Abstract
Urinary tract infections (UTIs) are a common reason for hospitalization in infants younger than 60 days, and the optimal approach to intravenous (IV) antibiotic therapy upon UTI diagnosis in this cohort is unknown. We determined whether there was an association between IV antibiotic therapy duration (long [>3 days] vs short [≤3 days]) and treatment failure via a retrospective review of infants with confirmed UTIs receiving IV antibiotics at a tertiary referral center. A total of 403 infants were included; 39% were treated with ampicillin and cefotaxime, and 34% with ampicillin and gentamycin or tobramycin. The median IV antibiotic duration was 5 (interquartile range: 3-10) days, and 5% of patients experienced treatment failure. The treatment failure rate was similar in both short- and long-course IV antibiotic groups (P > .05), and there was no significant association between treatment duration and failure. We conclude that treatment failure for infants hospitalized with UTI is uncommon and not associated with IV antibiotic duration.
Introduction
Urinary tract infections (UTIs) are a common reason for hospitalization in young infants, with more than 20 000 infants (<1 year of age) hospitalized annually in the United States.1,2 In children aged 2 to 24 months, the American Academy of Pediatrics guidelines from 2011 (reaffirmed in 2016) state that “initiating treatment orally or parenterally is equally efficacious.”1,2 Infants younger than 60 days often initially receive parenteral antibiotics as part of an evaluation for invasive bacterial infection. The optimal approach to intravenous (IV) antibiotic therapy when a UTI is diagnosed in these younger infants is not known. 1 As a result, significant practice heterogeneity exists both within and between institutions.3-6
Prescribing patterns and the appropriate duration of antibiotic therapy are a priority for the Centers for Disease Control and Prevention, hospital systems, and individual care providers alike as it has a big impact on antimicrobial resistance. 7 Moreover, a short-course IV therapy could provide additional potential benefits compared with a long-course therapy. Shorter hospitalizations would mean lower medical costs and increased hospital bed availability and may be less disruptive to parent-infant bonding. 8 In addition, shorter hospitalization could reduce the risk of nosocomial infection 9 and adverse events, such as IV infiltrates. This approach is also in keeping with value-based health care delivery.
The goal of this study was to determine the association between short-course (≤3 days) and long-course (>3 days) IV antibiotic therapies and treatment failure, defined as readmission from date of discharge within 30 days. We hypothesized that a short-course IV antibiotic therapy followed by oral antibiotics is safe, with no difference in treatment failure between short-course and long-course IV antibiotic therapy.
Methods
The study was approved by the institutional review board at our pediatric tertiary care center (REB #10000062307). We retrospectively reviewed the medical records of all infants <60 days old with confirmed bacterial UTIs who were admitted and received IV antibiotics between January 1, 2008, and December 31, 2017. Primary and secondary diagnosis codes for acute UTI or pyelonephritis (International Classification of Diseases, Ninth Revision, Clinical Modification, code 590.10, 590.11, 590.80, or 599.0) were included in the initial cohort.
The diagnosis of UTI was deemed certain if urine cultures obtained through suprapubic aspiration showed gram-negative bacteria (any amount) or >10 × 106 colonies per liter of gram-positive bacteria or if urine cultures obtained through bladder catheterization showed >50 × 106 colonies per liter of a single pathogen (excluding lactobacilli, corynebacteria, and coagulase-negative staphylococci) or >10 × 106 colonies per liter of Pseudomonas species.10,11 Those treated for UTI who did not meet the aforementioned criteria were excluded for the purpose of this study. We also excluded any infants with catheter-associated UTI, prior urological procedures, mixed-growth urine cultures, fungal growth, and bacterial meningitis. Covariates of interest included patient age, sex, prematurity (defined as gestational age <37 weeks), comorbidities, clinical parameters such as systolic blood pressure, creatinine level, presence of fever (ie, a reported temperature ≥38.0 °C [100.4 °F] at home, in an outpatient clinic, or documented in the emergency department and obtained via any method), ill appearance, causative pathogens, blood culture, imaging modalities, type and length of antibiotic treatment (oral/IV), caring team, and length of stay. Ill appearance was defined as any of the following physical examination terms documented on arrival to the emergency department: “ill appearing,” “toxic,” “limp,” “unresponsive,” “gray,” “cyanotic,” “apnea,” “weak cry,” “poorly perfused,” “grunting,” “listless,” “lethargic,” or “irritable.” 12 If none of these terms were documented, the infant was classified as alert. Nosocomial UTI was defined as any infant who developed UTI during hospital stay for other reasons, except those with catheter-associated UTI.
Short-course IV antibiotic treatment was defined as the receipt of IV antibiotics for ≤3 calendar days, and long-course treatment was defined as the receipt of IV antibiotics for >3 calendar days. One or more doses of an antibiotic on a given calendar day was considered a full day of treatment. The main outcome was readmission for a UTI, which is defined as any readmission within 30 days of the index discharge with an International Classification of Diseases, Ninth Revision, Clinical Modification, code related to UTI present at admission. We chose 30 days because this time would allow more than 2 weeks’ period without antibiotics for all infants who completed a typical 10- to 14-day course, including oral antibiotic treatment at home. A UTI diagnosis within this period likely would be related to treatment failure. 13
Descriptive statistics were performed for categorical and continuous variables, and data were summarized as median (interquartile range [IQR]), counts and percentages. For comparison of each group, chi-square and Fischer’s exact tests were used for categorical variables and Mann-Whitney U test for nonparametric continuous data. Univariate and multivariable logistic regression were used to examine the association between the primary exposure variable (short vs long IV treatment) and the odds of readmission for a UTI and for all causes within 30 days. Statistical analyses were performed with STATA (16.1; StataCorp, College Station, Texas). All P values were calculated by 2-tailed distribution, and values <.05 were considered significant.
Results
During the study period, 403 patients were included (Figure 1), of which 236 (59%) received long-course IV antibiotics, and 167 (41%) patients received short-course IV antibiotics. The characteristics of patients in both groups are presented in Table 1.
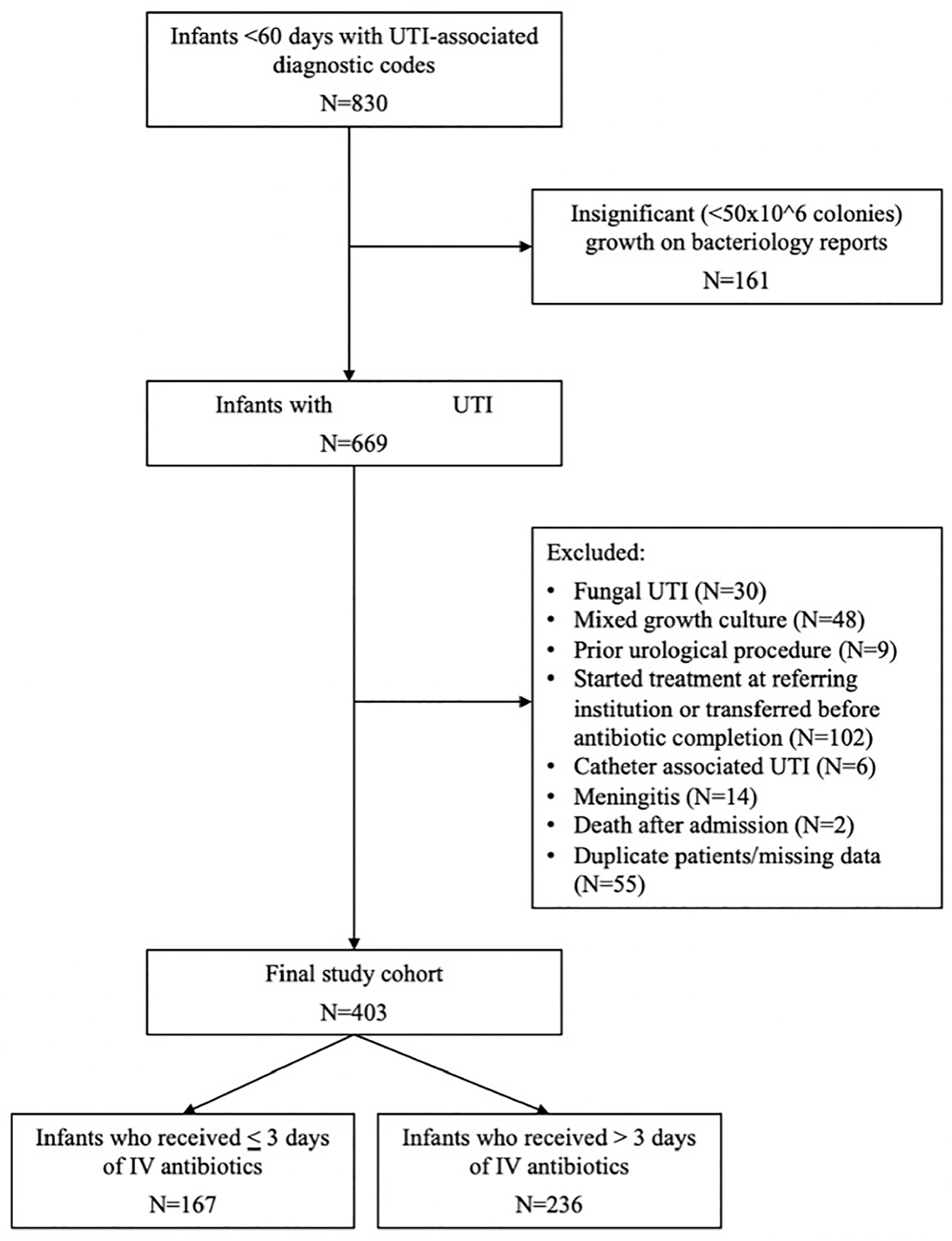
Algorithm of infants selected for the study.
Descriptive Characteristics of the Study Population.
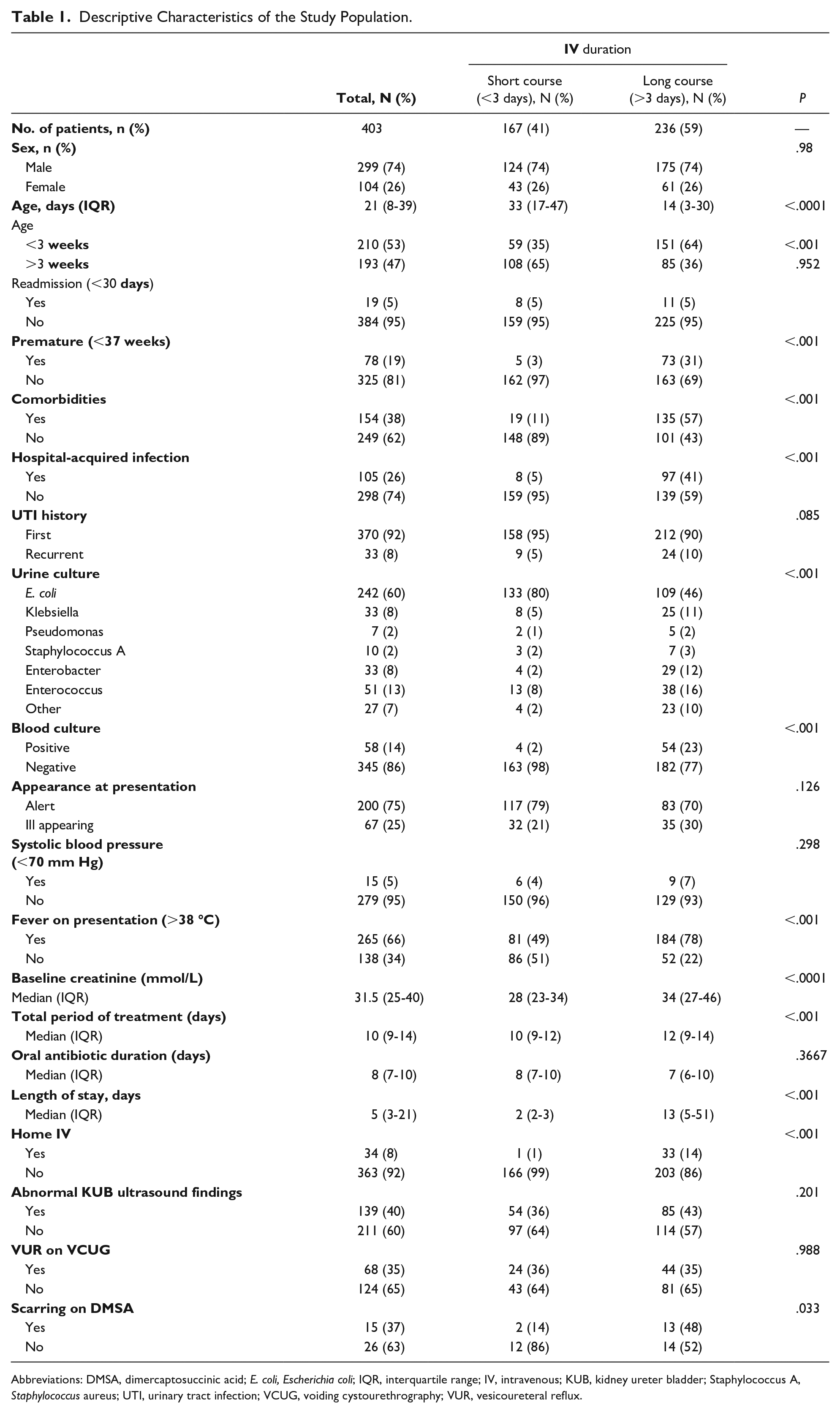
Abbreviations: DMSA, dimercaptosuccinic acid; E. coli, Escherichia coli; IQR, interquartile range; IV, intravenous; KUB, kidney ureter bladder; Staphylococcus A, Staphylococcus aureus; UTI, urinary tract infection; VCUG, voiding cystourethrography; VUR, vesicoureteral reflux.
Out of the total cohort, 299 (74%) were male, and the median age was 21 (IQR: 8-39) days. The microorganisms isolated in the urine culture were as follows: Escherichia coli in 242 (60%) cases, Klebsiella species in 33 (8%) cases, Enterobacter species in 33 (8%) cases, Enterococcus faecalis in 51 (13%) cases, and Pseudomonas aeruginosa and Staphylococcus species in 7 and 10 cases, respectively (2%). Blood cultures were done for all patients, of which 58 (14%) proved positive. In all cases, the microorganism isolated in the blood culture matched that of the urine culture. In 240 (60%) cases, lumbar puncture was performed, and none of these cases had a positive cerebrospinal fluid culture. One hundred fifty-five (38%) patients were treated with ampicillin and cefotaxime, and 135 (33%) with ampicillin and gentamycin or tobramycin. The median IV antibiotic duration was 5 (IQR: 3-10) days. In 351 (87%) cases, a renal and bladder ultrasound (US) was performed, and 139 (40%) had abnormal findings such as hydronephrosis and/or a dilated ureter. Voiding cystography was carried out in 192 (47%) patients, with only a third of those patients positively demonstrating vesicoureteral reflux. Dimercaptosuccinic acid (DMSA) scanning was carried out on 41 (10%) children, with a median time to DMSA scan of 176 (IQR: 108-368) days.
Oral cephalexin and amoxicillin were used in the continuation phase in 48% and 30% of the cases, respectively, and the total period of treatment (oral and IV) was 10 (IQR: 9-14) days. The median length of stay was 5 (IQR: 3-21) days, and only 34 (8%) patients needed long-term home IV antibiotics. Of the 403 children in the final study cohort, only 19 (5%) experienced treatment failure, defined as readmission within 30 days after discharge. Over the course of the study period, the proportion of children exhibiting treatment failure per year ranged from 2.5% to 8.8%. The treatment failure rate was similar (5%) between both groups; short-course versus long-course IV antibiotic exposure (P > .05). The median time to readmission was 17 (IQR: 8-27) days.
We found in the bivariate analyses (Table 1) that nosocomial UTI, age younger than 3 weeks, children with a higher baseline creatinine level, premature children, the presence of comorbidities, and children with a positive blood culture were each associated with increased likelihood of receiving a long-course IV antibiotic treatment (P < .001). Children with comorbidities and hospital-acquired UTI had longer length of stay (28 vs 3 days [P < .0001] and 56 vs 3 [P < .0001], respectively). In retrospect, the rate of renal scarring on DMSA scan was significantly higher in patients that had received long-course IV antibiotics (2 vs 13 days, P = .03). There were no statistically significant associations between IV antibiotic duration and patient sex, UTI history, appearance at presentation, low systolic blood pressure, abnormal KUB US findings, and presence of vesicoureteral reflux on voiding cystourethrography.
Following multivariate adjustment, there was no significant association between specific treatment group and treatment failure, with an odds ratio (OR) for long- versus short-course treatment of 0.42 (95% confidence interval [CI]: 0.11-1.63). Additionally, prematurity, positive blood cultures, and the presence of comorbidities were also not predictive of treatment failure. However, the odds of treatment failure increased if the patient had documented fever on admission (OR 3.37, 95% CI: 1.05-10.86), UTI history (OR 6.87, 95% CI: 1.71-27.15), and length of stay more than 15 days (OR 6.56, 95% CI: 1.65-26.09) (Table 2).
Multivariate Logistic Regression.
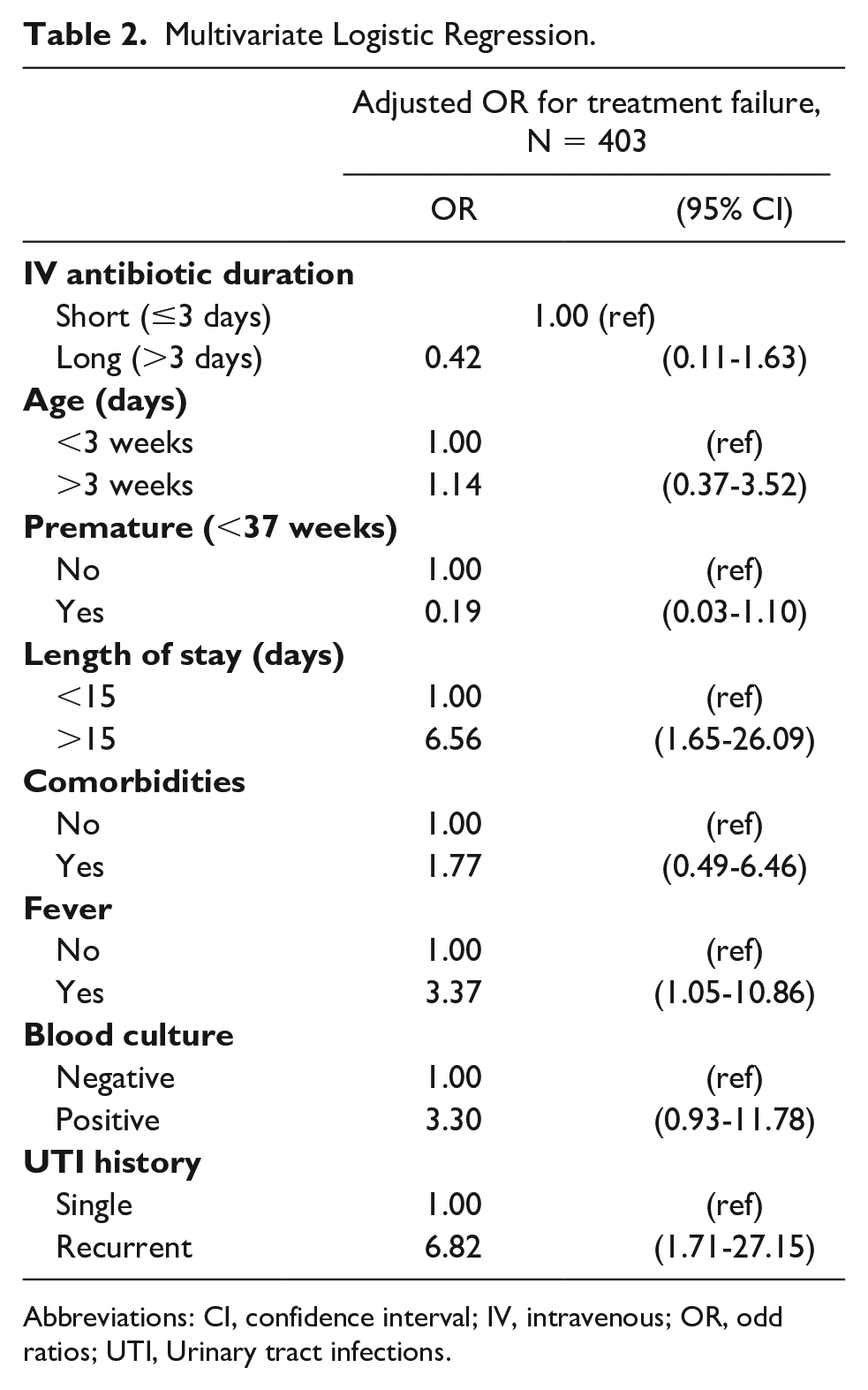
Abbreviations: CI, confidence interval; IV, intravenous; OR, odd ratios; UTI, Urinary tract infections.
Discussion
In this large retrospective cohort study of infants younger than 60 days, we examined the association between the duration of IV antibiotic therapy for UTIs and the risk of treatment failure. We observed no association between the receipt of short-course IV therapy (≤3 days) and increased odds of readmission for UTIs compared with the receipt of longer courses. There are no available published recommendations on the duration of parenteral treatment for UTI in infants younger than 60 days. Data on parenteral antibiotic use can be gleaned from existing studies and suggest that prolonged parenteral antibiotic courses offer no clear benefit. However, these studies are limited by small numbers, lack of inclusion in some cases of infants younger than 1 month, or lack of focus on clinical outcomes.3,6,14-17
In a recent retrospective chart review of 112 neonates <28 days old hospitalized for UTI, 18 the median IV antibiotic duration was 49 hours (51% received IV antibiotics for >48 hours). No demographic features or laboratory values correlated with IV antibiotic duration apart from age <7 days. The odds of long IV antibiotic duration increased if the neonate had a secondary diagnosis extending hospitalization (adjusted OR [aOR] 3.2, P < .002, 95% CI: 1.2-8.7), subspecialty consult (aOR 4.79, P < .001, 95% CI: 1.87-12.3), or an abnormal renal US (aOR 2.26, P < .02, 95% CI: 1.01-5.08). Only 1 neonate experienced treatment failure.
In a multicenter retrospective study conducted by Desai et al in a similar age group with bacteremic UTIs, infants receiving a short course of parenteral antibiotics did not experience more-frequent UTI recurrence or hospital reutilization than infants receiving a long-course parenteral therapy. However, short-course parenteral antibiotic duration was defined in this study as ≤7 days. 19 Another multicenter retrospective study by Schroeder et al 20 of infants ≤90 days of age with bacteremic UTIs revealed that infants with recurrent UTI did not receive shorter parenteral antibiotic treatment than infants without recurrent UTI in an unadjusted analysis. Schroeder’s findings are similar to those in this study, with infants who received longer courses of parenteral antibiotics more likely to be younger and have a positive blood culture. In 2007, Magín et al 14 reviewed 172 infants (median age of 19 days) with UTIs, finding that early transition to oral antibiotics had no increased rate of treatment failure, relapses, or renal complications.
Historically, bacteremic UTIs have been treated with longer courses of IV antibiotics because of concerns for severity of infection, renal scarring, hospital readmission, and recurrent UTIs.6,14,16,21 It was therefore not surprising that in our study, infants with bacteremia received a longer duration of IV antibiotic therapy than those without bacteremia although there are no available published recommendations on duration of parenteral treatment. Many studies in the literature revealed variation in duration of parenteral antibiotics for treatment of UTI and bacteremic UTI. 6
Angeles et al 22 performed a more than 10-year retrospective analysis of otherwise healthy infants younger than 60 days of age with a UTI and identified an increasing trend toward short-course parenteral antibiotics, as well as no differences in readmission rates as the parenteral antibiotic duration shortened. Total antibiotic courses in that particular study were not analyzed.
In a randomized clinical trial of 309 infants aged 1 to 24 months with fever and UTI, patients received initial therapy with either oral cefixime alone or IV cefotaxime. Thirteen infants, after randomization, were found to be bacteremic. The study found no significant differences in time to defervescence, symptomatic reinfection, or renal scarring at 6 months, regardless of the presence of bacteremia. 3 Significantly, in this study, none (0 of 13) of the infants with bacteremia were found to have renal scarring by 6 months of age; however, 10% (26/288) of the nonbacteremic population did have evidence of scarring. A recent systemic review was published in pediatrics 23 including several of the aforementioned studies. In this review, the largest two studies on bacteremic UTI found no difference in the rates of 30-day recurrence between those treated with ≤7 days versus >7 days of IV antibiotics. For nonbacteremic UTI, there was no significant difference in the adjusted 30-day recurrence between those receiving ≤3 days versus >3 days of IV antibiotics in the largest 2 studies identified. A Finnish study including 134 children with bacteremic UTI matched with children with UTIs and negative blood culture results found that infants with bacteremia often had no difference in clinical presentation or recovery and that the presence of positive culture results did not add new microbiologic information. 16
The presumption that IV antibiotics are superior to oral antibiotics remains a driving force for the treatment in many pediatric and neonatal conditions. 5 Yet, in studies in which neonates are specifically addressed, oral antibiotic therapy has not been associated with treatment failure or recurrence of infection despite concerns of decreased enteral absorption. 24
The present study is not without its limitations, namely the retrospective design, and there is a potential for misclassification in infants with recurrent UTI if the recurrence was managed in the outpatient setting or at a different institution and would, therefore, not have been captured using our data-collection methodology. Based on the young age of the infants and the fact that we are the only children’s hospital in Toronto, we suspect this is likely to be very uncommon, but this misclassification could lead to an underestimation of treatment failure. Given this, we acknowledge that the low incidence of treatment failure within our cohort posted limitations to our statistical analysis, particularly in the case of variable selection for our multiple logistic regression models.
Although we collected data on a variety of demographic, clinical, laboratory, and imaging variables, there may be others that were not included such that they could have impacted IV treatment duration, including response to treatment, resolution of fever, vomiting, or other clinical symptoms. Moreover, patients who are premature and/or present with comorbidities may not have the same outcomes with a shorter IV therapy. In addition, it is possible that there is a subset of infants in which there are high risk factors and in whom longer courses of parenteral antibiotics are appropriate. Future studies should focus on identifying which populations of young infants, if any, may benefit from longer courses of parenteral antibiotic therapy. Lastly, we did not collect information on multidrug-resistant organisms. As such, this study does not address or make specific recommendations relating to therapy nor duration of therapy for infants with culture results growing multidrug-resistant pathogens.
Conclusion
Treatment failure for infants hospitalized in the first 2 months of life with UTI is uncommon and does not change if the duration of parenteral antibiotic therapy is more or less than 3 days. Treating more infants of this age group with short courses of IV antibiotic therapy might decrease the length of hospitalization and resource use without affecting the readmission rates. The relationship between longer antibiotic duration and prematurity, comorbidities, bacteremia, younger age group (<3 weeks), hospital-acquired UTI, and other clinical parameters should be explored further because these factors were significantly associated with longer antibiotic duration.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
