Abstract

Introduction
The severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) has been reported in the literature to cause persistent symptoms and long-term complications lasting for more than 1 month following acute infection known as long COVID or post-COVID syndrome. 1 However, knowledge of the long-term effects of the SARS-CoV-2 virus infection is still evolving. Better understanding of the varied long-term effects is essential to ensure that health care needs and expectations are met in patients with long COVID and support the development of dedicated medical and psychological services to look after patients experiencing long-term sequelae of the disease. 1
The cell-surface protein angiotensin-converting enzyme 2 (ACE2) has been identified as the main receptor for the SARS-CoV-2 spike protein. Its widespread distribution in various tissues accounts for the manifold manifestations of the disease in several organs. It has been shown to be highly expressed in the intestinal tract as well as the urothelium of the bladder.2-4
The purpose of this report is to present a small case series of 5 children presenting with new-onset bladder and bowel incontinence following COVID-19 infection and a review of the literature for reported potential pathophysiological mechanisms. To the best of our knowledge, this is the first report of this morbidity in children.
All cases are anonymized, and informed consent was obtained from each parent/patient prior to publication.
Cases
The summary of the cases is presented in Table 1.
Cases Summary.
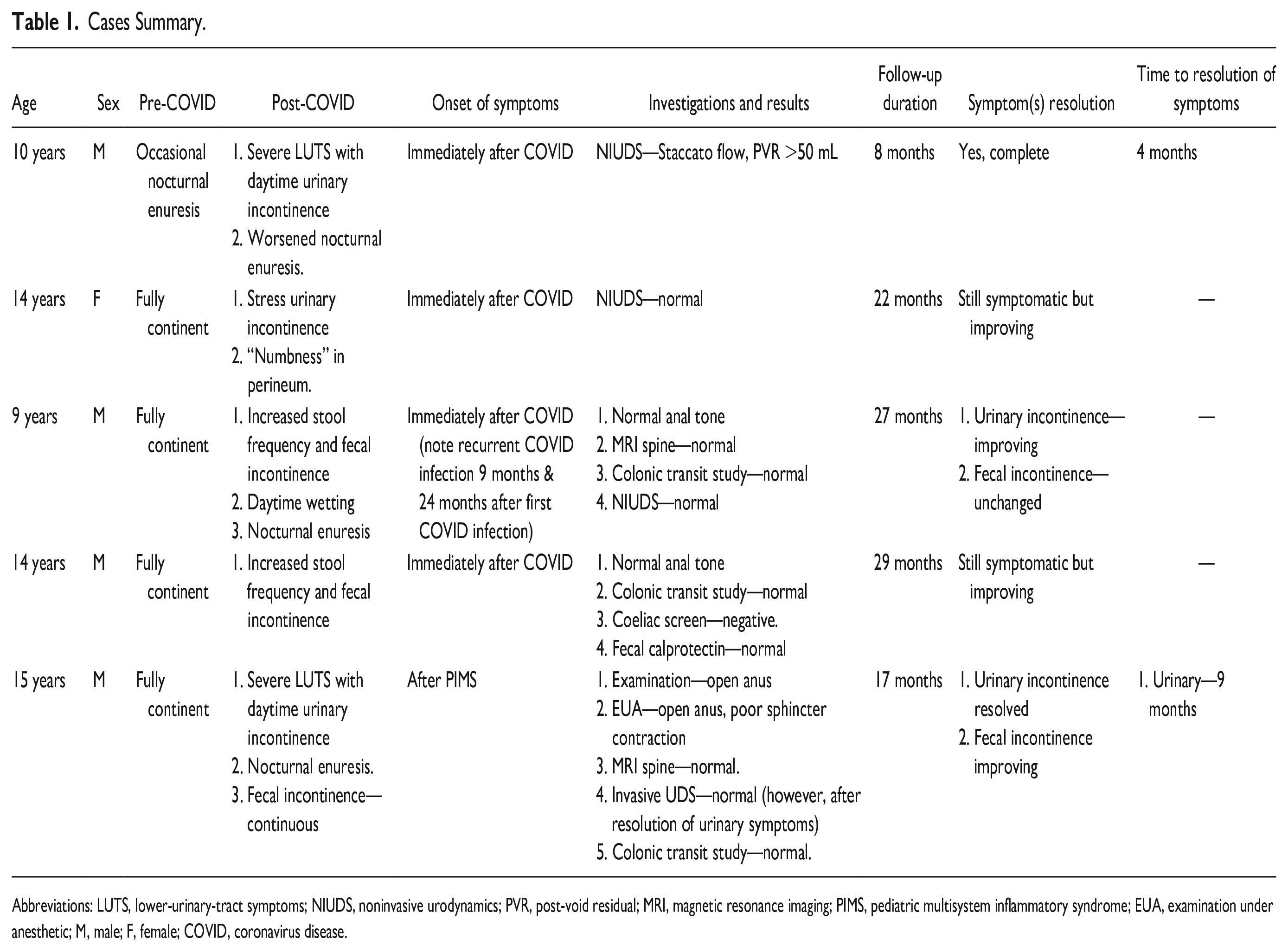
Abbreviations: LUTS, lower-urinary-tract symptoms; NIUDS, noninvasive urodynamics; PVR, post-void residual; MRI, magnetic resonance imaging; PIMS, pediatric multisystem inflammatory syndrome; EUA, examination under anesthetic; M, male; F, female; COVID, coronavirus disease.
In our series, we had 4 males and 1 female presenting with new-onset urinary and/or fecal incontinence following COVID-19 infection. Two of the patients had urinary incontinence only, 1 had fecal incontinence only, and 2 had both. Two of the patients referred for fecal incontinence disclosed symptoms of urinary incontinence on direct questioning only. The majority developed symptoms immediately following the acute infection, with 1 patient developing the symptoms after subsequent pediatric multisystem inflammatory syndrome. In all patients, there appears to be spontaneous improvement of symptoms with only 1 complete resolution during follow-up.
There is increasing recognition of long COVID morbidities including myocarditis, pulmonary fibrosis, encephalitis, thromboembolic events, psychiatric illness, neurological sequelae, and persisting symptoms such as dyspnoea, cough, and fatigue. 1 Our observation of new-onset urinary and fecal incontinence in children may be part of this growing list of long-term morbidities associated with COVID-19.
We reviewed the current literature to explore possible pathophysiological mechanisms that would explain our observation. We summarize reported hypothesized mechanisms below.
Gastrointestinal Tract Invasion By the SARS-CoV-2 Virus
Data show that the ACE2 protein, which has been proven to be a cell receptor for SARS-CoV-2, is abundantly expressed in the gastrointestinal tract, notably in the stomach, duodenum, and rectum. The invasion of the rectum probably accounts for the occurrence of diarrhea in patients with COVID-19. This diarrhea has been shown to persist for up to 90 days after the infection. In the observational report of Weng et al, 2 17 (15%) of the 117 patients reviewed 90 days after COVID reported diarrhea. Constipation was excluded, as was sphincter incompetence. Increased stool frequency was a prominent symptom in 2 of our patients who presented with fecal incontinence.
COVID-19-Associated Cystitis
Chancellor et al 3 were the first US group to identify severe and ongoing genitourinary symptoms in patients with confirmed SARS-CoV-2 infection termed COVID-19-associated cystitis. They demonstrated increased frequency, urgency, and nocturia via an overactive bladder assessment tool in 53 patients discharged following COVID-19 infection compared with 12 asymptomatic controls. Nocturia was the most bothersome symptom. This was also observed in our series, with 3 patients experiencing new or worsening nocturnal enuresis. In addition, Chancellor et al noted increased expression of proinflammatory biomarkers in the urine, further substantiating a potential argument of an inflammatory process in the bladder. The exact mechanism by which this occurs is still unclear. It has, however, been shown that the ACE2 receptor is expressed in the bladder urothelium. Three possible infection routes have been hypothesized: infection via capillaries especially in the setting of viraemia; infection via urine as SARS-CoV-2 has been detected in urine elsewhere; and due to local inflammation (eg, endotheliitis). 4 The mechanism of cystitis may explain the isolated urinary incontinence and absence of fecal incontinence in two of our cases.
Autonomic Dysfunction
Autonomic nervous system (ANS) dysfunction may play a role in the post-COVID bladder and bowel symptoms. Buoite Stella et al 5 administered a Composite Autonomic Symptom Score-31 questionnaire to 180 patients who had recovered from COVID-19 4 weeks to 9 months earlier. Of the 180, 79% and 35% reported gastrointestinal and urinary symptoms, respectively. Gastrointestinal symptoms included diarrhea and nausea while urinary symptoms included involuntary voiding and voiding difficulty. Fourteen percent also reported sexual impairment. This dysautonomia was independent of the presence or absence of neurological symptoms, further highlighting that ANS dysfunction may be an isolated long-term complication of COVID-19.
Proposed mechanisms for COVID-19 associated dysautonomia include the sympathetic overactivation inducing proinflammatory cytokines, immune response producing antibodies to autonomic receptors, and endothelial dysfunction from affectation of the microcirculation, which in turn affects the ANS.5,6
Pudendal Nerve Neuropathy
Neurological complications have been reported in COVID-19 patients. These include headache, anosmia, hyposmia, dysgeusia, impaired consciousness, seizures, encephalitis, and demyelinating neuropathy. SARS-CoV-2 can damage neurons by invading the central and peripheral nervous systems, leading to various neurological complications including demyelination seen following the infection. It is believed that the proinflammatory “cytokine storm” caused by the virus causes neuroinflammation, which eventually results in neuron demyelination.7,8 It also is known that certain viral infections cause various demyelinating diseases in animals and humans. In addition, the acute demyelinating inflammatory polyneuropathy, Guillain-Barré syndrome, has been reported as a complication of COVID-19. 9
Due to the innervation of the urethral and anal sphincters by the pudendal nerve, demyelination of this nerve and subsequent neuropathy lead to bladder and bowel incontinence. With human immunodeficiency virus and herpes zoster infections having been previously known to cause pudendal neuropathy, it is possible that the SARS-CoV-2 virus can cause specific pudendal nerve demyelination. 7 One patient in our series presented with features of a pudendal neuropathy with urinary incontinence and continuous soiling secondary to markedly reduced anal sphincter tone. We were unable to investigate the competence of his urinary sphincter as urinary symptoms had resolved before the invasive urodynamics.
In conclusion, our case series and literature review suggest that the SARS-CoV-2 virus may have long-term effects on bladder and bowel leading to incontinence. Awareness of this potential underreported morbidity is important in order to raise the index of clinical suspicion, identify patients early, and help develop appropriate informed services to support patients suffering long-term effects of COVID-19.
Author Contributions
Victor Emordi: Literature search, Resources, Writing original draft.
Andre Lo: Resources, Literature search.
May Bisharat: Conceptualization, Review and editing, Supervision.
Georgina Malakounides: Conceptualization, Review and editing, Super-vision
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
