Abstract
This article reviews otitis media with effusion (OME) in children with cleft palate (CP), the current evidence and recommendations regarding management of OME in cleft palate with a focus on interdisciplinary communication, and the impact this condition has on patients and caregivers. OME is nearly ubiquitous in children with CP due to the anatomical variation of the palatal musculature and resultant Eustachian tube dysfunction. If untreated, OME can negatively affect hearing, speech development, cognitive development, and social integration. Patients with CP have the potential to be negatively impacted by the otologic, cognitive, and psychosocial consequences of untreated chronic OME if not actively managed for this condition. Multidisciplinary management, appropriate intervention, shared decision-making, and appropriate surveillance optimize the wellbeing, cognitive development, and social progression of CP patients with OME.
Introduction
Otitis media with effusion (OME), defined as the presence of fluid in the middle ear without signs or symptoms of acute infection, is estimated to occur in up to 60% of all children by the age of two.1-3 Its prevalence in children with craniofacial anomalies such as cleft palate is even greater, up to 97%, and is well documented.4-8 Orofacial clefting is common, with an incidence in the United States of 1 in 700 live births. OME occurs in the presence of a cleft palate due to an abnormal insertion of the tensor veli palatini muscle causing failure of the eustachian tube to open leading to poor ventilation of the middle ear.2,9
Recurrent OME almost always leads to conductive hearing loss and if left untreated has been associated with delayed speech acquisition and language development as well as hindered social development.5,10-16 Thus, recent efforts have been focused on optimizing the identification, timing of management, and monitoring of OME in this population.
When treating any child born with cleft palate and craniofacial conditions, a multidisciplinary team approach is the standard of care. There is some variation in the structure and membership of a cleft team, depending on local factors, however the core principle is that the various specialists who treat the numerous needs of the patient collaborate to achieve the best possible outcomes. Core cleft palate team members include, at a minimum, a surgical specialist (plastic surgery, oral and maxillofacial surgery, otolaryngology), a speech and language pathologist (SLP), and an orthodontist. Other specialists who may be core members or consulting members include audiologist, dentists, pediatricians, geneticist, psychologist, social workers, and nurses. These team members share their expertise with each other to promote care that is timely, safe, and effective.
The societal burden of OME in cleft palate patients specifically is largely unstudied, though some inferences can be made from the data on OME patients in general. The current evidence describes both direct and indirect costs of OME in the United States as 4 billion USD annually, most notably in the treatment of hearing loss, increased assistance in school, and loss of caregiver productivity. 17 Overall though, more work is needed to understand the cost of OME in cleft palate patients specifically.
In this manuscript, we will review the anatomic causes of OME in cleft palate, treatment recommendation, treatment complications from the viewpoint of otolaryngology, plastic surgery, audiology, and SLP. Cleft palate may occur as an isolated finding, or may occur along with cleft lip, and can be unilateral, bilateral, complete, or incomplete. All of these variations have the same otologic considerations, and so in this article, they will be understood to be included within the term “cleft palate.”
Eustachian Tube Anatomy and Function
Located in the parapharyngeal space, the eustachian tube is a partially osseous and partially cartilaginous canal which connects the middle ear to the nasopharynx. It is lined by respiratory epithelium and courses from the anterior middle-ear space to its opening in the lateral wall of the anterior nasopharynx. It holds a variety of functions including the regulation of middle-ear pressure and allowing mucociliary clearance of middle-ear secretions.18,19 Additionally, the eustachian tube has been shown to influence the local immune constitution and oxygenation of the middle ear. It is typically collapsed under normal circumstances and intermittent opening allows for ventilation of the middle-ear space.
There are six muscles with recognized roles in the function of the eustachian tube including the levator veli palatini (LVP), tensor veli palatini (TVP), salpingopharyngeus (SP), medial and lateral pterygoids, and the tensor tympani. The TVP originates at the medial pterygoid plate and the wall of the eustachian tube and creates a tendinous sling around the medial pterygoid plate to insert on the palatine aponeurosis, which functions to open the eustachian tube. The LVP originates on the petrous portion of the temporal bone and the medial wall of the eustachian tube and inserts on the palatine aponeurosis, acting as a muscular sling for the soft palate and functions to dilate the eustachian tube (Figure 1). The SP originates from the medial and inferior eustachian tube and functions to lower the inferior eustachian tube wall during swallowing opening the tube.11,18,19 When inserted correctly, these muscles will intermittently open the eustachian tube in the nasopharynx in order to equalize the pressure and ventilate the middle ear.
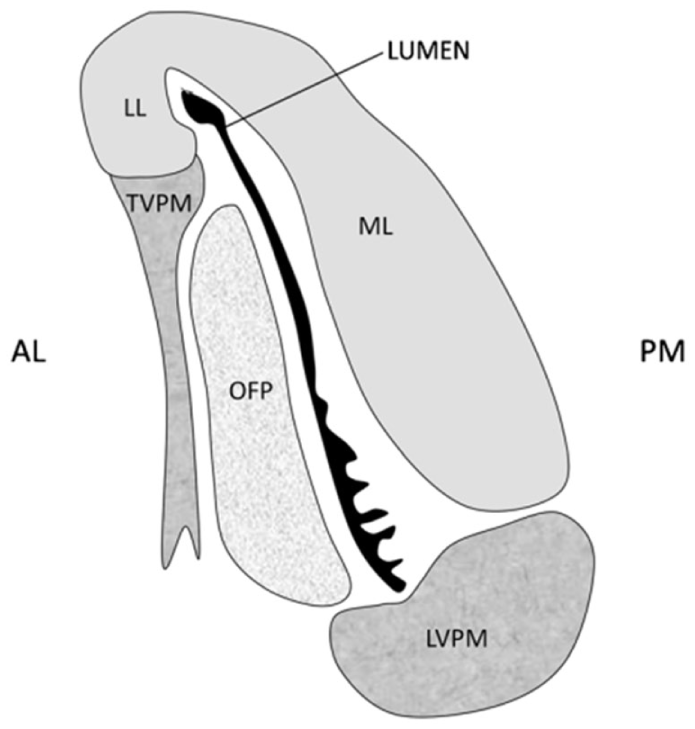
A schematic of a transverse section through the ET in the midcartilaginous section, as seen in the closed state, with the collapsed lumen shown in black. The mucosal folds in the inferior part of the lumen can be seen.
The function of the eustachian tube is dependent on the proper development of the palate. Disruption in the formation of the palate during embryogenesis resulting in cleft palate also results in abnormal insertion and function of the LVP and TVP muscles.13,20 With mal-alignment of the LVP and TVP in children, the natural muscular sling is disrupted, resulting in inadequate opening of the ET during swallowing or yawning. This leads to continuous negative pressure, inadequate fluid clearance, insufficient ventilation, and poor pressure equalization in the middle ear putting the child at high risk for development of recurrent OME.1,11,21-23
Submucous cleft palate (SMCP), characterized by clefting in the palatal musculature with intact overlying mucosa, can also result in eustachian tube dysfunction in the same manner as those with frank cleft palate. The appearance of the palate in SMCP may be relatively normal, leading to delay in diagnosis. Physical findings in SMCP may include a bifid uvula, a palpable notch in the posterior hard palate bone edge, and a zona pellucida, which is a nearly transparent membrane in the midline of the soft palate. It occurs when the palatal muscles are abnormally inserted in the edge of the hard palate and do not meet to form a midline muscular sling while the mucosal coverage remains intact (Figure 2).24,25 Children born with SMCP are sometimes not identified until an older age when they can present with hyper-nasal speech, chronic middle-ear effusion, impaired hearing, recurrent middle-ear infections, and nasal reflux of food and liquids.26,27 A significant number of children with SMCP are asymptomatic, and its presence alone is not an indication for treatment, nor is the risk of OME as high in SMCP alone. 28
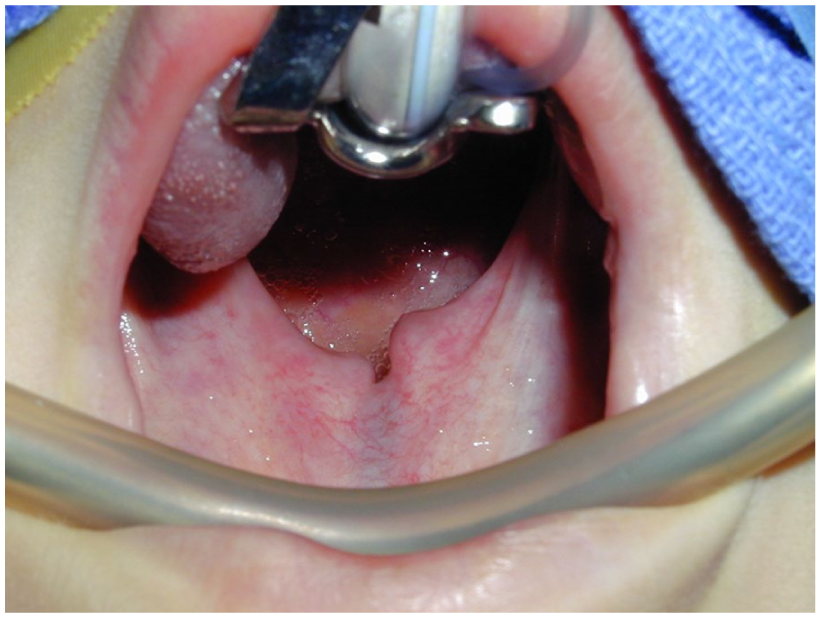
Author’s photograph of submucous cleft palate with bifid uvula and zona pellucida.
Monitoring and Management
In accordance with the Early Hearing Detection and Intervention initiatives, newborn children are generally screened for hearing loss prior to leaving the hospital. This initiative includes newborn hearing screening, formal audiologic evaluation for those children who fail the screening, and intervention by age 6 months for the patients who fail the formal audiologic evaluation. 29 In children, there is a unique urgency to address hearing loss as language acquisition and auditory cortex development is in an accelerated phase in the early years. Timely surgical repair of a child’s palate has been shown to have favorable outcomes in speech intelligibility and development, while timely placement of ear tubes in patients with OME improves language acquisition.30-32
The role of medical management in the treatment of OME is minimal, with current society guidelines recommending against the routine use of antihistamines, decongestants, anti-reflux therapy, and topical nasal steroids given the lack of evidence for their efficacy. These guidelines also recommend against the use of systemic steroids and antibiotics as the side effects of these medications outweigh their minimal benefit.33-35
Unfortunately, there is a paucity of evidence regarding ideal timing of tympanostomy tube insertion in children with cleft palate. The argument could be made for consideration of ear tube placement at the first sign of OME given the propensity for this condition to persist in children with cleft palate. The clinical practice guidelines published by the American Academy of Otolaryngology–Head & Neck Surgery (AAO) focuses on children born without clefts palate, reducing the applicability of the consensus recommendations. 36
It is common practice of many cleft lip and palate teams to concurrently insert myringotomy pressure equilization (PE) tubes at the time of palatoplasty while the child is under general anesthesia, usually at 9 to 10 months of age, but occasionally delayed until 18 months of age. The literature suggests differing opinion about the value of ear tube placement at the time of palatoplasty. Based on retrospective observational studies, Ponduri et al 6 concluded that there is inadequate evidence to support routine tube insertion at the time of surgical palatal repair. Conversely, Yang et al 37 compared ear tube placement before and after 1 year of age in children with cleft palate and observed better hearing outcomes with earlier intervention. Hubbard et al 38 reported superior hearing outcomes with PE tubes inserted at 3 months in children compared to 30 months.
Timing of first anesthesia exposure differs for children born with both clefts lip and cleft palate because cleft lip is typically repaired at a much younger age. At our institution, it had been historical practice to insert PE tubes at 10 weeks of age during surgical lip repair for those patients born with concurrent clefts of the lip and palate, however, the insertion of PE tubes at this time lead to a high incidence of otorrhea in our practice, introducing an additional care burden for families. Ultimately this practice was changed to defer tube placement until palatal repair (9-10 months of age). In a review of our experience, we found that deferring tympanostomy tube placement from the time of lip repair, between 2 and 3 months of age, to the time of palate repair, between 9 and 10 months of age, did not significantly impact hearing outcomes at ages one and two. 39 Moreover, the incidence of otorrhea was significantly decreased. This decrease in otorrhea may substantially reduce the overall burden of care for caregivers, whether via missed days of work due to appointments, phone calls, or cost of additional treatment (eg, co-pays, costs of prescription antibiotics, and travel expenses).
The process of deciding when ear tube placement should take place in a child with cleft palate is one of shared decision-making among caregivers and involved clinicians. Parents should be counseled on the negative effect of persistent OME on language development due to prolonged OME as well as alternatives to PE tubes, including close observation and hearing aid therapy. Regardless of the timing of ear tube placement, children require long-term otologic routine monitoring throughout childhood to obtain optimum speech and language outcome and to avoid complications of OME and chronic eustachian tube dysfunction.23,40
At our institution, children with cleft palate and PE tubes in place are evaluated every 6 months by ENT and audiology. Standard audiologic assessment includes behavioral assessment with pure tone, and when possible, speech audiometry, as well as physiologic testing with acoustic emittance testing and otoacoustic emissions (OAEs). After PE tubes have extruded, they are followed at least yearly at team until age 6 and at least every 2 to 3 years thereafter through adolescence. Families are also educated to report frequent infections, chronic ear drainage, or symptoms of poor hearing.
Sequelae Specific to the Tympanic Membrane and Middle Ear
Eustachian tube dysfunction and the associated chronic or recurrent OME is associated with a number of adverse sequelae related to the tympanic membrane and middle ear in patients with cleft palate. Chronic negative middle-ear pressure and chronic effusions may result in hearing loss and damage to the tympanic membrane (Figure 3). The hearing deficit related to middle-ear effusion or tympanic membrane retraction may be as great as 50+ dB across normal hearing frequencies.2,5,9,41,42 Chronic OME can also result in the need for multiple sets of standard PE tubes (grommet ventilation tubes) or long-term PE tubes (T-tubes). One third of patients with chronic OME associated with CP require at least 3 sets of PE tubes over their lifetimes and often more. Placing standard PE tubes on multiple occasions can lead to scarring and devascularization of the tympanic membrane which increases the risk of tympanic membrane perforation. Placement of long-term PE tubes increases the risk of chronic tympanic membrane perforation significantly more than placing standard grommet tubes—up to 15% risk as opposed to a 1% risk with standard PE tubes.43,44 When evaluating all comers children with cleft palate have a 7.7 times increased risk of tympanic membrane perforation compared to patients without cleft palate. Tympanic membrane perforation may cause significant conductive hearing loss depending on the size and location of the perforation. 23 Recurrent PE tube placement can also lead to scarring of the tympanic membrane known as tympanosclerosis. Tympanosclerosis is a white calcific scar in the middle fibrous layer of the tympanic membrane which may lead to conductive hearing loss.23,30,45-47
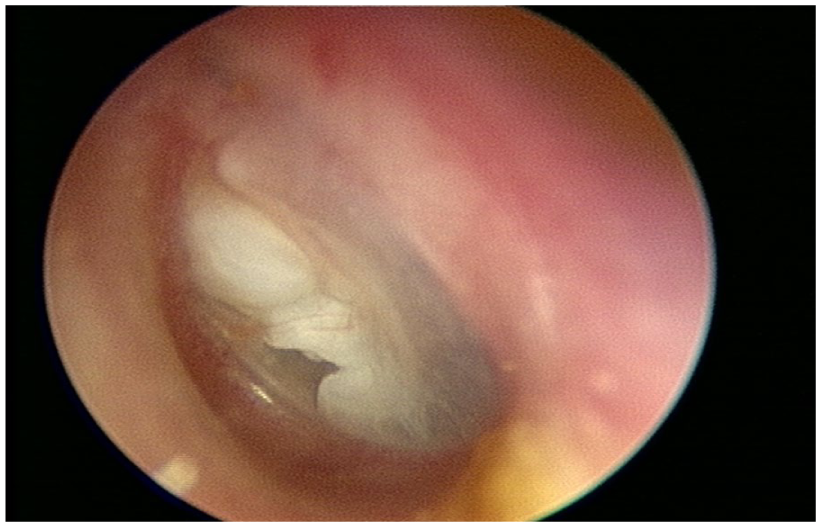
Author’s photograph of a primary acquired cholesteatoma.
When eustachian tube dysfunction progresses to retraction of the tympanic membrane the possibility of cholesteatoma formation arises. Patients with cleft palate are at a higher risk for cholesteatoma formation (Figure 4).
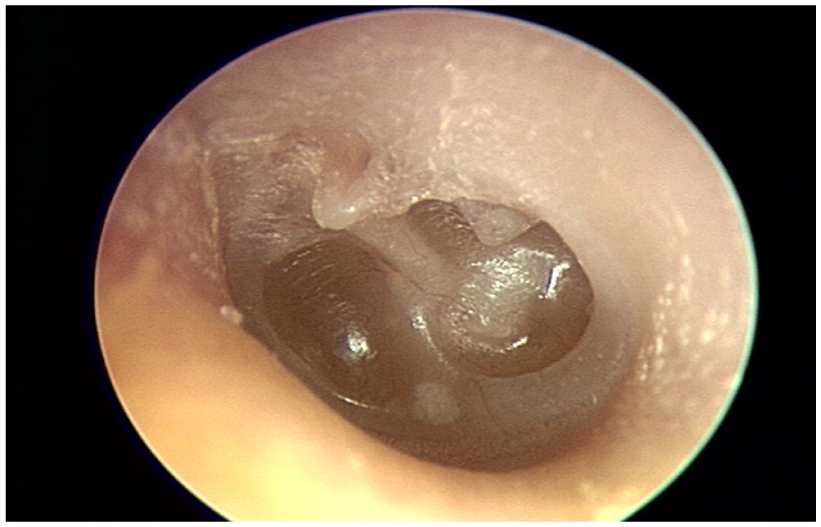
Author’s photograph of a deep tympanic membrane retraction.
A cholesteatoma is an abnormal collection of skin cells in the middle ear that arises from an invaginated tympanic membrane that continues to desquamate skin cells from its outer squamous layer hence forming a cyst of trapped keratin debris. Cholesteatoma can also arise from a chronic tympanic perforation when the squamous layer of the tympanic membrane grows into the middle-ear space (Figure 5). When left untreated, cholesteatoma can lead to chronic middle-ear drainage and infection, severe hearing loss with erosion of the ossicles, extension into the mastoid, chronic or acute mastoid infections/mastoiditis, meningitis, and less frequently, semicircular canal or labyrinthine fistulas. 45 In the general population the incidence of cholesteatoma is estimated to be less than 10 in 100,000 people, however, in the cleft palate population nearly 6% of patients will develop cholesteatoma.48,49 Among children with cleft palate who are of the age range of 4 to 17 years, the mean age of cholesteatoma development is 10 years. 50
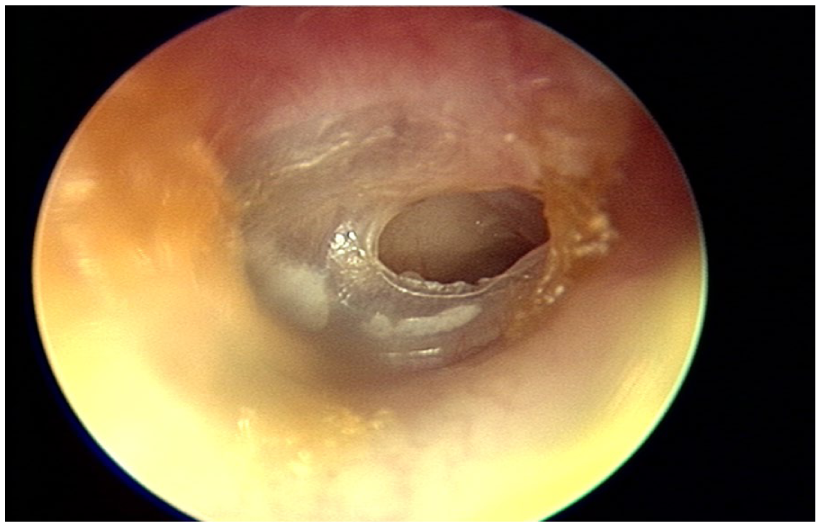
Author’s photograph of a dry tympanic membrane perforation.
Cleft Palate–Associated Ear Sequelae Speech and Language Deficits
Outside of the well-documented issues of speech articulation and phonemic generation associated with the anatomic anomalies of cleft palate, the presence of concurrent middle-ear pathology and its attendant hearing loss increases the likelihood of delay in routine language acquisition in these children. Hearing loss from any cause during a child’s critical first 5 years of language development is associated with speech delays, decreased cognitive ability, and language impairment.2,5,9,41,42 OME lasting greater than 3 months has been associated with poorer school performance and reduced quality of life.2,51,52 The increased OME duration seen in cleft palate has also been associated with lower scores on the Wechsler Intelligence scale for children including scores for articulation and use of morphologic markers.53,54 When compared to peers, children with cleft palate and early hearing loss perform worse when examining speech and language on standardized testing.11,54,55 Deficits in speech and language in children with cleft palate is also associated with psychosocial sequelae such as shyness, dependence, and poor creativity. 5
Patients with and without cleft palate tend to have significantly less OME as they age; however, patients with cleft palates tend to have evidence declines in middle-ear disease much later than children who are not affected by cleft palate. Children without cleft palate tend to evidence a decline in OME between ages 2 and 4 years while children with cleft palate evidence a decline in OME between ages 6 and 7.56,57
Role of SLP, Audiologists, and Parents on the Cleft Team
SLPs are integral to assure optimal speech and language development in patients with cleft palate. They play a vital role in evaluation and diagnosis of velopharyngeal dysfunction, resonance disorders, and other conditions that will cause difficulties in speech.26,32,58 In addition, they assess for language disorders and often can identify the need for specialized learning evaluation. Early identification of cleft palate patients with speech and language delay related to middle-ear dysfunction and hearing loss improves outcomes. SLPs play an integral role in early identification of cleft palate patients with these issues.
Audiologists are vital for surveillance of hearing changes related to eustachian tube dysfunction, otitis media, and cholesteatoma and provide tympanometric data to help guide the decision for tympanostomy tube placement.29,36 Monitoring children through appropriate and frequent hearing tests is important to detect early hearing loss or changes to hearing, so that interventions can be swiftly implemented to preserve a child’s functional status.59,60 When persistent hearing loss occurs, the audiologist provides recommendations for amplification, and also advocates for appropriate educational services, such as preferential seating and FM systems. The audiologist and the SLP work together to advise the cleft team on recommendations to optimize communication for the child with cleft palate.
While a multidisciplinary team and care plan are necessary for successful care of the child with cleft palate, it must be noted that success starts and ends with the caregiver. It is of the upmost importance for caregivers to be properly educated not only on potential outcomes for the child, but on maintaining vigilance throughout the child’s development. Due to persistent otologic manifestations of children with cleft palate within the first 10 years of life, surveillance by caregivers to report on any changes in hearing to optimize outcomes. 14 Decline in grades, listening to the television or devices louder than usual, or not responding when called are some specific alarm examples that caregivers can be made aware of to identify potential hearing loss sequelae of cleft palate. Caregivers and school-based SLPs should also educate teachers on the unique challenges presented by children and when it may be necessary for those educating the child to speak up and call attention to issues of inattentiveness.
Conclusion
Patients born with cleft palate have a significantly increased risk of eustachian tube dysfunction leading to middle-ear pathology such as chronic or recurrent OME, tympanic membrane atelectasis, or cholesteatoma. These children will benefit from early identification of these issues, followed by intervention, then surveillance utilizing a multidisciplinary approach. It is important to consider the entire gamut of otologic sequelae in these at-risk patients, as well as the potential neurocognitive and psychosocial effects of hearing loss related to cleft palate. Due to variability in individuals’ presentation and progression of disease, education for all caregivers is important for optimal outcomes. Regular evaluation by the multidisciplinary cleft palate team and optimization of caregiver communication with shared decision-making and an individualized management plans will ensure optimized outcomes for each patient.
Author Contributions
All authors made substantial contributions to the conception and design of the review, the acquisition and interpretation of the literature, and the drafting and critical revision of the manuscript. All authors approved the final version submitted for publication.
Footnotes
Ethical Considerations
Our study did not require an ethical board approval as it is a subject review.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data presented in this report has not been published or presented elsewhere.
