Abstract
Systemic lupus erythematosus is defined as an autoimmune and inflammatory disease that is distinguished by the involvement of diversified multisystem and a persistent course with unpredictable flares. Immune complex-mediated glomerulonephritis is the main presentation of renal involvement and is commonly regarded as lupus nephritis (LN). Although the long-term renal prognosis has been considerably improved, a significant morbidity and mortality is still experienced by children and adolescents with LN. Furthermore, present literature is insufficient with regard to data related to pediatrics, resulting in clinicians to depend entirely on therapeutic approaches employed for adults. Treatment protocols have been produced by various international groups that recommend an intense duration of immunosuppression to stop the process of acute renal inflammation, with subsequent maintenance therapy and close monitoring for disease improvement and rapid assessment of disease flares. Prevalence of renal remission is suboptimal indicating only 40% to 60% of patients attaining complete remission. The rate of chronic kidney disease (CKD) stage V has been observed to be at least 15% and the presence of LN has an association with high mortality rates. In ascertained renal failure, renal transplantation is implied to be the most favorable modality of renal replacement therapy in this specific patient group. This review aims to describe childhood-onset LN and provide an overview on the pathogenesis, therapeutic approaches, emerging therapies and present renal outcomes for this disease with the objective of recognizing the key features that might aid in reducing the risk of long-term CKD.
Keywords
Introduction
Systemic lupus erythematosus (SLE) is defined as an autoimmune and inflammatory disease that is distinguished by the involvement of diversified multisystem and a persistent course with unpredictable flares. Although it affects each organ system, blood cells, the central nervous system, joints, skin, and renal involvements are customary. 1 Childhood-onset systematic lupus erythematous (cSLE), occurring at less than 18 years of age, comprised 10% to 20 % of all cases. The severity of cSLE is higher than adult SLE with regard to both the disease flares and aggressive onset. 2
Immune complex (IC)-mediated glomerulonephritis is the main presentation of renal involvement and is commonly regarded as lupus nephritis (LN), which depicts one of the most critical symptoms of SLE. Childhood-onset systematic lupus erythematous represents higher frequency of LN than adult SLE, progressing in 32% to 55% of cases and also more critical with a substantial risk of chronic renal damage.3-10 From the 1980s, kidney prognosis has been noticeably improved after the launch of cytotoxic drugs in the pharmaceutical market, with novel studies depicting 92% and 86% 5- and 10-year renal survival, respectively. 11 However, notable morbidity and 19 times greater mortality is experienced by young aged patients with LN than healthy ones, 12 thus emphasizing the critical requirement for proper management of LN for these patients.
Although progression to chronic kidney disease (CKD) stage V is less frequent in childhood-onset lupus nephritis (cLN) than in adult disease, it remains a clinically relevant outcome, particularly in patients with proliferative (class III/IV) nephritis. Long-term pediatrics cohorts report progression to kidney failure (KF) in approximately 5% to 15% of affected children, with risk strongly influenced by histological severity, chronicity, and response to therapy.13-15 Importantly, cSLE displays marked ethnic predilections, with higher incidence, more severe renal involvement, and poorer kidney outcomes observed among African, Hispanic, Asian, and Indigenous populations compared with White children.5,16-20 These disparities likely reflect a complex interplay of genetic susceptibility, immune dysregulation, and social determinants of health.16-18
Childhood-onset systematic lupus erythematous exhibits a strong female predominance that becomes more pronounced with advancing age. Population-based studies report that overall incidence in girls is substantially higher than in boys (≈1.2 vs 0.2 per 100 000 children), with female patients accounting for the vast majority of pediatric cases. 21 Age-stratified data further demonstrate that the female-to-male ratio increases from approximately 3 to 4:1 in pre-pubertal children to over 7:1 in adolescents, consistent with the peak onset of disease around the peri-pubertal years (median = 11-12 years).22,23 Although cSLE can occur at any age before 18, diagnosis before 5 years is rare and may suggest a monogenic lupus phenotype. 24 Regarding SLE being predominant in female, its dependence on genetic and hormonal alterations is still not clearly understood.
There is a rapid evolution in managing LN due to numerous advances in current research, which is largely related to novel therapies. Also, there is a greater apprehension of pathogenesis, a superior histological definition, the recognition of prospective biomarkers for the purpose of risk stratification, and novel targets for various therapies at the time of follow-up. However, pediatric patients are not included in most of the clinical trials. Due to the scarcity of data, particularly in children suffering from LN, recommendations and clinicians depend entirely on treatment approaches employed for adults. Most of them recommend the off-label use of new treatments in children for collecting particular data and developing clinical experience, while awaiting recent clinical trials for proving the ideal perspective for LN children. 25
Pathogenesis
The comprehension of SLE has aced recently, which is largely related to new genetic factors, variants associated with epigenetics and expression of genes linked with sporadic and monogenic SLE types. As a matter of fact, SLE is a composite disease arising from genetic vulnerability and exposure to environmental factors that persuade epigenetic modifications, for example, air pollutants, viral infections, medications, hormonal abnormalities, and sunlight.26-29
Systemic lupus erythematosus arises from a breakdown of immune tolerance driven by the interaction of genetic susceptibility and immune dysregulation. Crow et al highlights in a contemporary synthesis that impaired clearance of apoptotic material and ICs leads to persistent exposure of nuclear antigens, which in turn promotes auto-antibody production and immune-complex deposition in target organs, such as the kidney and skin. Rather than a single pathway, SLE represents a convergence of several dysregulated innate and adaptive immune responses that amplify inflammation and tissue injury over time. 30
A central unifying mechanism in this process is chronic activation of the type I interferon (IFN-I) pathway. Type I interferon promotes dendritic cell activation, B-cell differentiation, and auto-antibody production, creating a self-sustaining inflammatory loop. Elevated interferon signatures are particularly prominent in cSLE and are associated with higher disease activity and more severe organ involvement, including LN. This interferon-driven immune activation provides a mechanistic explanation for the aggressive disease phenotype often observed in pediatric patients.31,32
Importantly for pediatric clinicians, these immune abnormalities translate directly into organ-specific disease. Immune-complex deposition and complement activation within the glomerulus drive the histopathological patterns seen in LN, while similar processes underlie cutaneous and hematologic manifestations. Understanding SLE as an interferon-mediated, immune-complex–driven disease helps link pathogenesis with emerging targeted therapies and explains why early recognition and immunomodulation are critical in childhood-onset disease.30,32
Interestingly, recent studies have failed to indicate IFN signature role in adults for classification and assessment of disease activity. However, a recent study showed that patients with cSLE and normocomplementemia reported elevated IFN scores and lesser disease severity compared with hypocomplementemia patients, which on the contrary demonstrated an elevated titer of anti-dsDNA antibodies. A new disease model was hence suggested for cSLE comprising 2 different subsets of disease. The elevated IFN, normocomplementemic, and probably milder phenotype group of children sets nearer to auto-inflammation in the pathogenic continuum accompanying autoimmunity, which is usually prevalent in SLE. 33 The identification of this sub-group may aid in classification of patients in upcoming trials and assist in the investigation of agents targeting the IFN route. 34 Our observation that SLE severity can be stratified according to IFN-I intensity is consistent with genetic data implicating gain-of-function changes in this pathway as a major driver of disease. Multiple studies have shown that both common risk alleles and rare high-penetrance variants in genes regulating IFN-I production or signaling are associated with increased IFN activity and a higher likelihood of developing SLE, often with more severe clinical manifestations, including nephritis.32,35 Monogenic “interferonopathies” and monogenic forms of lupus, in which constitutive activation of the IFN-I pathway is a core feature, can be viewed as extreme examples of the same biology, presenting with early-onset, severe disease. 36 In this context, our IFN-I-based severity classification likely reflects the phenotypic expression of cumulative genetic influences on the IFN-I pathway, together with environmental and immunological triggers, rather than opposing mechanisms.
There is also an involvement of adaptive immunity in pathogenesis of SLE, with B-cell and/or T-cell phenotypic or functional irregularities that trigger loss of immune tolerance and contributes to the proinflammatory level. Effector T-cells are influenced by impaired interleukin (IL)-2 production, such as the T-helper 17 (Th17) phenotype, decreasing regulatory T-cells. 31 Immunosuppressive drugs, such as azathioprine and mycophenolate mofetil impedes T-cell function. The primary source of IL-17 are the Th17 cells, which engage B-cells and neutrophils, triggers innate immunity as well as aggravate tissue injury. 37 CD8+ T-cells have recently gained interest as they have been identified in the renal interstitium, suggesting their role in pathogenesis of LN. The most active cells for triggering B-cell differentiation are the T follicular helper (Tfh) cells. 38 Furthermore, elevated levels of B-lymphocyte stimulator are characteristics of cSLE, which is accountable for the generation of auto-antibody, the vital attribute of SLE. 39 Drugs targeting the B-cell pathway, including belimumab and rituximab are the evident therapies. Belimumab, a Food and Drug Administration (FDA)-approved B-cell targeting agent, is considered the first-line agents for active pediatric LN, particularly in patients with persistent serological or extrarenal activity.40,41
High amounts of auto-antibodies undergo a reaction with self-antigens and result in the formation of circulating ICs that deposit in several tissues, specifically in the glomerulus, resulting in the first insult to determine the development of LN. The subsequent activation of neutrophil, macrophage, classical complement, and consumption of complement components that leads to a classical decrease in both C4 and C3 blood levels cause the generation of profibrotic and proinflammatory cytokines, for example, IL-4, converting interferon-gamma, tumor necrosis factor, and growth factor-beta, resulting in epithelial hypercellularity, mesangial endocapillary, and podocyte injury, deposition in the extracellular matrix, and ultimately renal impairment.42-44 Amyloidosis, IC-mediated tubulointerstitial nephritis, and thrombotic microangiopathy (TMA) are some of the other kidney disorders occurring in SLE other than LN. 45 In cSLE, TMA most commonly occurs in association with antiphospholipid antibodies, consistent with secondary antiphospholipid syndrome. Antiphospholipid antibody-associated TMA represents the predominant etiology in this population and should be clearly distinguished from complement-mediated TMA, as the underlying mechanisms, management strategies, and clinical outcomes differ substantially. 46
Renal histopathological classification represents a critical link between LN pathogenesis and therapeutic decision-making. The International Society of Nephrology/Renal Pathology Society classification stratifies LN into 6 classes based on the location and extent of immune-complex deposition, degree of glomerular involvement, and associated inflammatory and chronic lesions. 47 This classification has direct clinical implications, as proliferative forms (classes III and IV, with or without concomitant class V) are associated with more aggressive disease, poorer renal outcomes, and the need for intensive immunosuppressive therapy, whereas mesangial and membranous patterns often follow different clinical courses and management strategies.48,49 In addition, histological activity and chronicity features provide prognostic information beyond class alone and inform treatment intensity and expectations of reversibility, particularly in childhood-onset disease. 48 Accordingly, histopathological assessment remains the cornerstone for risk stratification and therapeutic planning in LN and provides essential context for the treatment approaches discussed in the following section.47-49
Treatment Approaches
There is a scarcity of large clinical trials for guiding the treatment of children with LN. Moreover, most of the current studies suggest application of adult regimens to the pediatric population. Recommendations were developed for pediatric LN by Childhood Arthritis and Rheumatology Research Alliance (CARRA) and Single Hub and Access point for pediatric Rheumatology in Europe (SHARE) in 2012 and 2017, respectively.13,50 Because of the pending pediatric trials, clinicians depend on adult studies and recommendations, including the 2021 Kidney Disease: Improving Global Outcomes (KDIGO) and 2019 EULAR and European Renal Association-European Dialysis and Transplant Association (EULAR/ERA-EDTA).51,52 Nevertheless, it is important to address issues specific to age group, such as fertility, growth, treatment, and psychosocial problems while treating LN in children. Below, we are summarizing the current standard of care at present, after which we will discuss the literature related to emerging therapies.
General Treatment
General treatment of cSLE includes sunscreen protection, a balanced diet, physical activity on regular basis, and control of cardiovascular risk factors including dyslipidemia, obesity, and type 1 diabetes.53,54 As these patients have elevated infection risk, which can trigger disease flares and constitute a vital cause of mortality, 55 it is strongly recommended that they are immunized with inactivated vaccines. Attenuated vaccines are not administrated in immune-compromised patients, despite the fact that no severe adverse reactions were seen in a case series of patients with cSLE when given measles, mumps, rubella, and varicella-zoster vaccinations. 56 The treatment of adjunct arterial hypertension and proteinuria is also suggested because they influence the long-term prognosis as one-third of cSLE patients are still affected with these complications during the third year of their disease. 57 The preferred reno-protective treatments include angiotensin receptor blockers and angiotensin-converting enzyme inhibitors. 50 Finally, issues related to mental health, such as depression and anxiety, which affects greater than one third of cSLE patients should be referred to psychologists or psychiatrists. 58
Immunosuppressive Treatment
The treatment goals are reduction of disease activity, prevention of flares and chronic renal injury, minimization of drug toxicity, and improvement in quality of life of the patient. 59
Initial treatment
Hydroxychloroquine, an antimalarial drug, is recommended by all adult and pediatric guidelines for all patients suffering with cSLE, provided that it decreased end-stage kidney disease (ESKD), risk of flares, and death.50,60,61 For the avoidance of retinopathy, the daily dose should be less than 5 mg/kg. In the absence of pediatric studies regarding classes I and II LN, a low-dose oral glucocorticoid therapy (0.25-0.5 mg/kg/day, maximum 30 mg/day) is recommended by SHARE initiative, the dose of which should be tapered over a time period of 2 to 6 months. Mycophenolate mofetil or azathioprine, the disease-modifying antirheumatic agents (DMARDs), may be used in class II LN in scenarios after re-evaluating renal biopsy for exclusion of misclassification, particularly where tapering of glucocorticoids in impossible after three months or proteinuria is persistent. 50
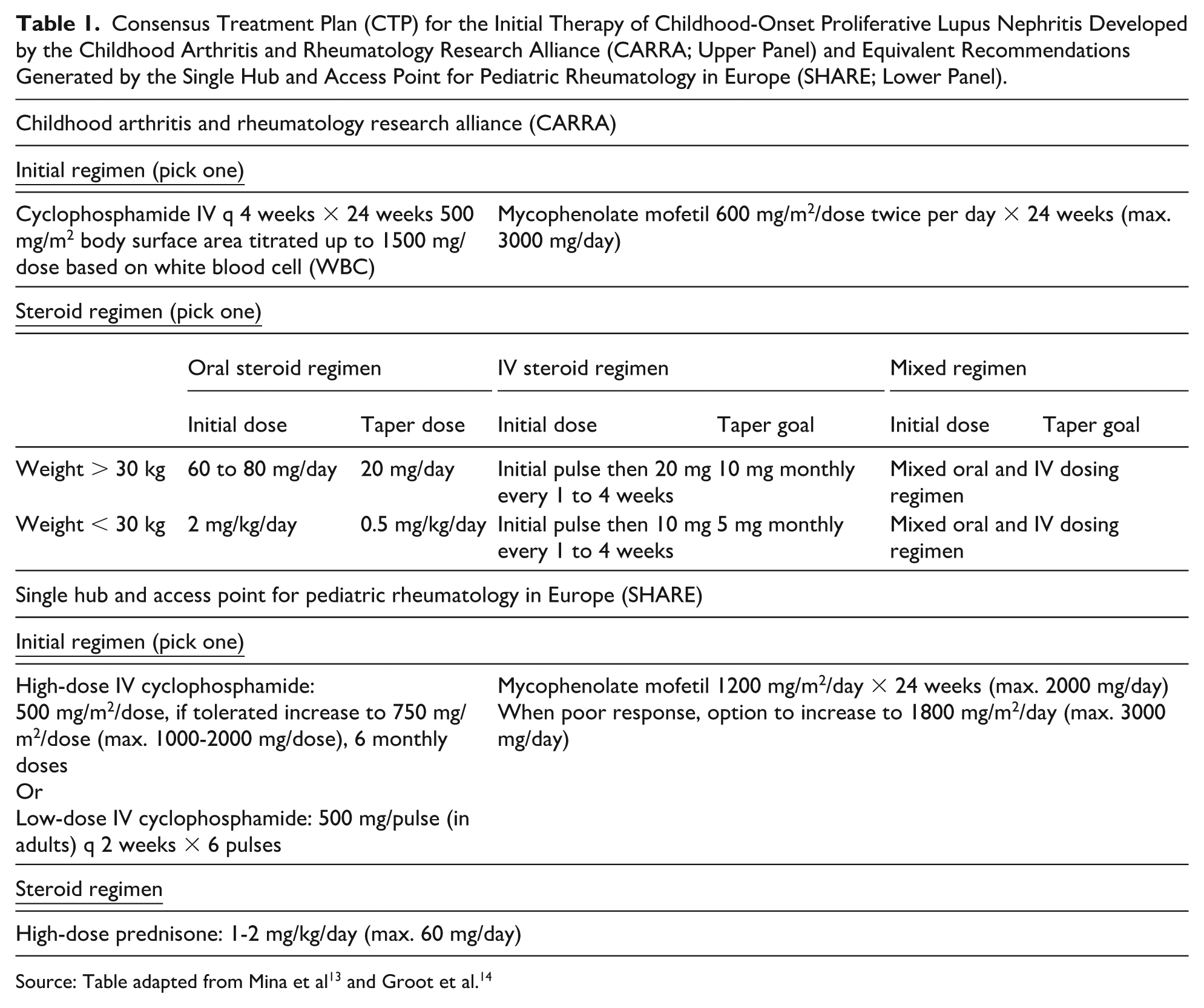
Table 1 summaries a Consensus Treatment Plan (CTP) for the initial therapy in cLN. According to the recommendations of CARRA and SHARE, initial treatment for LN classes III and IV should include high-dose glucocorticoids in combination with either intravenous (IV) cyclophosphamide or mycophenolate mofetil (1200 mg/m2/day, maximum 2000 mg/day; if response is poor, 1800 mg/m2/day, maximum 3000 mg/day). In the Euro-Lupus Nephritis trial, similar efficacy and better side-effect profile of low-dose IV cyclophosphamide in comparison with high-dose IV cyclophosphamide has been seen. 62 It has been indicated by SHARE recommendations that, if cyclophosphamide is used, either low-dose (6 pulses every 2 weeks, 500 mg/pulse) or high-dose (6 monthly pulses, 500-700 mg/m2, maximum 1000-2000 mg/dose) IV cyclophosphamide should be given. Although cyclophosphamide is the only therapeutic agent with long-term data availability that has exhibited efficacy in preserving renal function,63,64 its well-recognized toxicity profile—including delayed adverse effects, such as myelofibrosis, hematologic malignancies, and infertility—has limited its use, particularly in children. Moreover, accumulating evidence indicates that early achievement of renal response is strongly associated with favorable long-term kidney outcomes.65,66 Mycophenolate mofetil is another alternative to be used, particularly in children. Other mycophenolic acid analogues may be given if adverse gastrointestinal effects of mycophenolate mofetil occurs. 67
The optimal initial dosing regimen for glucocorticoids remains insufficiently studied. For patients with acute and severe renal function impairment, nephrotic range proteinuria, a vascular lesion on renal biopsy, and/or an increased ratio of crescents, IV methylprednisolone pulses (3 daily doses of 250-1000 mg each) is recommended. Current clinical trials suggest the use of glucocorticoid pulses, after which a lower oral initial dose and/or quickly tapered off.68-70 However, it is important to avoid excessive exposure of steroid in children, given the implications for drug adherence, growth, and psychosocial problems.71,72 Calcineurin inhibitors, rituximab, and IV cyclophosphamide are some other options.73-75
Consensus Treatment Plan (CTP) for the Initial Therapy of Childhood-Onset Proliferative Lupus Nephritis Developed by the Childhood Arthritis and Rheumatology Research Alliance (CARRA; Upper Panel) and Equivalent Recommendations Generated by the Single Hub and Access Point for Pediatric Rheumatology in Europe (SHARE; Lower Panel).
Maintenance treatment
In both adults and children, the maintenance treatment mainly comprises of either mycophenolate mofetil or azathioprine. No treatment is recommended by SHARE as there are conflicting evidences regarding the evaluation of both these drugs in pediatric LN.76-80 KDIGO and EULAR/ERA-EDTA has recommended mycophenolate mofetil as the first-choice drug for maintenance treatment.51,68
Refractory disease and relapse
Due to higher chances (>60%) of non-adherence in SLE patients, 81 compliance needs to be assessed along with periodic evaluation of serum concentrations of a drug. This is considered as the first recommended step in refractory disease cases. According to majority of the guidelines, it is important to switch between first-line agents, with the possibility of adding calcineurin inhibitors in combination with mycophenolate mofetil and glucocorticoids,82-85 or rituximab, as proved by observational researchers in adult and pediatric LN.86-89 There is a paucity of high-quality, pediatric-specific evidence to guide the management of refractory and relapsing LN, with most treatment approaches extrapolated from adult studies or small observational pediatric cohorts.
Emerging therapies
Emerging targeted therapies for cSLE are increasingly focused on interferon and B-cell survival pathways, as well as kidney-directed regimens in pediatric LN. A dedicated pediatric phase III trial is underway for anifrolumab (anti-IFNAR1), building on adult efficacy demonstrated in TULIP-2, with the ongoing BLOSSOM trial evaluating pharmacokinetics, efficacy and safety in participants aged <18 years.90,91 For cSLE/LN, intensification of B-cell targeting beyond belimumab is also being evaluated; obinutuzumab (type II anti-CD20) is in a pediatric/adolescent LN study including an adolescent randomized component and a younger pediatric open-label cohort, supported by strong adult LN efficacy data.92,93 In parallel, BAFF/APRIL pathway modulation is expanding in pediatrics; telitacicept is being studied in cSLE (including pediatric PK trials), and early pediatric reports suggest improvements in disease activity and serologic markers when added to standard care.94-96 Finally, specifically for pediatric LN, a calcineurin inhibitor strategy is being tested with voclosporin, with ongoing adolescent/pediatric trials informed by the AURORA-1 adult LN results.97,98 Table 2 represents emerging therapies for LN, which also includes those under trial in pediatric population.
Emerging Therapies for Childhood-Onset Lupus Nephritis.
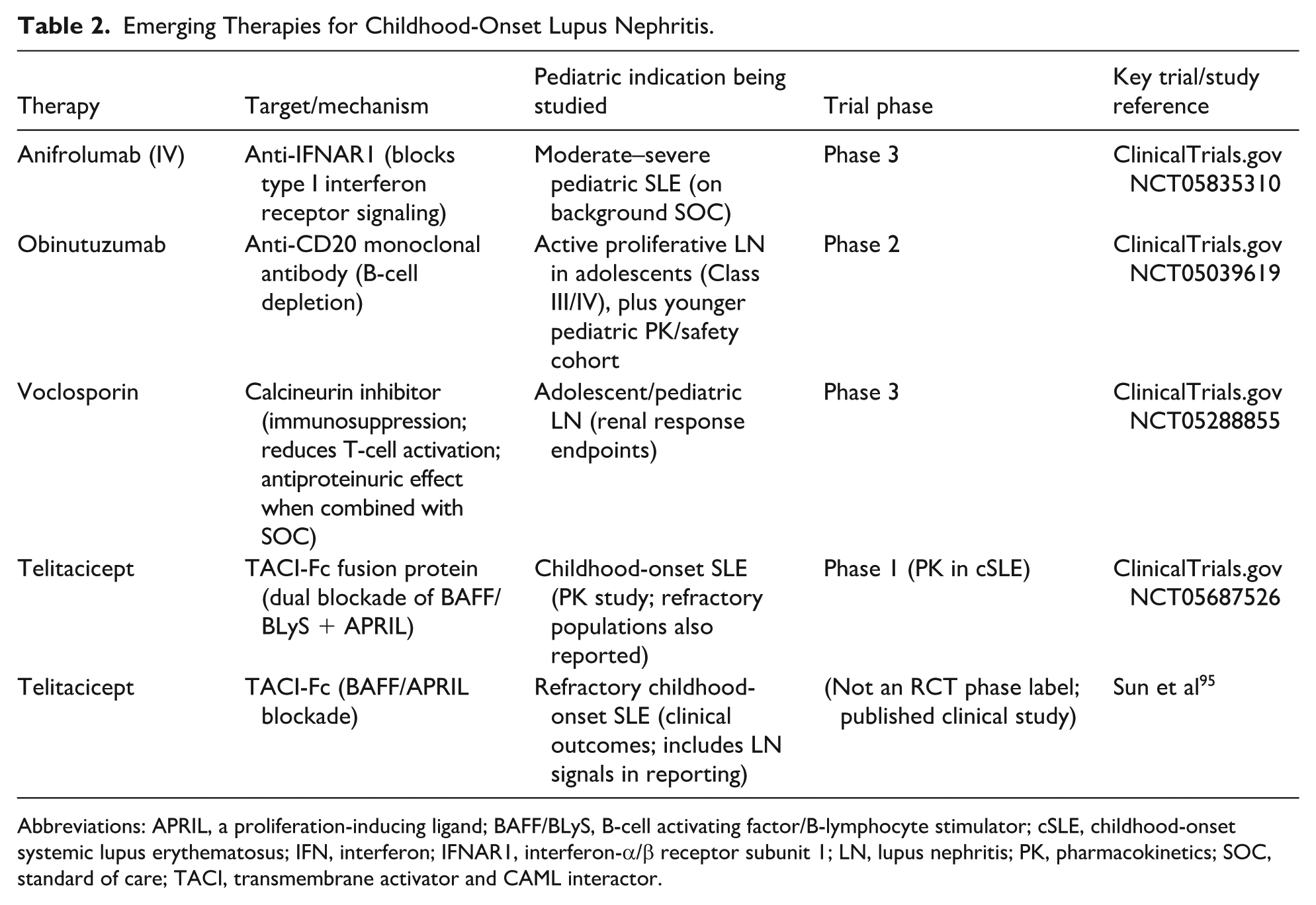
Abbreviations: APRIL, a proliferation-inducing ligand; BAFF/BLyS, B-cell activating factor/B-lymphocyte stimulator; cSLE, childhood-onset systemic lupus erythematosus; IFN, interferon; IFNAR1, interferon-α/β receptor subunit 1; LN, lupus nephritis; PK, pharmacokinetics; SOC, standard of care; TACI, transmembrane activator and CAML interactor.
Renal Outcomes in LN
CKD and LN
Lupus nephritis, a disease defined by histopathological involvement, affects approximately 50% to 82% of children, compared with 20% to 40% of adults.9,99,100 The treatment choices are often dictated by the degree of renal involvement in children with lupus. Histological appearances are used to guide the treatment decisions as per the classification of 2003 ISN/RPS. 47 Figure 1 represents a proposed treatment protocol for the induction and maintenance management of classes III, IV, and V LN in children as based on published recommendations.13,14,101-103 Several treatment protocols have been generated by international groups, which include intensified immunosuppression therapy, followed by maintenance therapy carried out with close examination for improvement along with subsequent inspection of disease flares.13,14,101-104 The cue to carrying out a renal biopsy in pediatric LN is based on the consensus recommendations, which suggests that there should be a urine protein-creatinine ratio (UPCR) of <50 mg/mmol (approximately <0.45 mg/mg) in the early morning sample or a reproducible proteinuria of higher than 0.5 g every 24 hours.14,105 When viewed histologically, a poor renal outcome is usually predicted from the characteristics of chronicity and disease activity. 48
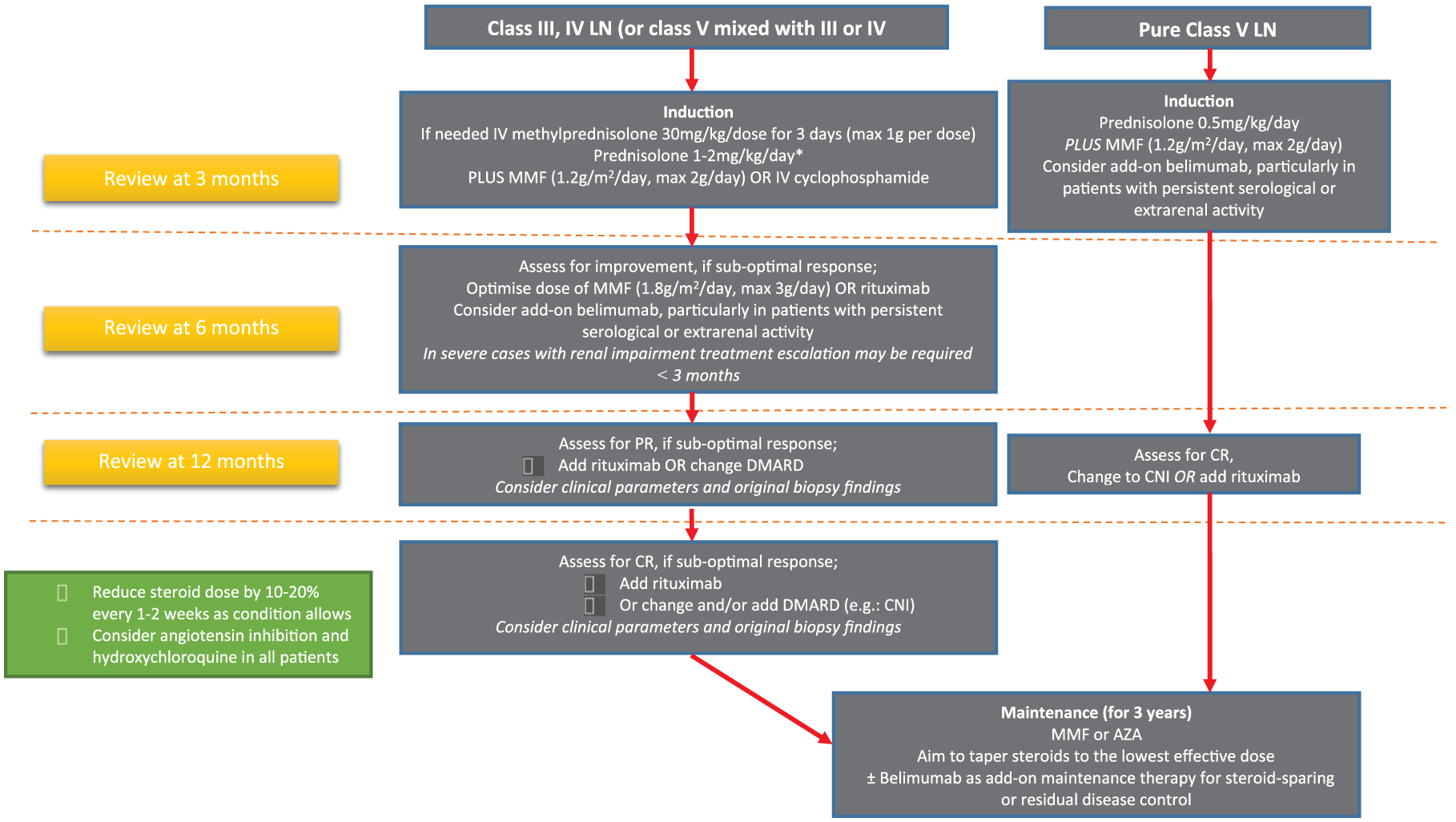
A proposed treatment protocol for the induction and maintenance management of histologically classes III, IV, and V lupus nephritis in children as based on published recommendations.13,14,40,101-103 *See the green box in the figure; CR, complete response (UPCR < 50 mg/mmol, normal kidney function); PR, partial response (> 50% reduction in proteinuria, not nephrotic, normal kidney function).
Response to treatment in LN has paradoxically been examined by employing “renal remission,” the measurement of which is carried out from different clinical components for the creation of a sequential end point, ie, partial remission, complete remission or no remission. The timing and definitions of these endpoints vary across studies, and their validity as surrogates of disease improvement has been questioned, in part because several novel—predominantly biologic—therapies have failed to consistently achieve these endpoints in clinical trials. 106 Intriguingly, new trials are efficient enough in the demonstration of efficacy in SLE, especially those reporting adjusted primary endpoints. 90 Despite these changes, it is evident that renal remission rates reside in the suboptimal range (complete remission in 40%-60% individuals by employing the definition of urine protein less than or equal to 0.5 g/day and normal range of serum creatinine). 107 Significantly, most of the pediatric nephrologists try to seek entire resolution mitigation of proteinuria in young patients in lieu of accepting less than or equal to 0.5 g/day. 108
Lower rates of complete treatment response are associated with an increased risk of CKD, likely reflecting ongoing inflammatory injury and progressive renal damage. This association was demonstrated in a retrospective study of 25 children with LN who underwent repeat kidney biopsy 1 year after the initial diagnosis. Despite the relatively short follow-up period, a higher histological chronicity index was observed, indicating early progression of irreversible renal injury. 109 These results demonstrate that present treatment schedules are not able to entirely stop the renal inflammatory process in most of the patients that adds to overall damage.
Specified published data are scarce with respect to risk of development of CKD before the need of renal replacement therapy, especially in children with LN. In a separate analysis of a UK childhood-onset SLE cohort comprising 399 patients, 3.8% developed CKD stages II to V over a mean follow-up period of 6.6 years. 9
Development of CKD Stage V in LN
In SLE, renal involvement has been noted for approximately 50 years and as per previous research, it has association with increased mortality rates. At present, a long-term follow-up is suggestive of CKD stage V at a rate of up to 15% as a result of LN during childhood. 110 Table 3 represents a summary of pediatric cohorts with LN and the frequency of CKD stage V or KF.
A Summary of Pediatric Cohorts With Lupus Nephritis and the Frequency of Chronic Kidney Disease CKD Stage V (or Kidney Failure (KF)).

Source: Adapted from Oni et al. 110
Generally, no specified contraindications have been listed regarding the employment of any kind of renal replacement therapy in young patients suffering from CKD stage V as a result of LN, however, it is crucial to consider the coagulopathies that are commonly linked with LN. A study from Taiwan involving 94 young adults with ESKD (CKD stage V) secondary to LN evaluated the use of different renal replacement therapy modalities, including kidney transplantation, hemodialysis, and peritoneal dialysis. The renal transplantation group demonstrated superior survival outcomes; accordingly, as in other causes of ESKD, kidney transplantation appears to be the preferred renal replacement therapy for this patient population. 112
Renal Transplantation in LN
Progression to KF occurs in a minority of children with LN but remains a major contributor to long-term morbidity in cSLE. For patients who reach KF, kidney transplantation is considered the preferred renal replacement therapy and is associated with improved survival and quality of life compared with long-term dialysis. 113 Cohort studies of patients with childhood-onset LN followed into adolescence and adulthood demonstrate that patient and graft survival after kidney transplantation are comparable with those observed in children transplanted for other primary kidney diseases, provided that systemic disease activity is well controlled at the time of transplantation. Importantly, transplantation is generally deferred until a period of sustained clinical quiescence is achieved, reflecting the need to minimize the risk of disease recurrence and extrarenal flares.18,110,112
Recurrence of LN in the transplanted kidney is uncommon but well recognized, with reported rates varying widely according to diagnostic criteria, duration of follow-up, and the use of protocol biopsies. Most studies suggest that recurrent disease is usually mild and infrequently leads to graft loss, while graft outcomes are more strongly influenced by factors, such as rejection, infection, and medication adherence.111,114,115 Long-term observational studies from different regions indicate that graft survival in LN is similar to that seen in other causes of KF, and that transplantation should not be withheld solely on the basis of an underlying diagnosis of LN. These findings support early referral for transplant evaluation in eligible adolescents with progressive disease and reinforce the importance of multidisciplinary care involving pediatric rheumatology and nephrology teams.116-118
Improving Renal Outcomes in LN
Transformable risk factors linked with worse renal prognosis that are likely to be affected include the early identification of active renal disease, as hindrance leads to accruement of damage. Current international efforts in this area focus on the identification and validation of novel urinary biomarkers that outperform conventional clinical markers. The integration of biomarker panels into routine practice may enable earlier diagnosis and more timely management of disease. 119 Besides these novel biomarkers, standardization of various features of care might be an aid. This should involve histological elucidation while performing a renal biopsy. According to the recent recommendations of an international consensus for young age LN, including these features and incorporating these directions into clinical scenarios should be prioritized. 14
Due to the heterogeneous nature of this autoimmune disease, studies aimed to target a particular biological pathway might render enhanced rates of absolute response in various patients. Individualized immune profiling may be explored within a treat-to-target framework, particularly in patients who fail to achieve complete remission, to identify key pathogenic immune components—such as cytokines, complement pathways, or B-cell-mediated responses—that could be selectively targeted with tailored therapeutic strategies. A thorough scientific apprehension of diverse biological routes concerned with active LN will be required, and profiling of immune cells that is considered as a clinically vigorous method, needs to be designed and oriented to the present biological treatments. This process can be elevated if genetic risk elements can individually be identified. Furthermore, patients from high-risk ethnic groups, including Hispanic, Asian, and African ancestry populations, may require more proactive and intensive treatment strategies, as these groups experience poorer outcomes than Caucasian patients, in whom early aggressive therapy may be particularly beneficial. 120 The present induction therapy can be made better by additional agents, for example, concurrent calcineurin inhibitor or targeting B-cell at an early stage. 121
At a global level, one of the greatest challenges remains the marked disparity in outcomes of cSLE, particularly in low- and middle-income countries, where differences in healthcare infrastructure, political priorities, and social determinants of health substantially limit equitable access to pediatric care worldwide.
Conclusion
Although the long-term renal prognosis has been considerably improved, a significant morbidity and mortality is still experienced by children and adolescents with LN. Many new developments have been made in comprehending the pathogenesis of SLE with novel treatments that are effective, safe and tolerable leading to improved management of pediatric LN despite the unavailability of detailed information for children and adolescents.
Children characterized by elevated interferon activity, normal complement levels, and a relatively milder clinical phenotype may lie closer to the auto-inflammatory end of the pathogenic continuum, rather than the classical autoimmune spectrum traditionally associated with SLE. The identification of this sub-group may aid in classification of patients in upcoming trials and assist in the investigation of agents targeting the IFN route. The age-linked B-cells constitute an emerging subset of B-cells. Their expansion and auto-antibody production are carried out by the TLR7 route and not by the B-cell receptor. The characterization of their cell surfaces may present new therapeutic targets. Belimumab, an FDA-approved B-cell targeting agent, is considered the first-line agents for active pediatric LN. Rituximab or a regimen based on 3 drugs including glucocorticoids, mycophenolate mofetil, and calcineurin inhibitors is considered as the second-line treatments for refractory LN. To avoid excessive steroid exposure, current clinical trials suggest the use of glucocorticoid pulses, after which a lower oral initial dose and/or a quicker taper of oral glucocorticoid can be used.
Renal involvement is independently associated with an elevated risk of mortality and additional damage accruement. It is essential to quickly recognize and manage LN. Although an international consensus protocol exists, there is an insufficiency of the evidence supporting LN management in children and the renal outcomes are suboptimal. Hence, it is essential to improve various aspects in relation to clinical care in pediatric population, and our comprehension associated with this disease, specifically assessing novel therapies as adjuncts to present treatment to enhance outcomes.
Author Contributions
A.S. conceived the review, performed the literature search, analyzed and interpreted the data, and drafted and revised the manuscript. The author approved the final version of the manuscript and is accountable for all aspects of the work.
Footnotes
Ethical Considerations
Not applicable.
Consent to Participate
Not applicable.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All data generated or analyzed during this study are included in this published article.
