Abstract
Introduction
Venous thromboembolism (VTE) has an annual incidence of 117 per 100 000 individuals, and obesity is a risk factor that confers a six-fold increase in risk of developing a VTE compared to non-obese individuals. 1 The prevalence of obesity has risen over the years in the United States, now affecting nearly half of all adults. 2 Obesity is frequently a comorbidity in hospitalized patients, and medication dosing in this population can be challenging due to unpredictable pharmacokinetics. 3
Intravenous (IV) unfractionated heparin is an anticoagulant commonly used for treatment of VTE. Although the CHEST Antithrombotic Therapy Guideline for VTE Disease recommends a weight-based heparin dosing regimen, there is a lack of guidance regarding dosing in obese patients.4-6 Heparin’s volume of distribution is mainly limited to plasma with minimal distribution into adipose tissue, complicating heparin dosing in obese individuals.7,8 Previous retrospective studies have demonstrated that morbidly obese patients (body mass index (BMI) ≥ 40 kg/m2) were more likely to exhibit supratherapeutic activated partial thromboplastin time (aPTT) values compared to non-obese patients when dosed based on total body weight (TBW) in a standardized heparin nomogram.9-11 Additional retrospective studies have analyzed alternative dosing strategies, such as adjusted body weight (ABW) in obese patients. Researchers found that ABW dosing in obese patients resulted in similar therapeutic aPTT values compared to non-obese patients dosed using TBW.12,13
However, certain gaps in the literature regarding heparin dosing in obese patients remain, such as the effects on coagulation markers at multiple time points and the consideration of reagent dependent variance in aPTT assays. To further characterize appropriate dosing strategies, this study seeks to evaluate the achievement of therapeutic anticoagulation with IV heparin utilizing TBW versus ABW in obese patients across various timepoints and utilizing the heparin correlation value (HCV), a standardized reporting of aPTT levels that corrects for reagent-based assay variance. 14
Methods
Study Design and Patient Population
This was a single-center, retrospective, observational cohort study conducted at a large, tertiary care academic medical center and approved by the institution’s Institutional Review Board. Included patients were obese (BMI ≥ 30 kg/m2), at least 18 years of age, initiated on and receiving a continuous IV heparin infusion following a standard nomogram utilized for the treatment of active thrombosis for at least 6 hours, and admitted between January 1, 2020, to May 20, 2023. Exclusion criteria included patients with a prolonged aPTT at baseline (>40 seconds), heparin infusion initiated at an outside institution, history of genetic prothrombotic disorders (ie, protein C or S deficiency, factor V Leiden, antithrombin III deficiency, and antiphospholipid syndrome), use of additional anticoagulants at the time of the heparin infusion, and active pregnancy. Two cohorts were defined according to the initial dosing weight selected for the heparin infusion (TBW vs ABW, where ABW = Ideal Body Weight (IBW) + 0.4 × (TBW − IBW) 12 ).
Heparin Protocol
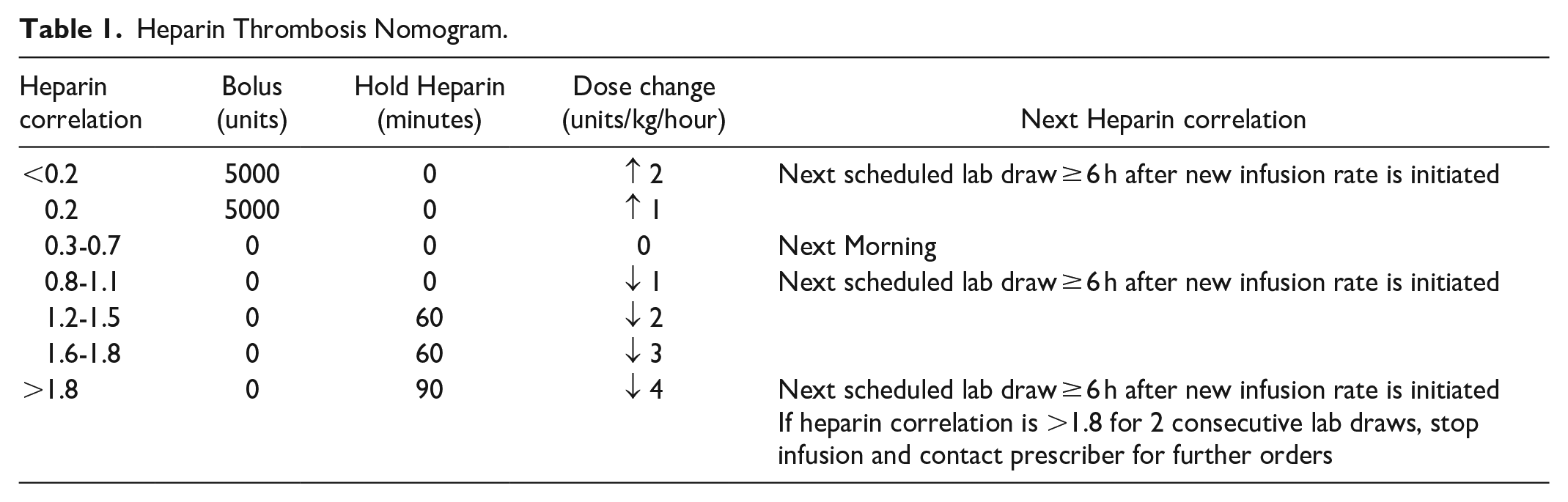
A standard weight-based institutional nomogram for heparin monitoring and dose adjustment was followed in all patients. This nomogram, typically used in patients with new VTE or acute limb ischemia, consists of an infusion of 18 units/kg/hour with or without an initial loading dose of 80 units/kg; neither the initial bolus nor initial rate are dose capped. The nomogram defaults to recorded TBW for dose determinations, but providers can adjust the dosing weight during order entry based on clinical expertise.
Heparin monitoring was based on HCV which, similar to the INR, has the benefit of standardizing reference ranges such that therapeutic HCV ranges can be maintained and the relationship of HCV to aPTT is adjusted when laboratory reagents change rather than adjusting the goal aPTT range. 14 Dose adjustments were performed per nursing protocol, targeting a therapeutic HCV range of 0.3 to 0.7 (Table 1). HCV was monitored per nomogram, with an initial level checked 6 hours after initiation of the heparin infusion and subsequent timing of levels determined by the HCV.
Heparin Thrombosis Nomogram.
Data Collection and Study Outcomes
Patients were identified using the Carolina Data Warehouse for Health (CDWH) database. Data was collected from electronic medical records in patients who met inclusion criteria and included baseline demographics, heparin medication order and administration details, relevant laboratory values, and signs of bleeding complications.
The primary outcome was the achievement of HCV value within the therapeutic range at the first laboratory check after heparin initiation. Secondary outcomes included achievement of HCV within the therapeutic range at any time point, time to first HCV within the therapeutic range, heparin infusion rate (units/kg/hour) required to achieve HCV within the therapeutic range, incidence of subtherapeutic, therapeutic, or supratherapeutic HCV at 24 hours, number of dose adjustments required in the first 24 hours, and incidence of supratherapeutic HCV during any point in therapy. The primary safety outcome evaluated incidence of major bleeding as defined by the International Society of Thrombosis and Homeostasis (ISTH), which included any fatal bleeding and/or symptomatic bleeding in a critical area or organ, bleeding causing a fall in hemoglobin level of ≥2 g/dL, or leading to transfusion of ≥2 units of whole blood or red cells. 15 Because follow-up imaging to evaluate for new, residual, or recurrent thrombosis is not routinely performed, efficacy endpoints were not evaluated.
Statistical Analysis
Patients were stratified according to obesity class into the following categories: Class 1 (BMI 30-<35 kg/m2), Class 2 (BMI 35-<40 kg/m2) and Class 3, or morbid, obesity (BMI ≥ 40 kg/m2). Descriptive statistics were used to evaluate demographic and clinical patient data. A power calculation was not performed as the dosing weight utilized was imbalanced during the study timeframe. Analysis of categorical data was conducted using a chi-square test and analysis of continuous data was conducted using an unpaired t-test or Mann-Whitney U test. A significance level of <.05 was assumed for all statistical analyses.
Results
Screening and Baseline Characteristics
A total of 688 patients were screened during the study timeframe and 477 were included in the primary analysis, with 453 (94.9%) patients in the TBW dosing cohort and 24 (5.1%) patients in the ABW dosing cohort (Figure 1). Patients dosed using TBW had a mean BMI of 36.8 ± 7.4 kg/m2 whereas patients dosed using ABW had a mean BMI of 42.1 ± 9.3 kg/m2 (P < .001). Patients with Class 3 obesity represented 20.1% (n = 91) of the TBW cohort, compared to 54.2% (n = 13) of the ABW cohort (P < .001). An initial heparin loading dose was administered in 50.3% (n = 228) and 66.7% (n = 16) of patients in the TBW cohort and ABW cohort, respectively (P = .119, Table 2).
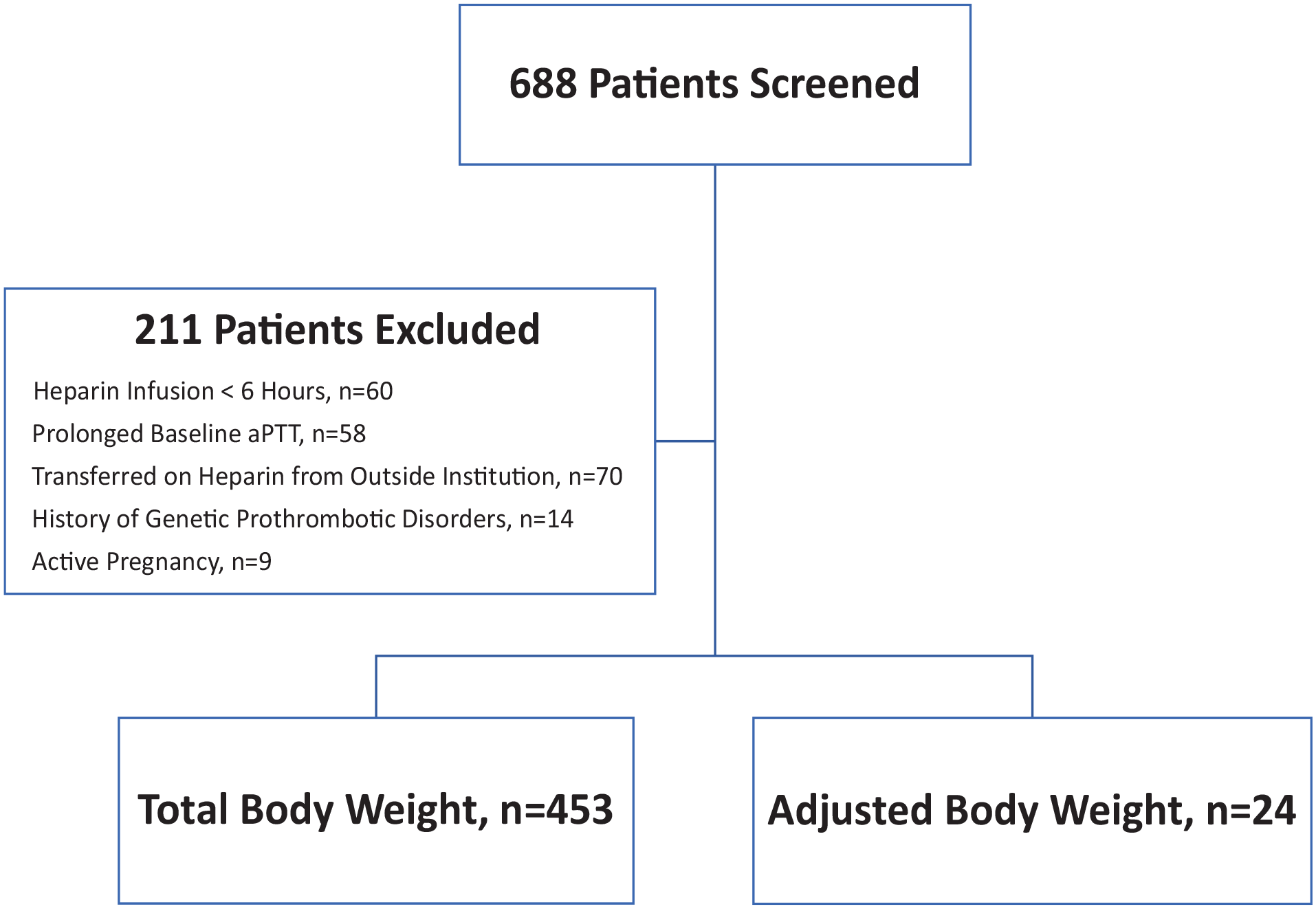
Consort diagram of included patients.
Patient Characteristics at Baseline.
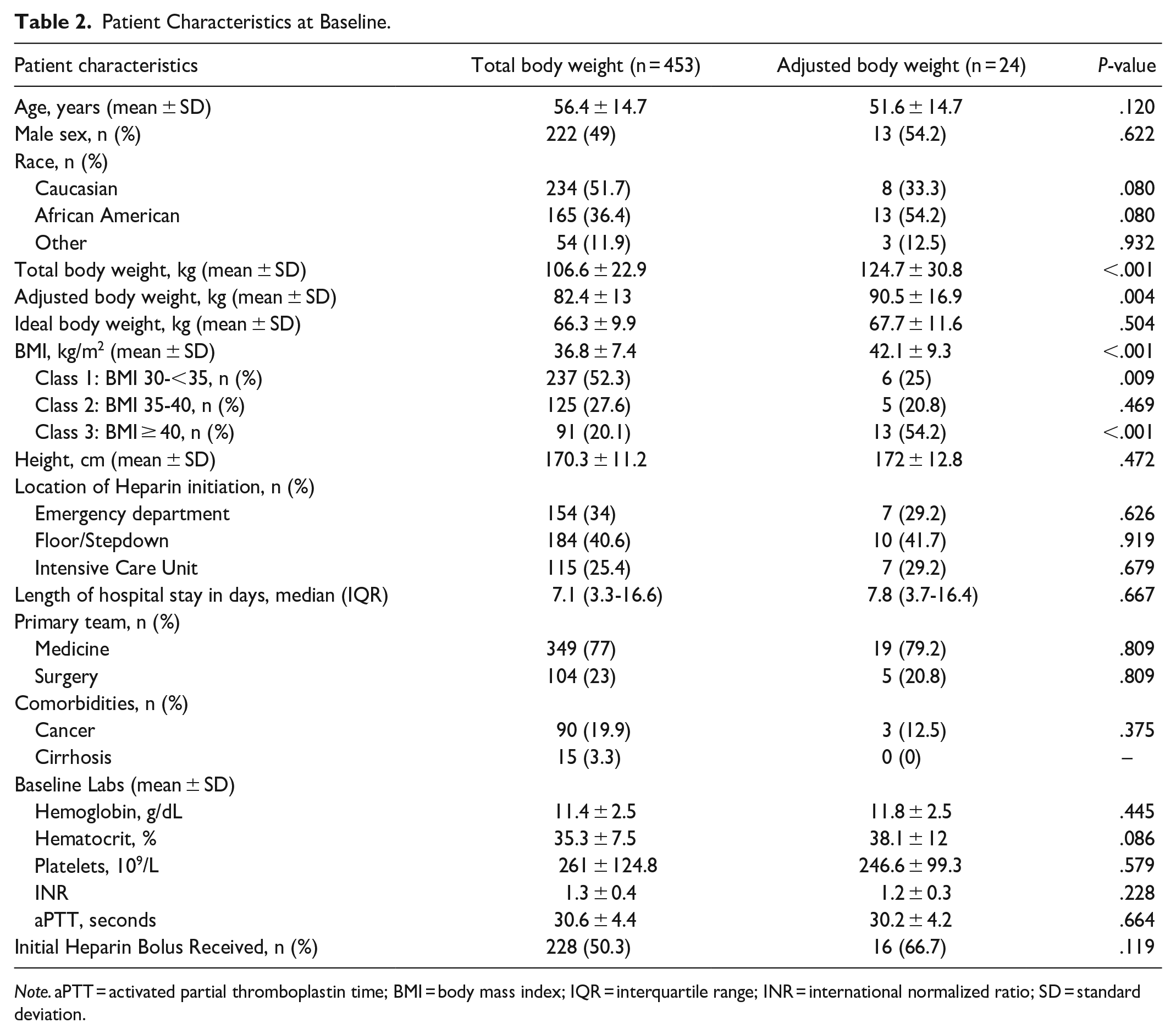
Note. aPTT = activated partial thromboplastin time; BMI = body mass index; IQR = interquartile range; INR = international normalized ratio; SD = standard deviation.
Outcomes
The primary outcome of initial HCV within the therapeutic range was achieved in 41.9% (n = 190) and 54.2% (n = 13) of patients in the TBW cohort and ABW cohort, respectively (P = .238). When this outcome was evaluated based on patients who received an initial heparin bolus, there was no statistical significance seen between cohorts (TBW, 74/228 [32.5%] vs ABW, 8/16 [50%], P = .151). Furthermore, there was no statistically significant difference in initial HCV classified as subtherapeutic (P = .773) or supratherapeutic (P = .320) between both cohorts (Table 3).
Primary, Secondary, and Safety Outcomes.
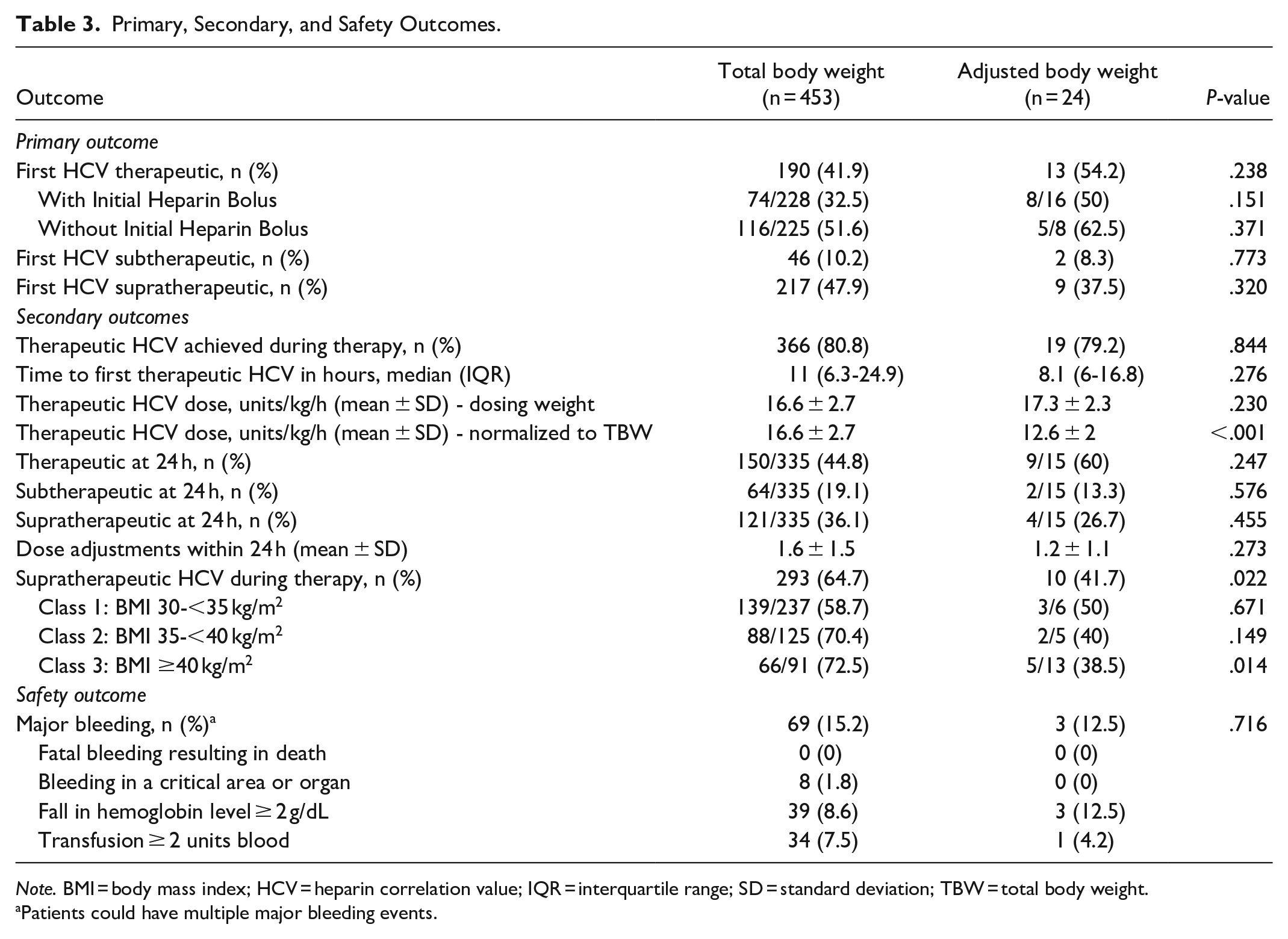
Note. BMI = body mass index; HCV = heparin correlation value; IQR = interquartile range; SD = standard deviation; TBW = total body weight.
Patients could have multiple major bleeding events.
Therapeutic HCV at any point during heparin therapy was achieved in a majority of patients. Time to reach therapeutic HCV was similar between cohorts (TBW, 11 [6.3-24.9] hours; ABW, 8.1 [6-16.8] hours; P = .276). The heparin infusion rate required to achieve HCV within the therapeutic range averaged 16.6 ± 2.7 units/kg/hour (based on TBW) in the TBW cohort compared to 17.3 ± 2.3 units/kg/hour (based on ABW) in the ABW cohort (P = .230). However, when the therapeutic heparin infusion rate in the ABW cohort was expressed in units/kg/hour using TBW, the rates required to achieve a HCV in the therapeutic range were significantly different between the groups (TBW, 16.6 ± 2.7 units/kg/hour based on TBW; ABW, 12.6 ± 2 units/kg/hour based on TBW; P < .001).
At 24 hours, there was no difference between the TBW and ABW cohorts regarding therapeutic (44.8% vs 60%, P = .247), subtherapeutic (19.1% vs 13.3%, P = .576), or supratherapeutic (36.1% vs 26.7%, P = .455) HCV results. The number of dose adjustments required within the first 24 hours were similar between TBW and ABW cohorts (1.6 ± 1.5 vs 1.2 ± 1.1, P = .273).
Supratherapeutic HCVs during any point in therapy occurred more frequently in the TBW cohort (64.7% vs 41.7%; P = .022). In an analysis of relevant subpopulations, this effect was most pronounced among subjects categorized as Class 3 obesity (BMI ≥ 40 kg/m2), with 72.5% (n = 66/91) of the TBW cohort and 38.5% (n = 5/13) of the ABW cohort experiencing a supratherapeutic HCV at some time during therapy (P = .014, Figure 2).
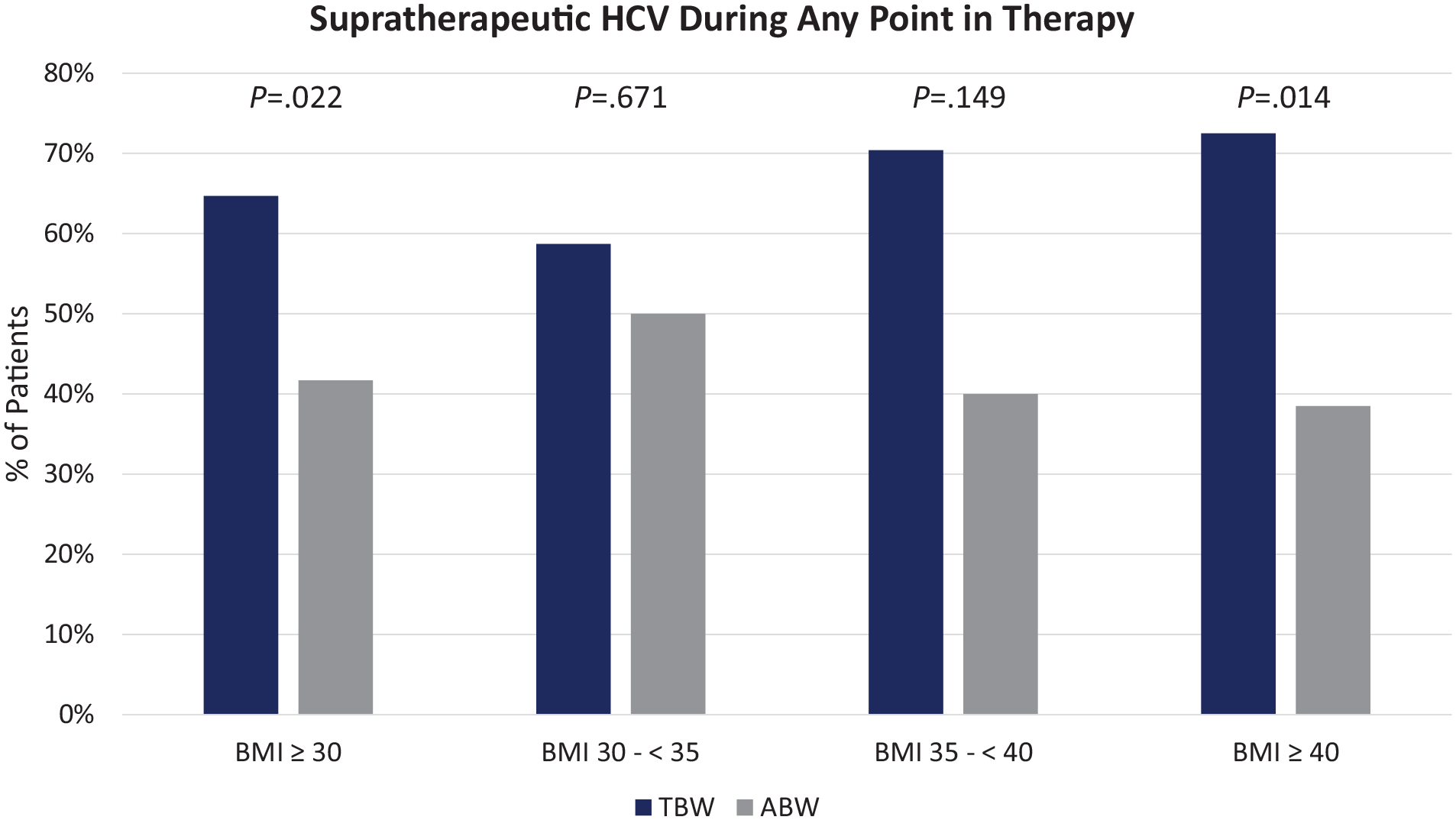
Secondary outcome & subgroup analysis—Comparison of supratherapeutic HCV during any point in therapy based on TBW versus ABW dosing and stratified according to BMI category.
Major bleeding events were similar and occurred in 15.2% (n = 69) of patients in the TBW cohort and 12.5% (n = 3) in the ABW cohort (P = .716), with the majority of bleeding events being attributed to bleeding associated with a fall in hemoglobin level ≥ 2 g/dL (TBW, 8.6%; ABW, 12.5%).
Discussion
Our results demonstrated that utilizing a heparin dosing strategy based on ABW versus a dosing strategy based on TBW in obese patients did not result in a statistically significant difference in frequency of initial HCV values within the therapeutic range. However, considering the 12.3% difference in rate of initial therapeutic HCV and limited sample size, a meaningful difference cannot be excluded. Given the importance of time to therapeutic heparin levels, it is worth noting that even at 24 hours there was a persistent 15% difference in attainment of therapeutic HCV between the TBW and ABW groups, though this difference did not reach statistical significance.
A notable finding from our study is the increased risk of supratherapeutic levels of anticoagulation in obese patients receiving heparin infusions dosed based on TBW, which was consistent across multiple evaluated timepoints. This is reflected in the 10% higher rate of supratherapeutic HCV on initial check, the 9% higher rate of supratherapeutic HCV at 24 hours, and the 23% higher rate of supratherapeutic HCV at some point during heparin therapy. While only the last result was statistically significant and there was no discernible difference in major bleeding events in our study, the clinical significance of these results is worth consideration given the potential for bleeding with prolonged exposure to supratherapeutic levels of anticoagulation. As one would expect, the risk of supratherapeutic HCV becomes more pronounced as the BMI increases, reflecting the known distribution of heparin. The significantly higher risk of supratherapeutic HCV at some point during heparin therapy for patients with Class 3 obesity (BMI ≥ 40 kg/m2) dosed based on TBW compared to patients dosed based on ABW (72.5% vs 38.5%, respectively) argues for consideration of empiric dose adjustment at the time of heparin initiation in this patient population.
Potential strategies for empiric heparin dose adjustment in obese patients include adjusting the initial weight-based infusion rate, placing an upper limit on dosing weights or resulting units/hour initial infusion rates, or selecting a dosing weight more reflective of the volume of distribution of heparin (ie, using ABW instead of TBW). While this study did not directly compare these dosing strategies, results do suggest that dosing weight adjustment may be the most appropriate approach. At baseline, the ABW cohort had a greater average TBW, greater average BMI, and higher percentage of patients with BMI ≥ 40 kg/m2 than the TBW cohort. When comparing heparin infusion rates between the cohorts as calculated based on TBW, rates required to achieve HCV within the therapeutic range were significantly lower in the ABW cohort than in the TBW cohort (12.6 vs 16.6 units/kg/hour, respectively), suggesting that the TBW weight-based dose required to achieve a HCV within the therapeutic range decreases as weight and BMI increase. This result implies that adjusting the dosing weight may be the more consistent and reliable method for empiric heparin dose adjustment. Future studies directly comparing these dosing strategies are warranted.
This work attempts to address important gaps in current literature. Several studies have examined various heparin dosing strategies in obese patients, though these studies are limited by small sample sizes, varied heparin dosing regimens, inconsistency in defining obesity, and reliance on the aPTT, a reagent-dependent value, for heparin monitoring. Fan et al performed a retrospective pre-post cohort study evaluating a revised institutional heparin nomogram that utilized ABW-dosing in obese patients (>100 kg) compared to TBW-dosing in non-obese patients (<100 kg). They found no significant difference in percentage of first aPTT values within therapeutic range (16.9% vs 20.9%, P = .58); however, obese patients were more likely to have subtherapeutic aPTT values (39% vs 19.6%, P = .003) and less likely to have supratherapeutic aPTT values compared to non-obese patients (44.2% vs 59.5%, P = .04). 12 These results align closely with those of the present study, although a higher percentage of patients in our cohort had an initial therapeutic HCV, which may suggest that ABW-dosing may be more appropriate for obese patients based on BMI classification instead of a kilogram threshold. Hosch et al evaluated the outcomes of dosing heparin infusions utilizing ABW in obese patients (>20% IBW) compared to TBW-dosing in non-obese patients. This protocol consisted of an 80 units/kg bolus followed by an 18 units/kg/hour infusion with a dose cap. Researchers found that dosing with ABW resulted in similar times to therapeutic aPTT (15 vs 15.7 vs 15.4 hours, P = .506) and no differences in rates of bleeding amongst non-obese, obese, and severely obese patients, respectively (14% vs 13.6% vs 7.9%, P = .453). 13 Although these results are similar to our findings despite a dose cap implemented in the aforementioned study, it is difficult to characterize the implications of these results as achievement of an initial therapeutic aPTT may not imply steady state or a full anticoagulation effect was achieved.
Our study is the first to evaluate different heparin dosing weight strategies in obese patients and the effects on HCV, a standardized reporting of the aPTT that eliminates the need to revise a heparin nomogram if there is a change in the aPTT reagent used. Additional strengths of this study include assessment of HCV measures at different time points, stratification of patients by obesity class, inclusion of a high proportion of patients with Class 3 obesity, and assessment of patient-centered safety outcomes.
This study also has notable limitations. The small sample size of this study does not address the problem of small sample sizes represented in other studies and limits the ability to evaluate differences in relevant subgroups, including the critically ill and patients with risk factors for bleeding, surgical patients, and patients who did versus did not receive the initial loading bolus of heparin. Use of a convenience sample over a pre-specified timeframe and study group imbalances over that timeframe limited statistical power to detect meaningful differences in both heparin monitoring-based outcomes and patient-centered outcomes, including major bleeding events. Allocation to the ABW dosing group was dictated by provider preference, potentially introducing confounder bias in treatment selection which may have led to more patients with Class 3 obesity or more patients with risk factors for bleeding receiving heparin dosed based on ABW, leading to between-group differences. The design of the study cannot exclude a provider effect, in which some providers were more likely to prefer dosing based on ABW while others more often chose to dose based on TBW. The retrospective nature of this study and missingness of relevant data elements precluded evaluation of additional clinical outcomes, including efficacy endpoints, as well as the assessment of all possible risk factors that may contribute to bleeding, VTE development, or alterations in HCV values. This was also a single center study utilizing a specific heparin order set and nomogram, which may limit generalizability.
Conclusion
Utilization of ABW compared to TBW for dosing of heparin infusions in obese patients receiving treatment for thrombosis indications did not lead to a higher attainment of therapeutic anticoagulation at the first monitoring timepoint. Increased rates of supratherapeutic levels of anticoagulation in obese patients dosed based on TBW warrant consideration of empiric dose adjustment in this patient population. Future prospective studies in obese individuals should compare different heparin dosing strategies including dosing weight selection, modifications to the initial bolus and/or rate, and dose capping.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
