population, identifying the aetiology of, and risk factors for, stroke is crucial (Dash et al., 2014). Post-stroke care is key too, as inadequate post-stroke care may reduce long-term chances of recovery, lead to families’ changed economic circumstances and impact the country’s economic growth (Huffman et al., 2011).
As part of our National Institute for Health and Care Research Global Health Research Group on IMPROVIng Stroke carE (IMPROVISE) in India collaboration, we are working towards building capacity among healthcare professionals in India to enhance the clinical and cost-effectiveness of stroke care. To determine whether the costs of the programme are worth the improvement in patient outcomes, we are developing an economic evaluation. For this economic evaluation, we need to collect direct and indirect data on the costs of hospital care. In a preliminary search, we identified systematic reviews on the economic burden of cardiovascular disease (CVD), hypertension or non-communicable diseases in low- and middle-income countries and South Asia (Brouwer et al., 2015; Gheorghe et al., 2018; Rijal et al., 2018). However, literature reviews on the extent and quality of the current evidence base on the costs of stroke in India were lacking.
The aim of this scoping review was to identify sources of cost data related to the delivery of stroke care in hospitals in India, summarise the current evidence and consider how the information might be used in future economic evaluations.
Data and Methodology
Our scoping review was designed to follow the methodological guidance on the conduct and reporting of scoping reviews (JBI, 2020; Tricco et al., 2018), with its methods outlined in an unpublished protocol.
Search Strategy
We identified studies through searches of nine electronic databases, including MEDLINE, Embase, Web of Science, CINAHL, ABI/INFORM, Academic Search Complete, Cochrane Library, Global Index Medicus, and Centre for Reviews and Dissemination (CRD) databases. Following initial scoping searches, an information specialist developed a search strategy adopting the Cochrane Stroke strategy (Cheyne, 2020), the Canadian Agency for Drugs and Technologies in Health (CADTH) search filter for economic evaluations (CADTH, 2016), and including terms for costs, stroke and India. Databases were searched from their inception to July 2020 (Table S1 includes the MEDLINE search strategy). Additional references were identified from previously held libraries, contact with experts, and citation screening from identified studies.
Study Selection
Studies were eligible for selection in the scoping review based on specific criteria described through the population, concept and context (PCC) framework (JBI, 2020) and on their study design. We adopted a healthcare sector and broader societal perspective, as we aimed to capture data on personal expenditure and lost income on the part of the patients and their caregivers.
Specifically, studies were eligible if.
their focus was on patients who had had a stroke and were admitted to a private or public secondary (e.g., hospitals) or tertiary (e.g., neurological centres) care setting in India (we did not place limits on the length of the follow-up period); the authors considered any stroke care intervention delivered in those settings; among the outcomes, different types of costs were presented, including direct medical costs (e.g., medications) and non-medical costs for stroke care (e.g., travel, food), and indirect costs (e.g., lost income for the patient); and the authors reported these outcomes as part of an economic evaluation, econometric, cost-of-illness or studies where costs of stroke care had been included.
Although abstracts containing cost data were included, we excluded editorials and commentaries. We did not search grey literature or online sources such as government reports but only published peer-reviewed sources. All studies had to be reported in English.
Studies were selected through a two-stage process (using criteria specified prior to screening) by one co-author (VB; Table S2). First, titles and abstracts were screened, with full manuscripts of those appearing to meet the selection criteria retrieved and assessed in the second stage. Any uncertainties around decisions were discussed and resolved with other co-authors (DC/AC). The outcome of the selection process is reported using the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) flowchart for scoping reviews (Tricco et al., 2018).
Data Extraction and Studies’ Quality
Data from the selected studies were extracted independently by two co-authors (VB/JS), with each co-author’s extraction being validated by the other co-author. Any disagreements were successfully resolved by the two co-authors. The co-authors extracted the data using a pre-piloted form adapted from an existing template (Gheorghe et al., 2018), including: publication details; high-level study detail; attributes of economic analysis; cost data characteristics; additional details (e.g., limitations, funding); and costs in study currency (aggregate economic estimates, direct costs and indirect costs). While lists of individual data items for each of the above areas had been initially set, these lists were continually updated as new relevant items were found while the co-authors extracted the data. The final, detailed data extraction list is included in Table S3.
We assessed the quality of studies using selected questions included in the Consensus on Health Economic Criteria (CHEC) list (Evers et al., 2005) and in a quality assessment tool used in a previous economic systematic review (Gheorghe et al., 2018). Questions were selected if they were related to the broad study design and to the way cost data were reported, in line with the focus of our scoping review. The full list of questions is presented in Table S4. Decisions on study quality were made independently by two co-authors (VB/JS), who resolved any differences through discussion and also ensured a shared understanding of the decisions with DC and AC.
Data Synthesis
The main characteristics of the included studies were discussed with all authors, summarised in tables and then narratively synthesised. We provide a detailed description of the study designs, geographical locations, types of healthcare setting, units of analysis, number of healthcare facilities/services investigated, sample sizes and the types of economic estimates reported.
All extracted costs data were converted into one common currency (Indian rupee, INR) for the reference year 2020 using the EPPI-CCEMG Cost Calculator (Shemilt et al., 2010). Point, dispersion and interval estimates extracted for each individual cost item (e.g., cost of medications) and aggregate cost (e.g., total healthcare costs over 12 months post stroke) were then grouped, where possible, or were presented separately in a table and were analysed descriptively. Study quality was discussed narratively in the context of the findings of the studies, with the results tabulated.
Results
Search Results
With our search strategy, we identified 2,990 papers which, after de-duplication, resulted in 2,510 papers for inspection. Following screening of titles and abstracts, 31 records were identified for retrieval for full-text screening. Although eight papers met the selection criteria, these included a systematic review (Gheorghe et al., 2018), with only one study relevant to our scoping review (Huffman et al., 2011). A decision was made to exclude the systematic review and include only the relevant study (Huffman et al., 2011), resulting in eight studies being included in the scoping review (Bahuguna et al., 2020; Basu et al., 2015; Chhabra et al., 2019; Gopi et al., 2017; Huffman et al., 2011; Kwatra et al., 2013; Marfatia et al., 2014; Singla & Singla, 2016). Details of the records excluded following full-text screening (with related reasons) are presented in Table S5. The full process of identification and inclusion is illustrated in an adapted version of the PRISMA flowchart for scoping reviews (Tricco et al., 2018; Figure S1).
Overall Summary of Included Studies
Each included study met the inclusion criteria, and a summary of their characteristics is provided in Table S6. The studies were characterised by various designs, including cost-of-illness studies (n = 3 [Chhabra et al., 2019; Kwatra et al., 2013; Marfatia et al., 2014]), economic evaluations (n = 2 [Basu et al., 2015; Gopi et al., 2017]), a cross-sectional costing study (n = 1 [Huffman et al., 2011]), a costing study based on econometric modelling (n = 1 [Bahuguna et al., 2020]) and a policy/clinical review (n =1 [Singla & Singla, 2016]).
The included studies were set in broad and specific geographical locations, ranging from the whole of India (n = 1 [Basu et al., 2015]) to specific states or regions—Delhi National Capital Region (n = 1 [Singla & Singla, 2016]) and Punjab, Haryana, Tamil Nadu, Odisha, Himachal Pradesh and Kerala (n = 1 [Bahuguna et al., 2020])—and cities, that is Guntur (n = 1 [Gopi et al., 2017]), Gurgaon, Mumbai and Bengaluru (n = 1 [Marfatia et al., 2014]), Trivandrum (n = 1 [Huffman et al., 2011]), Ludhiana (n = 1 [Kwatra et al., 2013]) and an unspecified location in Punjab (n = 1 [Chhabra et al., 2019]).
The range of healthcare settings was large in some studies—covering primary, secondary and tertiary care settings (n = 1 [Basu et al., 2015]) or primary and secondary care settings (n = 1 [Bahuguna et al., 2020])—and more specific in others—covering secondary care settings only (n = 1 [Huffman et al., 2011]), tertiary care settings only (n = 2 [Kwatra et al., 2013; Marfatia et al., 2014]), neurological departments (n = 2 [Chhabra et al., 2019; Gopi et al., 2017]) and a stroke care unit (n = 1 [Singla & Singla, 2016]).
In almost half of the studies, the individual patient or caregiver was the unit of analysis (n = 5 [Chhabra et al., 2019; Gopi et al., 2017; Kwatra et al., 2013; Marfatia et al., 2014; Singla & Singla, 2016]), while in the remaining studies the analyses were based on income groups (n = 1 [Huffman et al., 2011]), healthcare facility (n = 1 [Bahuguna et al., 2020]) and the general population (n = 1 [Basu et al., 2015]).
More variation emerged in relation to the number of healthcare facilities and sample sizes considered. The number of healthcare facilities ranged from studies set around a single facility (n = 3 [Chhabra et al., 2019; Gopi et al., 2017; Kwatra et al., 2013]) to a study considering 81 facilities, of which 15 were hospitals (Bahuguna et al., 2020). Sample sizes naturally differed according to the types of design (for instance, modelling or cross-sectional), units of analysis (for instance, individual patient or healthcare facility) and number of healthcare facilities chosen, making sensible comparisons difficult. For instance, the three studies set around a single facility had between 100 and 189 individuals in their samples (Chhabra et al., 2019; Gopi et al., 2017; Kwatra et al., 2013), while the study based on national-level modelling did not consider specific healthcare facilities and had a very large (simulated) sample (Basu et al., 2015).
Summary of Cost Estimates of Stroke for India
The economic estimates presented in the studies were variable. In general, a mix of estimates for overall or aggregate costs and individual items were reported. Overall or aggregate costs to treat a patient with stroke were presented in five studies which accounted for the costs incurred by both the healthcare provider and the patients or their families (Basu et al., 2015; Chhabra et al., 2019; Huffman et al., 2011; Kwatra et al., 2013; Marfatia et al., 2014). In some of these studies, estimates for disaggregated costs were also included, ranging from the higher level (i.e., direct medical, direct non-medical and indirect costs [Chhabra et al., 2019]) to the more granular level (e.g., individual inpatient, outpatient and indirect costs, such as medication, rehabilitation and lost income [Basu et al., 2015; Kwatra et al., 2013; Marfatia et al., 2014]). Differences emerged in respect of the time horizons adopted, spanning six months (Chhabra et al., 2019; Kwatra et al., 2013), 12 months (Marfatia et al., 2014) and 15 months (Huffman et al., 2011) post admission or event. In another three studies, the focus was more specific, including admission and staff costs (Bahuguna et al., 2020), specific medications (Gopi et al., 2017) and procedures (Singla & Singla, 2016). The remaining study was a modelling study where the authors simulated the effect of expanding government healthcare insurance in the long term, up to 2035 (Basu et al., 2015).
Tables 1 and S7–S19 include the cost estimates relevant to our scoping review as extracted from the eight included studies. Each table presents data for a separate category of cost regarding the delivery of stroke care, with estimates accompanied by information on the study, including notes and caveats. Where possible, estimates were grouped with reference to a common economic outcome (e.g., mean total costs per patient). Where this grouping was not possible, individual estimates were presented separately for each study.
Total Costs
Within the six months post admission, the mean of the total cost per patient ranged from ₹19,428.86 (n = 130), as measured in a neurology department of a tertiary care government hospital in an unspecified location in Punjab (Chhabra et al., 2019), to ₹118,040.15 (n = 189; 95% confidence interval (CI): ₹106,270.11 to ₹129,698.91) in a private tertiary care centre (stroke unit and neurology clinic) in Ludhiana (Kwatra et al., 2013) (Table 1).
Estimates of Total Costs Extracted from the Included Studies.
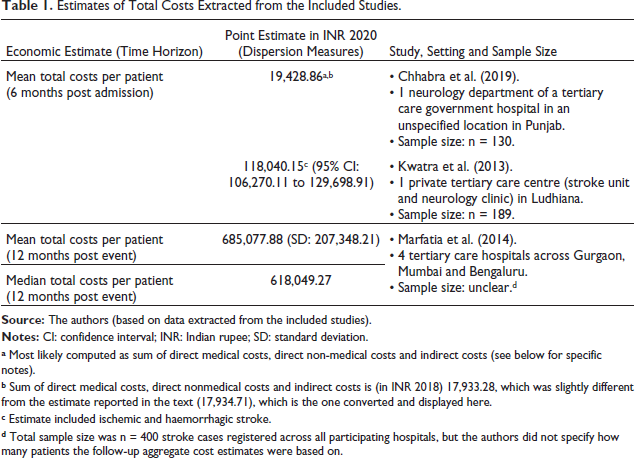
Estimates of Total Costs Extracted from the Included Studies.

a Most likely computed as sum of direct medical costs, direct non-medical costs and indirect costs (see below for specific notes).
b Sum of direct medical costs, direct nonmedical costs and indirect costs is (in INR 2018) 17,933.28, which was slightly different from the estimate reported in the text (17,934.71), which is the one converted and displayed here.
c Estimate included ischemic and haemorrhagic stroke.
d Total sample size was n = 400 stroke cases registered across all participating hospitals, but the authors did not specify how many patients the follow-up aggregate cost estimates were based on.
Within the 12 months post event, the mean total direct cost per patient in four tertiary care hospitals across Gurgaon, Mumbai and Bengaluru was ₹685,077.88 (standard deviation [SD]: ₹207,348.21; median: ₹618,049.27; Marfatia et al., 2014).
OOP Expenditures
The total OOP expenditures related to CVDs was estimated for different income groups as measured in six secondary care settings with outpatient follow-up in Trivandrum (Huffman et al., 2011) (Table S7). The estimated medians total OOP CVD-related expenditures were ₹18,148.88 (n = 66; interquartile range (IQR): ₹3,826.01 to ₹32,485.84) for the low-income group, ₹37,400.97 (n = 99; IQR: ₹8,966.47 to ₹65,816.70) for the middle-income group and ₹68,464.39 (n = 335; IQR: ₹24,786.89 to ₹138,534.29) for the high-income group.
These OOP expenditures may produce a decrease in individual median monthly income following hospitalisation, estimated as ₹873.17 (n = 66; IQR: ₹0.00 to ₹3,309.61) for the low-income group, ₹0.00 (n = 99; IQR: ₹0.00 to ₹2,840.16) for the middle-income group and ₹0.00 (n = 335; IQR: ₹0.00 to ₹610.28) for the high-income group.
The effects on household monthly income were also estimated, with median decreases of ₹992.88 (n = 66; IQR: ₹0.00 to ₹2,159.46) for the low-income group, ₹0.00 (n = 99: IQR: ₹0.00 to ₹2,840.16) for the middle-income group and ₹0.00 (n = 335; IQR: ₹0.00 to ₹704.17) for the high-income group.
Total Direct Costs
We calculated the mean total direct costs per patient within the six months post admission as ₹84,023.00 (n = 189), adding up the estimates reported for total direct medical (inpatient and outpatient) costs and for direct non-medical costs presented by Kwatra et al. (2013) (Table S8).
Total Direct Medical Costs
Total mean inpatient and outpatient direct medical costs per patient within the six months post admission were estimated in two studies as ₹11,169.23 (n = 130; Chhabra et al., 2019) and ₹76,956.29 (n = 189; 95% CI: ₹69,289.21 to ₹85,386.26; Kwatra et al., 2013) (Table S9).
Mean total outpatient direct medical costs per patient were estimated to be ₹272,236.02 (SD: ₹150,046.86) within the 12 months post event (Marfatia et al., 2014).
The cost of service for primary and secondary stroke care originated from a modelling study (Basu et al., 2015). In this study, the authors simulated the effect of expanding government healthcare insurance to incorporate risk factors for ischemic heart disease and cerebrovascular disease, likelihood of experiencing both myocardial infarction or stroke, and the probability of receiving treatment before and after such events. Costs, which included screening, monitoring and patient costs per patient per year, were presented as a 95% CI from ₹104.69 to ₹209.38 (this range represented the 95% CIs across the different age categories considered).
We integrated Table S9 with sub-tables, covering: admission and care costs (e.g., generic index hospitalisation and outpatient expenses) in Table S10; medication costs in Table S11; tests, investigations and procedures costs in Table S12; staff costs in Table S13; outpatient consultation costs in Table S14; rehabilitation costs in Table S15; and outpatient nursing care services costs in Table S16.
Total Direct Non-medical Costs
Mean total direct non-medical costs per patient within the six months post admission ranged from ₹2,924.40 (n = 130; Chhabra et al., 2019) to ₹7,066.71 (n = 189; 95% CI: ₹6,035.84 to ₹8,157.62; Kwatra et al., 2013) (Table S17).
We integrated Table S17 with a sub-table covering travel and home modification costs (Table S18).
Total Combined Lost Income for Both Patients and Carers/Family Members
Estimates of the mean total combined lost income for both patients and carers/family members within the six months post admission ranged from ₹5,335.22 (n = 130; Chhabra et al., 2019) to ₹34,015.69 (n = 189; 95% CI: ₹29,296.25 to ₹39,206.63; Kwatra et al., 2013) (Table S19).
We integrated Table S19 with a sub-table covering separate lost income for patients and for carers/family members (Table S20).
Quality Assessment of the Included Studies
The aggregate and disaggregated results of our quality assessment are shown respectively in Tables S21 and S22. In almost all the included studies the study population and research question were clearly defined, and the perspective for the economic analysis was appropriately chosen (n = 7; 87.50% for all these questions). On other factors, there was less certainty. Only in half or less of included studies, appropriate levels of identification (n = 4; 50.00%), explanation of sources (n = 4; 50.00%), measurement (n = 3; 37.50%), valuation (n = 4; 50.00%) and reporting (n = 2; 25.00%) of cost data were reached. The generalisability of the results was discussed in most studies (n = 6; 75.00%), but only in half of the studies productivity costs, uncertainty and/or heterogeneity were addressed as part of the analyses (n = 4; 50.00%). Lastly, in only five studies no potential conflicts of interests between the authors and the funders were reported (62.50%).
The availability of general cost information related to the Indian healthcare system (Bahuguna et al., 2020), and for the different facets of stroke care specifically (Agrawal et al., 2019; Chhabra et al., 2019), is a recognised problem. In this scoping review, we found that the number of costing studies to quantify the costs of stroke for India has been evolving since 2011 but remains limited. In the eight included studies we identified an array of cost data which could be useful to assist the design of future economic evaluations of stroke care interventions in India. Our findings complement evidence from systematic reviews on the economic burden of CVD, hypertension or non-communicable diseases in low- and middle-income countries and South Asia (Brouwer et al., 2015; Gheorghe et al., 2018; Rijal et al., 2018), by providing cost data specific to the Indian context for studies published up to July 2020. However, limitations need to be highlighted on the included studies and extracted data. As such, we identified three aspects which deserve attention by future research: comparability, transparency on data sources and generalisability.
Comparability Across Studies
Despite the wide range of estimates provided by the included studies, only a few presented estimates on a seemingly comparable cost measure. A key difference across the studies which complicates comparisons is the extent of economic estimates being reported.
In Table S23 we displayed which cost estimates were extracted from the included studies, as measured against the range of types of estimates we were expecting to capture in our data extraction template. The authors of some studies reported data solely on individual stroke care cost items such as admission and salary of staff (Bahuguna et al., 2020), medication (Basu et al., 2015; Gopi et al., 2017), tests, procedures and investigations (Singla & Singla, 2016). On the other hand, in other studies, aggregate estimates related to the overall direct and indirect costs were provided, either in conjunction with individual cost items (Kwatra et al., 2013; Marfatia et al., 2014) or alone (Chhabra et al., 2019). The study by Huffman et al. (2011) represents an exception to this categorisation, since its focus was onto OOP expenditures, reporting various estimates on the financial burden of stroke to the patients and their families.
Across those studies presenting similar types of estimates, which is the case for the three cost-of-illness studies reporting total, direct medical, direct non-medical and indirect costs (Chhabra et al., 2019; Kwatra et al., 2013; Marfatia et al., 2014), a comparison of the magnitude of estimates and extent of the cost data reported could be attempted. But even in this case, the different locations (i.e., different cities or states), settings (i.e., private vs government hospitals) and time horizons (i.e., 6 months vs 12 months post admission or event) make comparisons difficult.
Another complication is given by authors identifying categories of costs with the same name but in reality including different cost items, such as different types of investigations (for direct medical costs), expenditures (for direct non-medical costs such as home modifications, food or travel) and people for which lost income was estimated (for indirect costs related to the patients, family members and carers). This highlights a lack of standardisation in the definition and identification of the costs belonging to a specific category. While the presentation of aggregate estimates remains good practice, future costing studies on stroke care set in India should endeavour to report estimates for disaggregated cost items too (as is the case for Kwatra et al. (2013) and Marfatia et al. (2014)), so as to let the reader recognise which specific cost data were identified and used in the analyses and (more importantly) which were not.
This lack of standardisation affects the presentation of the statistical measures too, since in some studies only the point estimates or the dispersion measures were reported. As a result, the interpretation of half-reported results will cause uncertainty, and more complete reporting is thus advocated in future studies.
Lastly, given the variability of the cost estimates across the studies, it should be noted that smaller costs are not necessarily led by a more efficient use of resources. Variations in costs may arise from differences in terms of the treatments provided to the patients (e.g., thrombolysis or thrombectomy) and may reflect diversions from an ideal treatment pathway. The lack of effective treatments may lead to early deaths which may reduce length of stay in hospitals and thus short-term healthcare costs. However, at the same time, the lack of effective treatments may contribute to poor long-term recovery and dependency, which in turn would increase long-term costs for the healthcare system, as well as for the patients and their families.
Transparency of Data Sources
The details provided on the data sources were reported differently across the studies, as highlighted by our quality assessment. In those studies where the authors relied on secondary data, they generally provided sufficient details on where the cost data came from, particularly the two modelling-based studies (Bahuguna et al., 2020; Basu et al., 2015). On the other hand, there was less uniformity across the studies using primary data. While in all these studies the general methods of data collection were indicated (typically hospital bills [Chhabra et al., 2019; Kwatra et al., 2013], medical chart records [Huffman et al., 2011; Marfatia et al., 2014], interviews [Chhabra et al., 2019; Kwatra et al., 2013], questionnaires [Huffman et al., 2011; Marfatia et al., 2014] and data collection forms [Gopi et al., 2017; Marfatia et al., 2014]), only two studies (Kwatra et al., 2013; Marfatia et al., 2014) also detailed how the data for each cost item was specifically sourced.
The availability of information on the data sources used naturally impacts the quality of the data presented in the studies. Given the shortcomings in some of the included studies, authors of future studies should aim to report their data sources transparently and comprehensively. The authors of four of the included studies (Bahuguna et al., 2020; Basu et al., 2015; Kwatra et al., 2013; Marfatia et al., 2014) provided more details on their data sources compared with the other studies included in our scoping review, making their data more reliable for secondary use and, at the same time, representing examples of good practice for future studies.
Generalisability
There seems to be varying degrees of generalisability across the cost estimates presented by the studies. Even for studies with bigger samples and considering more diverse settings, concerns emerged. One of these concerns is the quality of the data used and reported. This was an issue in the national-level simulation study by Basu et al. (2015), where a battery of cost estimates with relevance to the delivery of stroke care were presented, but we found problems with the way data from other literature sources were used, which limited the amount of data that we could extract from this study.
Another concern is around the specificity, as in the study by Bahuguna et al. (2020), where estimates with a broad connection to the delivery of healthcare, but without a specific connection to stroke care, were presented.
The sample composition could limit generalisability too. This is the case of the studies by Huffman et al. (2011), where the sample (n = 500) appeared to be dominated by participants belonging to one income group (i.e., high-income group, n = 335), and the studies by Kwatra et al. (2013) and Chhabra et al. (2019) which were based on a single setting. The study of Marfatia et al. (2014) was perhaps the one with the most generalisable results among the studies specifically focusing on stroke care, as it was based on four tertiary care hospitals across Gurgaon, Mumbai and Bengaluru with 400 patients in the analysis (albeit different sub-samples were then considered for different cost items). The strength of future studies could be increased by considering diverse geographical locations and settings (for instance, a mix of rural and urban, privately and government-run care centres) able to account for reasonably sized and representative sample sizes. Also, the Indian national health insurance scheme Ayushman Bharat may increase the accuracy of cost estimates in the future.
It is important that future research on the costs of stroke in India should address these limitations to ensure the data will be more reliable and usable in economic evaluations of stroke care interventions in India. These efforts would need to align with standardised approaches for data collection in economic evaluations of stroke care, as set out by international guidance (Cadilhac et al., 2020).
Strengths of our review include the systematic search and approach recommended for scoping reviews (JBI, 2020; Tricco et al., 2018). Nevertheless, we recognise that our scoping review process may potentially have missed relevant records including publications not written in English. Also, new studies on this field continue to emerge (Goswami et al., 2020; Rajasulochana & Kar, 2021) and should then be incorporated in future updated reviews.
Conclusions
With our scoping review, we aimed to compile and evaluate the availability of cost data in relation to stroke care provided in India. We identified only a few relevant studies which nevertheless contained a broad range of aggregate and disaggregated cost estimates. Importantly, these estimates cover not only direct but also indirect costs, since the impact of the latter can be burdensome, with OOP expenditures covering over 80% of health spending in India (Downey et al., 2018).
The estimates found in the included studies could assist in populating economic models on stroke care interventions in India. However, more costing studies are needed to increase the quantity and quality of cost data available. The estimates extracted from the included studies come with caveats regarding how comparable and generalisable they were, and which data sources they were based on. Our analysis and discussion suggest that future costing studies may contribute to provide more robust estimates as long as they are based on standardised definitions of direct (medical and non-medical) and indirect costs, as well as using reasonably sized samples involving participants from diverse settings characterised by different financial governances (private vs public care settings) and located in different sites (urban vs rural, or in multiple Indian states).
Supplemental Material
Supplemental material for this article is available online.
Supplemental Material for Costs of Hospital Care for Strokes in India: A Scoping Review by Valerio Benedetto, Dominique A. Cadilhac, Joseph Spencer, Andrew Clegg, Catherine Elizabeth Lightbody, Pallab K. Maulik, Madakasira Vasantha Padma Srivastava, Jeyaraj Pandian, P. N. Sylaja, Caroline L. Watkins, in The Indian Economic Journal
Footnotes
Acknowledgements
We gratefully thank Catherine Harris (Information Specialist at the University of Central Lancashire) for her help in defining the search strategy and running the multi-databases searches for our scoping review.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article:
This research was funded by the NIHR (16/137/16) using UK aid from the UK government to support global health research. Valerio Benedetto, Andrew Clegg and Caroline L. Watkins were funded by the National Institute for Health and Care Research (NIHR) Applied Research Collaboration North West Coast (ARC NWC); Caroline L. Watkins was funded by the National Institute for Health and Care Research, UK, and Arthritis Research, UK; Jeyaraj Pandian was funded by the Indian Council of Medical Research, India, National Institute for Health and Care Research, UK, NHMRC, Australia, Population Health Research Institute, Canada, and Department of Biotechnology, India; Catherine Elizabeth Lightbody was funded by the National Institute for Health and Care Research, UK; Pallab K. Maulik was funded by NIHR grant (GHR Group [16/137/16])—Developing a Global Health Research Group for Stroke, NHMRC grant (APP1143911)—Systematic Medical Appraisal, Referral and Treatment for Common Mental Disorders in India—SMART Mental Health, and MRC grant (MR/S023224/1)—Adolescents’ Resilience and Treatment Needs for Mental Health in Indian Slums (ARTEMIS); P. N. Sylaja was funded by the Indian Council of Medical Research.
The views expressed in this publication are those of the author(s) and not necessarily those of the NHS, NIHR, Department of Health and Social Care or the UK government.
These funding bodies had no role in study design, data collection, data analysis, data interpretation or writing of the review.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
