Abstract
Hereditary angioedema (HAE) is a rare autosomal-dominant disorder; most cases are characterized by low plasma levels of C1 esterase inhibitor (C1-INH). Clinical manifestations of HAE due to C1-INH deficiency include unpredictable, acute, recurrent episodes of nonpruritic swelling that can affect the face, trunk, limbs, and the respiratory, gastrointestinal, and genitourinary tracts. Attacks can be disfiguring, disabling, painful, and even life-threatening if laryngeal swelling occurs. Symptoms of HAE generally manifest in childhood. Effective medications are available and approved to treat HAE in children. However, evidence informing use of these medications in pediatric clinical practice is limited. Hereditary angioedema management plans are critical to optimize outcomes and should address on-demand treatment for acute attacks and plans to prevent potentially fatal laryngeal attacks. The plan should also comprise a holistic approach to address nonclinical aspects of HAE, including quality of life (QoL) and psychological issues. This article provides an overview of HAE management principles that health care providers can apply to treat pediatric patients to improve their QoL.
Introduction
Hereditary angioedema (HAE) is a rare autosomal-dominant disorder; most cases are associated with mutations in the SERPING 1 gene that codes for C1 esterase inhibitor (C1-INH). 1 Two types of HAE are associated with a dysfunction in plasma C1-INH. Type I HAE, which accounts for ~85% of cases, is characterized by low plasma levels of C1-INH. Type II HAE, which accounts for the remaining ~15% of cases, is characterized by normal levels of C1-INH protein that are dysfunctional. 2 Hereditary angioedema due to C1-INH deficiency is characterized by unpredictable, acute, recurrent episodes of nonpruritic swelling.3,4 Swelling can affect tissues of the face, trunk, limbs, and the respiratory, gastrointestinal, and genitourinary tracts.3,4 Attacks can be disfiguring, disabling, painful, and even life-threatening if laryngeal swelling occurs.3-5 These episodes may occur spontaneously or in response to triggers such as trauma, psychological stress, or infections.3,4 This disease is a result of overproduction of the vasoactive peptide bradykinin secondary to C1-INH levels that are insufficient to inhibit the enzymes plasma kallikrein and activated Factor XII.6,7
Symptoms of HAE generally manifest in childhood, with the age of symptom onset ranging from 4.4 to 18 years.4,8 While on rare occasions attacks may occur in the first year of life, there has been a single documented case of an HAE attack occurring in utero in a mother who had a facial attack while in active labor.9,10 The mean age of the first HAE episode is 10 years, and approximately 40% of patients experience their first episode by age 5 years.3,4,8
The onset of clinical symptoms at an early age is associated with a more severe disease course, including more HAE episodes per year and more hospitalizations for HAE-associated symptoms. 10 Hereditary angioedema episodes typically become more frequent at puberty. 11 Both early onset of HAE and frequency of attacks are associated with a greater negative impact on a child’s quality of life (QoL) and mood.11-13 The disease itself and HAE attacks impact QoL, including physical, emotional, and psychosocial domains. 14 While no studies have specifically examined the psychological impact of HAE in children and adolescents, multiple studies have documented the negative impact of chronic illness on psychological domains in children and adolescents. 15
The median time from symptom onset to HAE diagnosis is estimated at 8.5 years. Diagnosis is often delayed, even among those with a family history of HAE. 16 Diagnostic delays in pediatric patients may be due to the fact that HAE is a rare disorder and that many HAE symptoms can mimic those of more common conditions. Furthermore, 25% of patients have no family history of angioedema. 17 Delays in diagnosis can defer effective treatment and result in inappropriate treatment or unnecessary surgical interventions. 16
Effective medications are available and approved to treat HAE in children. However, evidence informing use of these medications in pediatric clinical practice is limited because clinical trials of these agents have focused mostly on adults and have underrepresented pediatric patients. 1
Management plans are critical to optimize outcomes and should address treatment, on-demand treatment for acute attacks, and forestall potentially fatal laryngeal attacks. The plan should also comprise a holistic approach to address nonclinical aspects of HAE, including QoL and psychological issues.
The objective of this article is to provide an overview of HAE management principles that health care providers can apply to treat pediatric patients to improve their QoL.
Diagnosis
Early identification of HAE is critical to reducing mortality and morbidity and to developing an appropriate management plan. The first HAE episode may be a laryngeal attack, which can be fatal.1,5,18 An analysis of 70 patients with HAE who had asphyxiated during a laryngeal attack showed that mortality was higher in patients with undiagnosed HAE (63 cases) than in patients with diagnosed HAE (7 cases).2,5 Upper airway swelling is also more severe in pediatric patients due to the smaller diameter of the airway, which increases the risk of asphyxiation. 1
Barriers to early diagnosis include poor recognition of HAE symptoms by health care professionals, a lack of appropriate family screening tools, and a lack of access to simple laboratory tests to help establish a diagnosis.16,19-21 Symptoms typically present in childhood, and delays in diagnosis can subject patients to inappropriate treatment and unnecessary medical interventions. 16
Despite its familial association, many family members of patients with HAE are not screened for the disorder. 21 A survey of patients with HAE found that only 48% of immediate family members and 26% of extended family members had been tested for the condition. 21 With the availability of effective therapies, screening of family members has increasingly been emphasized as a way to reduce the substantial morbidity and mortality associated with HAE. 21 Each child of a parent with HAE has a 50% chance of inheriting the disorder; therefore, all infants with a family history of HAE should be considered to have the disorder until the diagnosis is ruled out.1,21
Clinical Presentation
The clinical course and presentation of HAE vary. However, the hallmark symptoms of all forms of HAE include recurrent angioedema causing cutaneous swelling, abdominal symptoms from gastrointestinal angioedema, and respiratory symptoms due to airway involvement. 1 In children, bowel wall edema with resultant abdominal pain and swelling of extremities are the most frequently reported initial symptoms.1,22 Hereditary angioedema is not associated with urticaria or pruritus, but patients can exhibit prodromal symptoms of erythema marginatum, an evanescent, nonpruritic macular rash. 1 In contrast to histamine-induced angioedema, swelling due to HAE is protracted; untreated HAE symptoms often last 3 to 5 days from start to resolution. Protracted episodes are associated with considerable morbidity and mortality if not treated with effective medication. 1 The lack of a clinical response to treatment with antihistamines, corticosteroids, or epinephrine should raise suspicion of HAE. 1 Table 1 lists signs and symptoms that can alert health care providers to the possibility of HAE. Figure 1 outlines the World Allergy Organization (WAO)/European Academy of Allergy and Clinical Immunology (EAACI) recommended diagnostic workup in patients suspected to have HAE. 20
Signs and Symptoms That may Suggest HAE. 1

Abbreviation: HAE, hereditary angioedema.
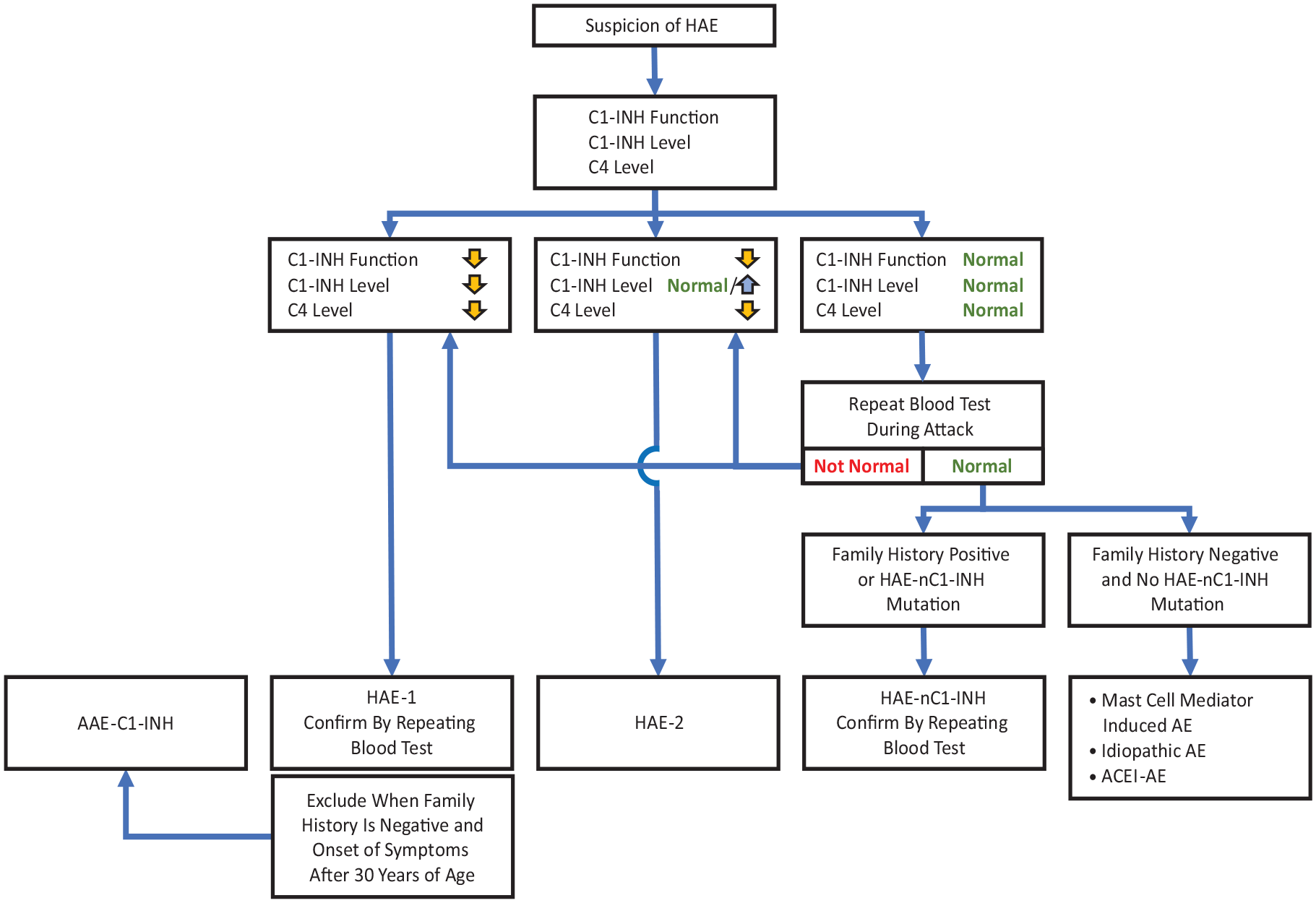
HAE workup algorithm.
Blood Testing
Measurement of C1-INH antigenic and functional levels is required to identify or exclude HAE-C1-INH. 1 Measuring quantitative (antigenic) and functional activity of C1-INH and plasma complement protein 4 (C4) generally will be sufficient to establish a diagnosis. 2 Although low C4 and C1-INH functional levels are clinically diagnostic, genetic mutation analysis is the ultimate confirmatory step; however, this last step is usually cost-prohibitive. 1 A normal C4 test during an episode of angioedema excludes a diagnosis of HAE-C1-INH in most instances because C4 levels decrease further during episodes of swelling.1,7 Blood levels of C4 proteins and C1-INH are low at birth, so testing for these markers is recommended after 1 year of age. 1
Genetic Testing
Genetic testing may be necessary in specific clinical scenarios, including differentiation of HAE-C1-INH from acquired C1-INH deficiency, prenatal testing due to family request, or situations where repeated biochemical C1-INH test results are ambiguous. 1
In patients with a known family mutation for HAE, DNA analysis from cord blood or peripheral blood is sufficient to quickly establish a diagnosis. Even if the family mutation is not known, most children will have the same genetic abnormality as the parent. Therefore, both the parent’s and child’s genes should be tested simultaneously. 2
If antibody-based testing is not conclusive for C1 functional deficiency but a diagnosis is still suspected, a chromogenic C1-INH functional assay is more sensitive than an ELISA and can help provide a definitive diagnosis. 1
Treatment Options for Pediatric Patients
The principles guiding pharmacologic management of HAE include the following:1
Availability of effective on-demand acute therapy for all patients;
Early treatment to prevent attack progression;
Treatment of attacks irrespective of the site of swelling;
Incorporation of long-term prophylaxis (LTP) based on shared decision-making.
Children with HAE should have access to ≥2 standard doses of US Food and Drug Administration (FDA)-approved medicines to treat unpredictable angioedema episodes. 2 While on-demand treatment alone can be sufficient for some patients, first-line prophylactic therapy together with on-demand treatment of breakthrough attacks may be indicated for others. 2
Currently, 7 products are approved by the FDA for use in pediatric populations: 3 are indicated for acute HAE attacks (on-demand treatment), and 4 are indicated for prophylaxis. Therapies approved in the United States for treatment of HAE in pediatric patients are listed in Table 2.
On-Demand and Prophylaxis Medications Approved in the United States and Recommended by Guidelines for Treatment of HAE in Pediatric Patients. 1
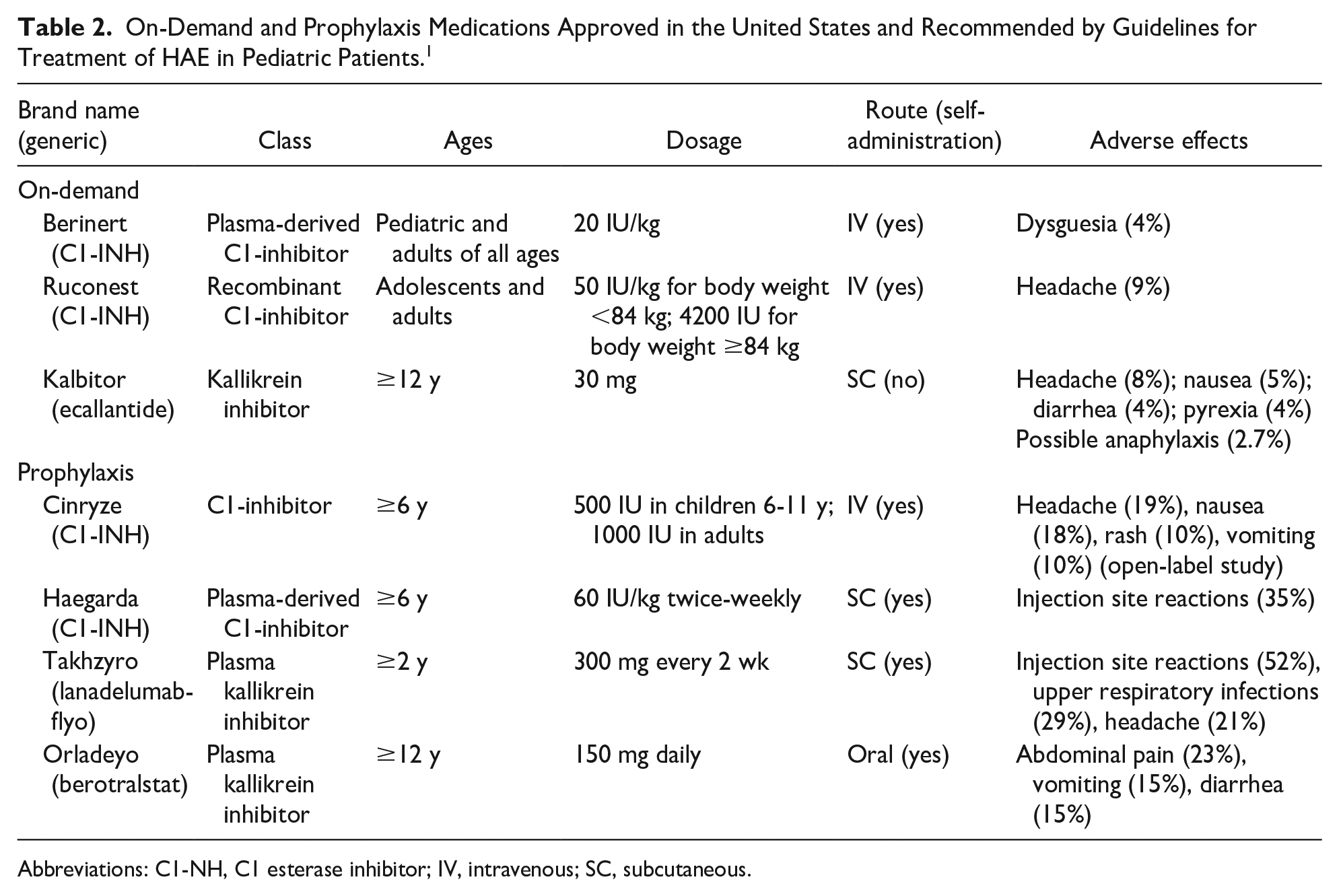
Abbreviations: C1-NH, C1 esterase inhibitor; IV, intravenous; SC, subcutaneous.
On-Demand Treatment
The goal of on-demand acute therapy for HAE is to minimize morbidity and prevent mortality from an ongoing swelling episode. 1 While on-demand treatments prevent swelling from progressing, it may take between 30 minutes and 2 hours for swelling to resolve after the medication is administered. 1 It is imperative to convey to parents that early treatment leads to better symptom control. 1 A second dose generally is not needed unless the swelling continues to worsen. 1
Based on improved understanding of the pathophysiologic mechanism of swelling in HAE-C1-INH, 23 3 products have been approved for on-demand treatment in pediatric populations (Table 2). Each product has been shown in randomized, controlled studies to be effective and safe,1,24-26 and these findings have been confirmed in subsequent open-label extension studies and patient registries.1,27-31
C1 esterase inhibitors
Four C1-INH medications are available in the United States: 3 are derived from human plasma and 1 is a recombinant form. The 3 human plasma–derived C1-INH medications for HAE available in the United States are the following: Berinert (C1-INH [human]; CSL Behring, LLC) is approved for on-demand treatment in children 32 ; Haegarda (C1-INH subcutaneous [human]; CSL Behring, LLC) is approved for prophylaxis in children 33 ; and Cinryze (C1-INH [human]; ViroPharma Biologics, Inc.) is approved for prophylaxis in adults and adolescents 34 and has been studied for acute episodes in children, although it is not approved for on-demand therapy in the United States. A recombinant form of C1-INH, Ruconest (C1-INH [recombinant]; Pharming N.V.), is approved for treatment of acute episodes in adolescents and adults. 35 Haegarda is given subcutaneously, and the other agents are given intravenously. Dosages of these medications are based on weight.
Purified plasma-derived C1-INH (Berinert) is the only on-demand therapy that has been approved for children of all ages by the FDA. 2 As the fundamental problem in HAE is decreased C1-INH function, this therapy directly addresses the cause of swelling episodes. Plasma-derived C1-INH has been used in Europe for decades, and numerous studies have demonstrated the efficacy and safety of plasma-derived C1-INH in pediatric patients. 2 The 2017 WAO guidelines for the treatment of HAE recommend plasma-derived C1-INH as the preferred therapy for on-demand treatment in pediatric patients. 20
Ecallantide
Ecallantide (Kalbitor; Dyax Corp.) is a plasma kallikrein inhibitor approved for treatment of HAE episodes in patients aged ≥12 years. 36 An analysis of pooled data suggests that ecallantide is effective for treating HAE episodes in younger patients and has an acceptable safety profile.2,37 Ecallantide has been associated with hypersensitivity reactions in a small percentage (3%-4%) of recipients and therefore has a boxed warning that states it must be administered under the supervision of medical personnel. 2
Prophylaxis
Prophylaxis can be considered for both short-term and long-term treatment. The goal of short-term prophylactic treatment is to reduce the risk of swelling in a child undergoing a stressor or procedure (eg, a medical, dental, or surgical procedure) that could trigger an episode. Long-term prophylaxis aims to reduce the overall frequency, severity, and burden of angioedema episodes. 1
Prophylaxis treatment should be individualized and considered in all severely affected patients. Considerations include disease severity, frequency of attacks, QoL, availability of health care resources, and failure to achieve adequate control with appropriate on-demand therapy.
The plasma-derived C1-INH agents Cinryze and Haegarda, and the kallikrein inhibitors Takhzyro (lanadelumab-flyo; Dyax Corp) and Orladeyo (berotralstat; Biocryst Pharmaceuticals, Inc.) are approved for prophylaxis in children.38,39 Prophylactic treatment options differ with respect to route of administration, potential for adverse effects, and efficacy. Therefore, patient experience and preference are important considerations when deciding which therapy is the most appropriate. 1
Intravenous and subcutaneous formulations of C1-INH are available for routine prophylaxis in pediatric patients with HAE. 10 Using intravenous C1-INH for LTP requires repeated venous access, which may be difficult to sustain over time and can lead to local and systemic complications.10,40 By contrast, subcutaneous administration of C1-INH prophylactic therapy can provide a convenient alternative to intravenous therapy for younger patients and their caregivers.10,41
Plasma-derived C1-INH is the recommended therapy for LTP in children with HAE per the 2017 WAO guidelines. 20 The Hereditary Angioedema International Working Group 2017 Consensus Guidelines also consider plasma-derived C1-INH to be the safest long-term prophylactic option for children. Androgens and antifibrinolytics are not recommended in children. 10
A recent subgroup analysis of an open-label trial investigating the efficacy and safety of C1-INH (subcutaneously) in pediatric patients showed that those treated with the approved dosage of 60 IU/kg experienced a significant reduction in attacks compared with prestudy values (Table 3). 10 Data from pediatric patients treated with C1-INH (subcutaneously) for up to 2.55 years showed no differences in efficacy compared with adult patients. 10
Efficacy Outcomes in Pediatric Patients Treated Long-Term With C1-INH (SC) 60 IU/kg. 10
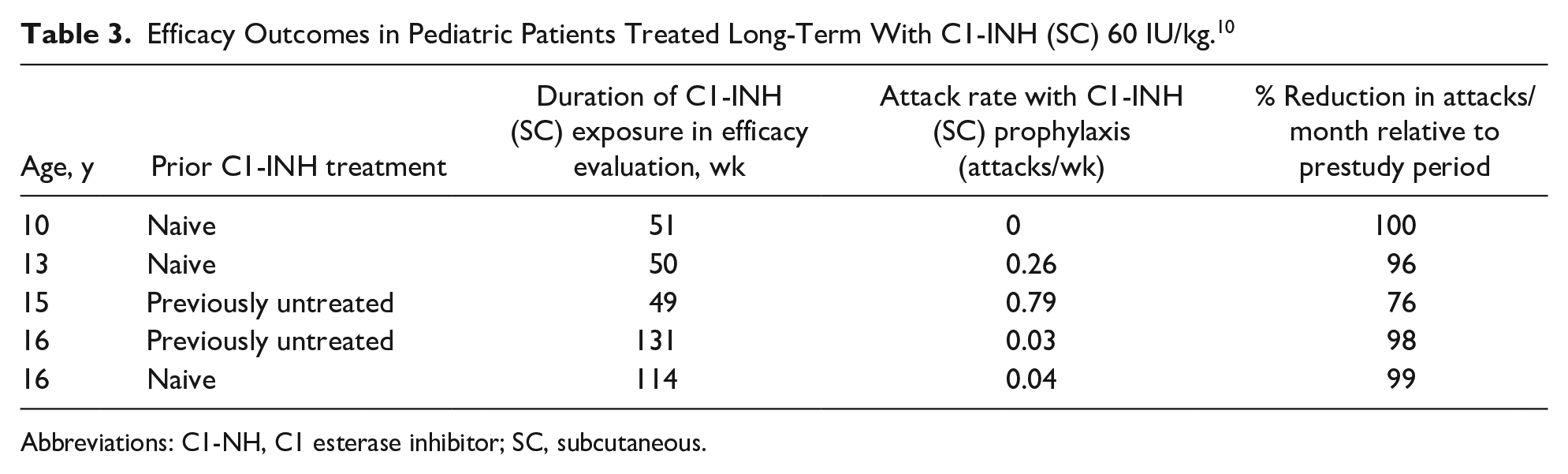
Abbreviations: C1-NH, C1 esterase inhibitor; SC, subcutaneous.
C1-INH (subcutaneously) was well tolerated within the pediatric subgroup. 10 Injection-site erythema was the most common treatment-related adverse event (Table 4). All injection-site reactions resolved, and all cases of injection-site erythema were mild except for one. No one in the pediatric subgroup discontinued treatment due to a treatment-related AE, and no incidents of serious AEs, thromboembolism, or anaphylaxis were reported. 10
Safety Profile of C1-INH (SC) 60 IU/kg in Pediatric Patients. 10
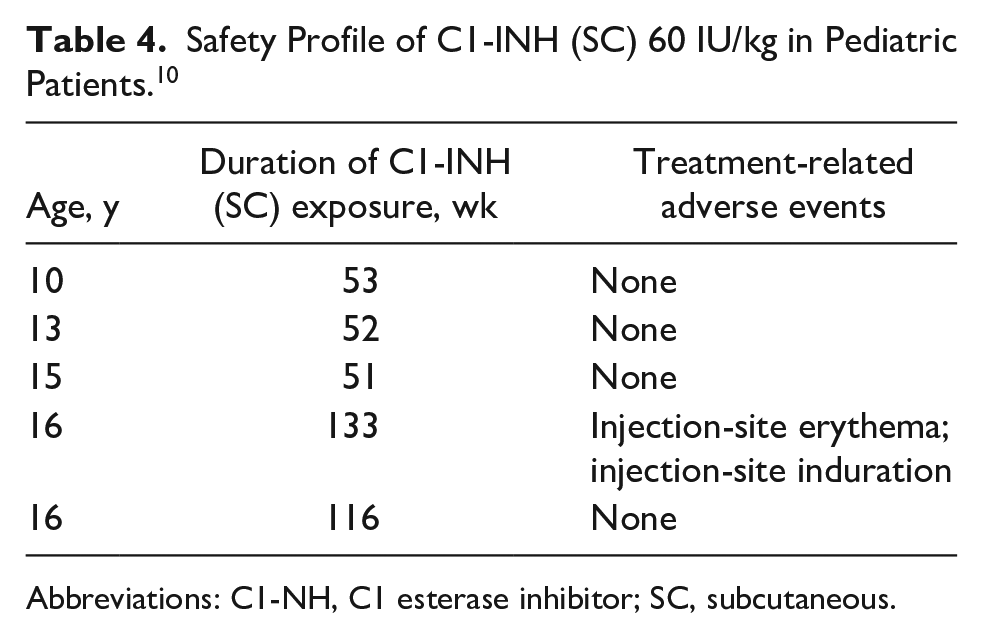
Abbreviations: C1-NH, C1 esterase inhibitor; SC, subcutaneous.
Rethinking Pediatric HAE Care: A Holistic Approach
As described in the introduction, children with HAE deal with a significant disease. Both HAE attacks and the disease itself impact QoL in several domains, including physical, emotional, and psychosocial. 14 The unpredictable nature of HAE episodes contributes in part to decreased QoL, so providing effective prophylactic treatment and reducing the frequency of HAE episodes is an important step toward improving pediatric patients’ QoL.14,42 Clinicians should consider the overall impact of effective management on the child’s QoL when planning treatment.
Prophylaxis can reduce the burden of illness associated with HAE. 39 The need for prophylaxis is not based on age; pediatric patients meeting eligibility criteria should be considered for prophylaxis and assessed in the same way as adults are.
In addition to providing effective LTP, clinicians also should consider an integrative holistic approach to HAE that offers opportunities to address the psychological and emotional needs of pediatric patients with HAE. Emotional stress is a known trigger for HAE episodes, and children may suffer significant emotional stress due to HAE attacks, which diminishes QoL. Anxiety and depression are also common among HAE patients, and QoL decreases as anxiety and depression become more severe. 43
Although standard medical therapy addresses the biological aspects of HAE, complementary integrative therapies (eg, hypnotherapy, music therapy) can support medical approaches and address psychosocial aspects of HAE to improve the patient’s overall experience and QoL.
Music therapy is one option among many complementary modalities that can address pain, anxiety, and stress in general. Music therapists help patients move their conscious thoughts away from the symptoms and divert their attention from stressful events, thereby reducing anxiety and perceived pain. 44 Music has been shown to calm patients before and during a variety of medical procedures, from injections to surgeries, and patients who listen to music before a procedure show fewer symptoms of anxiety. A meta-analysis of 29 observational studies involving children and adolescents undergoing invasive and noninvasive medical procedures demonstrated that music therapy can reduce pain, anxiety, and distress. 45 Because stress is a known trigger for HAE episodes, alleviating stress with complementary therapies could also lessen the disease burden and improve QoL.
In addition to stress reduction, another potential use of music therapy in the setting of HAE is to facilitate intravenous or subcutaneous administration by reducing a child’s anxiety and pain perception during administration. 46 Venipuncture and intravenous placement have been identified as the most painful and feared medical experiences among children.46,47 Several studies have assessed the impact of music therapy on pediatric patients receiving injections or infusions. Children (N = 64; ages 4-6.5 years) receiving music therapy during routine immunizations were less distressed and reported less pain than children who were not exposed to music therapy. 48 Another study of 42 children aged 3 to 11 years showed that those who listened to relaxing music during intravenous insertion reported significantly less pain, and some demonstrated significantly less distress, compared with children who did not listen to music. 46 Furthermore, 76% of health care providers reported that intravenous infusions were “very easy” to administer among children who were listening to music, compared with 38% of providers treating children without music. 46
Effective prophylaxis therapy improves biological, psychological, and social QoL in children with HAE. Therefore, any modality that makes it easier for a child to receive an injection or intravenous infusion, whether by directly reducing the child’s stress and fear or by reducing parents’ concerns about subjecting their child to multiple injections/infusions, supports overall QoL.
Summary
It is important to be aware of HAE as a cause of recurrent swelling in the absence of urticaria. The fact that 25% of patients do not have a family history is not an impediment to diagnosis because a C4 level and C1 inhibitor determination by protein and by function is diagnostic and has the potential to decrease the unnecessary lag time between clinical presentation and the initiation of appropriate therapy.
Hereditary angioedema cannot be cured, but effective treatments can reduce disease burden and improve QoL in children with HAE. Initial clinical symptoms of HAE typically occur in childhood. Early recognition by all health care professionals, particularly those caring for pediatric patients, reduces the risk of morbidity and mortality as well as unnecessary interventions. Alternative modalities, such as music therapy and other complementary therapies, can supplement medical management and improve a pediatric patient’s treatment experience by facilitating administration and reducing the child’s stress, anxiety, pain perception, and fear, and can relieve parental concerns about multiple injections. All these benefits contribute to an increased QoL for children with HAE.
Multiple therapies are available that can abort acute episodes of angioedema and therapy that can be administered regularly as prophylaxis to prevent them. These can be life-saving given the known mortality statistics when this disorder is untreated.
Author Contributions
RT: Contributed to conception and design; contributed to analysis; critically revised the manuscript. APK: Contributed to conception and design; contributed to analysis; critically revised the manuscript.
Footnotes
Acknowledgements
Editorial and medical writing assistance was provided by NCF Health, LLC, and funded by CSL Behring.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Raffi Tachdjian- Speaker or advisory board: Biocryst, Takeda, CSL Behring, Pharming; Research contracts: Biocryst, Takeda, CSL Behring, Pharming, Kalvista, Pharvaris, Ionis.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
