Abstract
Many pediatric assessment tools rely on limited percentile cut points, such as the 90th and 95th, forcing clinicians into broad risk categories and losing essential precision for tracking change. We propose a simple, robust method to impute continuous percentile scores using a piecewise-linear function. Validation against true percentiles in a longitudinal study of children at risk for pediatric feeding disorder (N = 234) yielded exceptionally strong correlations (r = 0.97–0.99). Although slight overestimation occurred in low-score ranges due to sparse cut points, the rank-ordering of scores was reliably preserved. Continuous percentile scores provide the fine-grained differentiation needed to observe subtle changes in developmental trajectory. As illustrated by a clinical case, this enhances data interpretation and allows for more precise tracking to alert the care team of worsening symptoms and facilitate timely, objective evidence for targeted referral. This reliable method is broadly applicable to many clinical tools, and improves the reporting of longitudinal research.
Keywords
Introduction
Interpreting and comparing assessment tool scores over time can be challenging, particularly when the scores are reported as non-standardized total scores or, when researchers must transition between different tools as the child ages. To enhance score interpretation and facilitate comparison across time, one highly effective method is to report percentiles, which indicate where a score falls relative to a normative sample. Within pediatric research, percentiles offer a second advantage: they are familiar to parents and well-understood by clinicians, as they are routinely used to monitor children’s growth in weight and height. Percentile tracking could similarly be used to monitor growth and development in other areas, such as feeding behaviors and skills in children at risk of pediatric feeding disorder (PFD).
Pediatric feeding disorder is defined as impaired oral intake that is not age-appropriate, lasting more than 2 weeks, and associated with medical, nutritional, feeding skill, and/or psychosocial dysfunction. 1 With an estimated prevalence of 1 in 23 to 1 in 37 children in the United States under age 5, 2 early identification and referral to appropriate feeding specialists are critical for effective treatment. An assortment of assessment tools is used to identify and measure the development and progression of PFD and evaluate treatment effectiveness. 3 These tools provide valuable information about a child’s strengths and weaknesses, informing clinical decision-making and facilitating inter-professional referrals. Measuring the impact of feeding on the parent and family has also been recognized as an important consideration when developing and testing pediatric feeding interventions. 4
While several feeding assessment tools provide percentile scores, published data are often restricted to only a handful of cut points (e.g., the 50th, 90th, and 95th percentiles). Although this facilitates grouping into risk categories, it risks the loss of critical detail that continuous percentiles offer. Using the Pediatric Eating Assessment Tool (PediEAT) as an exemplar, we illustrate this limitation. The PediEAT is a parent-report measure that assesses symptoms of problematic feeding across 4 domains for children 6 months through 7 years of age.5,6 To account for developmental changes in feeding behaviors, the PediEAT provides age-based normative values (established at standard well-child visit ages) 7 with cut points for the 50th, 90th, and 95th percentiles. 8
Using these cut points to categorize risk allows for comparison of a child with a reference population. For example, for 12- to 15-month-old children, the 50th percentile is a score of 77, the 90th percentile is 107, and the 95th percentile is 126. These cut points can be used to categorize a child’s symptoms of problematic feeding into groups (e.g., No Concern [<90th percentile], Concern [90th–95th percentile], or High Concern [>95th percentile]). However, this approach may oversimplify distinctions in scores. For example, a child with a score of 106 (just below the 90th percentile) may face a higher risk than a child with a score of 78 (just slightly above the 50th percentile), yet both would fall into the same “No Concern” risk group. This illustrates how categorization oversimplifies distinctions in scores and lacks precision for examining change over time.
To address this limitation, we propose a method to impute continuous percentile scores between published cut points. This method aims to provide more granular percentile reporting for more nuanced risk assessment and monitoring. We further aim to validate this approach and demonstrate the value of using continuous percentile scores across time with a case illustration.
Methods
All data used for analyses were approved by the Institutional Review Boards at the University of North Carolina at Chapel Hill and Boston College. The methodological approach described involves 3 phases: (1) imputation of continuous percentile scores from published percentile cut points using a simple piecewise-linear function, (2) validation of the imputation method against “true” percentiles from a normative sample, and (3) illustration of the utility of the continuous scores using a clinical case.
Sample
The validation method utilized a subsample of the New Thru Two (NT2) longitudinal study ([NINR R01NR018192, PI: Thoyre]). The NT2 study prospectively examined feeding behaviors and skills in children at risk for PFD, as well as the impact of feeding on the parent and family from neonatal intensive care unit (NICU) discharge through 24 months post-term age. All infants in this sample were considered at risk for PFD, based on inclusion criteria including premature birth (less than 35 weeks of gestational age [GA]) or identified developmental risk factors (infants born at 35 weeks GA or later). Parent-reported surveys were collected at 9 time points: 1, 2, 4, 6, 8, 10, 12, 18, and 24 months post-term age. 9
Subsamples for analysis
The following NT2 subsamples were used to perform the validation and illustration: (1) NT2 PediEAT Cohort (N = 234): NT2 sample with PediEAT data, used for longitudinal correlation analysis across 6 timepoints (6–24 months), (2) 12-month subsample (n = 177): A subset of the NT2 PediEAT Cohort at the 12-month time point, used for detailed visual and descriptive comparisons of true versus imputed percentiles, and (3) Case Illustration Sample: A single child selected from the NT2 study with complete data from 1 to 24 months post-term.
Measures
The PediEAT was the primary measure for validating the imputation method. The parent-reported PediEAT includes 78 items across 4 subscales: Physiologic Symptoms, Problematic Mealtime Behaviors, Selective/Restrictive Eating, and Oral Processing. Scores range from 0 to 390, with higher scores indicating more problematic feeding.
The case illustration includes 3 additional feeding-related measures used in the NT2 study:
Neonatal Eating Assessment Tool (NeoEAT)10-12: Assesses problematic feeding symptoms in children 0 to 7 months. Higher scores indicate more problematic feeding.
Child Oral and Motor Proficiency Scale (ChOMPS)13,14: Evaluates eating, drinking, and related motor skills (6 months to 7 years). Higher scores indicate higher skill levels.
Feeding Impact on Parent and Family Scales (Feeding Impact Scales) 15 : Assesses the negative impact of feeding on parents and families. Higher scores indicate greater impact.
The NeoEAT, ChOMPS, and Feeding Impact Scales were similarly developed, tested, and age-normed with similar percentile cut points published to identify levels of concern.
Statistical Analysis
Imputation of continuous percentiles (phase 1)
Continuous percentile scores between the published cut points were imputed using a piecewise-linear function. This simple approach was chosen to avoid the assumptions inherent to imposing a distributional constraint (e.g., normality). The PediEAT’s full normative data was available to compute “true” percentiles for all possible raw scores, enabling direct comparison with the imputed percentiles. The imputation formula is described in the “Results” section.
Validation analyses (phase 2)
The validation of the imputation method included 2 primary sets of comparisons using the PediEAT data:
Detailed Distributional and Descriptive Comparison (12-Month Subsample): To assess how closely the imputation tracked the true percentiles across the range of scores, we first visually compared the full distribution of imputed and true percentiles in the 12-month NT2 PediEAT subsample (n=177). We then quantified any potential estimation bias by comparing detailed descriptive statistics (mean, median, SD, range) between the true and imputed percentiles for the total score and all subscales in this same subsample.
Longitudinal Consistency and Correlation, including subscales (NT2 PediEAT Cohort): To evaluate the method’s performance in the total score and subscales at various ages, we computed Pearson correlations between the imputed percentiles and the true percentiles for the PediEAT total score and subscales at 6 time points (6–24 months) in the full NT2 PediEAT sample (N = 234). Correlations of ≥0.80 were set a priori as the threshold for acceptable linear agreement.
Case study (phase 3)
Finally, the clinical utility of the continuous percentile scores was demonstrated using data from a single participant. Continuous percentile trajectories were computed for the PediEAT, NeoEAT, ChOMPS, and Feeding Impact Scales across 1–24 months. For the NeoEAT, where only published cut points were available, our piecewise-linear method was applied to impute the continuous percentiles, highlighting the method’s practical use in clinical and research reporting.
Results
Sample Characteristics
Table 1 describes characteristics of N = 234 children from the NT2 study with completed PediEAT surveys. Based on the study site’s eligibility criteria for developmental follow-up, 46.6% were considered to be at high developmental risk, while the remaining were at moderate developmental risk. All children received neonatal intensive care for at least 3 days, and most were premature.
Descriptive Characteristics of the NT2 PediEAT Cohort (N = 234).
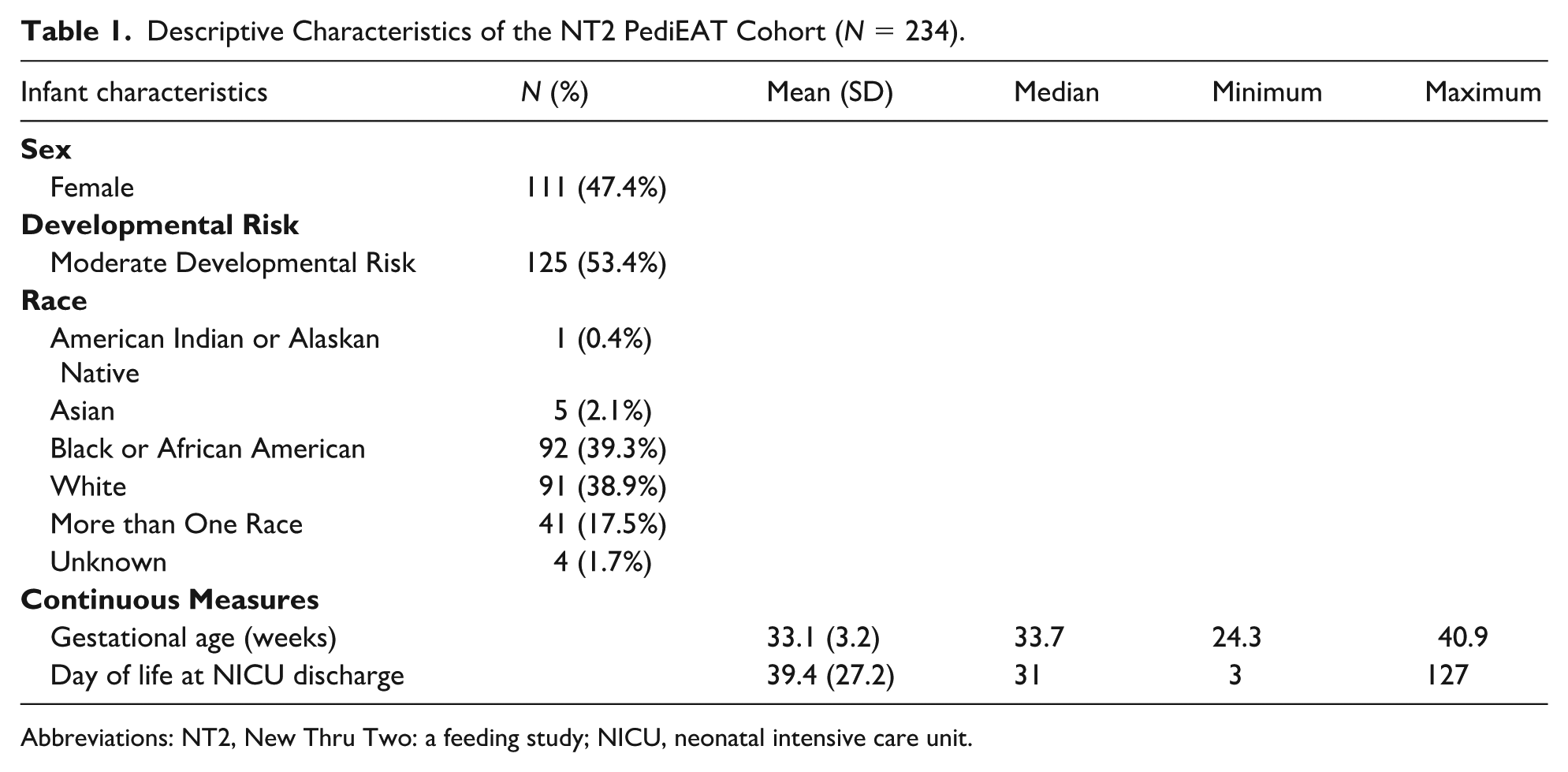
Abbreviations: NT2, New Thru Two: a feeding study; NICU, neonatal intensive care unit.
Imputation Method (Phase 1)
Continuous percentile scores were derived from published cut points using a piecewise-linear function. The resulting continuous percentile scores are illustrated in Figure 1, focusing on the total PediEAT score at 12 months.
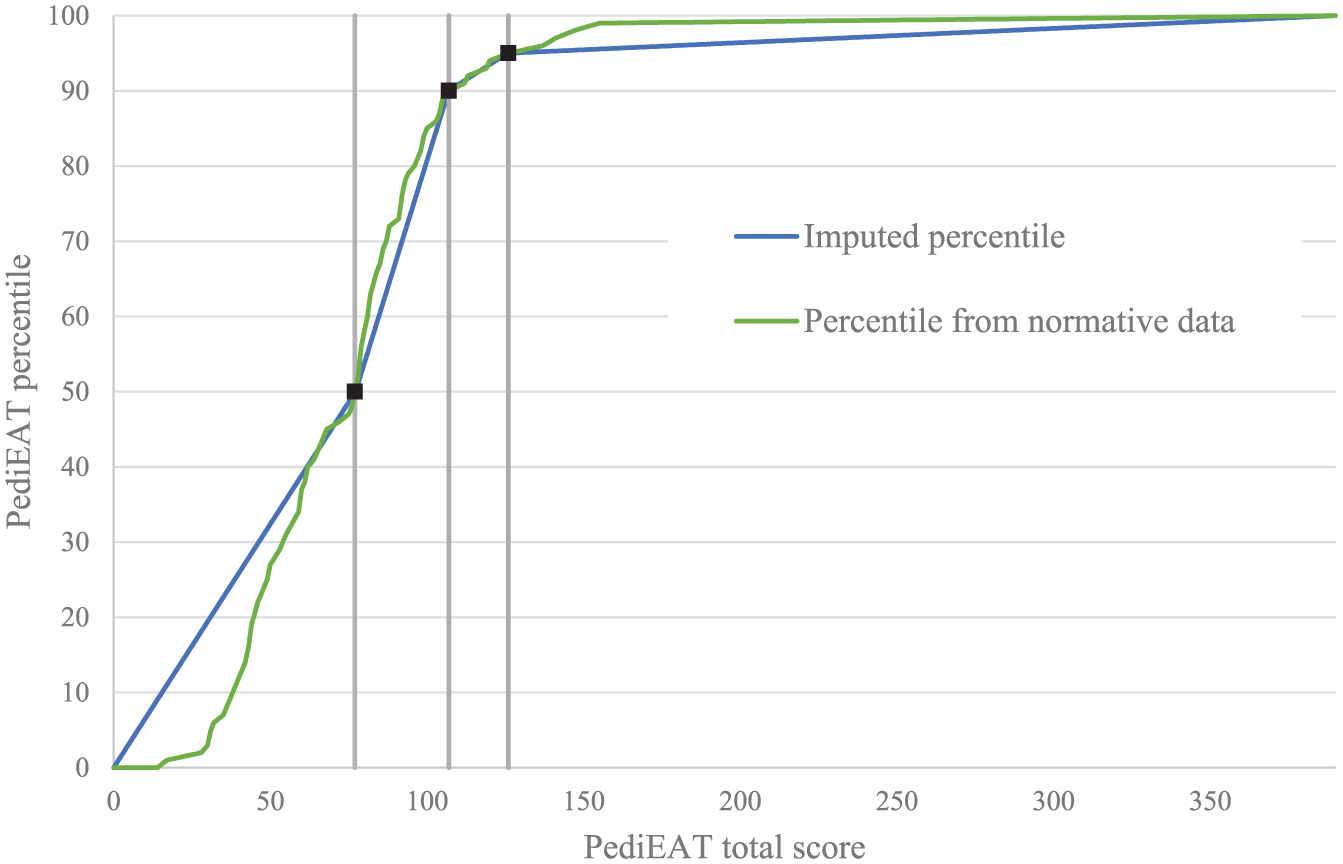
Comparison of true and imputed PediEAT total percentile scores at 12 months. The black squares indicate the published percentile cut points (50th, 90th, and 95th percentiles), which serve as anchor points for the piecewise-linear imputation method.
The imputed percentile, denoted P(x), for a given score (x) is computed using the following equation:
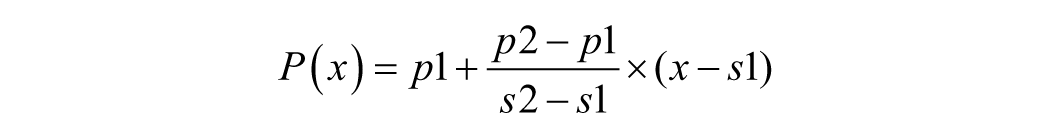
Here, p1, p2 are the lower and upper published percentile cut points immediately below and above x, and s1, and s2 are their corresponding raw scores. By identifying the cut point range relevant to a given x, we determine that x falls between scores s1 and s2 (where s2 > s1 ), and P(x) falls between percentiles p1 and p2 (where p2 > p1 ).
For example, using the PediEAT total score normative data for the 12- to 15-month age group, the 50th percentile corresponds to a score of s2 = 77. If a child has a PediEAT total score of
Validation of Continuous Percentile Scores (Phase 2)
The validity of the piecewise-linear imputation method was assessed through 2 complementary comparisons.
Distributional and descriptive comparisons (12-month subsample)
Figure 1 illustrates the relationship between the “true” percentiles (calculated from the full PediEAT normative data, green line) and the imputed percentiles (blue line) for the PediEAT total score in the 12-month NT2 subsample (n = 177). The 2 lines generally track closely from the 40th percentile onward, confirming overall distributional alignment. However, imputed percentiles tend to slightly overestimate true percentiles in lower score ranges (below the 50th percentile), where the published cut points are sparse.
To quantify estimation error, we compared detailed descriptive statistics (mean, median, SD, range) for the PediEAT total and subscale scores in this subsample (Table 2). The imputed percentiles tended to slightly overestimate the true percentiles for most subscales. As anticipated from Figure 1, this overestimation was consistently observed across subscales whose mean true percentiles fell below the 50th percentile, reflecting the characteristics of the linear imputation across the lowest score range.
Descriptive Comparison of True and Imputed PediEAT Percentile Scores in the 12-Month NT2 Subsample (n = 177).
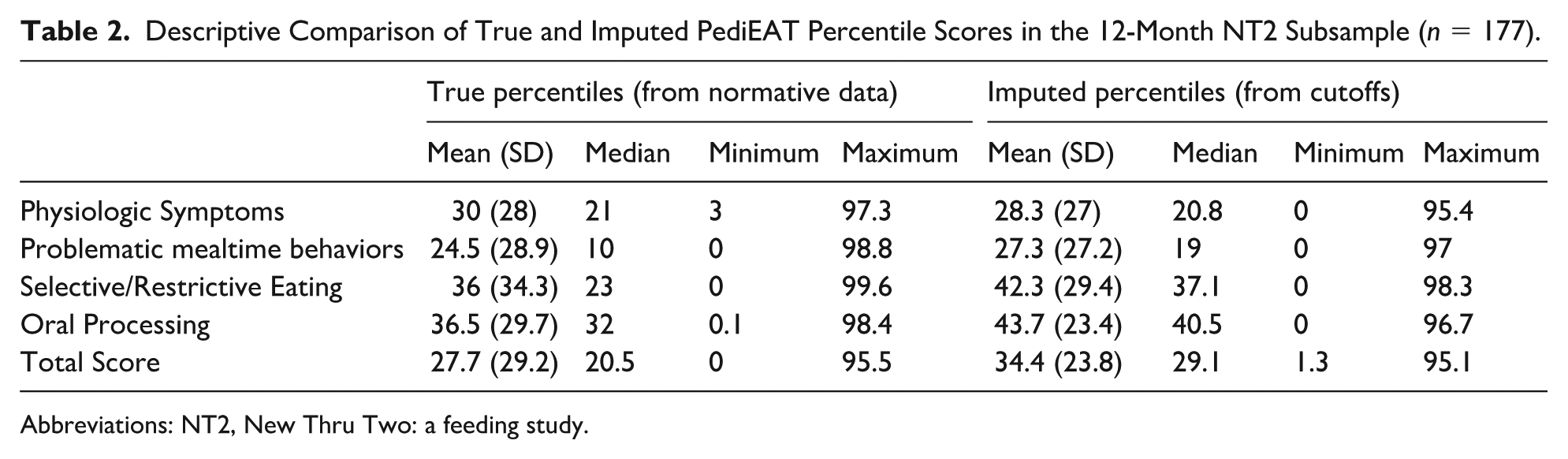
Abbreviations: NT2, New Thru Two: a feeding study.
Longitudinal consistency (correlational analysis)
We assessed the method’s consistency across time by calculating correlations between true and imputed PediEAT percentiles for the total and subscales across all 6 time points (6–24 months) in the full NT2 sample (N = 234). As shown in Table 3, correlations were exceptionally strong across all age groups and subscales, ranging from 0.97 to 0.999. These results confirm that the piecewise-linear imputation method produces scores that are highly linearly related to the true percentiles and adequately preserve the rank-ordering of scores for longitudinal analysis.
Correlations Between Imputed and True Percentiles of the PediEAT (Subscales and Total Score) Across Time.
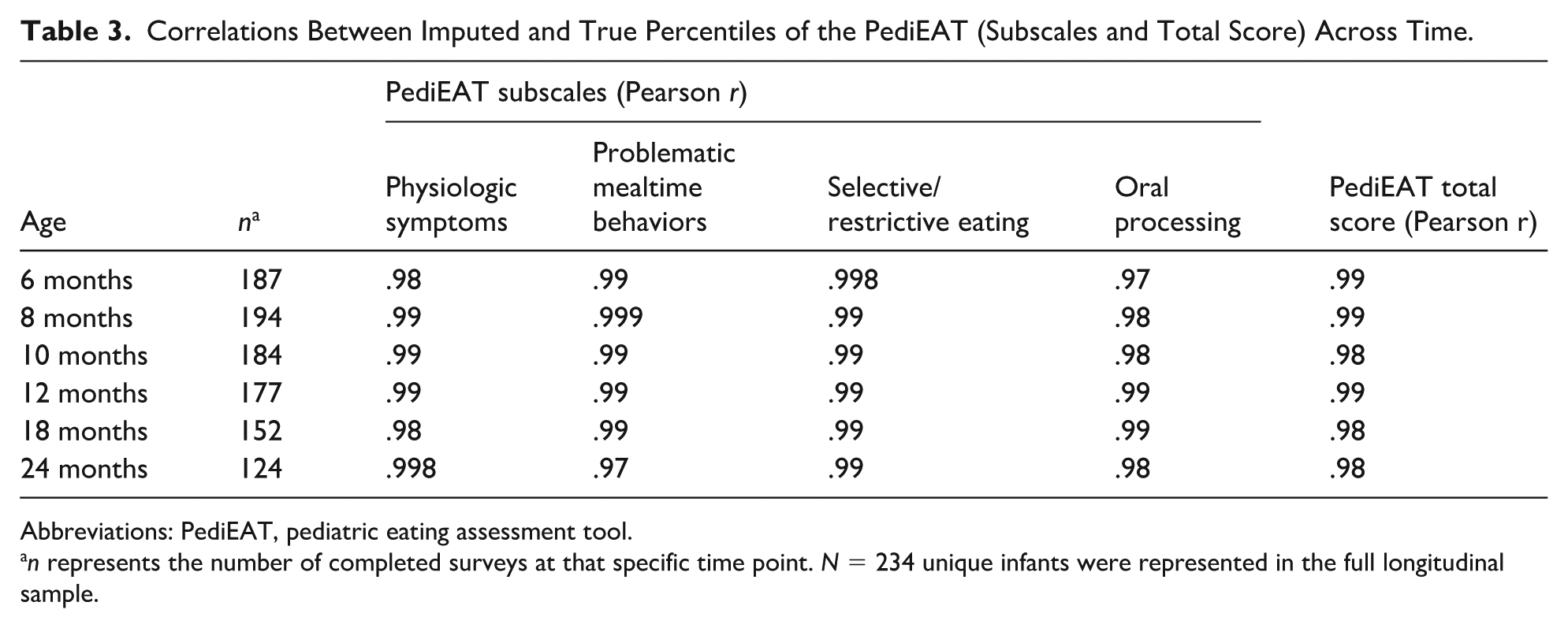
Abbreviations: PediEAT, pediatric eating assessment tool.
n represents the number of completed surveys at that specific time point. N = 234 unique infants were represented in the full longitudinal sample.
Case Illustration (Phase 3)
In Figure 2, we describe the trajectory of continuous percentile scores by tracking feeding measures for Joshua, a participant in the NT2 study. The piecewise-linear method was used to impute continuous percentiles for the NeoEAT, while continuous percentiles for the PediEAT, the ChOMPS, and the Feeding Impact Scales were derived from their full normative data.
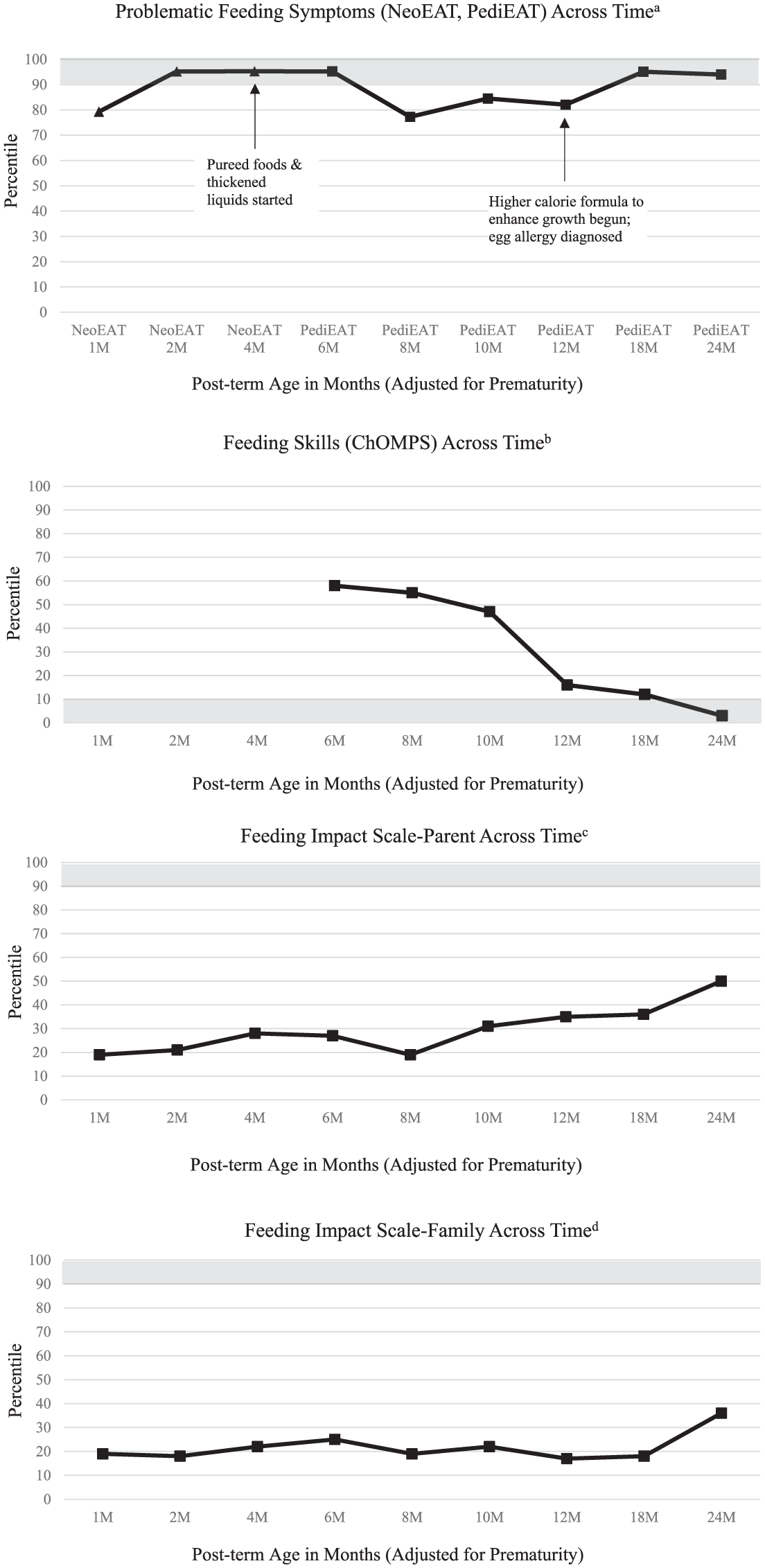
Case illustration using continuous percentile scores. The shaded area on each chart indicates the region of “concern” for that measure (>90th percentile for problematic feeding symptoms and the impact of feeding scales; <10th percentile for feeding skills).
Joshua was born prematurely at 33 weeks GA, weighing 1440 g. Prenatally, he was found to have intrauterine growth restriction and was determined to be small for gestational age at birth. He successfully transitioned to full oral feeding after 24 days and was discharged at 38 weeks post-menstrual age, receiving enhanced calorie formula. Post-discharge, his mother completed surveys at all study timepoints (Figure 2).
Joshua’s percentile scores, adjusted by age, showed a consistent trend of worsening symptoms of problematic feeding (NeoEAT at 1, 2, and 4 months; PediEAT thereafter). The PediEAT subscales help identify specific areas that require additional attention over time. For example, subscales that exceeded the 90th percentile included Physiologic Symptoms across all time periods, Selective/Restrictive Eating at 6, 18, and 24 months, and Problematic Mealtime Behaviors at 24 months. Simultaneously, his feeding skills, measured by the ChOMPS, declined over time, reaching the 16th percentile by 12 months. Notably, his Feeding Impact Scale scores (both parent and family scales) remained below the 50th percentile throughout the study period, indicating minimal subjective family distress despite the objective severity of the feeding problems.
Discussion
We have described a method for reliably deriving continuous percentile scores from published percentile cut points and validated it using the PediEAT. Our validation confirmed that while a piecewise-linear estimation may not perfectly capture true percentiles—particularly when cut points are sparse—the estimated percentiles were strongly correlated with the true percentiles (ranging from 0.97 to 0.999), allowing for accurate risk assessment on a continuous scale. We believe the method’s simplicity and high consistency outweigh minor imperfections in estimation, especially since the observed overestimation occurs in the lower-risk range, which is less critical than accurately identifying children at high risk.
Implications for Assessment and Reporting
Our illustration using a child at risk for PFD clearly highlighted the significant advantages of continuous percentiles over simple cut points or categories. The core issue with standard clinical thresholds (e.g., the 90th percentile) is that they are often arbitrary conventions rather than a definitive, criterion-referenced diagnosis, making reliance solely on these cut points inherently risky. Continuous percentiles, conversely, provide the fine-grained differentiation necessary for proactive clinical management. Using continuous scores allows clinicians to observe the longitudinal trajectory and rate of deviation in a child’s development, enabling intervention to be implemented before the child crosses a clinically severe threshold and their status deteriorates. As Joshua’s case illustrates, a simple dichotomy of concern vs. no concern masks critical changes. At 1 month, a dichotomous cut point would place Joshua’s NeoEAT scores simply as “below the 90th percentile.” However, the continuous percentile revealed his status was near the 80th percentile, a score already signaling proximity to the region of concern. More strikingly, at 12 and 18 months, his feeding skills (ChOMPS) were classified in the area of “no concern,” yet the continuous scores showed a worrying pattern of decline, reaching the 16th percentile at 12 months and the 12th percentile at 18 months. This consistent monitoring provided by the continuous score would trigger a timely clinical assessment.
This precision is critical for both communication and targeted intervention. Percentiles offer a common language shared by clinicians, researchers, and families (e.g., “Joshua’s feeding symptoms are currently at the 95th percentile, similar to being significantly above the normal range for his age”). While cut points provide static communication, continuous percentiles allow for dynamic discussions about the rate of change and the patient’s trajectory, which is vital for building consensus on interventions and supporting shared decision-making. Furthermore, continuous percentile scoring facilitates the targeting of interventions by enabling comparison across different tools. For example, at 6 months, Joshua’s symptomatic feeding behaviors (PediEAT) were very high (above the 95th percentile), while his feeding skills (ChOMPS) were within the expected range (58th percentile). As the complexity of foods offered increased at 10 and 12 months, the continuous ChOMPS percentile scores began to decline, directly suggesting potential difficulties that warranted clinical assessment by a feeding specialist. This precision allows clinicians to differentiate the primary source of impairment, directing them to the appropriate specialist—for instance, distinguishing between a motor-skill deficit requiring an occupational or speech therapist and a restrictive behavior requiring psychological intervention.
The data produced by this method is easy to use in practice, as continuous percentiles are generally familiar to clinicians and caregivers. If t-scores are desired by the clinician (standardized to have a mean of 50 and a standard deviation of 10), percentiles can easily be converted to t-scores using existing conversion tables or normed-score calculators. Whether presented as percentiles or t-scores, this approach enhances interpretation, facilitates comparison across different assessment tools, and provides a standardized, unit-independent measure.
Limitations
The piecewise-linear model preserves rank-ordering and produces estimates that are highly correlated with true percentiles, but there is the potential for estimates to be biased. The potential for bias is highest when fewer cut points are available (e.g., only the 90th percentile) or when the estimation spans a broad range of percentiles (e.g., 80th, 90th, and 95th percentiles) without lower values.
Conclusions and Future Direction
Continuous percentile scoring for assessment tools provides fine-grained differentiation that supports clinical decision-making, the evaluation of feeding interventions, and objective evidence for referral. Detailed scoring guidelines for the NeoEAT, PediEAT, ChOMPS, and the Feeding Impact Scales, including continuous percentiles and associated t-scores, are freely available from the authors and will be made publicly available via a dedicated resource website, currently housed at https://feedingflockteam.org. Future research will use these percentiles to report feeding outcomes of the participants in the NT2 study.
We encourage researchers developing new measures to prioritize the publication of percentiles for each possible raw score. Trajectories of percentile scores across time can improve the tracking of developmental processes and the reporting of research findings. In the absence of comprehensive normative tables, our method provides a viable and validated alternative for calculating continuous percentiles. This simple, reliable method may be applied to other tools with only cut points available to facilitate more precise interpretation of assessment tools.
Author Contributions
J. Crandell and S. Thoyre wrote the first draft of the manuscript without honorarium, grant, or other form of payment. Each author has seen and approved the submission of this version of the manuscript and takes full responsibility for the manuscript.
Footnotes
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Research reported in this publication was supported by the following; none had a role in the study design; collection, analysis, and interpretation of data; the writing of this manuscript or the decision to submit the paper for publication: The National Institute of Nursing Research of the National Institutes of Health under Award Number R01NR018192 (All authors); National Institutes of Health, National Institute on Child Health and Development (T32HD007376-27) and post-doctoral fellowship support from the Center for Developmental Science at the University of North Carolina at Chapel Hill (Estrem), the Charles L. Cahill Award for Faculty Research and Development (Estrem), the University of North Carolina at Chapel Hill School of Nursing and Francis Hill Fox Distinguished Term Professor funds (Thoyre), Boston College Research Incentive Grant (Park), and Boston College’s Start-up Research fund (Park).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data from the New Through Two study supporting this analysis will be provided by the first author upon reasonable request.
Prior Report of Data
There has been no prior presentation of study data as an abstract, poster, or publication.
