Abstract

Introduction
Urinary pneumococcal antigen is a rapid noninvasive test that may indicate recent invasive pneumococcal infection 1 or carriage, 2 and guide appropriate antibiotic therapy.3,4 It has good sensitivity (100%) and (although limited specificity, 56%) in children with suspected pneumococcal disease 3 but is confounded as a diagnostic test by nasopharyngeal carriage where up to 21% may be positive. 2 Urinary pneumococcal antigen may also be positive in 4% healthy nasopharyngeal culture-negative children. 2 In fact, because of the increased sensitivity of polymerase chain reaction (PCR) compared with culture, it is likely that nasopharyngeal culture-negative children would have pneumococcal carriage if tested by PCR. 5 Thus, a positive urinary antigen in a child with pneumonia and no pneumococcal nasopharyngeal carriage by PCR is likely to reflect invasive pneumococcal disease. 1 We therefore aimed to investigate the association of urinary pneumococcal antigen with pneumococcal infection in childhood pneumonia.
Methods
Participants
A prospective etiological study of childhood pneumonia was conducted from October 2009 to March 2011. Enrolled cases were children aged ≤16 years with clinical and radiological features suggestive of pneumonia. Patients were resident in North East England (excluding Cumbria) who presented or were transferred to the pediatric services at the Great North Children’s Hospital, the regional cardiothoracic center at Freeman Hospital, Newcastle or the James Cook University Hospital, Middlesbrough. Pneumonia was defined by signs and symptoms suggestive of lower respiratory tract infection and chest radiographic findings consistent with pneumonia as determined initially by the local pediatrician. Chest radiographs were first reported by local radiologists then reviewed by a second consultant cardiothoracic radiologist at the regional center. Radiographic findings were categorized into lobar, patchy, or perihilar according to World Health Organization criteria. 6 Exclusion criteria included clinical diagnosis of bronchiolitis, hospitalization in the preceding 3 weeks or normal chest radiograph after formal reporting by a radiologist. For cases, informed written consent was obtained from parents as well as assent from older children.
Because of logistic limitations on recruiting community controls, it was decided to collect the control urine samples from children who attended the pediatric renal service at the Great North Children’s Hospital during March to May 2010 for routine follow-up or investigations of previous urinary tract infection. Controls had no clinical evidence of concurrent infectious illness and urine microscopy performed to exclude acute infection. Ethical and Caldicott approvals were granted (Newcastle and North Tyneside Research Ethics Committee [No. 08/H0906/105], and Research Approval Board at South Tees Hospitals NHS Trust [No. 2008075]).
Laboratory Procedures
Extensive microbiological and virological testing informed the etiology of pneumonia in cases using defined diagnostic criteria and positive results were classified as definite/probable or possible (Table 1). Apart from testing urine for pneumococcal antigen, no other microbiological investigations were performed in controls. Blood samples were collected for serum, blood culture, (BacT/ALERT, bioMérieux, Basingstoke, UK) and pneumococcal PCR testing. Nasopharyngeal aspirates/swabs were collected from infants and older children, respectively. Expectorated sputum and tracheobronchial secretions (sampled via endotracheal tube or bronchoalveolar lavage) and pleural fluids were tested when collected.
Laboratory Investigations and Diagnostic Criteria for the Etiology of Pneumonia.
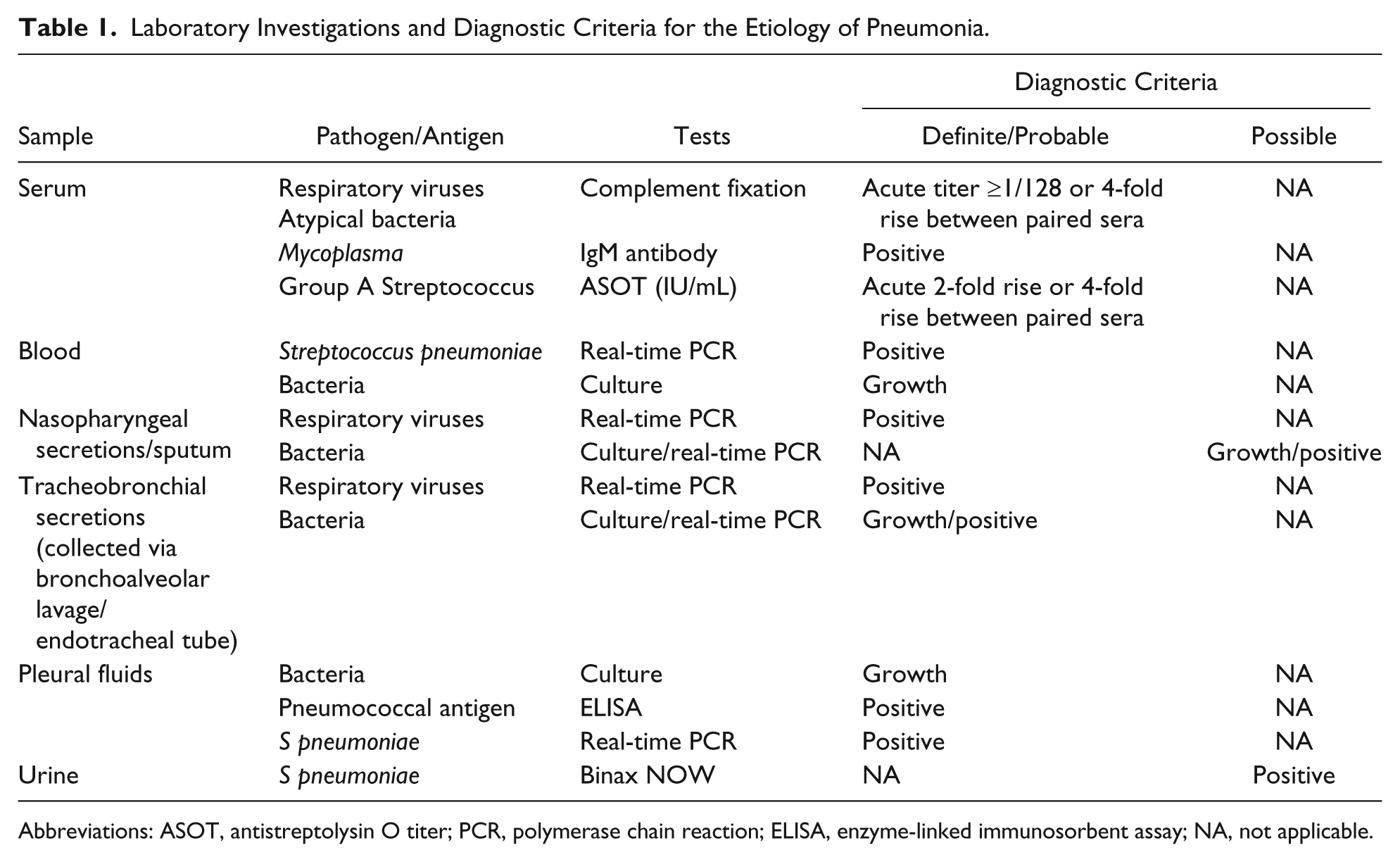
Abbreviations: ASOT, antistreptolysin O titer; PCR, polymerase chain reaction; ELISA, enzyme-linked immunosorbent assay; NA, not applicable.
Secretions were inoculated into plates of Columbia agar (Oxoid, Basingstoke, UK) supplemented with 5% horse blood (CBA) and Oxoid brain–heart infusion broth with 10% serum (Oxoid). Broths were incubated overnight at 37°C and subcultured (10 µL) into CBA plates for incubation at 37°C in 5% carbon dioxide for 48 hours. Pneumococcal isolates were identified by standard methods, including latex agglutination and API 32 STREP (bioMérieux). Urine samples were tested for pneumococcal antigen using Binax NOW (Inverness Medical Innovations Ltd, Ireland). Investigations were performed in the Microbiology Laboratory, Newcastle Hospitals NHS Trust and the Public Health England Newcastle Laboratory.
Total nucleic acid was extracted from blood, respiratory secretions, culture-negative pleural fluid, and pneumococcal antigen positive urine samples for PCR using an EasyMag automated nucleic acid extraction instrument (bioMérieux) and the resulting nucleic acid used as template in a Streptococcus pneumonia–specific PCR targeting the pneumolysin gene (and autolysin gene for pleural fluids). 7
Data Analysis
Detection rates of pathogens are expressed as proportions of those tested. Fisher’s exact test with odds ratios (ORs) and 95% confidence intervals (CIs) was used to evaluate group differences. Epi Info 7 was used for data analysis.
Results
A total of 160 cases of pneumonia were enrolled with a median age of 2.6 years (56% males and 69% aged <5 years). Not every case had all (but at least one) microbiological investigations performed. Their etiological data are presented and discussed in separate publications.1,8 Control urine samples were collected from 122 children with a median age of 4.7 years (37% males and 52% aged <5 years). Urinary pneumococcal antigen was detected in 28.3% (30/106) of cases, compared with 7.4% (9/122) in controls (OR = 0.2, 95% CI = 0.09-0.45, P = .00003). Among those aged <5 years, the urine antigen was positive in 23.5% (16/68) cases and 9.5% (6/63) in controls (OR = 0.3, 95% CI = 0.12-0.94, P = .037).
Streptococcus pneumoniae was the definite cause of pneumonia in 17.4% (24/138) of cases; 15% (14/93) and 22.2% (10/45) tested among those aged <5 and ≥5 years, respectively. Where performed among cases with identified pneumococcal infections, 75% (15/20) had positive urinary pneumococcal antigen (OR = 12.0, 95% CI = 3.76-38.26, P = .000008). S pneumoniae was identified from nasopharyngeal secretions by culture in 7% (10/141) and by PCR in 63% (76/121). Pneumococcal urinary antigen was not detected in any of the 40 children with nasopharyngeal pneumococcal carriage only (on culture and/or PCR) and no evidence of likely pneumococcal infection. Half (15/30) of those with Binax-positive samples also had S pneumoniae identified in a sterile site.
Discussion
Our study has shown that in children with radiologically confirmed pneumonia the urinary pneumococcal antigen is more likely to be positive than in healthy asymptomatic children. This indicates that positive urinary Binax test could be highly suggestive of this infection in healthy children, including young age-group who had no recent infections. Although previous findings showed poor utility of urine antigen test in distinguishing pneumococcal pneumonia from nasopharyngeal colonization in children,9,10 our findings showed significantly positive results of urinary pneumococcal antigen between children with pneumococcal infections and those with other causes of pneumonia. Also, with no association between urinary pneumococcal antigen and nasopharyngeal pneumococcal carriage in cases, urinary pneumococcal antigen is likely to indicate a pathogen than carriage.
We suggest that urinary pneumococcal antigen testing may be a useful investigation to help establish rapid diagnosis of invasive pneumococcal disease, particularly in low-resource countries where expensive PCR-based assays are not readily available. Used in this way this test may also prove a useful tool in clinical management and studies of the epidemiology of childhood pneumonia.
Footnotes
Acknowledgements
We thank the research nurses, Kerry Pollard and Pauline Singleton, for the assistance with data collection. We are grateful to Dr Fiona Hampton at the James Cook University Hospital, Middlesbrough for the facilitation of recruitment, and Audrey Nicholson at the Department of Microbiology, Freeman Hospital Newcastle for overseeing the laboratory logistics.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: DAS and JEC received unconditional research support from Pfizer. The sponsor had no role in the study design, data collection, analysis or interpretation, and writing of the article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by Pfizer Vaccines UK (No: 0887X1-4479).
