Abstract
Keywords
Introduction
The Institute for Safe Medication Practices (ISMP) publishes a list of high-alert medications in acute care settings, defined as medications that bear a heightened risk of causing significant patient harm when they are used in error. 1 Errors are not necessarily more common with these medications, however the consequences of errors are more likely to contribute to significant patient harm.1,2 Although this list serves as a reference for health-systems, the Joint Commission medication management standard MM.01.01.03 requires that hospitals develop an organizational list based on internal utilization and medication error data. 3
Methods of retrospectively and prospectively identifying hospital-specific high-alert medications have been described elsewhere.4-7 However, development of this list has been described as “relatively useless” without also identifying anticipated failures and implementing risk-reduction strategies to prevent, detect, or mitigate harm with each agent. 8 Every 2 years beginning in 2014, ISMP has issued “Targeted Medication Safety Best Practices for Hospitals” to identify and encourage adoption of consensus-based best practices that have been reviewed by an external Expert Advisory Panel and approved by the ISMP Board of Directors. 9 Each best practice addresses known medication safety issues that continue to cause fatal and harmful errors. 9 In 2022, ISMP introduced a new targeted medication safety best practice (TMSBP #19) to layer numerous strategies throughout the medication use process to improve the safety with high-alert medications. 9 The following 6 interventions to manage risk were recommended 9 :
Outline a robust set of processes for each high-alert medication
Ensure strategies are layered across each stage of the medication use process and apply to prescribers, pharmacists, nurses, etc.
Avoid reliance on low-leverage error prevention strategies
Limit the use of independent double checks to select high-alert medications
Review internal and external sources to routinely assess for risk
Establish outcome and process measures to monitor safety and effectiveness
Full compliance with ISMP’s new TMSBP #19 may prove difficult to fully comply with for many institutions. Despite long-standing recognition of the need to improve the safety with high-alert medications, a survey of 188 hospitals by ISMP in 2022 highlighted that only 64% of respondents reported full compliance with the recommendation to “outline a robust set of processes for each high-alert medication.” 10 Surveyed hospitals identified the following barriers to full compliance: significant time required to assess each high-alert medication, medication technology costs/limitations, lack of leadership support, and overreliance on internal voluntary reporting system to assess and monitor medication safety. 10 Additionally, 49% of respondents reported significant barriers to system change leading to an over-reliance on low-leverage strategies to prevent errors, such as education which has been described as “predictably disappointing.”10,11 ISMP’s hierarchy of effectiveness of risk reduction strategies defines low-leverage strategies as those which are easiest to implement, but least effective (eg, education, rules, and policies). 11 Medium-leverage strategies are harder to implement, but may need periodic reinforcement to be effective (eg, redundancies, checklists, standardization, warnings, and alerts). 11 Hardest to implement but most effective, high-leverage strategies improve system reliability by “designing out” hazards (eg, forcing functions, automation, barriers, and fail-safes). 11
Tracking observed institutional errors, identifying anticipated errors from external reporting, and cataloging implemented institutional system changes can be resource intensive. However, without an organized approach to layering system change strategies and tracking system change, institutions risk ongoing patient harm due to a lack of recognition of gaps in system support. Additionally, best practices established by unit-based or service-based system change to limit errors with a single agent or class of agents may not result in optimizing system-wide spread due to a lack of awareness. The objective of this study was to describe the development of a comprehensive framework of observed and anticipated errors coupled with safeguarding strategies for identified high-alert medications developed by a single academic medical center.
Methods
The Ohio State University Wexner Medical Center (OSUWMC) is an 1882-bed academic medical center composed of 7 hospitals in central Ohio including several full-service teaching hospitals and a freestanding cancer hospital. OSUWMC utilizes Epic Systems Corporation (Verona, WI) for its electronic health record system and has implemented bidirectional infusion pump interoperability. At the OSUWMC, adverse drug reactions and medication errors are monitored via a patient safety reporting system and severity assigned via the National Coordinating Council for Medication Error Reporting and Prevention (NCC-MERP) approach. Organizational high-alert medications take into consideration both the ISMP list and internal medication-related harm events (NCC-MERP E-I). NCC-MERP defines Category E-I as errors that reached the patient and may have contributed to or resulted in temporary or permanent patient harm or patient death. 12
At OSUWMC, medications and medication classes are designated as high-alert if the following criteria are met: (a) inclusion on ISMP List of High-Alert Medications in Acute Care Settings AND ≥1 reported medication-related event classified as NCC-MERP E-I in previous 4 years at OSUWMC; OR (b) ≥3 reported medication-related events classified as NCC-MERP E-I in previous 4 years at OSUWMC; OR (c) consensus of the Medication Safety Pharmacy & Therapeutics Subcommittee if internal vulnerabilities identified during routine review of external medication safety alerts. A list of the 19 medication classes designated as high-alert at OSUWMC can be found in Table 1. Medications are evaluated for addition to the high-alert medication list when considered for formulary addition. The high-alert medication list is reevaluated for addition/deletion of medications with any updates to the ISMP List of High-Alert Medications, if trends or vulnerabilities are identified from internal or external reporting of medication-related events, and every 2 years at minimum.
Organizational List of High-Alert Medications.

Note. IV = intravenous; SQ = subcutaneous.
Development of the comprehensive framework was initiated by first conducting a comprehensive literature search of each institutionally designated high-alert agent to identify externally reported errors. Additionally, internal voluntary reports were reviewed. Anticipated or frequently reported errors were categorized by error type based on the definitions for common cause errors defined in Table 2. Next, error reduction strategies already implemented institutionally were identified and categorized. This was done by: (1) compiling a list of organizational error-reduction strategies implemented for all medications whenever possible (Table 3); (2) thoroughly reviewing each individual high-alert medication and medication class by medication safety specialists and specialty practice pharmacists to identify all implemented safeguarding strategies to address each identified observed/anticipated error. The finalized framework of safeguarding strategies to address errors with organizational high-alert medications was embedded into our high-alert medication policy that is available to all staff.
Definitions of Common Cause Error Types.
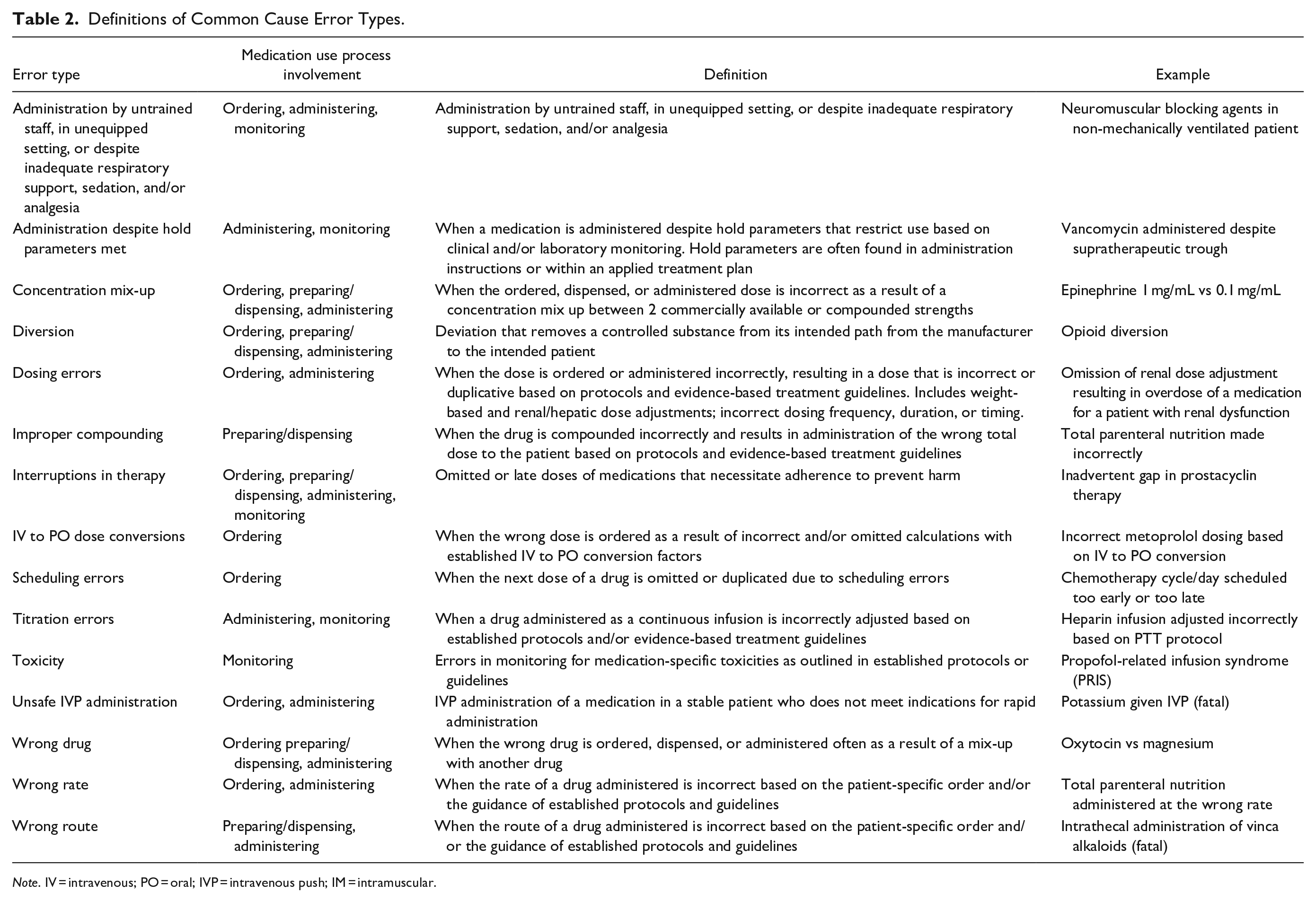
Note. IV = intravenous; PO = oral; IVP = intravenous push; IM = intramuscular.
Global Safeguarding Strategies Implemented for All Medications at OSUWMC.

Results
The review of externally and internally reported errors identified 101 observed/anticipated common cause errors across the 19 high-alert medication classes (median 5 error types per medication class, interquartile range 3-6). The top 8 errors associated most frequently across all medication classes were:
Concentration mix-ups
Dosing errors
Titration errors
Improper compounding
Toxicity
Interruptions in therapy
Administration despite hold parameters met
Administration by untrained staff, in unequipped setting, or despite inadequate respiratory support, sedation, and/or analgesia.
Safeguarding strategies specific to high-alert medications were identified in the following domains:
Separate or sequestered storage
Restricted ordering (eg, formulary restriction, restricted to select providers and/or consult services)
Active alerts (eg, best practice alerts, infusion pump clinical advisories)
Dispensing in patient-specific dosing, unit of use, or unit-dose packaging (eg, manufacturer premix or institutional premade products)
Dispensing from pharmacy only; auxiliary labeling
Level of care restriction (eg, intensive care vs general non-acute care)
Required monitoring (eg, telemetry, pharmacy-specific clinical scoring tools)
Independent double checks
Certification/privileging of staff
Specific guidelines for use/monitoring (eg, order sets and order panels, institutional policies, standard administration times)
Other/miscellaneous.
Identified safeguarding strategies were cumulative and represented inventions that had been implemented over several years (eg, at formulary addition, in response to an observed error, with a technology or practice advancement) and across many stakeholders (eg, information technology, pharmacy operations, pharmacy and therapeutics committee).
Identification of the observed/anticipated errors and the associated safeguarding strategies facilitated the development of a comprehensive tool which compiles all the safeguarding strategies in place for each individual high-alert medication or medication class. An example of this comprehensive tool for 1 medication class, prostacyclins, is shown in Table 4. Furthermore, this tool illustrates a visual framework of safeguarding strategies in place for each common cause error type. An example of this comprehensive framework for effective safeguarding strategies for 1 error type, improper compounding, is shown in Table 5.
Safeguarding Strategies to Address Anticipated Errors With High-Alert Medications: Prostacylins.

Note. ◆ = implemented safeguarding strategy; ♢ = recommended safeguarding strategy, not yet fully implemented
Framework of Safeguarding Strategies to Address Common Cause Errors: Improper Compounding.

Note. ◆ = implemented safeguarding strategy; ♢ = recommended safeguarding strategy, not yet fully implemented; IV = intravenous; PCA = patient controlled analgesia; STEMI = ST-elevation myocardial infarction.
Discussion
This comprehensive framework of safeguarding strategies to address anticipated errors with organizational high-alert medications is multifunctional. First, this framework can be leveraged by health-systems to take inventory of the current state of error-reduction strategies in place.
Safeguards are often implemented over several years and across many stakeholders (eg, information technology, pharmacy operations, pharmacy and therapeutics committee); therefore, comprehensively summarizing the safeguards can be a useful exercise for health-systems. Next, the framework can be used to prospectively identify gaps or opportunities to implement additional safeguarding strategies. For example, if a safeguarding strategy for an error type is in place for all high-alert medications except one, the health-system could evaluate if it would be feasible and effective to implement the safeguard for that high-alert medication. Additionally, the framework can be utilized when performing investigation of medication-related errors or root cause analyses to determine the safeguards that failed and/or were ineffective. Lastly, although this is just one approach to meeting The Joint Commission standard, it fulfills the requirement of following an organizational process for managing high-alert medications.
Simply having a high-alert medication list without associated risk-reduction strategies has been described as “relatively useless.” 8 Additionally, risk-reduction strategies should go beyond low-leverage risk-reduction strategies that are educational or informational due to their limited efficacy. 11 The authors believe the power of the proposed framework is that it includes a wide variety of low (eg, policies, auxiliary labels, competency requirements to certify/privilege staff), medium (eg, active alerts, independent double checks), and high-leverage strategies (eg, automation, ready-to-administer preparations to safeguard against compounding errors) that address multiple vulnerabilities throughout the medication use process. Furthermore, the safeguards are targeted across a wide range of healthcare practitioners including providers, pharmacists, and nurses. This multidisciplinary approach serves to effectively address anticipated errors at multiple steps in the medication use process as well as increase practitioner knowledge of high-alert medications and their associated safeguards. These characteristics demonstrate alignment with ISMP’s TMSBP #19 by layering risk-reduction strategies for high-alert medications throughout the medication use process, utilizing a multidisciplinary approach, and bundling low-leverage strategies with mid- and high-leverage strategies to prevent errors. 9
Cohen 13 previously described 3 guiding principles to safeguard the use of high-alert drugs: (1) reducing or eliminating the possibility of errors, (2) making errors visible, and (3) minimizing the consequences of errors. Furthermore, a framework was proposed for safeguarding the use of high-alert drugs which included the following strategies: fail-safes and forcing functions, constraints, externalize or centralize error-prone processes, access to information, standardize and simplify, differentiate/use warnings and reminders, redundancies/independent double checks, monitor patients and respond to drug effects, and failure mode and effects analysis. 13 When applying this framework to current practice at OSUWMC, the authors identified that many of the recommended safeguards had become standard of care for all medications (eg, use of flow control pumps for continuous infusions, computerized provider order entry); therefore, the authors sought to adapt the framework to one that captured safeguards beyond the standard of care for all medications. As a result, the proposed framework was designed to capture all the safeguarding strategies implemented specifically to address the observed/anticipated failures identified with each high-alert medication at an academic medical center with a fully integrated electronic health record and smart pump interoperability.
Development of this framework for safeguarding high-alert medications at a large, academic medical center was not without its challenges. First, while no additional labor resources were required at OSUWMC, an initial and significant investment of the medication safety team’s time was required to review internal and external error reports, identify observed/anticipated errors with each high-alert medication class, develop the proposed framework, and catalog all implemented institutional system safeguarding strategies. Specifically at OSUWMC, this process was completed longitudinally over approximately 1 year. Institutions facing resource constraints could consider leveraging learner resources (ie, residents and interns) to assist while also providing a valuable learning opportunity. Next, as the medication use process is ever-changing with newly approved drug classes/formulations and technology, maintaining an up-to-date framework will present a challenge to any institution. The authors suggest leveraging existing infrastructure, such as pharmacy and therapeutics committees and establishing a timeline for routine policy review (eg, annually). Finally, cataloging all institutional system safeguarding strategies across a wide range of medication classes for an entire health-system was a significant challenge. To accomplish this, input was solicited from a wide range of pharmacy specialists and leaders in operational, clinical, medication safety, and information technology roles. Therefore, the finalized framework comprehensively represents all locations across the health-system and also serves as a resource embedded in our high-alert medication policy that is available to all staff.
Although the implemented safeguards and common cause errors will vary between institutions, the proposed framework for identifying observed/anticipated errors for high-alert medications and their associated safeguards can be utilized by all health-systems. Notably, the step of identifying interventions beyond those implemented for all medications (eg, dose-error reduction software utilization within smart pumps at OSUWMC) will vary between institutions and may not be generalizable. Furthermore, the definition for high-alert medications at OSUWMC can be applied to and/or adapted by health-systems looking to improve their processes for identifying organizational high-alert medications. Ultimately, more research is needed to determine how to implement error-reduction strategies most effectively and judiciously. Categorizing observed/anticipated errors into common cause error types provides a visual framework that begins to define the playbook for error-specific safeguarding strategies. The authors envision leveraging this framework as a checklist of safeguards to implement when a new high-alert medication is added to formulary and/or a new observed/anticipated error is identified for an existing formulary medication.
Conclusion
A comprehensive framework of safeguarding strategies to address anticipated errors with organizational high-alert medications is proposed. Although individual safeguards are institution-specific, the framework can be leveraged by all hospitals in order to take inventory of error-reduction strategies and prospectively identify gaps to address common cause errors.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
