Abstract
Objective
This study proposes an AI-enhanced modular material selection approach for designing diabetic insoles. By customizing materials for forefoot and heel modules, the method enables personalized support and pressure redistribution, resulting in cost-effective insoles.
Background
Diabetic foot ulcers occur as a result of elevated plantar pressure and poor foot sensation. To mitigate this risk, developing insoles that redistribute plantar pressure can significantly lower the likelihood of ulcer formation.
Method
The insole was fabricated using cost-effective silicone molding, with functional personalization achieved via an AI-enhanced approach that selected optimal, interchangeable cushioning materials for forefoot and heel modules of each patient. The design integrates a ¾-length porous silicone upper layer with regionally optimized materials. Laboratory wear trials involving 23 diabetic patients compared the offloading performance of the hybrid insole (AIO) against barefoot, a PORON® Medical 4708 insole (PUR), and a cork-EVA insole (EVA).
Results
AIO demonstrated a 37.6% reduction in peak plantar pressure compared to barefoot condition while increasing contact area by 36.0% across the plantar surface. It significantly outperformed the commercial insole (EVA) and matched a therapeutic insole (PUR) overall, with superior regional offloading at the heel.
Conclusion
This study enhances diabetic foot care by combining efficiency of silicone molding with functionally personalized, modular design to achieve superior pressure redistribution. It establishes a paradigm of “modular personalization” for diabetic insoles, leveraging AI for patient-specific material selection within a standardized, biomechanically optimized geometry.
Application
The findings offer practical insights for clinicians and manufacturers seeking scalable, patient-specific solutions for preventing diabetic foot ulcers.
Keywords
Introduction
The growing prevalence of Diabetes Mellitus (DM) has become a critical global health challenge, with an estimated 589 million adults living with diabetes worldwide. Epidemiological models predict this number will rise dramatically to 853 million cases by 2050 (IDF, 2025). Amongst the most serious complications are diabetic foot ulcers (DFUs), which significantly increase the risk of lower extremity amputation if not managed promptly (Atosona & Larbie, 2019). The primary factor for developing DFUs is peripheral neuropathy, which leads to foot deformities (like claw toes and collapsed arch), loss of protective sensation (Uçkay et al., 2015), and impaired skin integrity (Gariani et al., 2014). Furthermore, poor circulation exacerbates delayed wound healing (Yachmaneni et al., 2023), underscoring the need for proactive foot health monitoring.
Diabetic foot care is crucial due to the high prevalence of DFUs and their devastating clinical consequences and socioeconomic burden (Kumbhar & Bhatia, 2024). Epidemiological studies indicate that 19–34% of individuals with diabetes will develop a DFU in their lifetime, of which 20% of cases progressing to amputation (Armstrong et al., 2017; McDermott et al., 2023). Beyond surgical risks, DFUs are associated with elevated mortality and substantially reduce patients’ quality of life, impairing physical mobility and mental health (Chen et al., 2023; Ghadeer et al., 2025). The condition also imposes a heavy burden on healthcare systems through costs related to prolonged hospitalizations and wound care (Rice et al., 2014). These multifaceted challenges underscore the urgent need for effective preventive solutions, such as improved therapeutic footwear.
Abnormal plantar pressure is a critical intermediary in ulcer pathogenesis. Neuropathy-induced structural deformities, combined with diminished protective sensation, leave patients vulnerable to repetitive trauma in high-pressure regions, particularly the metatarsal heads (MTHs) and heel. Research confirms 200 kPa as a crucial pressure threshold significantly elevating ulceration risk in those who have previously ulcerated (Bus et al., 2013). Detailed gait analyses reveal distinct regional plantar pressure patterns: the forefoot experiences peak pressures during propulsion, while the heel absorbs maximum impact at initial contact (Hessert et al., 2005; Hillstrom et al., 2013). Furthermore, the diabetic foot undergoes characteristic deformation and rotation during weight bearings in standing and walking (Zhang et al., 2023). Therefore, while an ideal intervention would modify both geometry and material properties, practical and scalable solutions must strategically prioritize the most critical factor. Given the direct link between high pressure and ulceration, intelligent pressure redistribution via material optimization becomes a paramount design objective for effective, accessible diabetic footwear/insole.
Current management of DFUs relies on advanced wound therapies (e.g., bioengineered skin substitutes and growth factor applications) and rigorous offloading strategies (e.g., total contact casts, therapeutic footwear, and custom insoles) (Boulton et al., 2008; Driver et al., 2010; Lavery et al., 2003). However, these approaches face critical limitations, with surgical interventions and advanced wound care exhibiting recurrence rates exceeding 40% within 1 year alongside high treatment burdens (Armstrong et al., 2017). While therapeutic footwear is a cornerstone of prevention, its efficacy is often compromised by poor patient adherence due to impractical designs (Zhu et al., 2024). Conventional insoles frequently employ uniform materials, failing to address the foot’s complex, region-specific biomechanical demands (Arts et al., 2015). While total-contact designs and zonal material properties show superior offloading (Owings et al., 2008), the high cost and infrastructure requirements of fully personalized manufacturing methods like 3D printing limit their accessibility (Chang & Choo, 2025).
This highlights a clear gap for a modular solution that balances customization (through patient-specific material selection for key regions), efficacy (in pressure redistribution), and scalability (through cost-effective fabrication). A modular design allows for functional personalization via interchangeable components, enhancing producibility and patient-specific fit. Silicone molding presents a viable alternative, offering a middle ground between customization and economic feasibility (Wortmann & Frese, 2022). To address this gap, this study presents an AI-enhanced modular material selection method to create cost-effective diabetic insoles with customized materials, enabling patient-specific pressure offloading and targeted support through interchangeable modules within a standardized, biomechanically optimized design.
Materials and Methods
Design and Fabrication of the Modular Insole
The hybrid diabetic insole (AIO) was developed by using a structured, two-stage design process that separates geometric standardization from material personalization. The term “hybrid” denotes the integration of a silicone-molded upper layer with interchangeable modular cushioning inserts. Stage 1 established a standardized, biomechanically informed geometry suitable for scaling (Zhang et al., 2023). Stage 2 achieved personalization through an AI-enhanced modular material selection system. Using previously validated plantar pressure prediction models (Zhang et al., 2024), optimal cushioning materials for the forefoot and heel regions were individually selected for each patient from a material library. This resulted in unique, patient-specific material combinations within the standardized template, enabling functional personalization focused on pressure redistribution.
Stage 1: Insole Geometric Standardization
The base geometry of the AIO insole was standardized, with biomechanical features (including a teardrop-shaped metatarsal pad, arch support, and heel cup) proportionally scaled across sizes (Figure 1). This geometry was derived from our prior dynamic foot shape analysis using a 4D scanning system (Zhang et al., 2023), which identified that this specific configuration provided optimal load distribution and accommodation of dynamic foot deformation during gait for diabetic patients. The geometry was proportionally scaled across European shoe sizes 38–41 to accommodate participant foot dimensions. No individual geometric customization was performed; instead, this standardized geometric template served as a platform for modular material selection strategy. Geometry of the ¾-length porous silicone upper layer
Stage 2: AI-Enhanced Material Selection and Fabrication
Material properties of the components used in the hybrid diabetic insole
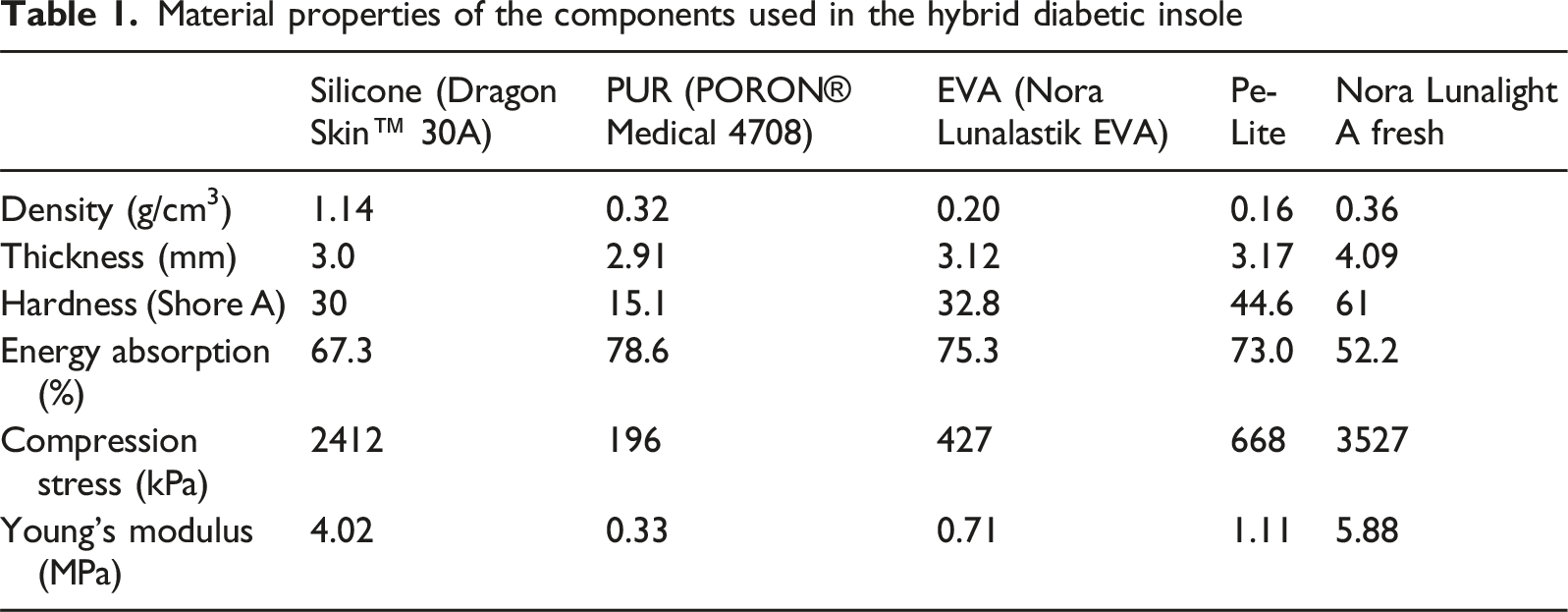
The final AIO design (Figure 2) integrated three distinct functional zones: (1) a porous silicone upper layer chosen for its flexibility, durability, and shock absorption (Mazurek et al., 2019); (2) the AI-enhanced cushioning material selection in the forefoot and heel modules; and (3) a semi-rigid EVA base layer for structural support. Silicone molding was utilized to fabricate the upper layer, leveraging its cost-effectiveness and ability to replicate complex geometries like the honeycomb structure (Kuo et al., 2020). Design schematic of the hybrid diabetic insole (AIO). The image depicts the standardized, biomechanically optimized geometry derived from dynamic foot scanning analysis (Zhang et al., 2023). The forefoot and heel regions represent modular cushioning zones; materials for these zones were individually selected for each participant via an AI-enhanced system (Zhang et al., 2024), enabling functional personalization within a standardized template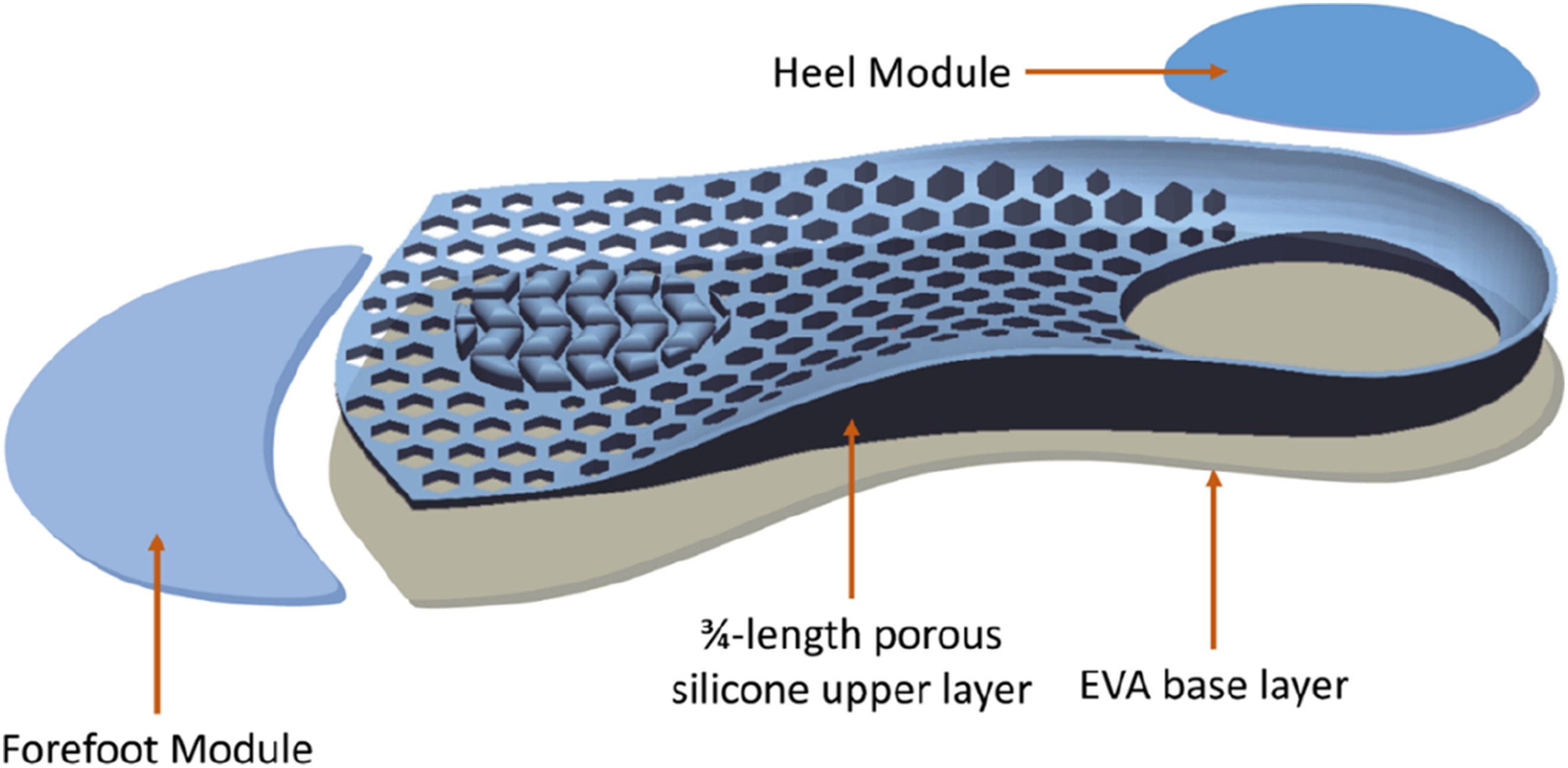
The fabrication process began with the creation of negative silicone mold (Mold Star™ 30) from a 3D-printed PLA master. The final upper layer was then cast using Dragon Skin™ 30A silicone. Following fabrication, the silicone upper layer was assembled with the AI-selected cushioning materials for forefoot/heel and an EVA base using a high-strength, flexible polyurethane adhesive (3M™ Transfer Tape) to ensure durability during gait. The key material properties for all components are summarized in Table 1.
Insole Evaluation
Participants
Participant characteristics (n = 23)
Experimental protocols and insole samples
Participants were required to walk through a distance of 8 m on a concrete surface at their natural pace for all the trials, with two timing gates (Brower Timing Systems, Utah, USA; precision: 0.01 s) positioned at 1 m and 7 m to determine the duration of each trial. Initial barefoot walking trials established baseline speeds, with 10 trials conducted to obtain the natural walking speed of each participant (6 m distance divided by time). Trials with walking speeds deviating by more than 5% from the established baseline were excluded; this accounted for fewer than 5% of all trials and was considered random (Burnfield et al., 2004). Recorded walking speeds across study participants fall between 0.58 m/s and 0.89 m/s (Brach et al., 2008).
Plantar pressure data were recorded using the Pedar® in-shoe system (Novel GmbH, Munich, Germany) under 4 conditions: barefoot and 3 insoles (Figure 3): Insole conditions: AIO (Newly designed hybrid insole), PUR (Double-layer PORON® Medical 4708), and EVA (cork-EVA insole). 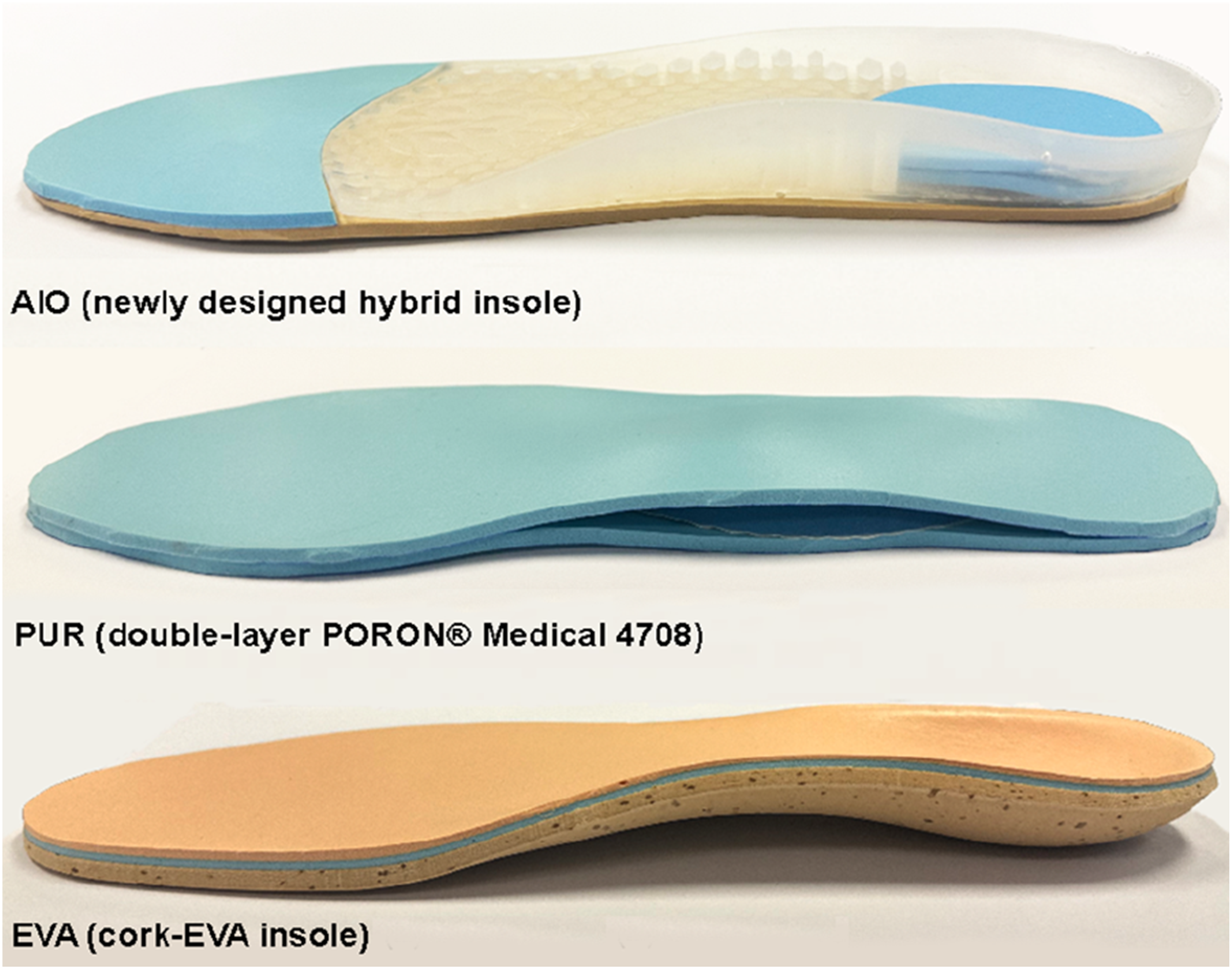
The system captured data at 50 Hz, with calibration before each test. For barefoot measurements (Figure 4), sensors were secured via cotton socks, while insole tests used standardized sports shoes (Kinghealth, Hong Kong). Each condition was tested three times in a randomized order, with 3-minute breaks between trials. After testing, participants rated the insole’s overall comfort on a 10-point scale (1: Very uncomfortable, 10: Very comfortable). This research complied with the tenets of the Declaration of Helsinki and was approved by the Institutional Review Board at The Hong Kong Polytechnic University (Ref: HSEARS20200128001). All participants provided written informed consent. Experimental setup for barefoot plantar pressure measurement (Pedar® sensor insole secured within a cotton sock to ensure consistent sensor positioning)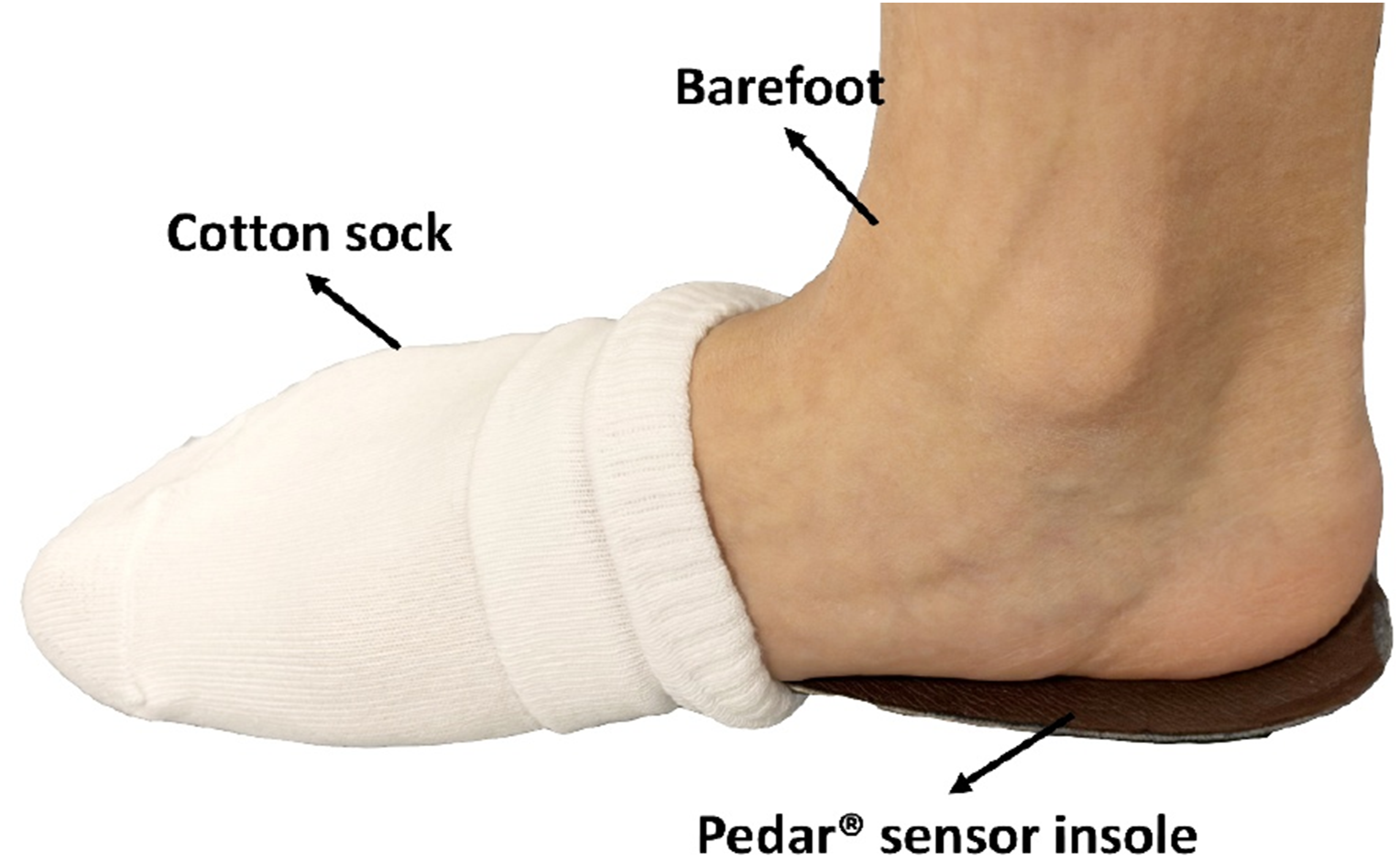
Data Analysis
Peak plantar pressure (PPP) and contact area (CA) of the different experimental conditions were statistically analyzed for the whole plantar surface and 5 regions (Figure 5) (Putti et al., 2008). The offloading performance and comfort ratings of AIO were compared to PUR and EVA. Data from 3 trials per condition (dominant foot, stance phase of 5 consecutive steps) were averaged to calculate PPP for each participant in each condition. PPP was defined as the average of the maximum pressure values recorded by each active sensor across the 15 total stance phases (5 steps × 3 trials). Statistical analysis used IBM SPSS 21. The Shapiro–Wilk test confirmed normal distribution. A one-way repeated-measures ANOVA identified significant differences (p < 0.05) among conditions. When a significant main effect was found, post-hoc pairwise comparisons were conducted for all condition pairs (Barefoot vs. AIO, Barefoot vs. PUR, Barefoot vs. EVA, AIO vs. PUR, AIO vs. EVA, PUR vs. EVA) using the Bonferroni correction to adjust for multiple comparisons and control Type I error. Effect sizes are reported as partial eta-squared (η2) for ANOVA results. Five regions of foot plantar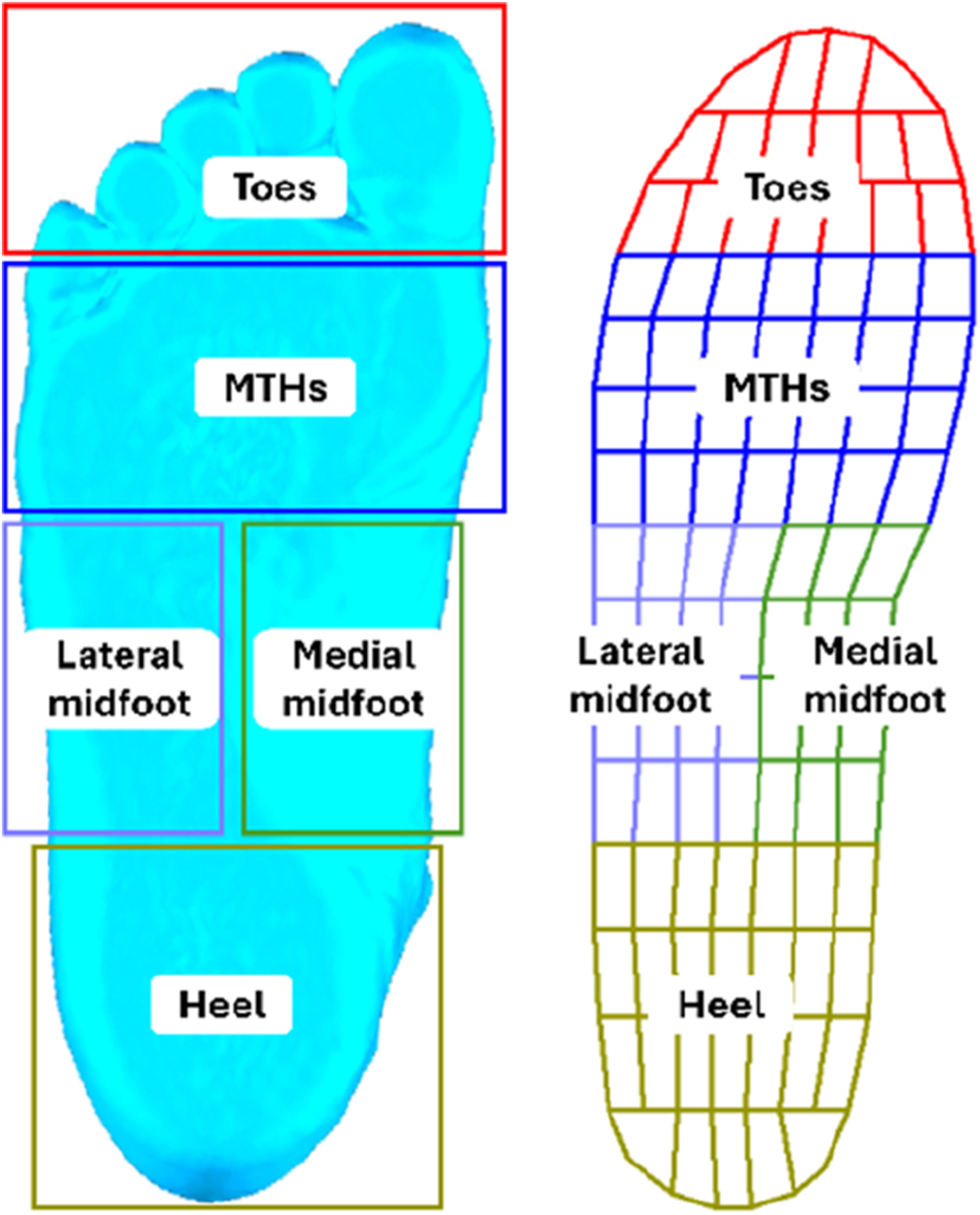
Results
AI Material Selection Outcomes
Prior to evaluating offloading performance, we characterized the material configurations generated by the AI selection system. Among the 23 participants, 7 distinct forefoot-heel material combinations were produced from the four-material library. The most frequently selected configuration was PORON® Medical 4708 for the forefoot paired with Nora Lunalastik EVA for the heel (N = 8, 34.8%), followed by the inverse arrangement (EVA forefoot + PORON heel; N = 6, 26.1%). Notably, 17 participants (73.9%) received heterogeneous insoles with different materials assigned to the forefoot versus heel regions, while only 6 participants (26.1%) received homogeneous configurations. This heterogeneity confirms that the AI system tailors material properties to individual pressure profiles rather than applying a uniform solution across the cohort.
Overall Offloading Performance of the Insoles
Mean peak plantar pressure (kPa) and percentage change relative to barefoot

n.s. = not significant (p ≥ 0.05). All p-values are from post-hoc pairwise comparisons with Bonferroni correction.
Mean contact area (cm2) and percentage change relative to barefoot

n.s. = not significant (p ≥ 0.05). All p-values are from post-hoc pairwise comparisons with Bonferroni correction.
Regional Offloading Performance of the Insoles
Regional peak plantar pressure (kPa) and percentage change relative to barefoot
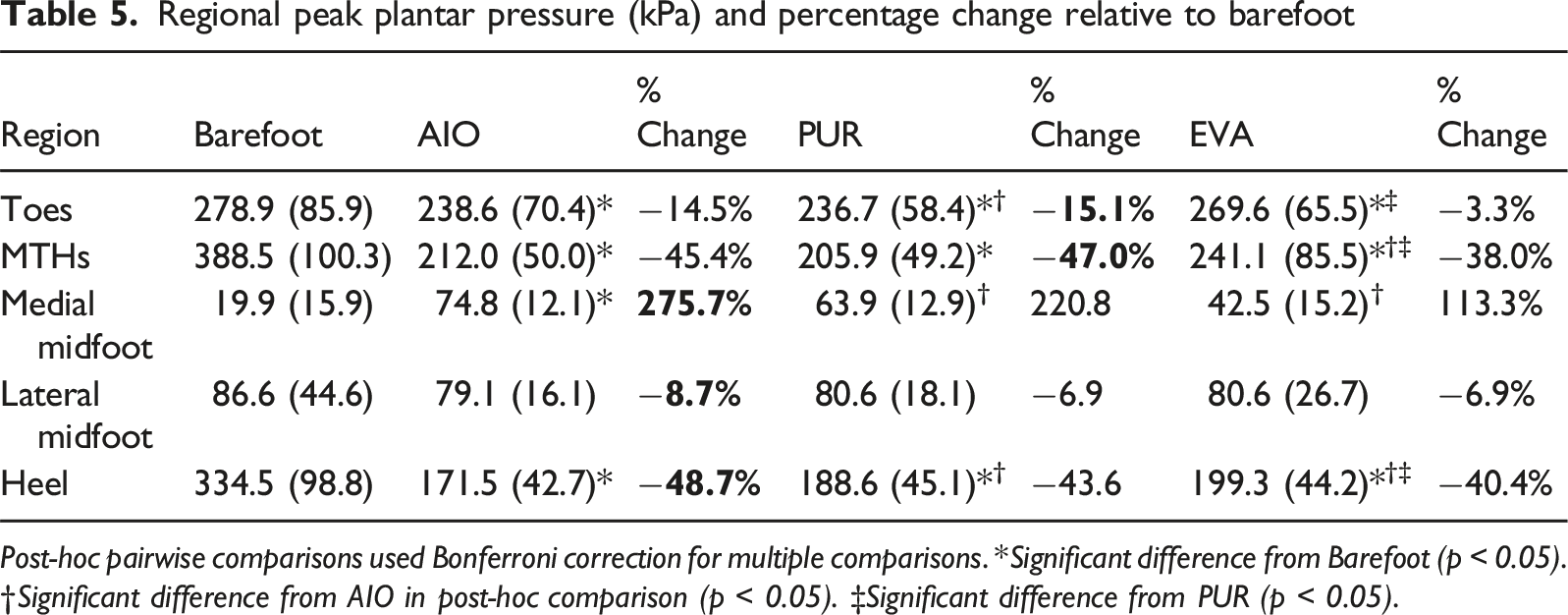
Post-hoc pairwise comparisons used Bonferroni correction for multiple comparisons. *Significant difference from Barefoot (p < 0.05). †Significant difference from AIO in post-hoc comparison (p < 0.05). ‡Significant difference from PUR (p < 0.05).
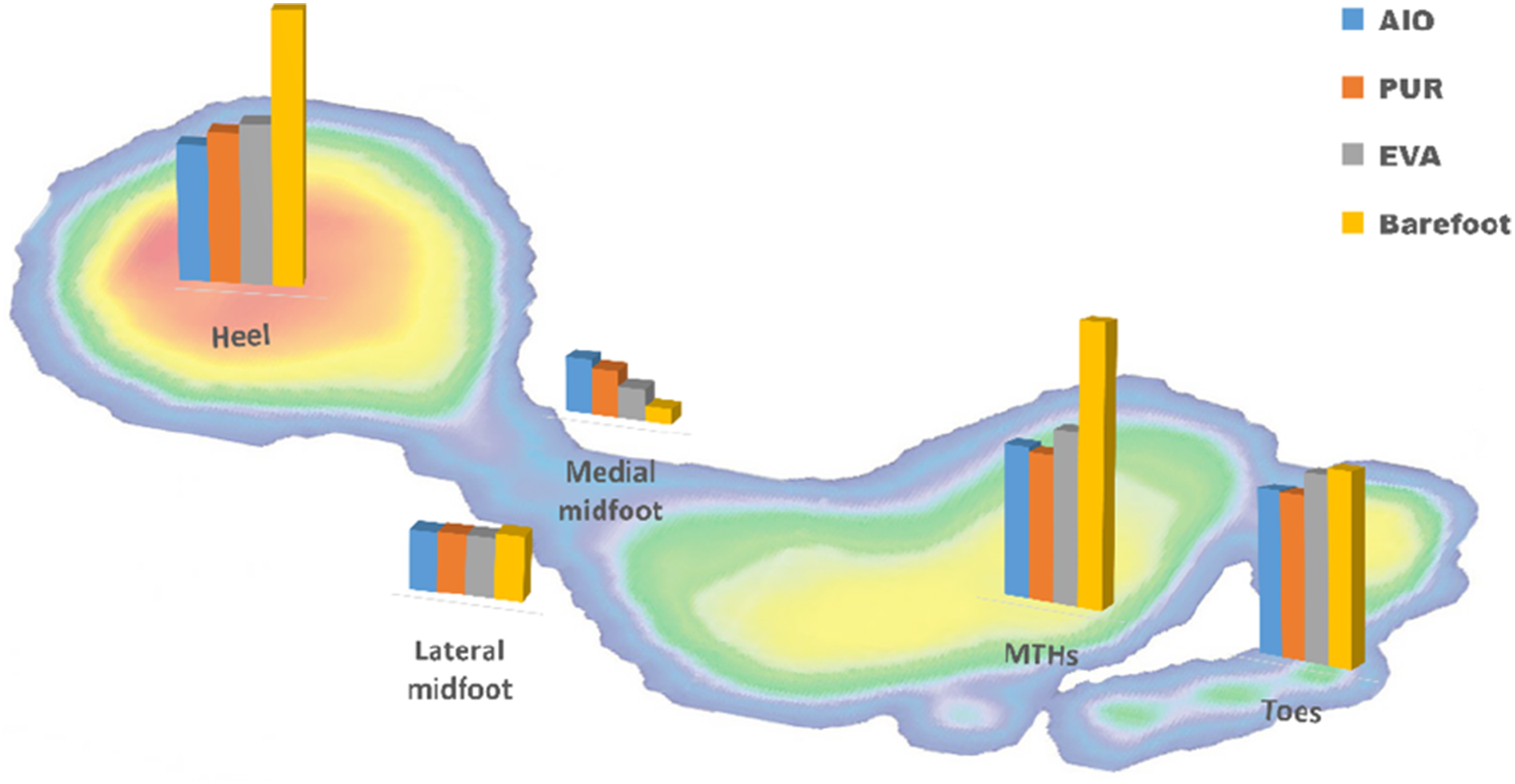
Regional peak plantar pressure at different foot/insole conditions
The post-hoc comparisons indicated that while both AIO and PUR provided superior offloading at the MTHs compared to EVA, only AIO achieved statistically significant greater pressure reduction at the heel compared to PUR and EVA (p < 0.05). This highlights the targeted efficacy of the AI-enhanced material selection in optimizing heel cushioning specifically, a critical region for ulcer prevention during heel strike.
Regional contact area (cm2) and percentage change relative to barefoot
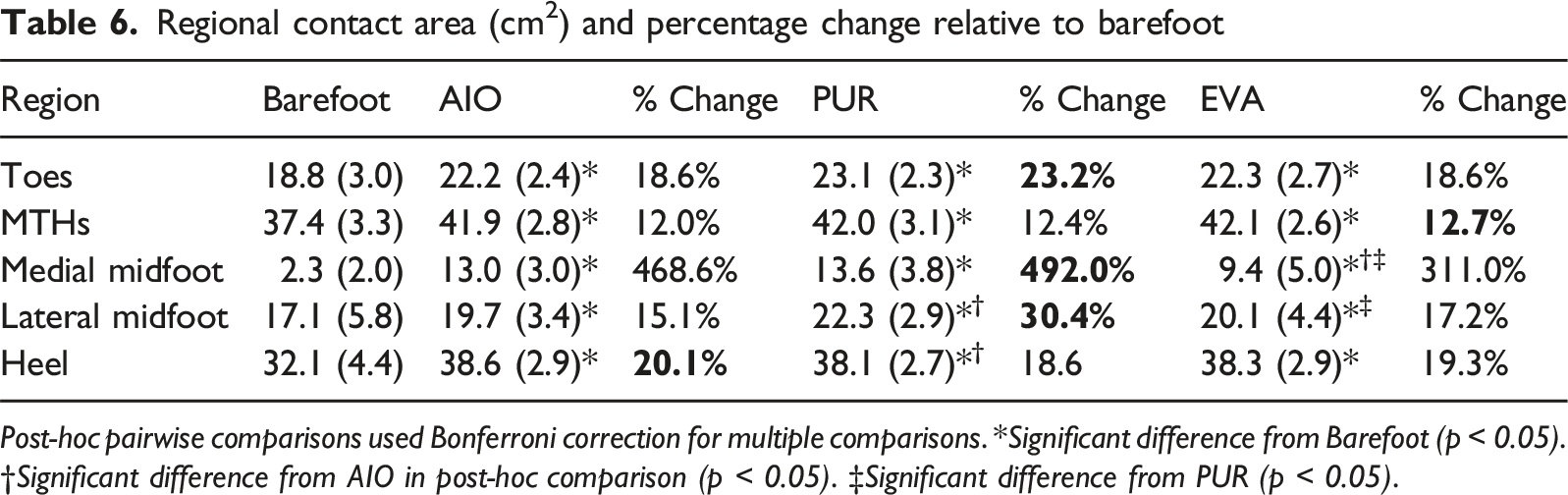
Post-hoc pairwise comparisons used Bonferroni correction for multiple comparisons. *Significant difference from Barefoot (p < 0.05). †Significant difference from AIO in post-hoc comparison (p < 0.05). ‡Significant difference from PUR (p < 0.05).
Subjective Comfort Evaluation of the Insoles
Subjective comfort ratings for the three insole conditions
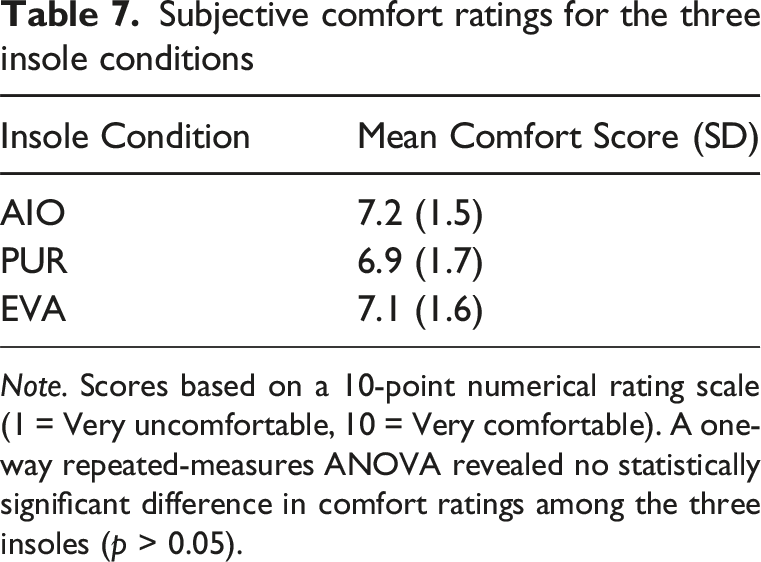
Note. Scores based on a 10-point numerical rating scale (1 = Very uncomfortable, 10 = Very comfortable). A one-way repeated-measures ANOVA revealed no statistically significant difference in comfort ratings among the three insoles (p > 0.05).
Discussion
The newly developed hybrid diabetic insole (AIO) demonstrated significant improvements in pressure redistribution compared to conventional designs while maintaining excellent wearability. This design reduced peak plantar pressure by 37.6% and increased contact area by 36.0% relative to barefoot walking. Most notably, it achieved targeted offloading of high-risk ulcer-prone zones, with pressure reductions of 45.4% under the metatarsal heads and 48.7% at the heel. These biomechanical improvements were accomplished with a mean comfort score of 7.2/10, suggesting strong potential for patient adherence—a critical factor for prevention efficacy (Armstrong et al., 2017; Zhu et al., 2024).
The regional pressure analysis confirms the insole’s biomechanical rationale. The substantial pressure increases in the medial midfoot was an intentional design outcome, demonstrating successful load transfer from vulnerable areas (metatarsal heads and heel) to more structurally robust regions. This aligns with findings that combined shape and pressure optimization outperforms uniform material designs (Owings et al., 2008). The insole’s geometric features—arch support, metatarsal pad, and heel cup—work synergistically to accommodate dynamic foot deformations during gait (Zhang et al., 2023), which is crucial for neuropathic patients, who lack protective sensation and rely on their footwear to prevent excessive tissue loading.
It is noteworthy that all three insole conditions demonstrated significant improvements in plantar pressure redistribution and contact area compared to barefoot condition. This finding is consistent with the established principle that any structured orthotic intervention can improve load distribution relative to unshod conditions in neuropathic feet (Bus et al., 2013). However, the critical distinction lies in the differential performance between insoles: while PUR and AIO performed similarly at the metatarsal heads, only AIO achieved significantly superior offloading at the heel. This highlights the value of region-specific, AI-informed material selection for optimizing protection at multiple high-risk sites simultaneously.
Modern diabetic foot care seeks greater personalization, currently explored through two divergent pathways. On one end, fully customized 3D-printed insoles offer high geometric and material tailoring but are limited by high costs, infrastructure demands, and production time, hindering accessibility (Chang & Choo, 2025; Cheng et al., 2021; Telfer et al., 2017). Recent work has advanced the understanding of load transfer mechanisms in custom accommodative insoles, demonstrating that 3D-printed lattice structures can achieve superior offloading compared to standard-of-care devices (Heino et al., 2025; Muir et al., 2022, 2025). Similarly, Hudak et al. (Hudak et al., 2022) introduced a workflow for patient-specific 3D-printed orthoses with personalized metamaterials. On the other, active offloading systems with sensors and actuators represent a frontier in dynamic pressure management (Castro-Martins et al., 2025; Erel et al., 2024), yet their complexity, cost, and power requirements pose significant barriers to widespread, everyday use. Our modular approach complements these innovations by offering a scalable alternative: rather than requiring full geometric customization via additive manufacturing, we achieve functional personalization through AI-guided material selection within a standardized, silicone-molded template. This may enhance accessibility in resource-limited settings while maintaining biomechanical efficacy. Between these extremes lies a clear gap for a passive, yet intelligently personalized, solution that balances efficacy, accessibility, and scalability.
This work introduces a novel design approach paradigm to fill that gap: a modular, AI-enhanced insole fabricated via cost-effective silicone molding. Unlike fully customized 3D-printed insoles (high cost, high customization) or fully standardized pre-fabricated insoles (low cost, low customization), our insole uses a standardized, biomechanically optimized design. Personalization is achieved not through geometric alteration, but through AI-enhanced selection of interchangeable cushioning modules for the forefoot and heel, based on individual plantar pressure predictions (Zhang et al., 2024). This core innovation—modular material personalization within a standardized template—enables functional personalization: the biomechanical properties of key load-bearing zones are tailored to the patient, while the manufacturing process remains streamlined and scalable.
Silicone molding offers potential cost advantages over fully customized digital manufacturing approaches. Based on our pilot production estimates, the material cost for our hybrid insole is approximately $15–25 USD per unit. By comparison, published reports suggest that single-material 3D-printed insoles may range from $50–150 USD in material and processing costs, while multi-material, patient-specific 3D-printed orthoses can exceed $200 USD depending on design complexity and production scale (Chang & Choo, 2025). Furthermore, mold creation for silicone molding (∼$100–200 USD one-time) represents a lower initial capital investment than acquiring industrial-grade multi-material 3D printing equipment, which typically requires investments exceeding $2,000 USD for entry-level professional systems. While our modular approach requires assembly labor, the standardized geometry and pre-characterized material library enable efficient batch production. This positions our solution as a potentially scalable, economically viable pathway to functional personalization for preventive diabetic foot care in resource-constrained settings.
The modular nature of the design is fundamental to its scalability and practical value. Forefoot and heel components, pre-fabricated from a library of characterized materials, can be efficiently selected and assembled based on AI recommendations. This process avoids the high equipment costs and material expenses of patient-specific 3D printing, positioning our solution as a more economically viable alternative. Consequently, the AI-material Optimized insole occupies a critical middle ground: it moves beyond the “one-size-fits-all” limitation of fully standardized insoles by offering patient-specific pressure offloading, yet avoids the prohibitive expense and complexity of fully customized digital or active systems. This provides a scalable compromise for delivering biomechanically informed preventive care in diverse clinical settings.
Practical Applications for Designers and Clinicians
Our findings offer several actionable insights for therapeutic footwear development. • Modular personalization is feasible and effective. Designers can achieve meaningful functional customization without full geometric personalization by focusing material optimization on high-risk regions (forefoot, heel) within a standardized, biomechanically informed template. • AI-enhanced selection can be implemented transparently. The two-stage workflow—AI prediction followed by interpretable selection rules—provides a practical framework for integrating machine learning into clinical decision support without creating “black box” recommendations. • Cost-effective manufacturing enables scalability. Silicone molding of standardized components, combined with modular assembly of pre-characterized materials, offers a pathway to personalized care that is accessible beyond specialized research centers. • “One size does not fit all” applies to materials, not just geometry. Even with identical geometry, varying material properties across patients yields measurable biomechanical differences. Designers should consider material libraries and selection algorithms as core components of personalized insole systems. • Regional analysis is essential. Global metrics (whole-foot PPP) can mask important regional effects. Design validation should include region-specific outcomes aligned with clinical risk profiles (e.g., heel pressure for ulcer prevention).
Limitations and Future Work
This study has several limitations. The relatively small sample size may affect the generalizability of the findings. The evaluation was conducted under controlled walking conditions, which may not fully replicate real-world scenarios involving varied terrains or prolonged use. Future studies should incorporate longitudinal real-world trials across varied scenarios (e.g., uneven surfaces and stairs) and longer durations. Furthermore, while PPP and CA are key metrics, future assessments should include long-term durability, real-world patient adherence, and direct impact on tissue health. Additionally, long-term material testing was not conducted in this study; therefore, the longevity and wear resistance of the silicone and modular components over extended periods remain to be validated in future work.
Conclusion
This study advances diabetic insole technology through a biomechanically driven hybrid design integrating three critical innovations: (1) anatomical specificity via a standardized, biomechanically optimized geometry from foot morphology analysis across dynamic stances, enabling optimized arch contouring, teardrop-shaped metatarsal head support, and heel cup geometry; (2) functional personalization through a modular design with AI-enhanced, region-specific material selection; and (3) manufacturing efficiency using cost-effective silicone molding. Laboratory trials demonstrate that the newly designed hybrid diabetic insole significantly outperformed the commercial insole (EVA) and matched the performance of a therapeutic insole (PUR) overall, while achieving satisfactory wearability. This approach thereby establishes a new framework for therapeutic footwear that addresses both biophysical requirements and healthcare accessibility challenges.
Key Points
• This study proposed a novel “modular personalization” paradigm for diabetic insoles, combining a standardized, biomechanically optimized geometry with interchangeable, AI-enhanced modular material selection for regional cushioning. • Personalization was achieved through an AI-enhanced system that selected optimal cushioning materials for modular forefoot and heel components for each individual. • The AI-material Optimized insole significantly reduced peak plantar pressure by 37.6% and increased contact area by 36.0% compared to barefoot, with targeted offloading of 45.4% at the metatarsal heads. • Laboratory wear trials confirmed its offloading performance was comparable to or better than a single-material insole and superior to a standard commercial insole while maintaining high subjective comfort (∼7.2/10). • The modular approach offers a scalable compromise between fully customized and fully standardized insoles, balancing functional personalization with manufacturing practicality for preventive diabetic foot care.
Footnotes
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research is funded by the InnoHK initiative of the Innovation and Technology Commission of the Hong Kong Special Administrative Region Government (Project: RP3.2 AiDLab), Hong Kong.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author [YKL], upon reasonable request.
