Abstract
Objective of this article is to describe differences in the demographic and clinical characteristics, severity of illness, and outcomes in pediatric patients with different SARS-CoV-2 variants. We conducted a retrospective study of pediatric patients admitted with COVID-19 during the 3 large waves of infection within a health network in New Jersey. We included demographic characteristics, clinical features, and outcomes and compared the data with respect to the different variants. Of 351 total patients included in this study, 74 were admitted during wave 1, 94 during wave 2, and 181 during wave 3. The median age of patients decreased from wave 1 (11.5 years) to wave 3 (3 years) (P = .0034). 87.7% of the patients were unvaccinated. The overall incidence of admissions due to pneumonia related to COVID-19 decreased in wave 3. COVID-19 bronchiolitis or croup admissions occurred mostly in wave 3. There was no significant difference in the number of patients requiring intensive care in any particular wave. Length of stay decreased across the waves (P < .0001). Treatments required did not vary between the waves except for a decrease in antibiotic use with each subsequent wave (P < .0001). The impact of COVID-19 on the pediatric population differs from the adult population, and the overall number of hospitalized children has mirrored the peak in cases observed during each infection wave. Our study illustrates the changes in clinical presentation and severity observed with the different coronavirus variants.
Keywords
Introduction
Although children have had overall lower rates of hospitalization for coronavirus disease 2019 (COVID-19) compared with adults, severe illness and death have been described.1-4 As of March 23, 2022, approximately 12 million cases of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection and 933 deaths due to COVID-19 have been reported in the United States in children less than 18 years of age. 5 At the time this manuscript was written, 3 SARS-CoV-2 variants have dominated the surges of infection in the United States, with Alpha predominance occurring between March 1, 2020, and June 26, 2021, Delta predominance from June 27 to December 18, 2021, and Omicron predominance from December 19, 2021, to January 31, 2022. 6 Although the proportion of hospitalized infants and children infected with COVID-19 admitted to an intensive care unit (ICU) was higher during Delta predominance, the rate of pediatric ICU admissions during Omicron predominance was approximately 3.5 times as high as that during Delta predominance, driven by the overall higher disease incidence. 6
The state of New Jersey (NJ) had 3 major waves of infection with the different SARS-CoV-2 variants. 7 A study published by our hospital network details the prevalence of the variants of concern (VoC) during each time period. Only 2 pediatric patients had infection with the gamma variant and were not included in this study. 8 During each separate wave of infection, children and young adults hospitalized had varying presentations and severities of illness. The objectives of this study are to describe differences in the demographic and clinical characteristics, severity of illness, and outcomes in pediatric patients during the different SARS-CoV-2 waves in our hospital network. Although there is already some literature from Canada and Chicago, for example,9,10 describing the presenting symptoms of pediatric patients during each VoC, this article adds to the growing body of literature describing clinical presentations and admission diagnoses of pediatric patients hospitalized during the Alpha, Delta, and Omicron predominant periods, respectively, in the Northeast United States which was one of the hardest affected areas in the country. 7 The presentations of the different VoC of this virus are important information for patients, families, and providers to have to determine risk status with different circulating variants.
Methods
This retrospective study was performed in 3 hospitals within a large academic health network in NJ. These 3 hospitals are located in varying geographic locations of the state. We defined the timelines as follows: Wave 1 occurred between March 1, 2020, and May 31, 2020; wave 2 occurred between November 1, 2020, and January 31, 2021; and wave 3 occurred between December 1, 2021, and February 28, 2022.
Inclusion criteria were as follows: patients between the ages of 0 and 21 years who were admitted to any of the hospitals with laboratory-confirmed SARS-CoV-2 infection via reverse transcriptase polymerase chain reaction (RT-PCR) during 1 of the 3 major COVID-19 infection waves in NJ were included in the study. These patients were identified using international classification of diseases diagnostic codes in our electronic medical record using the ICD-10 codes J12.82, J12.89, J20, U07.1, and M35.81. Patients hospitalized with post-COVID-19 inflammatory conditions such as Multisystem Inflammatory Syndrome in Children (MIS-C) were also included. We included all symptomatic patients, including those with coexisting bacterial or viral infections, because the presence of SARS-CoV-2 may have contributed to the overall severity of illness. Patients with acute appendicitis were included in the analysis if SARS-CoV-2 positive. Patients requiring high-flow nasal cannula (HFNC), invasive respiratory support, neurologic abnormalities, or hypotension were admitted to the pediatric ICU per institutional policy and were included in the study. We excluded patients who were asymptomatic but incidentally tested positive for SARS-CoV-2 and were hospitalized for unrelated indications such as trauma and surgical procedures.
This study was approved by the institutional review board.
Throughout the pandemic, standardized COVID-19 and MIS-C treatment guidelines were readily available to all network hospitals. Data were collected by the investigators from our network’s electronic medical record, EPIC (EPIC Systems Corporation). Demographic data included age and sex. Clinical parameters assessed included presenting symptoms outlined by the CDC for acute COVID-19 infection and MIS-C, body mass index (BMI) for children 2 years and older, comorbid conditions, and COVID-19 vaccination history. Vaccination history was obtained through a query of our state system in addition to parental reporting. Laboratory test and radiology study results were also recorded. The clinical course of the hospitalization, including mode of oxygen delivery if required, treatment with pharmacologic agents, length of stay, and complications including thrombotic events, concomitant bacterial infection, acute respiratory distress syndrome (ARDS), acute kidney injury (AKI), and residual restrictions at the time of discharge were also assessed. Patients with secondary bacterial pneumonia and ARDS were diagnosed by chest radiograph findings. AKI was defined as an elevation in serum creatinine level at least 0.3 mg/dL within 48 hours compared with baseline. Discharge without restrictions was defined as no new medical devices, no new therapy needs, and no residual physical disabilities.
We utilized standardized criteria to define severity of illness as mild, moderate, or severe. Severe illness was defined as admission to the ICU or transfer from the general pediatric unit to the ICU, need for mechanical ventilation, vasopressor use, or ARDS; moderate illness was defined as the need for high flow nasal cannula or those with acute kidney injury; patients requiring nasal cannula or none of these therapies/scenarios were classified as mild. We used the highest severity of a patient any time during the stay to classify our patient.
Statistical analyses were performed using JMP, Version 16.2.0 (SAS Institute Inc., Cary, NC, USA). Nonparametric continuous variables were analyzed using the Kruskal-Wallis test and Pearson’s chi-squared test for categorical variables. Histograms were used to visualize the incidence of events in each respective category.
Results
Study Population
A total of 351 patients were included in this study. Of these patients, 74 were admitted during wave 1, 94 during wave 2, and 181 during wave 3. The majority of patients for all 3 waves were admitted at the northern NJ hospital. The median age of patients decreased from 11.5 years in wave 1 to 3 years in wave 3 (P = .0034). There was equal sex distribution in waves 1 and 3. However, 59.6% of the patients in wave 2 were female (P = .0432). In all 3 waves, the most common race/ethnicity that our cohort identified as included White/Hispanic (42.2%), followed by White/non-Hispanic (30.7%). Sixty percent of the patients had Medicaid insurance. Most patients admitted to the hospital were unvaccinated (87.7%). Vaccines for children were not authorized in waves 1 and 2. Only 15 patients in wave 3 received 2 doses of the vaccine and 2 patients received 1 dose. The median BMI of patients admitted was significantly higher in wave 1 (23.6 kg/m2) compared with 20.8 and 20.9 kg/m2 for waves 2 and 3, respectively (P = .0353) (Table 1). Throughout all the waves, approximately 70% of the patients had no comorbidities. Of those with comorbidities, the 3 most common were history of asthma (19%), oncologic diagnosis (10%), and seizure disorder (8%). The most common risk factor for contracting disease during all 3 waves was having a close family contact with COVID-19 infection.
Patient Demographics.
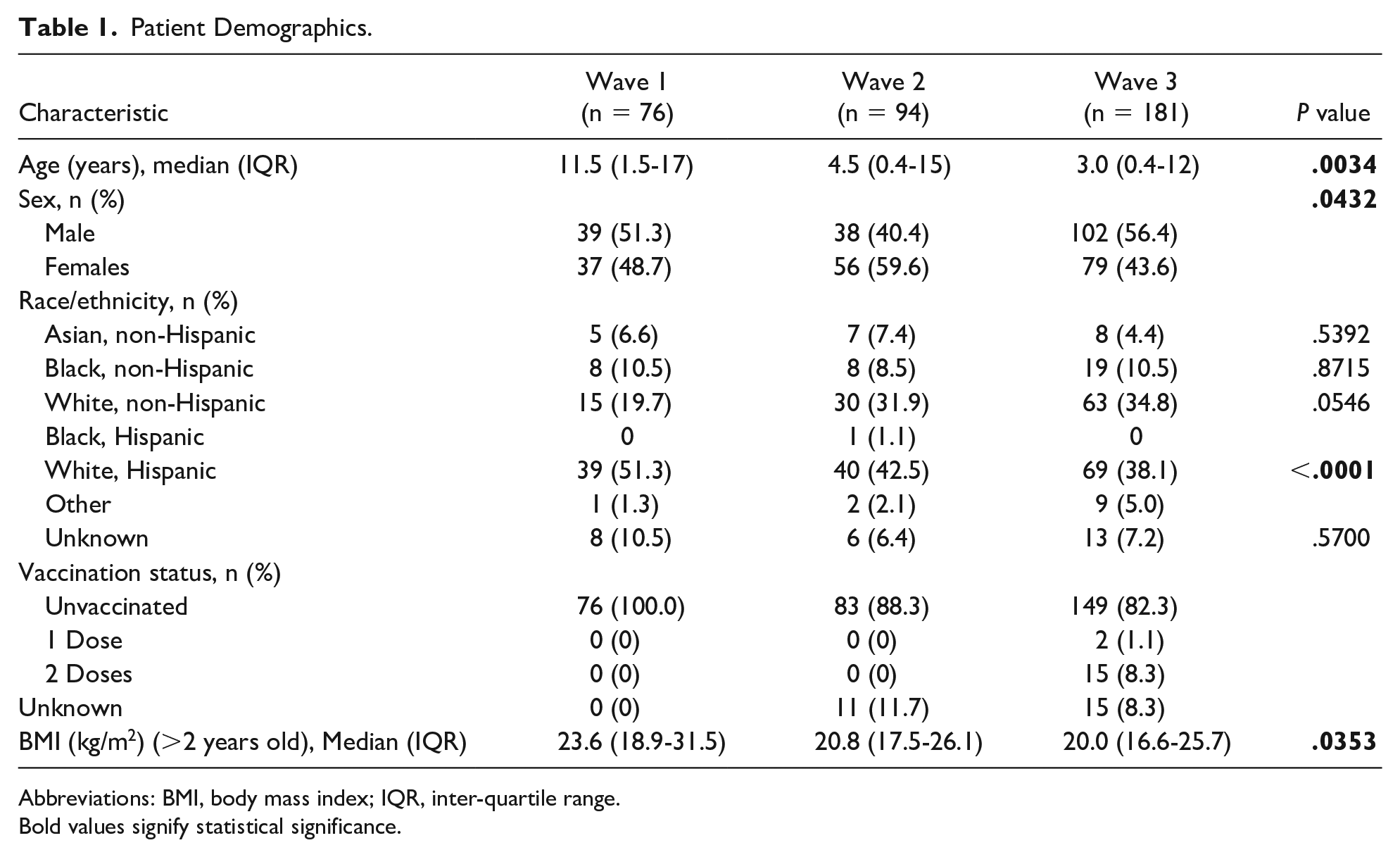
Abbreviations: BMI, body mass index; IQR, inter-quartile range.
Bold values signify statistical significance.
Clinical Presentations
For all 3 waves, the most frequently reported symptoms were fever, cough, and rhinorrhea. The occurrence of fever was significantly higher in wave 2 while the occurrence of rhinorrhea was significantly higher in wave 3 (Figure 1). In all waves combined, the top 4 admission diagnoses were COVID-19 pneumonia, COVID-19 bronchiolitis or croup, gastrointestinal (GI) symptoms, and MIS-C (Table 2). Of the 41 patients with GI symptoms, 4 had other viral co-infections including Adenovirus and 2 had urinary tract infections (UTIs). The overall incidence of pneumonia admissions decreased in wave 3 to 16.6% compared with approximately 34% of all admissions for waves 1 and 2 (P = .0063). COVID-19 bronchiolitis or croup admissions occurred mostly in wave 3 (P = .0063). Admissions due to concern for sepsis were highest in wave 2 (P = .0006). Thirty-eight patients (10.8%) had viral co-infections with the respiratory syncytial virus (RSV) and rhinovirus/enterovirus as the most common viruses identified. Fifty-eight patients (16.5%) had bacterial co-infections: 46 with secondary bacterial pneumonia, 10 with UTI and 4 with bacteremia. Of the 4 patients with bacteremia, 2 had coagulase-negative staphylococci and 2 had Streptococcus mitis isolated. The most common organisms isolated for the UTIs included Escherichia coli (n = 4), Enterococcus faecalis (n = 2), and Pseudomonas aeruginosa (n = 2). There were 40 patients admitted with MIS-C in this study. While the overall number of cases of MIS-C remained similar throughout all 3 waves, the overall proportion of MIS-C diagnosis decreased in wave 3 to 7% of the admissions, compared with approximately 15% of all admissions in waves 1 and 2 (P = .0311). Of the 40 patients with MIS-C, 4 had renal symptoms, 9 had neurological symptoms, and 34 had GI symptoms. The overall number of patients admitted with GI symptoms (without MIS-C) and neurological manifestations including seizures increased in wave 3, but these were not statistically significant (Table 2).
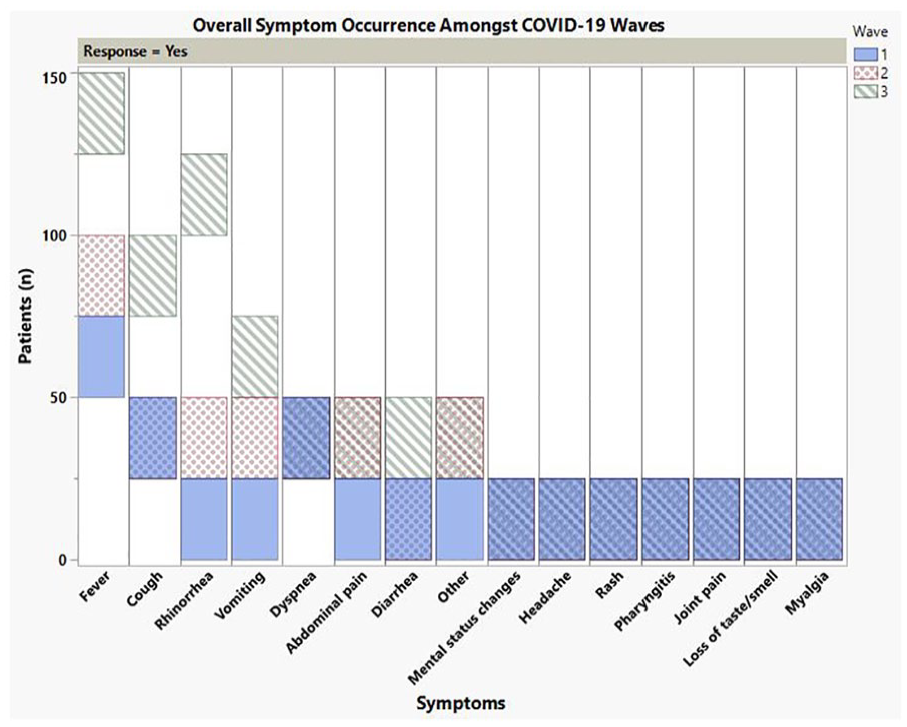
Presenting symptom occurrence amongst COVID-19 waves.
Reason for Hospitalization Per COVID-19 Wave.
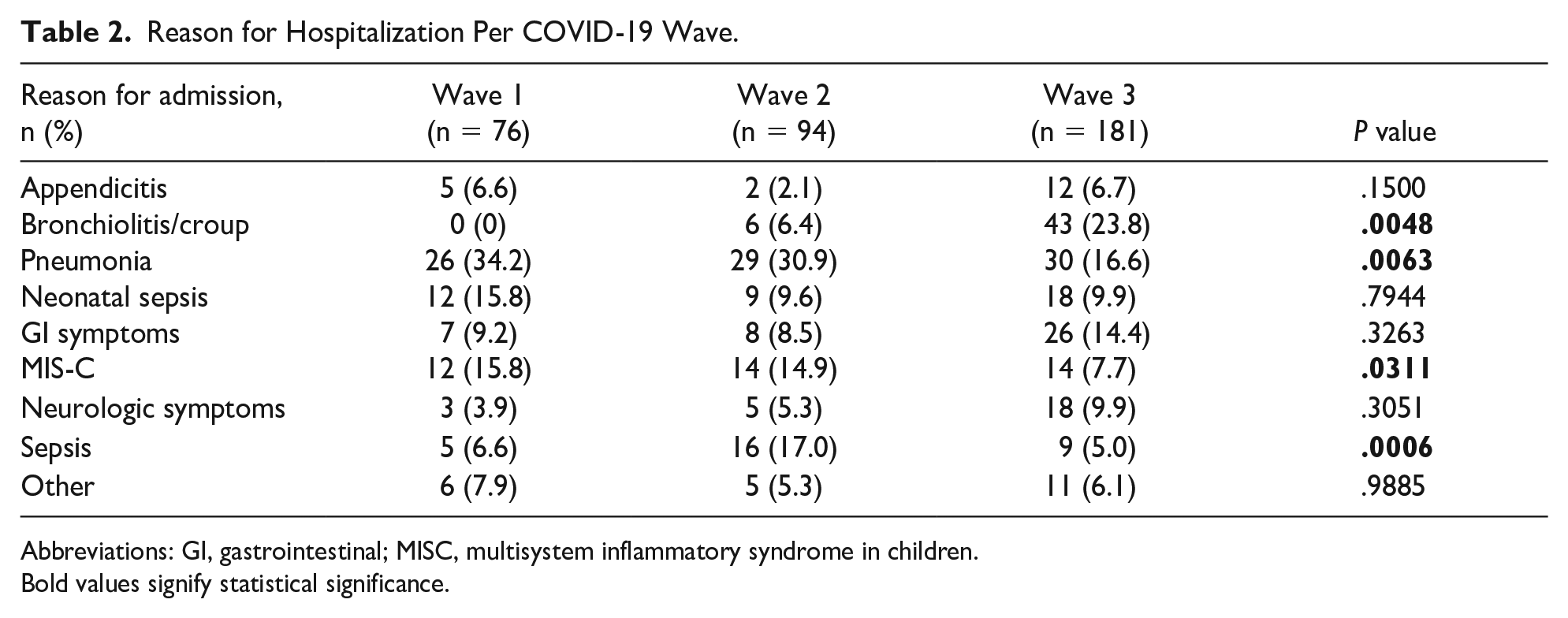
Abbreviations: GI, gastrointestinal; MISC, multisystem inflammatory syndrome in children.
Bold values signify statistical significance.
Hospital Course and Outcomes
Most patients (84%) were admitted to the general pediatric floor. There was not a significant difference in the number of patients requiring intensive care in any particular wave (P = .2188). Length of stay decreased across the waves, with wave 3 having the shortest length of stay in both general pediatric and intensive care units (P < .0001) (Figure 2). The interquartile range for wave 1 was 2 to 7 days, 2 to 5 days for wave 2, and 1 to 3 days for wave 3.
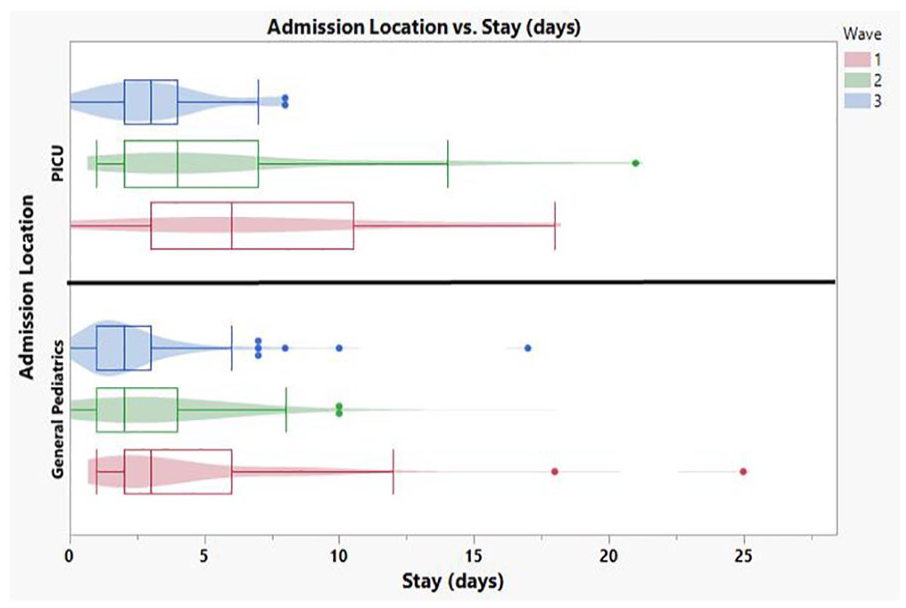
Length of Stay per COVID-19 wave.
Clinical complications including vasoactive medication use outside of the MIS-C population, thrombotic events, concomitant bacterial infection, ARDS, and AKI were rare in all waves. The most common clinical outcome was hypoxia requiring oxygen support during the hospital admission. This outcome showed decreased use throughout the waves (Table 3). Treatments required did not vary between the waves and most commonly included corticosteroids (33% of overall patients). The patients who received steroids included those with COVID-19 pneumonia or ARDS (n = 47), MIS-C (n = 32), and croup (n = 26). Use of antibiotics decreased throughout the 3 waves. In wave 1, 65.8% of patients received antibiotics during their stay; wave 2, 52.1% of patients received antibiotics during their stay; wave 3, 33.2% of patients received antibiotics during their stay (P < .0001). The patients who typically received antibiotics included those with superimposed bacterial pneumonia (n = 54), neonatal sepsis (n = 22), neutropenic fever/concern for sepsis (n = 19), and those with acute appendicitis (n = 19). Antibiotic prescriptions were based on institutional guidelines for each infection and overall, the most commonly prescribed antibiotic was ceftriaxone.
Oxygen Requirement During Hospitalization.

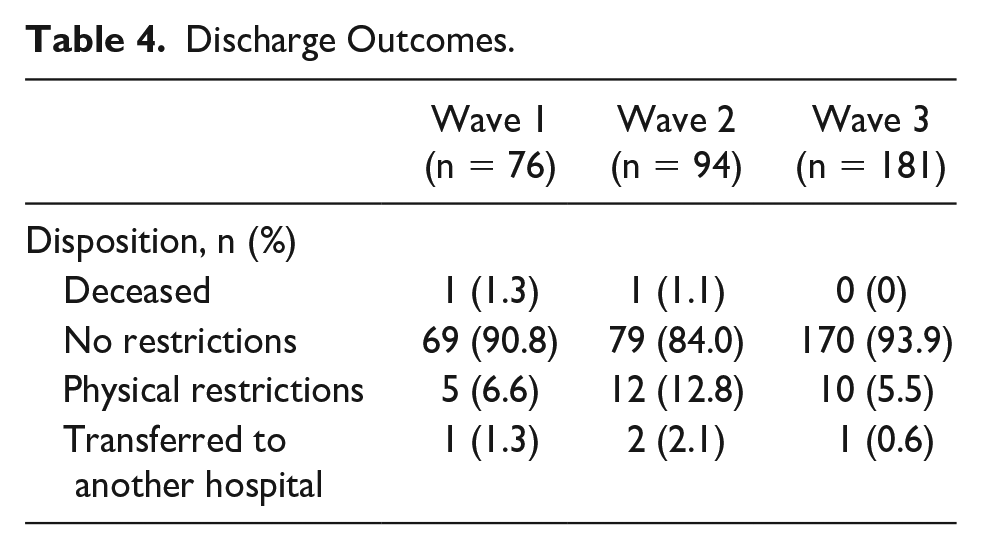
When comparing severity among the waves, only 3 patients met criteria for moderate disease. Severe illness declined from waves 1, 2, and 3 (35.5% [n = 27], 29.8% [n = 28], and 17.7% [n = 32]; respectively). This decrease was significant (P = .0057). Of the 87 patients with severe illness, 28 (32.1%) had bacterial co-infections and 9 (10.3%) had viral-co-infections. As a whole, patients in all 3 waves had favorable discharge outcomes. Two patient deaths were documented in this analysis, and their deaths were likely related to underlying illnesses rather than COVID-19 infection alone. A majority (91%) of our patients were discharged without any physical restrictions. In wave 1, 5 patients were discharged home on oxygen therapy. Wave 2 did have an increase in those discharged with physical restrictions as compared with the other waves with 6 patients that required home oxygen therapy and 6 patients required physical therapy (PT). In wave 3, 5 patients required home oxygen therapy and 5 patients required PT (Table 4).
Discharge Outcomes.
Discussion
COVID-19 cases among children spiked dramatically in 2022 during the Omicron variant winter surge, peaking at 1 150 000 reported cases in 1 week. 10 With the increase in overall pediatric cases, our study noted an increase in hospitalizations during the Omicron variant winter surge in NJ. A study from the Chicago area analyzed the severity of illness amongst the variants, and reported that patients infected with the Gamma variant and Omicron variant without a prior COVID-19 infection or vaccination were associated with increased COVID-19 severity. 11 Our study did not include patients with the Gamma variant as we only had 2 pediatric patients admitted to the hospital network with it compared with the other VoC. We found a statistically significant decrease in the median age of patients admitted in wave 3, compared with waves 1 and 2. One potential contributor to this shift is change in clinical presentation, where 23.8% of hospitalizations in wave 3 were attributed to COVID-19 bronchiolitis or croup which both affect younger children, compared with 0 cases in wave 1 and only 6.4% of cases in wave 2. This is consistent with other reports of increased frequency of croup with the Omicron variant.12-14 Our study also found that the overall proportion of patients admitted with MIS-C during wave 3 declined. A study published in Israel also noted a decline in the incidence and severity of MIS-C with the Omicron variant. 15 We also noted that patients with immunodeficiencies, malignancies and other chronic conditions were not at increased risk of hospitalization in any wave of our study.
Although the number of cases increased with wave 3 and more children across the country required admission, our cohort shows that the overall severity of disease decreased. Patients required less oxygen support, had lower length of stay, and were discharged without physical restrictions more frequently. Our severity classification utilizing specific criteria also showed a significant decrease in severity comparing our patients across the waves of admissions. This supports the assertion that the Omicron variant is less severe, as seen in adult studies across the country as well. 16
Treatment required during the hospitalizations did not change significantly; however, our use of antibiotics did decrease. This may be an indicator of increased understanding of the pathophysiology of COVID-19 amongst providers, but also a result of decreased numbers of sepsis and pneumonia as a reason for admission throughout the waves (Table 2). This portrays an evolving clinical confidence in the treatment of this once novel virus with less unnecessary antibacterial treatments. Limitations of this study include a small sample size with patients from a single network of hospitals in NJ. However, the hospitals are geographically dispersed throughout the state of NJ, and they care for patients of various ethnic backgrounds and socioeconomic statuses, which we consider a strength of our study. The patient population and different VoC recorded in our study may not be representative of that in other states in the United States. In addition, given the widespread community transmission, SARS-CoV-2 detection may also be coincidental with other diagnoses, such as acute appendicitis. Another limitation is that we did not include patients admitted in between the wave time periods and therefore, not every patient with acute COVID-19 and MIS-C was captured, including those with less prevalent variants.
Conclusion
The impact of COVID-19 on the pediatric population has differed from the adult population since the start of the pandemic, though the overall numbers of hospitalized children has mirrored the peaks seen in each infection wave. Our study illustrates the changes in clinical presentation and severity observed with the different coronavirus variants.
Author Contributions
SMB: Contributed to conception and design; contributed to acquisition; Drafted manuscript; critically revised manuscript; gave final approval; agrees to be accountable for all aspects of work ensuring integrity and accuracy.
KNC: Contributed to conception and design; contributed to acquisition; Drafted manuscript; critically revised manuscript; gave final approval; agrees to be accountable for all aspects of work ensuring integrity and accuracy.
HC: contributed to acquisition; critically revised manuscript; gave final approval; agrees to be accountable for all aspects of work ensuring integrity and accuracy.
JG: contributed to acquisition; critically revised manuscript; gave final approval; agrees to be accountable for all aspects of work ensuring integrity and accuracy.
RK: contributed to acquisition; critically revised manuscript; gave final approval; agrees to be accountable for all aspects of work ensuring integrity and accuracy.
TL: contributed to analysis and interpretation; critically revised manuscript; gave final approval; agrees to be accountable for all aspects of work ensuring integrity and accuracy.
SN: contributed to acquisition; critically revised manuscript; gave final approval; agrees to be accountable for all aspects of work ensuring integrity and accuracy.
MP: contributed to acquisition; critically revised manuscript; gave final approval; agrees to be accountable for all aspects of work ensuring integrity and accuracy.
MR: contributed to acquisition; critically revised manuscript; gave final approval; agrees to be accountable for all aspects of work ensuring integrity and accuracy.
PS: contributed to acquisition; critically revised manuscript; gave final approval; agrees to be accountable for all aspects of work ensuring integrity and accuracy.
AS: contributed to acquisition; critically revised manuscript; gave final approval; agrees to be accountable for all aspects of work ensuring integrity and accuracy.
CS: contributed to acquisition; critically revised manuscript; gave final approval; agrees to be accountable for all aspects of work ensuring integrity and accuracy.
CB: contributed to acquisition; critically revised manuscript; gave final approval; agrees to be accountable for all aspects of work ensuring integrity and accuracy.
Supplemental Material
sj-docx-1-cpj-10.1177_00099228231207314 – Supplemental material for Characteristics and Presentations of Hospitalized Children Due to 3 Predominate COVID-19 Variants Within a Health Care Network
Supplemental material, sj-docx-1-cpj-10.1177_00099228231207314 for Characteristics and Presentations of Hospitalized Children Due to 3 Predominate COVID-19 Variants Within a Health Care Network by Sejal M. Bhavsar, Katharine N. Clouser, Hailey Connolly, Jasmine Gadhavi, Ranbir Kaur, Tara Lozy, Srividya Naganathan, Margarette Pierre, Mariawy Riollano Cruz, Pooja Shah, Anita Siu, Carly Swenson and Cathleen Ballance in Clinical Pediatrics
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
The Hackensack University Medical Center IRB approved this study with waiver of patient consent.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
