Abstract
As individuals age, age-related biologic and physiologic changes, alterations to pharmacokinetic and pharmacodynamic variables, and multimorbidity place geriatric patients at high risk for drug-related problems. Drug therapy problems in these patients include polypharmacy, inappropriate medication use, and poor medication adherence often due to complex regimens and medication costs. Pharmacists, physicians, and advanced practice providers (APPs) are uniquely positioned to tackle drug therapy problems and must work in tandem to do so. For maximal success, routine drug assessment within the multidisciplinary team should occur across the care continuum from the intensive care unit to the general floor to the outpatient setting. Pharmacists must identify drug therapy problems through medication reconciliations performed at admission, discharge, and during transitions of care, assist with adherence strategies, and address barriers to medication access. Physicians and APPs must routinely review medication lists, deprescribe and taper pharmacotherapy when able, identify medications to avoid and their appropriate alternatives, and prevent the prescribing cascade. To do this, a variety of tools can be deployed such as the Fit for the Aged criteria, Medication Appropriateness Index, American Geriatric Society Beers Criteria, and Screening Tool of Older Persons’ Prescriptions and Screening Tool to Alert to Right Treatment criteria, to aid in medication selection in older adults. This review aims to describe key geriatric physiological changes, highlight the role of the inpatient pharmacist, physician, and APP and discuss methods for assessment and optimization of drug therapy regimens in geriatric patients.
Introduction
The development of acute and chronic medical problems is a normal part of aging. Geriatric patients often have multiple comorbidities, and with each new diagnosis another prescription medication is commonly added to treat or control it. The National Council on Aging reports that nearly 95% of older adults have at least one chronic condition and nearly 80% have two or more. 1
In addition to multiple comorbidities, geriatric patients often develop “geriatric syndrome” defined as clinical conditions that do not fit into discrete disease categories including delirium, instability (leading to falls), frailty, dizziness, urinary incontinence, and dementia. 2 As a result of these diagnoses and conditions, polypharmacy – defined as the use of five or more medications – is common, and it is reported that over one-third of men and women between ages 75 and 85 meet this definition. 3 Furthermore, older adults account for nearly 50% of intensive care unit (ICU) admissions and 60% of all ICU days.4,5
Due to the complexity of these patients, age-related physiological, functional, cognitive, and social changes that impact the safety, efficacy, and need for medications can easily be overlooked. Consequently, these patients are disproportionately affected by medication-related issues, creating a significant opportunity to optimize care. This review will describe the roles inpatient pharmacists and providers play in managing drug-related problems geriatric patients.
Physiological Considerations in Geriatric Patients
Age-related changes to biological systems occur within the central nervous (CNS), cardiovascular, pulmonary, renal, immune, and musculoskeletal systems. 6 These variations are described in detail in Figure 1. Clinicians should carefully consider these changes, as they increase the vulnerability of geriatric patients to adverse drug events (ADE).
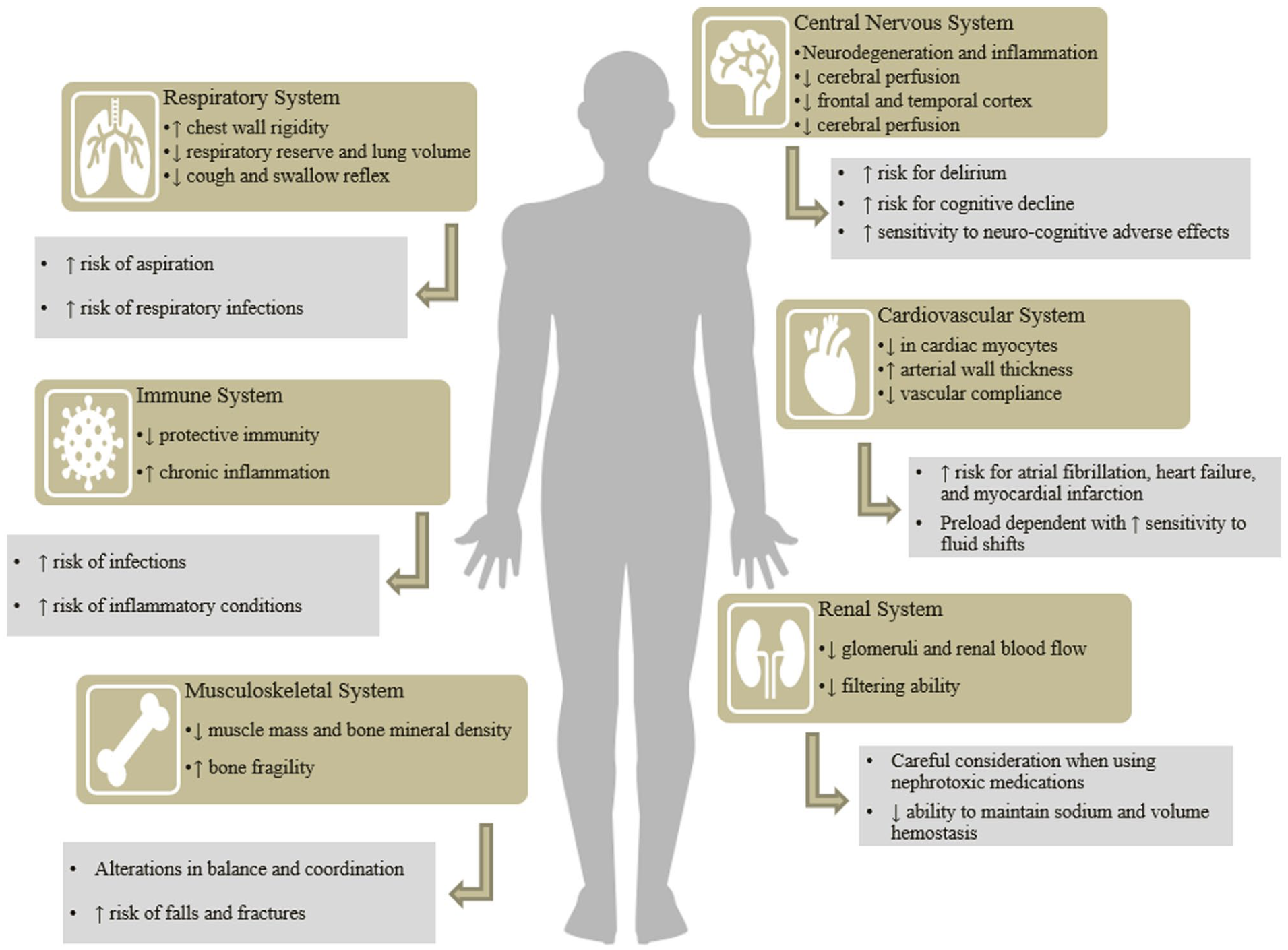
System-specific biologic changes and considerations for geriatric patients. Age-related changes to the central nervous, cardiovascular, respiratory, renal, immune, and musculoskeletal system are highlighted. In combination, these alterations affect medication-therapy management in elderly patients and should be considered by clinicians to guide therapy decisions.
In addition to biologic changes, geriatric patients also experience physiological changes which directly affect pharmacokinetic variables including absorption, distribution, metabolism, and excretion (Table 1). Absorption, the movement of a drug from its site of administration into systemic circulation most commonly occurring within the small intestine can either be increased or decreased in geriatric patients depending on drug-specific properties. 7 Distribution refers to the extent to which a drug disperses throughout the body relative to its plasma concentration. Drug distribution is influenced by factors such as protein size, lipophilicity, acidity, water solubility, charge, and molecular size.7,8 With aging, total body water decreases leading to smaller volumes of distribution and higher serum concentrations for polar, water soluble drugs. In contrast, nonpolar, lipid-soluble drugs have larger volumes of distribution in older adults, resulting in slower drug clearance. 7 Additionally, the blood-brain barrier becomes more permeable with age, increasing drug penetration into the CNS and heightening the risk of CNS adverse effects. 7
Pharmacokinetic Changes Associated with Aging and Possible Drug-Related Problems.
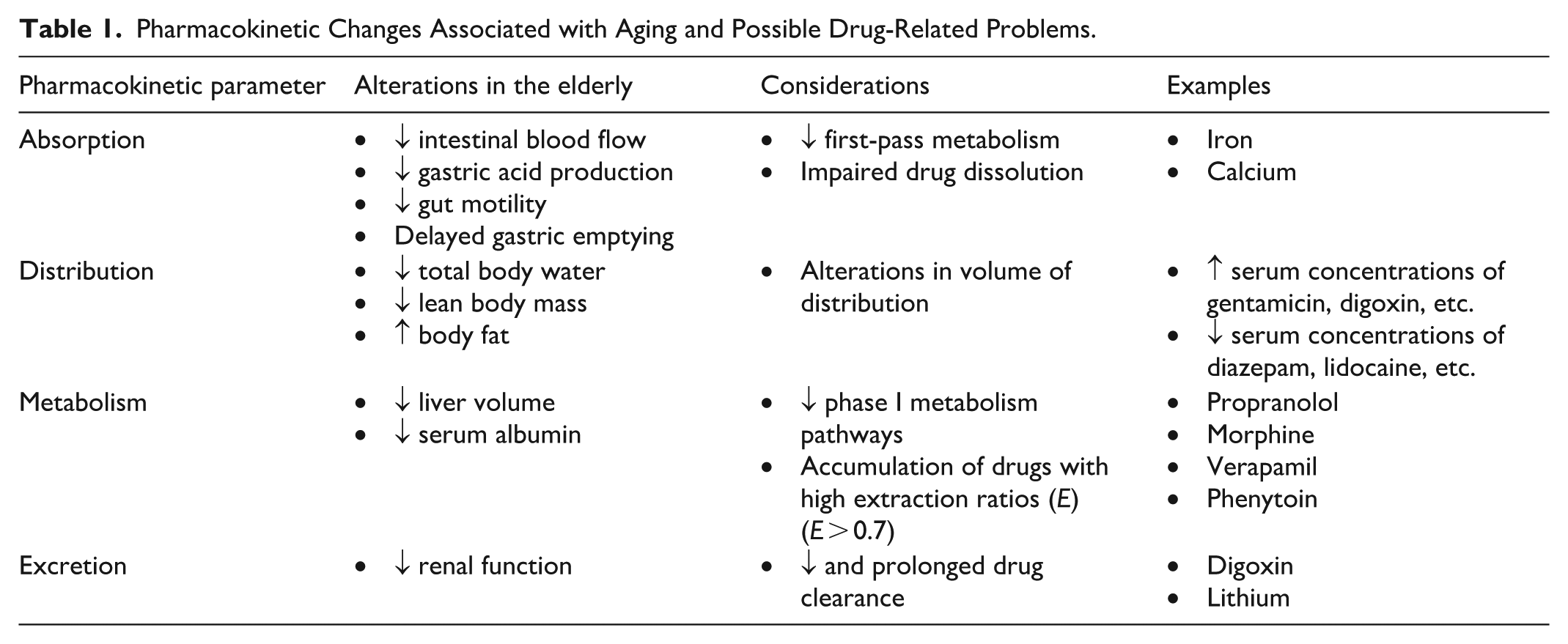
Metabolism refers to the breakdown of drugs through phase I enzymatic reactions (eg, oxidation, reduction, hydrolysis) or phase II conjugation reactions (eg, glucuronidation, sulfation acetylation). 9 Excretion, the elimination of metabolic waste from the body via the urine, feces, bile, or lungs, is also significantly affected by aging. Patients over 65 years of age typically have age-related decreased renal function, resulting in slower drug clearance and prolonged medication duration of action. Understanding these complex physiological changes is essential for the safe and effective management of medications in geriatric patients.
Role of the Inpatient Pharmacist
Inpatient pharmacists play an instrumental role in managing drug-related problems in geriatric patients, as they are uniquely trained to identify such problems, assess medication adherence, and navigate barriers to pharmaceutical care. As pharmacists are increasingly incorporated into interprofessional teams, access to pharmacy services continues to expand and the impact on patient care has become increasingly evident. In the ICU for example, critical care pharmacists have been shown to decrease medication costs, reduce medication errors, and prevent adverse events ultimately improving the quality and efficiency of care delivered.10,11
Medication cost is a critical barrier to adherence in all patients, but this is heightened in geriatric patients often due to fixed or limited income, rising out-of-pocket costs, and increasing pill burden. In a 2022 survey conducted by Vanderbilt University Medical Center, approximately 1 in 5 older adults reported cost-related medication non-adherence including delaying or not filling a prescription medication, using less or skipping doses of a medication to prolong supply, or using someone else’s medication. 12 Pharmacists are perfectly positioned to navigate cost barriers through enrollment in local and national patient assistance programs, optimizing medication therapy for cost-effectiveness (eg, combination regimens, dose adjustments, etc.), and by identifying medication therapies that may no longer be beneficial.
It is estimated that more than 400 000 preventable ADEs due to medication errors occur annually among hospitalized patients. 13 Common medication errors include omission (28.2%), wrong frequency (21.5%), wrong dose (20.0%), unnecessary medication (15.6%), and wrong route (14.7%). 14 Medication histories and reconciliations performed by pharmacists have consistently demonstrated lower rate of errors compared with those completed by other healthcare providers.15 -18 For example, in a quasi-experimental study conducted at an urban U.S. Veterans Affairs hospital in which both pharmacists and physicians conducted admission medication reconciliations, pharmacists documented statistically significantly more medication changes indicating a more comprehensive review. 16 Similarly, an interventional prospective study conducted in Italy involving 90 acutely admitted patients aged 75 and older found 455 drug-related problems among 911 prescriptions, most commonly actually inappropriate medications (AIMs) and clinically harmful drug-drug interactions. 19
Transitions of care, defined as the movement of a patient from one unit or healthcare setting to another, are also associated with a significantly increased risk of ADEs. 20 In a two-phase quality improvement project evaluating pharmacist-led discharge medication reconciliation for patients transferring to a skilled nursing facility, the intervention resulted in a 29.8% relative reduction in 30-day readmission rates (14.5% vs 20.6%). 21 Additionally, errors involving high-risk medications such as antibiotics, anticoagulants, and insulin accounted for 19.2% of pharmacist interventions. 22 Therefore, medication reconciliation should be performed by a pharmacist for all geriatric patients at admission, discharge, and during transitions of care, such as transfers from the ICU to the general medical floor.
Critical care pharmacist involvement in an ICU recovery clinic provides a unique opportunity for outpatient medication management following an ICU admission. In a prospective, single center, observational cohort study, a critical care pharmacist conducted a full medication review including medication reconciliation, interview, counseling, and interventions. All patients in the cohort had at least one pharmacy intervention and the median number of pharmacy interventions (eg, discontinuation, identification of ADE, initiation, etc.) per patient was four (IQR 2, 5). 23
Strategies to improve adherence and reduce medication errors may include alternate medication formulations such as extended- or sustained-release products, pill boxes, pill packs/bubble packs, and phone applications that provide reminders or alarms. Ensuring medication adherence is essential, as it has been estimated that 10% of hospitalizations and 23% of nursing home admissions are linked to medication non-adherence which ultimately increases healthcare costs and affects quality of life.24,25 Furthermore, the pharmacist must determine who is managing medications (ie, patient, caregiver, home health nursing, community pharmacy, etc.) to ensure conversations include the whole care team. To determine the most appropriate strategy for an individual patient, the pharmacist should use a structured communication framework such as the VALUE pneumonic: Value comments made by family, Acknowledge family emotions, Listen, Understand the patient as a person, Elicit family questions. 26 While not studied for pharmacist utilization, this approach has been shown to be beneficial for clinician conversations about death and dying in the ICU. 27 As such, pharmacists may utilize this pneumonic to foster patient and caregiver engagement and allows them to tailor interventions based on patient-specific factors, such as frequent medication changes or dexterity issues, to ensure optimal adherence and safety.
While utilizing the VALUE pneumonic can be a helpful way to initiate a conversation and tailor interventions, pharmacists must also consider patient baseline functional and cognitive function. The US Preventive Services Task Force recommends clinicians utilize validated tools such as the Mini-Mental State Examination (MMSE) and the Montreal Cognitive Assessment (MoCA) to assess global cognitive function. 28 Pharmacists should have a baseline understanding of these assessments and their results as they offer key insight into memory, attention, calculation and orientation which directly relate to safe medication management.
While pharmacists play a key role in medication management, they are also essential in educating providers regarding polypharmacy and deprescribing initiatives. By offering interprofessional education, clinical insight, and guidance on evidence-based deprescribing, pharmacists can work in tandem with providers to optimize medication regimens and enhance patient safety.
Role of the Physician and Advanced Practice Providers
Physicians and advanced practice providers (APPs) must be cognizant of the risks associated with drug therapy in geriatric patients and carefully weigh these risks against the anticipated benefits. Providers are uniquely positioned to identify, assess, and intervene on drug-related issues. To do so effectively, they must be empowered to make necessary medication changes, avoid the prescribing cascade, taper high-risk medications, and deprescribe when appropriate.
Avoidance of the prescribing cascade is essential in the management of geriatric patients. The prescribing cascade occurs when an initial medication causes an adverse effect that is misinterpreted as a new medical condition, leading to the prescription of an additional medication. A letter published by Brown University in 1995 stated, “Any symptom in an elderly patient should be considered a medication related problem until proven otherwise.” 29 Many adverse effects such as tiredness, constipation, diarrhea, incontinence, loss of appetite, and falls are frequently attributed to the normal aging process, causing medications to be overlooked as the underlying cause.30,31 Therefore, clinicians must shift their focus toward considering medications first and assessing for adverse effects before making a new diagnosis.
Assessing Drug Therapy in Geriatric Patients
Routine review of drug therapy should be conducted in all geriatric patients as they are particularly vulnerable to polypharmacy and often benefit from deprescribing. Deprescribing is an intervention aimed at identifying and discontinuing drugs for which the potential harms outweigh the current or anticipated benefit. It may also be appropriate when adverse effects are intolerable or negatively impacting quality of life.
Deprescribing is typically a collaborative process involving healthcare providers and the patient or family members, with shared goals of improving medication adherence, minimizing adverse effects, and enhancing quality of life. Figure 2 outlines key questions that clinicians should consider when tapering or discontinuing medications to ensure the process is safe and effective.

Questions clinicians should consider prior to tapering or deprescribing drug-therapy. By asking these questions, clinicians can work to determine if drug-therapy is necessary and determine the appropriate next steps for patient care.
While deprescribing is often considered an outpatient process, clinicians should be empowered to discontinue drug therapy throughout a hospitalization – especially in key transition periods such as prior to transfer from the ICU to the general medical floor. A multicenter, retrospective study of 958 patients transferred from the ICU to a non-ICU location in the Netherlands and United States found that 28.4% of patients had a medication continued, despite it having an ICU-only indication. 32 This finding reinforces the need for continual drug therapy review, particularly in this high-risk patient population.
Medications are considered a potentially inappropriate medication (PIM) when they meet any of the following criteria: the risks of treatment outweigh the benefits; they are prescribed for longer than clinically indicated or without a clear indication; or they are likely to interact with other medications or comorbid conditions. If the risk of harm associated with a PIM outweighs any potential clinical benefit, it is considered an actually inappropriate medication (AIM).33,34
The continuation of PIMs and AIMs, as defined by the 2003 Beers Criteria, was evaluated in 120 older adult ICU survivors. Medication use was identified throughout the hospital stay at five key timestamps during hospitalization: admission, floor admission, ICU admission, ICU discharge, and hospital discharge. At discharge, PIM categories with the highest positive predictive value for AIMs included anticholinergics (55%), non-benzodiazepine hypnotics (67%), benzodiazepines (67%), atypical antipsychotics (71%), and muscle relaxants (100%). Nearly two-thirds of AIMs were initiated in the ICU and inappropriately continued at discharge. 33
Several tools have been developed to determine whether a medication is appropriate in for individuals 65 years and older (Table 2). The Fit for the Aged (FORTA) provides a disease-related evaluation that identifies both over- and under-treatment. It categorizes medications using the following criteria: (A) benefit of efficacy/safety ratio proven in geriatric patients for a specific indication; (B) drugs with proven efficacy in the elderly, but limited efficacy or safety concerns; (C) drugs with questionable efficacy/safety profiles in geriatric patients which should be avoided or omitted in the presence of side effects; (D) avoid in geriatric patients. 35
Tools Used to Assess Medication Appropriateness in Elderly Adults.

The Medication Appropriate Index (MAI) is comprised of ten questions designed to help clinicians make informed decisions about deprescribing. These questions consider not only the medication in question but also patient-specific factors. The MAI can be applied to a single medication or an entire medication regimen. Each question is scored on a three-point Likert scale, and the total score reflects the overall appropriateness, with 0 indicating appropriate use and 18 indicating maximal inappropriateness. 36 The American Geriatric Society (AGS) Beers Criteria has guided clinicians in making safer prescribing decisions for older adults for more than three decades. It consists of a list of medications to potentially avoid or cautiously consider in geriatric patients to minimize the risks associated with drug therapy. 37 The Beers Criteria was first published in 1991 and most recently updated in 2023.
The Screening Tool of Older Persons’ Prescriptions and Screening Tool to Alert to Right Treatment (STOPP/START) criteria are two complementary resources designed to assist clinicians in evaluating the appropriateness of existing prescriptions (STOPP) and identifying beneficial new therapies (START) for geriatric patients. 38 Compared with the Beers Criteria, the STOPP/START tools not only recommend medications to avoid in older adults but also specify medications that should be initiated when clinically indicated. STOPP/START Version 1, published in 2008, has been cited 944 times as of April 2023. Version 2, released in 2015, expanded the criteria to include 114 evidence-based recommendations and had accumulated 1360 citations as of April 2023. Version 3, published in 2023, includes 133 mediations that should be discontinued in older adults. Notable additions to the “STOPP” recommendations include sodium glucose co-transporter (SGLT2) inhibitors in the setting of symptomatic hypotension, aspirin for primary prevention in cardiovascular disease, and levothyroxine for subclinical hypothyroidism. The updated START criteria contain 57 medications or medication classes appropriate for initiation, with key additions such as SARS-CoV-2 vaccines and osmotic laxatives for chronic persistent idiopathic or secondary benign constipation. 39
The Eliminating Medication through Patient Ownership of End Results (EMPOWER) trial, published in 2014, introduced a self-directed educational pamphlet on benzodiazepines that included tapering instructions for older adults. Distribution of this brochure resulted in a 27% reduction in benzodiazepine use, compared with only a 5% reduction in the control group. 40 A 2017 post hoc analysis of 261 community dwelling older adults demonstrated benzodiazepine discontinuation rates of 32% among participants with mild cognitive impairment and in 38% among those with normal cognition. 41 In 2019, the EMPOWER brochure was given to 50 geriatric inpatients who were chronic sedative users; following the intervention, sedatives were deprescribed in 64% of participants. 42
The Assess, Review, Minimize, Optimize, and Reassess (ARMOR) tool offers a structured, stepwise approach to evaluating medications in geriatric patients. The primary goals of ARMOR are to maintain and restore functional status while optimizing quality of life as a key consideration for modifying or discontinuing medications. 43 ARMOR also provides added value by assessing drug interactions, functional status, and the need for dose adjustment based on renal and hepatic function. 44
The Medication Regimen Complexity Index (MRCI) is a 65-item tool that measures the complexity of the medication regimen. The score is based on the number of medications, dosage from, dosing frequency, and additional or special instructions (eg, whether tablets must be crushed or split, timing of administration, and drug-food interactions). The tool was validated using 134 medication regimens from patients with moderate to severe chronic obstructive pulmonary disease. 45
The Good Palliative-Geriatric Practice (GP-GP) algorithm, developed by Dr. Doron Garfinkel, was designed to support decision making regarding medication discontinuation in older adults. A 2010 feasibility study demonstrated that 60% of medications taken by 64 geriatric patients were discontinued using the GP-GP approach. Of the 256 discontinued medications that were discontinued, only six were later restarted due to symptom reoccurrence. 46 A subsequent study evaluating a full-day educational workshop on the GP-GP tool showed a moderate reduction in polypharmacy among both inpatient and outpatient participants following the training. 47 The GP-GP algorithm is user-friendly and patient-specific; however, it does not address drug interactions, contraindications, therapeutic duplication, duration of therapy, allergies, or cost considerations. 44
While several tools exist to guide clinicians in deprescribing, it is important to recognize that some medications may also be inappropriately discontinued or unintentionally omitted during hospital admissions. A large population-based cohort study examined prescriptions for statins, antiplatelet and anticoagulant agents, levothyroxine, respiratory inhalers, and gastric acid-suppressing drugs using hospital and outpatient records from 396 380 Canadian patients. Patients were categorized into three groups: hospitalized without an ICU admission, hospitalized with an ICU admission, and non-hospitalized patients which served as the control group. Compared with the control group, hospitalized patients were more likely to have medications discontinued across all five categories and those admitted to the ICU were at highest risk of unnecessary discontinuation for four of the five categories. 40 These findings underscore the essential role of inpatient pharmacists and providers play in assessing and maintaining appropriate drug therapy for older adults throughout the care continuum. 48
Key Adverse Events and Drug Therapy Groups
Certain medication classes should be avoided in geriatric patients due to their high risk of adverse drug events (ADE); in such cases, alternative medications should be considered (Table 3). Medications with strong anticholinergic activity can cause delirium/dementia, dry mouth, and urinary tract dysfunction, and therefore should generally be avoided in older adults.49 -51 First-generation antihistamines, such as diphenhydramine, possess substantial anticholinergic activity.52,53 Anticholinergic agents may be used in the management of Parkinson’s disease; however, carbidopa-levodopa remains the drug of choice. 54
Medications to Avoid in Elderly Patients and Possible Alternatives.
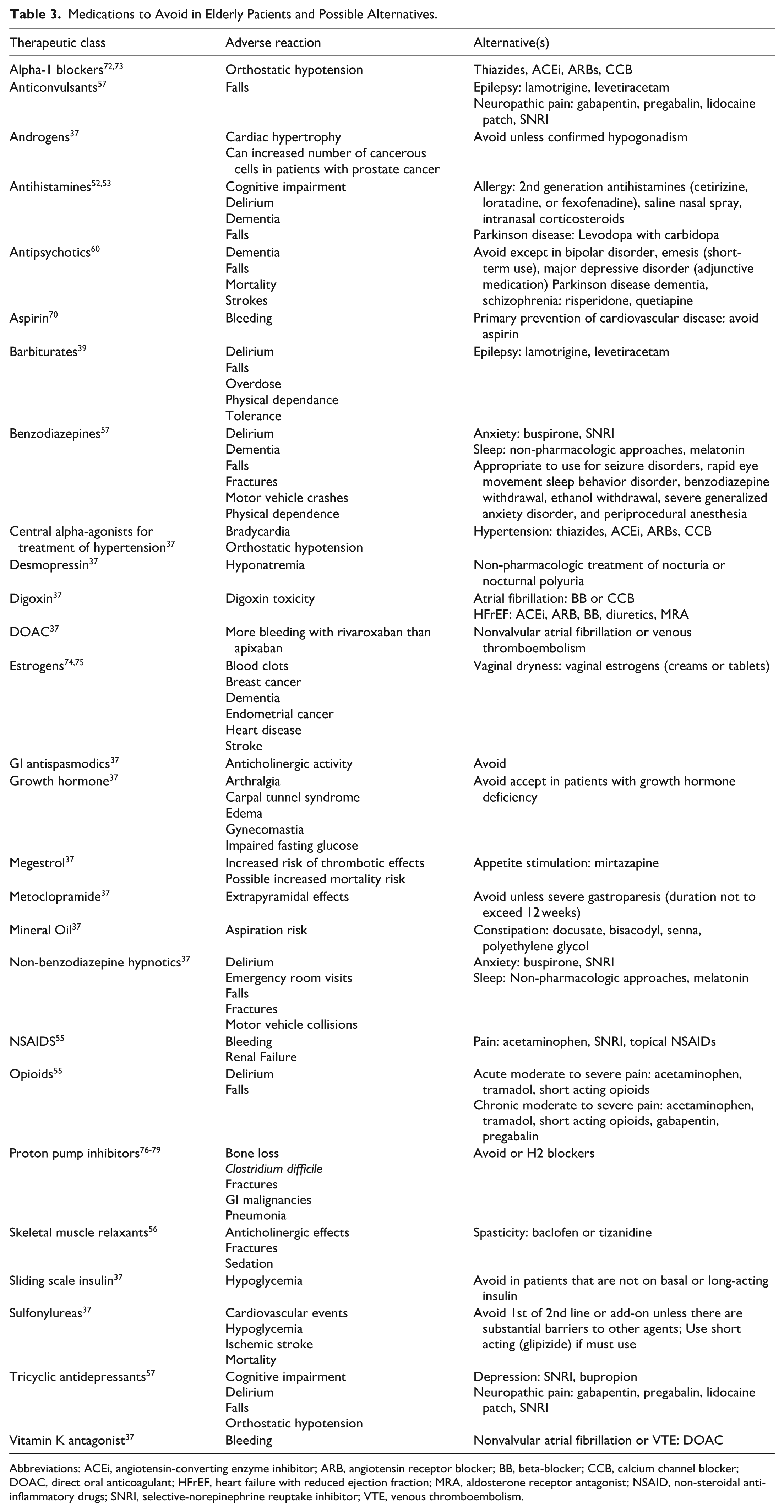
Abbreviations: ACEi, angiotensin-converting enzyme inhibitor; ARB, angiotensin receptor blocker; BB, beta-blocker; CCB, calcium channel blocker; DOAC, direct oral anticoagulant; HFrEF, heart failure with reduced ejection fraction; MRA, aldosterone receptor antagonist; NSAID, non-steroidal anti-inflammatory drugs; SNRI, selective-norepinephrine reuptake inhibitor; VTE, venous thromboembolism.
Pain management in older adults requires careful consideration. Nonsteroidal anti-inflammatory drugs (NSAIDs) can cause gastrointestinal bleeding and acute kidney injury, particularly in patients with chronic renal failure, hypertension, peptic ulcer disease, or heart failure. 55 Opiates can exacerbate delirium and should therefore be used for the shortest duration, with multimodal therapy deployed whenever feasible. 55 In hospitalized patients, pain should be assessed using validated tools such as the numeric rating scale (NRS) for patients who can self-report, and the Behavioral Pain Scale (BPS) or Critical Care Pain Observation Tool (CPOT) for those who are unable to verbalize. Skeletal muscle relaxants have significant anticholinergic side effects and can increase the risk of fractures in older adults. 56 Baclofen and tizanidine are reasonable alternatives to use in patients with spasticity. 37
Medications that act on the central nervous system such as antipsychotics, tricyclic antidepressants, antiepileptics, barbiturates, benzodiazepines, and non-benzodiazepines sleep aids not only increase the risk of falls in the elderly but also the risk of delirium and cognitive impairment. 57 The risk of falls is particularly high in elderly patients with a history of falls. 58 Tricyclic antidepressants may cause orthostatic hypotension and should be avoided. Benzodiazepines and non-benzodiazepine sleep aids have been shown to increase the risk of delirium. 59 Antipsychotics should be avoided in older adults due to their association with increased mortality. 60 Evidence demonstrates a lack of efficacy for antipsychotics in preventing or treating delirium.61,62 Despite this, these agents are frequently used during ICU stays and often continued after ICU discharge.63 -67 Sedative agents are similarly continued beyond the ICU stay, further increasing risk. 68 The ABCDEF Bundle (Assess, prevent, and manage pain, Both spontaneous awakening trials (SATs) and spontaneous breathing trials (SBTs), Choice of Analgesia and sedation, Delirium-assess, prevent, and mange, Early mobility and exercise, and Family engagement and empowerment) should be employed in ICU patients to minimize the exposure to sedatives and reduce delirium. 69
Anticoagulant therapy requires careful consideration in elderly patients. Aspirin for primary prevention may cause more harm than benefit. 70 Warfarin is associated with higher risk of intracranial bleeding in older adults with venous thromboembolism (VTE) or nonvalvular atrial fibrillation when compared to direct oral anticoagulants (DOACs). 71 Warfarin may be continued in older adults who have been stable on it long-term, have therapeutic INRs, and have not experienced adverse effects. In elderly patients with nonvalvular atrial fibrillation or VTE, apixaban should be considered over rivaroxaban due to rivaroxaban’s higher bleeding risk in this population. 37
Certain cardiovascular medications should be avoided in older adults. Alpha-1 blockers can cause orthostatic hypotension and should be avoided.72,73 Central alpha-agonists should likewise be avoided due to their risks of bradycardia and orthostatic hypotension. Digoxin should be avoided in elderly patients with atrial fibrillation or heart failure with reduced ejection fraction (HFrEF) due to the increased risk of digoxin toxicity secondary to age-related renal impairment. 37
Hormonal therapies and medications used to treat diabetes mellitus also require careful assessment in elderly patients. Androgens can cause cardiac hypertrophy and may stimulate cancer cell proliferation in males with prostate cancer. 37 Estrogen therapy increases the risk of breast and endometrial cancer and does not offer cardioprotective or cognitive benefits. Women older than 60 years who use hormone replacement therapy are at increased risk of heart disease, stroke, thromboembolism, and dementia.74,75 Sliding-scale insulin should be avoided in older adults who are not receiving basal insulin due to the risk of hypoglycemia. Sulfonylureas should be avoided unless all other alternatives have been exhausted. If sulfonylureas must be used, a short-acting agent such as glipizide is prefered. 37
Many medications that act on the gastrointestinal tract should be avoided in elderly patients. Proton pump inhibitors (PPIs) have been associated with Clostridium difficile infection, as well as pneumonia, gastrointestinal malignancies, bone loss, kidney disease, and fractures.76 -79 PPIs should be used for the shortest period of time possible and generally not longer than 8 weeks, unless the patient is also taking high-dose corticosteroids or chronic NSAIDs, has a documented need for maintenance therapy, or carries a diagnosis such as erosive esophagitis, Barrett’s esophagitis, or a pathologic hypersecretory condition. 37 These agents are frequently initiated in the ICU and continued without an appropriate indication.80 -83 This pattern highlights an important opportunity for deprescribing. Metoclopramide should be avoided for prolonged use due to its risk of extrapyramidal side effects and antispasmodic agents should not be used in elderly adults because of their significant anticholinergic activity. Mineral oil should also be avoided in elderly adults due to the risk of aspiration. 37
Opportunities for Intervention
There are numerous opportunities for healthcare providers to optimize care for geriatric patients, and research has shown that an interprofessional approach improves outcomes across the care continuum. Bergkvist and colleagues evaluated the role of integrated medicine management teams comprising of physicians, pharmacists, and nurses in 43 patients in Sweden and found a significant decrease in the number of inappropriate medications compared with the control group. 84 The successful identification and resolution of drug-related problems in older adults depends on effective collaboration between pharmacists, physicians, and APPs and establishing clear individual and collective roles is essential for optimal outcomes.
Conclusion
The increasing number of elderly adults poses a unique challenge for our healthcare system as the demand for specialized care – internal medicine to intensive care – continues to rise. All healthcare providers must be educated on the biologic, physiologic, pharmacokinetic, and pharmacodynamic changes associated with aging and the implications these changes have on drug therapy. Inpatient pharmacists, physicians, and APPs have an opportunity to utilize the plethora of tools available to optimize care, prevent harm, and improve quality of life for geriatric patients. Accordingly, clinicians must be aware of specific drug therapies to avoid, as well as preferred alternatives, when caring for this vulnerable patient population.
Footnotes
Acknowledgements
MP, JS, and WE all contributed equally to the manuscript writing.
Ethical Considerations
Ethical approval was not required for this review article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Dr. Ely has received a research grant from Dr. Franz Kohler Chemie GMBH and is supported by the NIH grants (R01 AG027472, R01 AG035117, R01 AG058639, T32 GM135094, I01RX002992).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
