Abstract
Background
Oral challenge is widely recognized as a safe and effective method for assessing penicillin allergies in low-risk population.1-3 However, there remains limited analysis on the timelines to and types of reactions that may occur during or after oral challenge interventions. This knowledge is essential not only for optimizing the safety and efficacy of oral challenge protocols but also for facilitating their integration into routine clinical practice. 4 By improving understanding of the characteristics, timelines, and predictors of hypersensitivity reactions, this work seeks to empower healthcare providers to confidently remove inappropriate penicillin allergy labels. In doing so, it will help reduce the unnecessary avoidance of first-line antibiotics, enhance antimicrobial stewardship, and improve patient outcomes across diverse healthcare settings.5-7 Specifically, this study aims to comprehensively describe the characteristics of oral challenge interventions, categorize the observed and reported reactions following oral challenge, and identify predictors of hypersensitivity reactions in studies designed to delabel penicillin allergies in patients with a low-risk of hypersensitivity.
Methods
This study leverages data from a previously published systematic review and meta-analysis focused on penicillin allergy delabeling in low-risk populations. 3 Details of the methods are available in both the published study protocol 8 registered in PROSPERO: CRD42022336457 and in a manuscript reporting the primary findings. 3 Briefly, a comprehensive search strategy was conducted across multiple databases (PubMed, Cochrane Database of Systematic Reviews, International Pharmaceutical Abstracts, Ovid MEDLINE, Ovid EMBASE, ClinicalTrials.gov and medRxiv) between their inception and February 2024. Two independent reviewers initially screened titles and abstracts prior to that of relevant full-text articles. This descriptive analysis uses data from all identified randomized trials and quasi-experimental studies evaluating individuals classified as having a low-risk penicillin allergy history who underwent oral challenge interventions, as these participants were directly evaluated for tolerability based on post-challenge outcomes. Low-risk was defined in the initial systematic review using thematic analysis of all eligibility criteria for inclusion in oral challenge arms across the 26 studies in which seven major themes were identified: non-severe cutaneous reactions, unknown reaction, side effects or intolerance, absence of anaphylaxis or angioedema, no personal reaction experienced, non-life-threatening reaction, and tolerated since index reaction. 3 Patients included in direct delabeling interventions without oral challenge were excluded from this analysis.
Baseline demographic and clinical characteristics, intervention protocols, and reported index reactions to penicillins (reason for penicillin allergy label) were extracted as described by the authors and reported by participants. The primary outcomes of interest for this study are the characteristics of reported reactions after oral challenge intervention, including the time to onset, classification of reported reactions, and potential predictors of hypersensitivity reactions. Any reaction to oral challenge or reported reason that led to lack of removal of the allergy label based on the discretion of the study authors were described as non-delabeled. The mean incidence of reactions (or non-delabeling) occurring after oral challenge administration was described in the original systematic review and meta-analysis but is also briefly reported herein. Given that none of the reactions were severe in nature, reactions were categorized into the following groups: (1) non-severe immediate reactions (within 1 hour of oral challenge), (2) delayed non-severe reactions (occurring more than 1 hour after oral challenge), and (3) other reported reactions.
Descriptive statistics, including means and ranges, were used to summarize patient demographics, intervention characteristics, and baseline reaction histories. A random-effects proportional meta-analysis was conducted, 9 heterogeneity of results across studies was assessed using the χ2 statistic and quantified using the I2 statistic and the DerSimonian-Laird approach was used to estimate between study heterogeneity. All oral challenge arms across all intervention types were pooled together for the purpose of this meta-analysis. Factors associated with an increased risk of post-oral challenge reactions, as reported by study authors, were synthesized and described narratively.
Results
Population Characteristics
As described in the initial systematic review and meta-analysis, there were 26 studies (2 RCTs, and 24 quasi-experimental studies) identified that included an oral penicillin challenge among low-risk patients. The mean age was reported in 24 studies (85%) with an average age of 56 years, ranging from 34 to 73 years.10-33 Most study participants were female (61%)10-25,28-33 with two studies including exclusively pregnant people.26,27 Five studies included both male and female participant demographic variables at baseline.10,17,19,25,29 Ten studies reported ethnicities of participants of which 88% were White.12-14,17,19,23-25,32,33
Index Reactions Leading to Allergy Labeling
The self-reported index reactions were described in 18 (69%) of the studies;11,13,14,16-22,24,26-28,30-32,34 details regarding all reported index reactions can be found in Table 1. Although index reactions were reported, these included both low and high-risk individuals without further stratification by allergy risk or correlation with tolerability of the oral challenge. Based on the reported data, diffuse rash occurring more than 10 years prior (or timeline not described) was reported in 40% of participants across 8 studies11,14,17,19,21,22,26,27 followed by childhood exanthem in 35% (min 9%, max 76%) across 5 studies13,17,20,22,34 and urticaria in 29% across 9 studies (min 9%, max 42%).11,14,16,17,19,26-28,30,31 Other types of index reactions were reported across various studies as described in Table 1.
Baseline Self-Reported or Reported Reactions to Penicillins (Reported in 18 of 26 Studies).
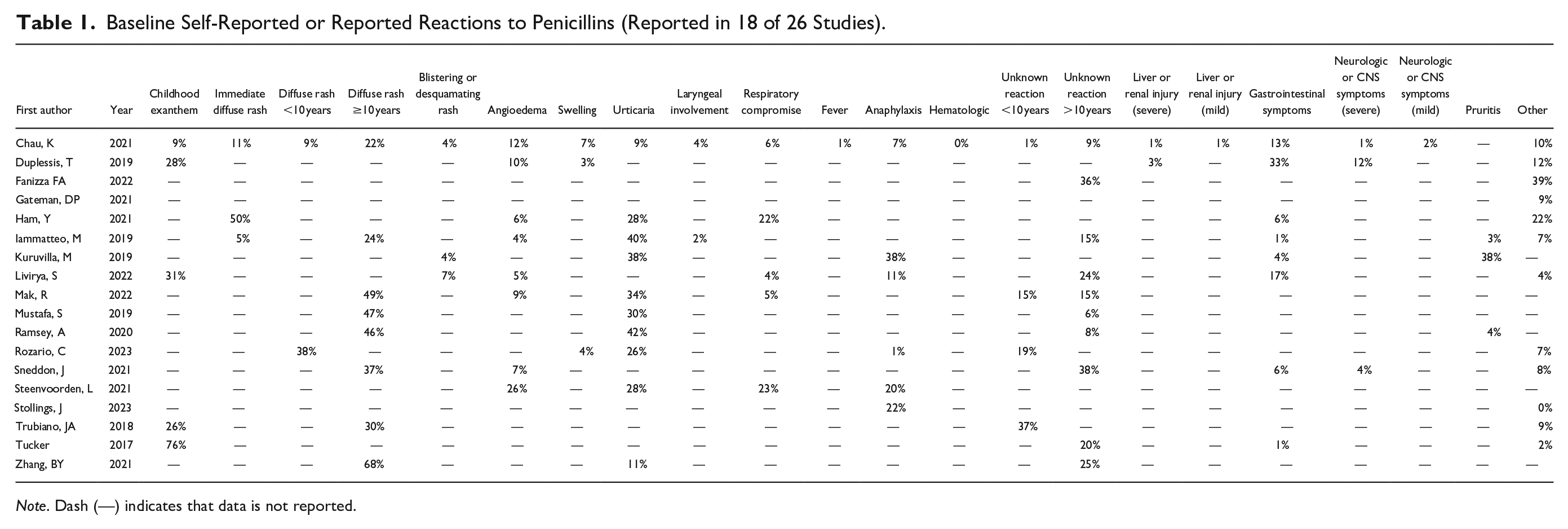
Note. Dash (—) indicates that data is not reported.
Oral Challenge Intervention Characteristics
In the 26 studies for which oral challenge in the setting of a low-risk penicillin allergy was performed, amoxicillin 250 mg as a single oral dose was an option in 42% of studies16,17,20,22-25,28,31,32,34 (Table 2). Amoxicillin 500 mg administered orally was described in 27% of studies;14,17,21,27,29,30,33 however, frequency of dosing varied from once daily to twice daily administration for multiple days. Graded oral challenge strategies were described in 19% of studies.11-13,18,35 In the study by Alnaes et al, 33 the penicillin for which the index reaction occurred was used if known, otherwise amoxicillin was selected. Other penicillins for oral challenge were considered in fewer studies as can be seen in Table 2. With respect to provider monitoring, nurses were involved in 27%,14,15,17,20,22,25,35 pharmacists in 15%,13,16,24,32 allergists or immunologists in 12%,11,26,30 primary care providers in 4%, 18 and other or non-defined providers in 38%.10,12,19,21,23,27,28,31,33,34 One study by Fransson et al 29 included patient self-completion.
Protocol Descriptions and Reported Reactions from 26 Studies Involving an Oral Challenge.
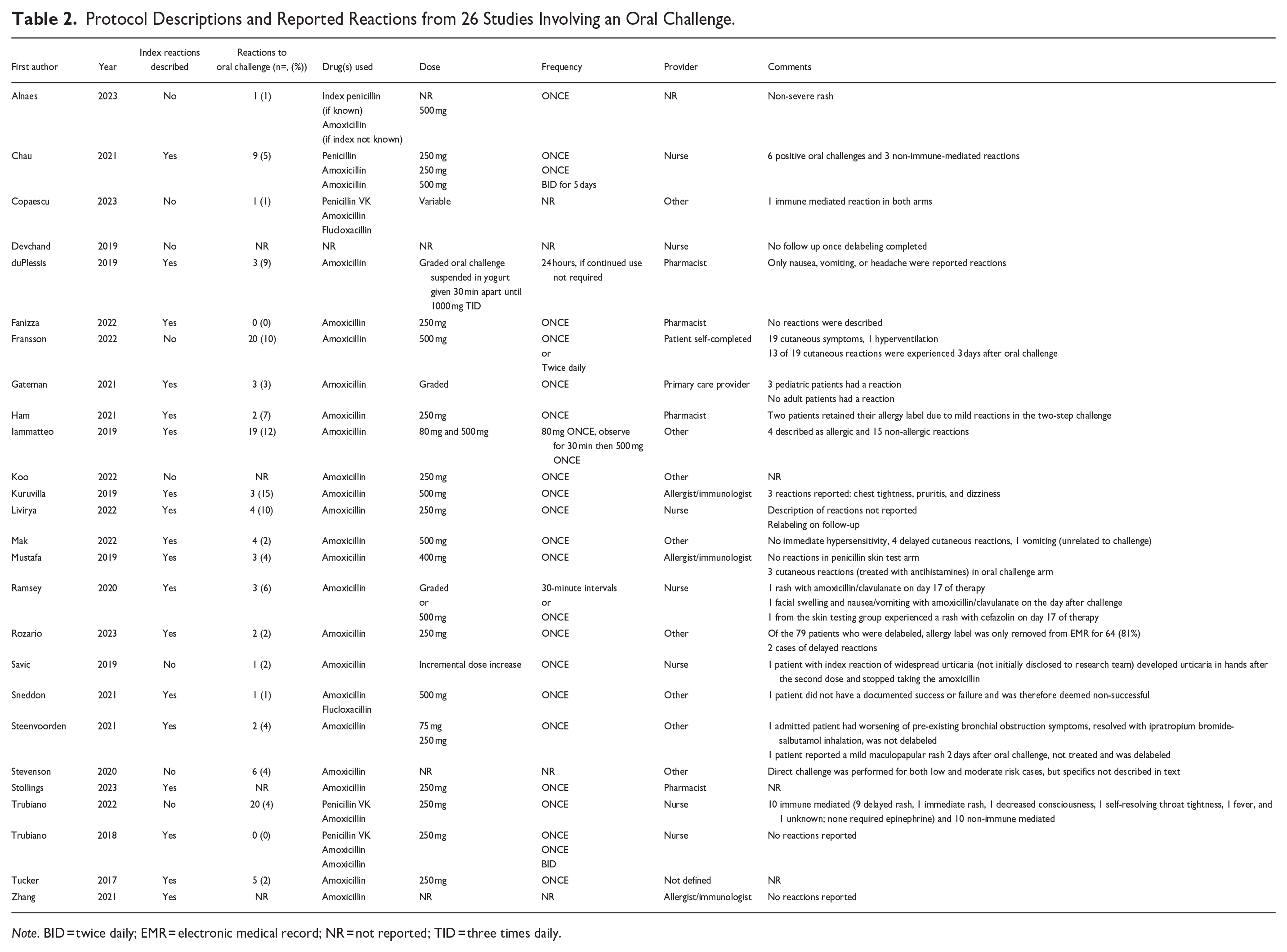
Note. BID = twice daily; EMR = electronic medical record; NR = not reported; TID = three times daily.
Incidence of Reaction to Oral Challenge
Of 26 studies in which participants received an oral challenge, the pooled weighted mean incidence of reaction or non-delabeling due to an experienced reaction, or non-delabeling at discretion of the study authors was 4% (95% Confidence Interval 3%, 6%), I2 = 43.2%, P = .0001 (Figure 1). None of the reported reactions were severe in nature. The reaction rate varied across studies with 6 studies describing all participants as tolerating oral challenge. In 13 studies, between 1% to 5% of participants were not delabeled; in 5 studies 6% to 10% of participants were not delabeled. Reactions to oral challenge occurring at a rate greater than 10% were described in studies by Kuruvilla et al 30 and Iammatteo et al 19 ; however, of the 19 reactions described in the Iammatteo study, 15 are deemed non-allergic in nature. Details regarding the reported reactions can be found in Table 2.
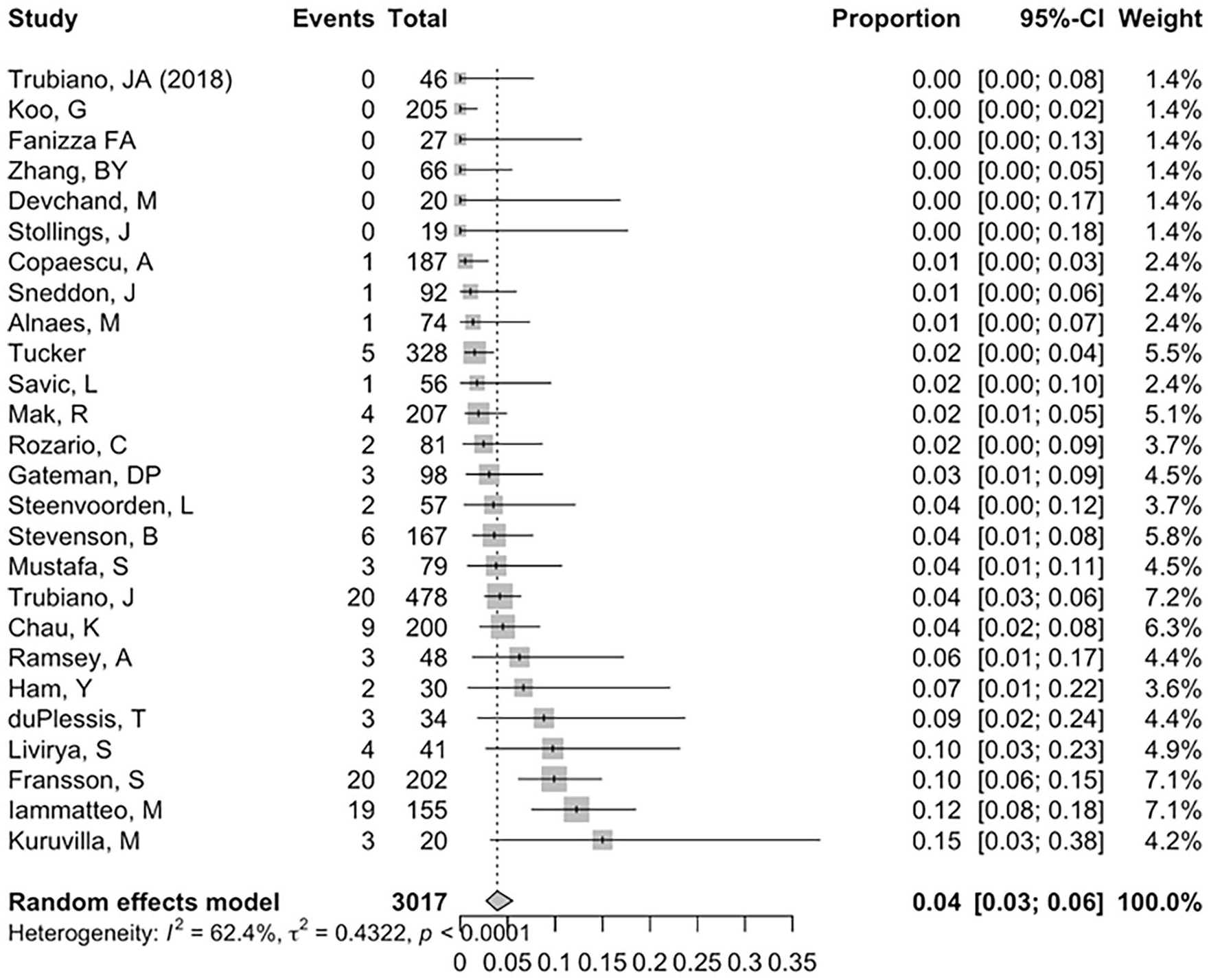
Proportion meta-analysis of oral challenge reactions.
Immediate Reactions
In 2 RCTs, reactions to oral challenges were infrequent and non-severe. Copaescu et al 12 reported immune-mediated reactions in 0.5% of participants (one per study arm) while Mustafa et al 11 reported three mild cutaneous reactions, all resolving with oral antihistamines. Of 24 quasi-experimental studies, including two involving pregnant individuals, 13 reported no reactions, mild non-allergic symptoms, or non-severe cutaneous reactions, with seven studies (27%) reporting no reactions during oral challenge.15,22-24,26,29,32 Mak et al, 27 in a study of pregnant individuals, reported 4 delayed maculopapular rashes treated with topical steroids and oral antihistamines and 1 case of vomiting attributed to pregnancy. Ham et al 16 found 98% of participants were delabeled, with two retaining their allergy label due to mild, non-IgE-mediated symptoms (lip tingling and gastrointestinal discomfort). Kuruvilla et al 30 observed 3 non-IgE-mediated reactions (pruritus, chest tightness, dizziness); all self-resolved without treatment. Rozario et al 28 reported that while 98% of participants were eligible for delabeling, only 64 of 79 who tolerated oral challenges opted to remove the allergy label from their electronic medical record.
Delayed Reactions
In 6 quasi-experimental studies, non-severe delayed reactions were reported. Chua et al 17 report that 97% of participants completed the oral challenge successfully, with 6 documented reactions: 3 non-immune-mediated (fever with baseline urosepsis, vomiting, and pruritus without rash) and 3 delayed immune-mediated reactions suspected to be T-cell-mediated, occurring 5 to 7 days after oral challenge intervention. du Plessis et al 13 reported no immediate reactions, but 3 cutaneous reactions occurred within 72 hours. Ramsey et al 14 found 97.9% of participants tolerated the intervention with 1 delayed rash after 17 days and 1 case of facial swelling, nausea, and vomiting the day after challenge with amoxicillin-clavulanate, managed successfully with antihistamines. Trubiano et al 25 reported a 95.8% delabeling rate, with 10 presumed immune-mediated reactions (9 delayed rashes, 1 immediate rash, and others including throat tightness and fever), with none requiring epinephrine. Fransson et al 29 observed 19 of 20 reactions involved skin symptoms, with 13 occurring 3 days post-challenge. In the only study with 6-month follow-up, 20 10% of participants had allergy labels re-applied without detailed explanations.
Other Reported Reactions
Unique patient factors contributed to reactions in 2 studies, and 1 study described broad treatment for notable reactions. In the Steenvorden et al 31 study, a patient with bronchial obstruction experienced worsening respiratory symptoms, which resolved with ipratropium-salbutamol inhalation; this was deemed unlikely to be IgE-mediated, although the patient was not delabeled. In the study by Savic et al 35 one patient developed hand urticaria after the second dose of amoxicillin. Further questioning revealed a history of widespread urticaria, which the patient did not disclose, as they were eager to participate in testing. Tucker et al 34 studied marine recruits, of whom five (2%) experienced reactions: two with rashes, two with unknown reactions, and one case of hives; in these instances, epinephrine was administered empirically to prevent escalation although anaphylaxis was not reported.
Risk Factors that Correlated with Reaction to Oral Challenge
Two studies explored risk factors linked to positive oral challenges and defined populations deemed “low risk.” Iammatteo et al 19 found a correlation between the number of baseline reported allergies or intolerances and oral challenge outcomes. Patients with allergic reactions had a median of 3 baseline allergies or intolerances (IQR 2.5-3.5), compared to 2 (IQR 1-4) in those with non-allergic reactions and 1 (IQR 1-2.5) in those with no reaction (P = .02). 19 Stevenson et al 10 evaluated 8 definitions of low risk, including timing and severity of index reactions. The optimal model, suited for outpatient settings, identified low-risk reactions as those occurring over 1 year prior with mild symptoms (eg, benign rash without mucosal involvement). Among these low-risk patients, 97.1% tolerated oral challenge without any cases of anaphylaxis.
Discussion
This study examines the characteristics of participants undergoing oral challenge to delabel low-risk penicillin allergies, the interventions used, and post-exposure adverse reactions. The findings support oral challenge as an effective, safe, and practical strategy, with low adverse reaction rates and high success in delabeling. In this adult population, the mean age was 56 years with a notable female predominance (61%), reflecting the higher reported prevalence of penicillin allergies in women. Ethnicity data were inconsistently reported, with most participants identifying as White and all studies were conducted in higher income countries. This underscores the need to fully ascertain participant ethnicity and include more diverse populations in future studies to ensure equitable access to care and improve the generalizability of rash assessments during penicillin oral challenge interventions across all skin tones, particularly as cutaneous reactions are the most reported reaction to oral challenge. Furthermore, availability of resources to conduct oral challenge interventions in low and middle-income countries requires further study to determine the best strategies to implement these interventions in resources limited settings.
Amoxicillin was the preferred agent for oral challenge, likely due to its accessibility, tolerability, and safety profile. Most studies used single-dose or short-duration challenges at doses of 250 or 500 mg, with less frequent use of graded challenges or other penicillins. Based on the ease of administration of a single dose of amoxicillin and its association with successful delabeling, the use of a single 250 mg dose is reasonable for assessing tolerability. Variability in dosing and provider roles suggests the need for standardized protocols. Nurses and pharmacists were commonly involved in monitoring, demonstrating the feasibility of non-physician healthcare professionals in scaling delabeling interventions.
No clear link emerged between the agent or dose used in oral challenges and the risk of reactions. However, based on tolerability information on follow-up with patients who were prescribed penicillins after oral challenge, there may be a correlation between the use of amoxicillin-clavulanate and intolerance or mild hypersensitivity reactions. 14 This is supported by a study that used spontaneous reporting of adverse drug reactions in Italy by Salvo et al, 36 which found higher rates of gastrointestinal (13% vs 7%), hepatic (4% vs 1%), and hematologic (2% vs 1%) reactions with amoxicillin-clavulanate compared to amoxicillin, despite skin reactions being more common with amoxicillin (82% vs 76%). Furthermore, a study and case report show a correlation between oral challenge with amoxicillin/clavulanate and the development of hepatotoxicity and pancreatitis.37,38 Therefore, amoxicillin-clavulanate is a beta-lactam characterized by a unique association with hepatopancreatic adverse drug reactions.
The absence of severe reactions supports oral challenge as a first-line strategy, even in vulnerable populations such as pregnant individuals who are assessed as having a low-risk penicillin allergy. This was supported by a cohort study conducted by Wong et al, 39 which evaluated penicillin oral challenges during the antepartum and postpartum phases and found the approach to be safe and effective in the antepartum phase. The authors also noted that deferring oral challenges to the postpartum period poses a significant barrier to penicillin allergy delabeling. 39 However, even though oral challenge was relatively well-tolerated across the studies included in this analysis, follow-up is important as mild delayed reactions such as maculopapular rashes were reported. Studies with extended follow-up indicated some relabeling, emphasizing the importance of patient education and robust follow-up to sustain benefits and understand long-term sequalae associated with delabeling interventions. One study suggested a correlation between the number of reported allergies and reaction likelihood, indicating potential for risk stratification. 19 Timing since the index reaction and a benign nature of the reaction were also important factors, supporting their inclusion in future risk assessment tools. 10 An area for focus in future study designs would be to consider potential predictors of hypersensitivity reactions as covariates in regression models across all studies to better understand factors that may impact both the presence of an allergy label and the true incidence of reaction.
This research has some limitations. Inconsistent reporting of demographics, comorbidities, and equity factors, along with heterogeneous challenge protocols, complicates cross-study comparisons. Many studies lacked standardized definitions for allergic versus non-allergic reactions, and long-term outcomes were often underreported. The inclusion of participants with low and high-risk baseline reactions without linkage to eligibility for oral challenge leads to the inability to clearly interpret how reported baseline reactions correlate with outcomes. Also, the underrepresentation of vulnerable and diverse populations limits generalizability. Most studies relied on short-term follow-up and patient-reported data, which are prone to recall bias. Further, the research was largely conducted in high-income countries with few randomized controlled trials, highlighting the need for more rigorous, standardized, and equity-informed studies in this field.
Conclusion
This descriptive analysis underscores the critical role of oral challenge interventions in improving antibiotic stewardship by safely delabeling low-risk penicillin allergies. Most oral challenge interventions involve a single 250 mg dose of amoxicillin, with most participants tolerating the challenge. Reactions were mild and primarily cutaneous in nature, with few delayed reactions having occurred. Implementing standardized, evidence-based protocols in diverse healthcare settings, including primary care and community pharmacies, is possible and has the potential to enhance accessibility to penicillin allergy assessment interventions.
Footnotes
Data Availability Statement
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work has not received financial support. Mira Maximos has received a CIHR Doctoral award.
