Abstract
Key Points
Colorectal surgery (CORS) patients are at the highest post-operative venous thromboembolism (VTE) risk of any general surgery population and are also at an increased risk for post-operative bleeding.
Standard compared to reduced dose enoxaparin for VTE prophylaxis for CORS patients under 50 kg demonstrated increased bleeding and thrombotic event rates.
This study aims to update the institutional anticoagulation management guidelines in addition to provider order sets to reflect the option for using reduced dose enoxaparin post-operatively for low body weight CORS patients.
Introduction
Post-operative venous thromboembolism (VTE) is defined by The Centers for Medicare and Medicaid Services (CMS) as an occurrence of any deep vein thrombosis (DVT) or pulmonary embolism (PE) following a surgical procedure in the same encounter. It is well established that surgical complications are associated with post-operative VTE events and in addition contribute to an increased cost of hospitalization and length of stay. In 2011, it was estimated that post-operative VTE events increased the cost of hospital admission related to the surgical procedure by approximately $5000. 1 In a report from 2018, post-operative bleeding increased the cost of hospital admission related to the surgical procedure by almost $17 000. 2 Estimates from a 2002 report demonstrated that post-operative VTE events accounted for nearly 25% of all reported VTE events in the United States annually. 3
Colorectal surgery (CORS) patients have the highest rates of VTE of any general surgical specialty, a 30% risk compared to a 20% risk, respectively. 4 Oncology patients are also known to have an increased risk of VTE that is nearly twice as high as patients without malignancies and this risk increases when having extensive abdominal or pelvic surgeries. 5 Ninety day VTE rates have previously been reported in The American Society of Colon and Rectal Surgeons Clinical Practice Guideline for the Prevention of Venous Thromboembolic Disease in Colorectal Surgery as 2.2% with 39% of events occurring following the procedural event discharge. 4 In addition, there is also data to suggest that CORS patients, especially those undergoing procedures related to earlier stages of cancer, are also at an increased risk for bleeding post-operatively. 6 Post-operative bleeding may also be a serious and potentially fatal complication with a bleeding incidence ranging from 1% to 14%. 2
Several studies have investigated weight based enoxaparin prophylactic dosing in other surgical patient populations, such as trauma, bariatric, and orthopedic surgery. Numerous institutions have protocols that follow guideline directed therapy regarding adjustment of enoxaparin based on indication, medical/surgical patient population, and body mass index (BMI) or weight for obesity. Trials conducted have evaluated weight based enoxaparin protocol directed therapy in comparison to guideline adherence in the trauma population, but presently the data supporting weight based prophylactic enoxaparin dosing primarily focuses on dosing adjustments in surgical patients with obesity rather than underweight surgical patients. 7
Additional dosing strategies based on the utilization of anti-factor Xa levels for dosing guidance in the obese surgical population are also present. Shelkrot et al determined that patients with obesity undergoing bariatric surgery may require higher standard doses of enoxaparin to achieve therapeutic anti-factor Xa levels and lower the risk of post-operative VTE occurrence. 8 Al Otaib et al conducted a prospective study using weight based enoxaparin dosing in morbidly obese surgical patients and monitored anti-factor Xa levels. This study determined that the weight based doses resulted more often with therapeutic anti-factor Xa levels without an increase in VTE events while minimizing bleeding adverse events. 9
A limited number of studies were available to date that examined the specific surgical patient population of interest which included CORS patients, but did not compare dosing strategies focused on underweight individuals in regards to both bleeding and thrombotic outcomes. A prospective, observational study conducted by Kramme et al demonstrated that oncology patients undergoing open or laparoscopic abdominal surgery receiving enoxaparin dose adjustments based on peak anti-factor Xa levels compared to fixed dose enoxaparin 40 mg daily demonstrated a lower rate of in-hospital VTE events without increasing the rate of bleeding events. 10 Lastly, a prospective, non-randomized trial designed by Pannucci et al assessing anti-factor Xa levels for CORS patients of all body weights identified that weight was inversely proportional to anti-factor Xa levels and more breakthrough VTE events occurred from inadequate prophylaxis with the fixed standard dose enoxaparin 40 mg every 24 hours compared to real time adjustments based on anti-factor Xa levels.11,12 Routine anti-factor Xa level monitoring is not conducted within the enterprise and therefore not collected during this study as it is a send out lab which may result in delayed dosing adjustments for the studied population.
In contrast to the body of literature supporting weight or anti-factor Xa based prophylactic enoxaparin dosing in several surgical specialties post-operatively, the data in CORS patients based on actual body weight is lacking, especially when considering dosage reductions for low body weight individuals due to bleeding and thrombotic risk. Although, enoxaparin is commonly used for VTE prophylaxis at a standard dose of 40 mg subcutaneously (SQ) every 24 hours for medical and surgical patients with normal body weights, at this time, major clinical guidelines such as the CHEST guidelines do not provide recommendations on optimal dosing strategies for the underweight, surgical population requiring VTE prophylaxis with LMWH. 13 The manufacturer also recommends clinical monitoring when using enoxaparin for VTE prophylaxis for low body weight patients, but does not include specific recommendations for dosage reductions in underweight patients. 14
This study aimed to evaluate the pattern surgical providers within the enterprise had recognized for patients with an actual body weight less than 50 kg being at a higher risk of post-operative bleeding complications compared to a similar patient population weighing equal to or greater than 50 kg. Current practice for post-operative CORS patients within the enterprise is enoxaparin 40 mg SQ every 24 hours. Enoxaparin dose reductions to 30 mg every 24 hours are reserved for patients with a creatinine clearance (based on the Cockcroft-Gault equation) less than 30 mL/min. Although there is literature describing the bleeding and thrombotic risk among patients who receive post-operative VTE prophylaxis, the literature is sparse when evaluating bleeding and thrombotic risks associated with VTE prophylaxis in CORS patients with reduced actual body weight. The purpose of this study is to assess both the safety and efficacy of standard versus reduced dose enoxaparin prophylaxis in CORS patients weighing less than 50 kg without any other indications for reduced enoxaparin dosing.
Methods
This study was a multicenter, retrospective chart review. It was conducted within an institutional enterprise in Northeast Ohio that encompassed a large, academic medical center in combination with 14 affiliated regional, teaching hospitals. Data was collected on post-operative CORS patients over a 10-year period from January 1, 2011 through January 1, 2021. CORS patients were identified via the electronic medical record (EPIC®) using International Classification of Diseases, 9th and 10th codes (ICD-9 and ICD-10) and Current Procedural Terminology (CPT) CORS procedural codes for both open and laparoscopic colorectal surgical procedures.
Data was collected by a single reviewer using a standardized, electronic data collection form and was stored in a password protected Excel document. The STROBE case-control checklist was used when writing the report. 15 The data underlying this article are available in the article. This study was approved by the institutional review board and met the guidelines of the responsible governmental agency. Descriptive statistics were performed using mean ± standard deviation (SD) or median [interquartile range, IQR] for continuous variables and frequency and percentages for categorical variables. All descriptive statistical analyses were performed using SAS software (version 9.4, Cary, NC). This study was performed as a longitudinal pharmacy resident research project over the course of 1 year and researchers involved in this study were caregivers within the institution.
Since this was a retrospective chart review, evaluation and severity of post-operative bleeding and VTE events was extracted from provider progress notes in the electronic medical record. Patients were not scored for VTE risk using validated scoring methods such as the Padua Prediction Score or Caprini Score as a VTE estimation tool. The notes did not provide measurements on post-operative blood loss per volume if there were bleeding complications nor use a validated scoring tool such as the Bleeding Academic Research Consortium (BARC) or Thrombolysis in Myocardial Infarction (TIMI) criteria for bleeding event data collection. Instead, data on intra-operative blood loss in milliliters (mL) was recorded in progress notes, therefore there were no severity classifications associated with bleeding events reported post-operatively in this study. Bleeds are instead reported by type as described in the notes, drop in hemoglobin, as well as, by medical or surgical intervention. Evaluation of bleeding or VTE events was only related to the inpatient admission for the surgical procedure.
The primary endpoint evaluated the incidence of post-operative bleeding in CORS patients weighing less than 50 kg who had received post-operative VTE prophylaxis with enoxaparin standard dose 40 mg SQ every 24 hours compared to reduced dose prophylaxis with 30 mg SQ every 24 hours. The secondary endpoint evaluated post-operative thrombotic event rates in the same population receiving standard versus reduced dose enoxaparin. The investigators hypothesized that patients receiving the standard dose compared to reduced dose enoxaparin would demonstrate an increased rate of post-operative bleeding events without a notable difference in regards to thrombotic event rates. This hypothesis was generated based on the pattern surgical providers had noticed within the enterprise. The hypothesis was supported by prior literature proposing that over-anticoagulation may occur with standard prophylactic LMWH dosing in low body weight patients based on anti-factor Xa level monitoring and that bleeding risk may also increase with advanced age and reduced body weight.16,17
Results
Patients were included if they were 18 years of age or older at the time of CORS procedure, weighed less than 50 kg based on actual body weight, and received at least 1 dose of enoxaparin 30 mg or 40 mg SQ post-operatively. Figure 1 depicts patient enrollment. Initially 190 patients were identified; meeting inclusion criteria. Patients were excluded from the trial if they had an actual body weight greater than or equal to 50 kg, had a prior diagnosis of pre-existing coagulopathies, had used antiplatelet or anticoagulant agents pre-operatively prior to admission, had experienced intra-operative bleeding greater than 400 mL, had a creatinine clearance less than 30 mL/min, were less than 18 years of age, were pregnant, had a supratherapeutic INR with no prior anticoagulant use, or had received doses of enoxaparin that differed from the investigational doses. Overall, 44 patients were excluded, therefore 146 total patients were included in the study.
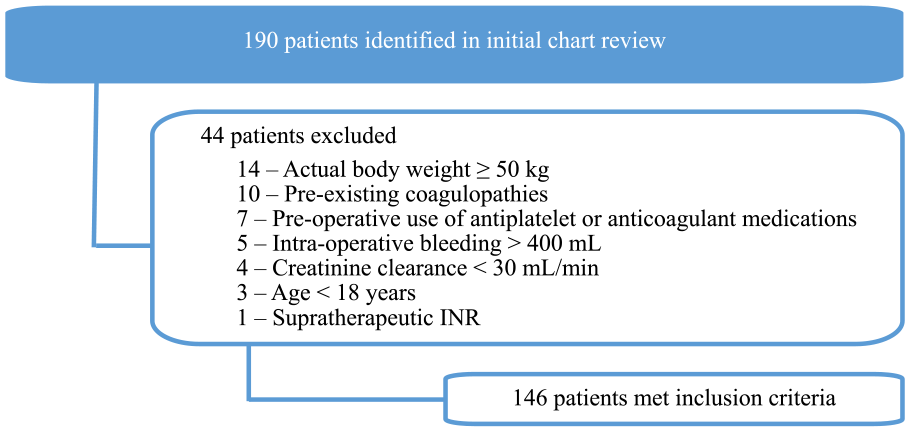
Exclusion criteria for study population.
Table 1 depicts the demographic information for the study population. The average age of the patients was 58 years old [36.0, 67.0]. The majority of patients were female (91.8%). Patients had an actual body weight of 46.7 kg [43.5, 49.0]. Patients included did not have renal impairment and were not on renal replacement therapy during the study period.
Baseline Characteristics of Study Population.

Note. Statistics presented as Mean ±SD, Median [P25, P75], N (column %).
Of the total population (n = 146) included in this study, 129 (88.4%) received enoxaparin 40 mg SQ every 24 hours and 17 (11.6%) received enoxaparin 30 mg SQ every 24 hours post-operatively. Table 2 represents the outcomes for each group evaluated. For the purpose of this trial, clinically significant major VTE events were any confirmed VTEs unrelated to a catheter occlusion. Overall, there were 9 reported bleeding events (7.0%) and 2 major, clinically significant VTE events (1.6%) all within the 40 mg group. In the 30 mg group, there were no bleeding events and the only thrombotic event reported was an occluded peripherally inserted central catheter (PICC) that was cleared with a medical intervention using a single dose of 2 mg intravenous alteplase.
Descriptive Statistical Analysis of Outcomes Data for Study Population.
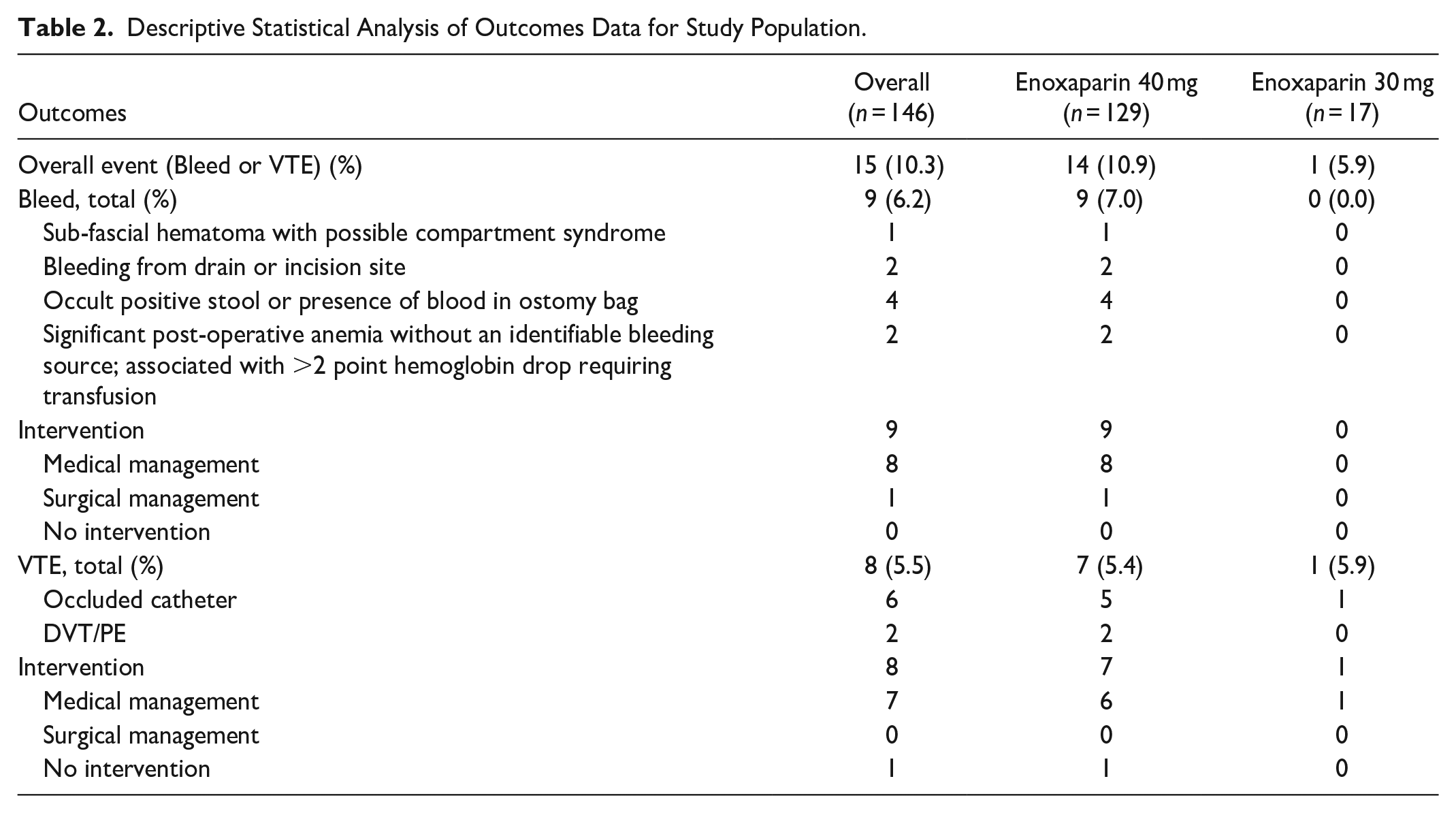
Bleeding events in the standard enoxaparin group included 1 sub-fascial hematoma with possible compartment syndrome, 2 bleeding events from the drains or incision sites, 4 patients with occult blood positive stool or presence of blood in ostomy bag, and 2 patients with significant post-operative anemia without an identifiable bleeding source and associated with greater than a 2 point hemoglobin drop requiring a packed red blood cell (pRBC) transfusion. In the reduced enoxaparin dose group no medical or surgical interventions were performed based on event rates compared to 89.0% requiring medical interventions and 11.0% requiring surgical interventions for bleeding events in the standard enoxaparin group. Medical interventions included holding or discontinuation of enoxaparin, transfusion with 1 to 2 units of pRBCs, administration of injectable or oral phytonadione, or a change to SQ heparin after initially holding enoxaparin 1 to 3 days. Surgical intervention included endoscopic clipping or a return to the operating room for managing bleeding complications.
In the standard enoxaparin group there were 7 thromboembolic events which included 2 DVTs and 5 clotted catheters requiring 1 to 4 doses of alteplase 2 mg intravenously. For the 2 confirmed DVTs in the standard enoxaparin group, 50.0% required medical intervention and were therefore changed to an unfractionated heparin infusion for the clinically significant VTE which was classified as a subclavian, axillary, and brachial vein DVT. The remaining 50.0% received no medical or surgical intervention for the VTE event defined as a non-occlusive superior mesenteric vein thrombus and non-occlusive sub-segmental portal vein thrombus.
Discussion
Post-operative VTE and bleeding events are associated with increased costs of hospitalizations, length of stay, and may be the initial insult in a cascade of complications impeding surgical recovery and leading to poor outcomes. 2 Since the majority of CORS patients require multiple procedures, minimizing costs and complications of each procedure is of considerable importance. Based on the established guidelines and protocols followed by the institution, there are recommendations for reduced dose enoxaparin regimens in medical patients without indications other than an actual body weight less than 50 kg but there are no similar recommendations for surgical patients weighing less than 50 kg at this time. There is a need for the development of standardized, evidence-based guideline directed dosing for underweight surgical patients that supports clinical decision making by providers and pharmacists.
This research focus was hypothesis generating; that there may be an increased bleeding risk with standard dose enoxaparin regimens in patients weighing less than 50 kg who had no other qualifier for reduced dose enoxaparin post-operative VTE prophylaxis. The question of optimal dosing of post-operative enoxaparin prophylaxis with standardized versus weight based dosing has yet to be established. Therefore, this study attempted to evaluate both safety and efficacy of these 2 VTE prophylactic dosing strategies. Studies utilizing anti-factor Xa levels and measuring rates of bleeding and VTE events have been tested in another surgical populations like plastic and reconstructive patients which found that weight based dosing using anti-factor Xa levels minimized over-anticoagulation and bleeding events and was more effective with regards to under-anticoagulation and thrombotic events, but guideline directed therapy and institutional protocols have yet to universally adopt this approach. 18
While this study, investigated a novel low body weight surgical patient population, limitations were present due to the nature of the study design being a 1 year longitudinal pharmacy resident research investigation. Limitations of this study include the retrospective chart review trial design, inability to meet power based on the sample size with an unequal distribution of patients receiving the standard regimen (88.4%) compared to those receiving the reduced dose regimen (11.6%), and the omission of data collection on additional confounding factors. Confounding factors include the length of surgery, co-morbidities, bleeding and thrombotic baseline risk classification, lack of validated, standardized outcome assessment tools for bleeding and thrombotic events, omission of provider rationale or categorization for choice of enoxaparin dose prescribed, inpatient medication interactions for increased bleeding or thrombotic adverse effects, smoking status, mobility status, anti-factor Xa levels, and the time to administration of first post-operative dose of enoxaparin which may introduce bias and variability in determining the primary and secondary endpoints. Manual data collection of bleeding and thrombotic events was limited to provider progress note documentation rather than use of a validated scoring tool. The descriptive nature of the event introduces subjectivity and potential variability in event reporting. Also, race and ethnicity were not collected in this study and since there were more females to males included the external validity of this trial primarily is applicable to female patients.
Alternatively, since data involving low body weight CORS patients is extremely limited, a major strength of this study was the specific patient population of interest evaluated over a 10-year period within a multicenter enterprise. This study also reported a 7.0% bleeding event rate and 5.4% thrombotic event rate in patients weighing less than 50 kg and receiving enoxaparin 40 mg SQ every 24 hours after surgery compared to no bleeding events, in addition to 1 thrombotic event, in the same patient population receiving a reduced dose of enoxaparin 30 mg SQ every 24 hour post-operatively.
Despite the fact that there is an abundance of literature evaluating safety and efficacy of VTE prophylaxis among different pharmacologic regimens for surgical patients and dosage adjustments based on weight, BMI, and anti-factor Xa levels that are supported primarily in the obese surgical population, there is a lack of literature comparing different prophylactic dosing regimens based on actual body weight for enoxaparin in this subset of low body weight CORS patients. Although this study had a small sample size, it was similar in sample size to other studies of this nature that assessed bleeding and thrombotic risk with enoxaparin dosing for low body weight patients.17,19 While this trial noted increased bleeding and VTE events with standard compared to reduced dose enoxaparin, further studies should be conducted evaluating the same outcome in a similar population of patients on a larger scale since statistically and clinically significant conclusions cannot be made based on this sample size alone. Future considerations in these trials include designing a study that is prospective, has an adequate sample size to achieve power, and accounts for additional confounding variables that could impact bleeding and VTE risk. This hypothesis generating study highlights the need for future systematic reviews and meta-analyses on this topic for underweight surgical patients in order to apply evidenced based medicine to practice, reduce medication and surgical complications post-operatively, and improve patient care outcomes in this CORS population.
Conclusion
To our knowledge, this was the first study to investigate the incidence of bleeding and thrombotic events associated with VTE prophylaxis using standard versus reduced dose enoxaparin in low body weight colorectal surgery patients. This trial demonstrated that the current recommended dosing regimen of enoxaparin 40 mg SQ every 24 hours for VTE prophylaxis in post-operative CORS patients weighing less than 50 kg had higher rates of bleeding and thrombotic events requiring intervention in comparison to a post-operative reduced dose regimen of enoxaparin 30 mg SQ every 24 hours. Findings from this research are aimed at updating the institutional anticoagulation management guidelines in addition to provider order sets to reflect the option for using reduced dose enoxaparin post-operatively for low body weight CORS patients. It also aims at providing interdisciplinary education to clinical pharmacists, nurses, colorectal surgeons, surgical residents, and affiliated colorectal surgery midlevel prescribers. These findings may offer an opportunity for reducing post-operative risk and complications including bleeding and VTE events in this unique low body weight CORS patient population. Additionally, these clinical findings may encourage future investigation, development and implementation of evidenced-based medicine changes into practice, and improvement in patient care.
Footnotes
Acknowledgements
The authors thank Kathryn Willner, MSN, APRN.CNP, clinical colorectal surgery consultant, and Lu Wang, MS, statistician for the contributions to this research study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
