Abstract
Australia has a National Numeracy Learning Progression (NNLP) that is strongly aligned with the Australian Curriculum: Mathematics. This article examines how a sub-element within this progression could be impacting students’ learning of Science. This sub-element is firmly based on Mathematics education research as to how students build their understanding of geometric measurement (the structure of length, area and volume). Mathematics educators subsequently researched children’s measurement of mass and included it within the same sub-element of the NNLP. The contexts in which mass and volume are measured in Mathematics are different to those used in teaching Science. This article presents two studies that used variation theory and task-based interviews of children in Years 5 and 6, to explore their thinking about mass and volume in a Science context. The findings suggest that mathematical constructs in geometric measurement could be constraining the development of scientific ideas about matter. This research has implications for furthering the development of the NNLP to encompass scientific aspects of measuring matter.
Introduction
The development of a learning progression is seen as a means of synthesising decades of educational research to inform the design of curriculum and assessment and assist instructional practices. In 2016, the Australian Curriculum and Reporting Authority (ACARA) began to consult with researchers to synthesise common pathways of learning concepts and acquiring skills in the domains of literacy and numeracy. These pathways, called the National Literacy and Numeracy Learning Progressions (ACARA, 2021a) are periodically reviewed and refined based on large-scale written tests. However, these tests can only reflect what students have already had the opportunity to learn (Siemon et al., 2019), not what they could learn if the curriculum was transformed.
The sequencing of levels within the National Numeracy Learning Progression (NNLP) aligns with year level expectations of the Australian Curriculum: Mathematics (AC: M); however, the NNLP does not replace the curriculum. Its primary purpose is to support teachers in identifying and addressing the numeracy needs of individual students. Growth in numeracy is signified by moving up levels of the NNLP. The writers of the NNLP do not state its purpose as supporting curriculum areas outside Mathematics. Rather, they view the NNLP as assisting teachers ‘to identify opportunities to support students’ numeracy development and ‘to provide meaningful contexts for the application of numeracy skills identifying the numeracy demands of each subject in the Australian Curriculum’ (ACARA, 2021b, p. 1).
Elements and sub-elements of the national numeracy learning progression, version 3.
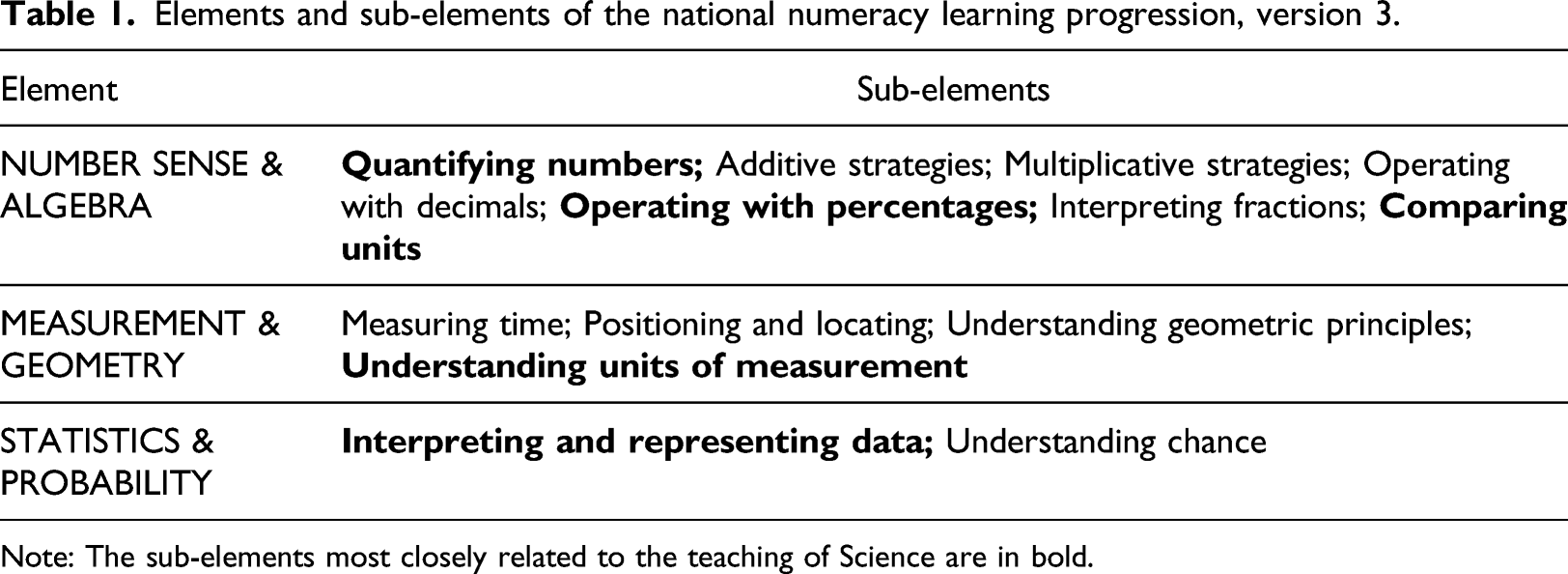
Note: The sub-elements most closely related to the teaching of Science are in bold.
The research reported in this article relates to the measurement of mass and volume. The developmental levels for measuring these attributes lie within the sub-element ‘Understanding units of Measurement’ (UuM). This topic is of particular interest because for the primary school years (Foundation to Year 6), the measurement of mass and volume is developed entirely within the AC: M while the application of this learning to real-world contexts is expected to occur largely in Science. We argue that this division of learning could be contributing to students’ difficulties in understanding the concepts of mass and volume, thereby causing a barrier to their learning in Science.
We report on two studies that used in-depth, one-to-one task-based interviews with students in Years 5 and 6 to explore their thinking about the mass and volume of materials and the relationship between them, which is ‘density’. Our aim was to reveal how children’s thinking about science contexts might have been influenced by their learning of measurement concepts in Mathematics. The levels of the NNLP are aligned with the AC: M, not with the AC: S. This research has implications for the ongoing development of the NNLP.
Literature review
The term ‘learning progression’ is used to describe the development of students’ core science ideas and practices. Given recent intermingling in the usage of the terms ‘learning trajectory’ and ‘learning progression’ (Confrey, 2019), we use the term ‘learning progression’ to refer to both. ‘Learning progression’ was chosen because this term is used by ACARA, the author of Australia’s NNLP (ACARA, 2021a).
We begin by describing the development of a science learning progression for ‘matter’ and a learning progression for ‘measurement’, which is the sub-element UuM in the Australian NNLP. Following this, the topic of chemical sciences in the AC: S and measurement in the AC: M and are examined and compared.
Science learning progressions for matter
Matter is the scientific term for what children commonly refer to as ‘stuff’ (Skamp, 2020a, p. 420). All matter has the attributes of mass and volume. Attributes of matter take part of their meaning from each other: mass the measure of how much matter there is; weight the force of gravity acting upon matter; volume the measure of the space occupied by matter and density the mass of matter in relation to its volume.
Smith et al. (2006) proposed a sequence of learning about matter and atomic-molecular theory to inform national standards and large-scale assessment in the United States from the time students start school through to Year 8. They named it the Learning Progression in Matter (LPM). They suggest that during the first 3 years (up to Grade 2), students should begin to construct measures of weight and volume, explain the properties of objects in terms of the material they are made from, and learn what properties change when an object is re-shaped or melted. During the next 3 years (Grades 3 to 5), students should learn that materials are composed of invisible particles, that all matter has weight and takes up space, that these properties can be measured, and that properties such as density are characteristic of the material and independent of the size of the material. They also learn that the amount of matter and its weight are conserved across a range of transformations, including heating. During the middle years of schooling (Grades 6 to 8), this learning is developed so that they can differentiate between weight and mass, understand the characteristics of materials in greater depth, and know that there are empty spaces (a vacuum) between atoms so that mass and weight (but not volume) are conserved when a material is heated.
Wiser et al. (2012) revised the LPM for Years 3 to 5 and added a detailed curriculum through The Inquiry Project (TERC, 2021). In subsequent years, the LPM has been extended up to the final year of school and re-organised by splitting it into four related ideas – chemical reactions; physical states and their changes; atoms, molecules and particle systems; and conservation (Hadenfeldt et al., 2014).
More recently, Hadenfeldt et al. (2016) systematically reviewed and analysed a large body of research concerning students’ progression in their understandings of matter. They identified five levels of understanding but did not link these to a particular age or year level. Important aspects of each level are described below.
At Level 1, students’ focus is on everyday perceptual experiences of matter. They perceive a property such as hardness, rather than an underlying property like density (Smith, 2007). When visually comparing two objects, they will often think the object with the larger volume is heavier (MacDonald, 2010; McDonough et al., 2013). ‘Heaviness’ and ‘heaviness for size’ (which is density) are conflated to one undifferentiated weight concept. When hefting two objects to compare their weight, the object made from denser material is often perceived as weighing more (Smith et al., 2006). When placing objects in water, the object that sinks or sinks faster is perceived as being heavier (MacDonald, 2010; McDonough et al., 2013). For materials such as air, ‘weight’ and ‘space occupied’ cannot readily be perceived (Skamp, 2020a; Smith, 2007) so students at Level 1 do not see weight and volume as essential properties of all matter.
At Level 2, students go beyond perceptual judgements to create measures of weight and the amount of space occupied. They consent to the existence of all matter being composed of particles that are too small to see and believe that the number of particles remains the same for the same amount of material. They may think, however, that the properties of the particles can change and that the particles are embedded in materials (Hadenfeldt et al., 2014).
Students at Level 3 can use the particle concept to explain a change in matter such as thermal expansion. At Level 4, they can use a more differentiated particle concept to explain basic chemical reactions and at Level 5, they hold a systematic view of matter that enables them to explain macroscopic properties, chemical reactions and the conservation of matter as a result of interactions between sub-atomic particles.
Smith and Wiser (2013) posit that students’ reconceptualisations of concepts such as mass, weight, volume and density, necessitate revisions in their understanding of measurement. In the primary school years, the important move required is a move from personal perception (Level 1) to trusting data from measuring instruments (Level 2).
Mathematics learning progressions for measurement
In mathematics, measurement learning progressions focus on the conceptual understanding of units and on comparison as a means of relating measures to units. The contexts commonly used in establishing measurement progressions have been the spatial attributes of length, area and volume (Barrett et al., 2011). Kim et al. (2017) developed a learning progression for these geometric measurements in one, two and three dimensions, in which understanding develops both vertically and horizontally because the same key concepts underlie an understanding of measurement for all three attributes. The tasks they designed to examine students’ understanding of volume measurement were restricted to measuring the volume occupied by a solid object or the space within a rigid container (a box). In these contexts, the volume being measured has a clear boundary and ‘volume’ is always conserved.
The measurement of mass and weight has received considerably less attention in mathematics education research than geometric measurement (Cheeseman et al., 2012; Clarke et al., 2003). Confrey et al. (2020) report that in the U.S., 62 learning trajectories grouped into 24 clusters, are being provided to teachers at scale to guide instruction in Mathematics for Years 6 to 8. Of these 24 clusters, three involve measurement concepts and none of these are applied to attributes other than length, area, volume and angle. They do not consider the measurement of mass or weight to be an aspect of mathematics teaching. Measuring mass or weight is more commonly found in science education literature (Cheeseman et al., 2014).
In Australia, Outhred et al. (2003) pioneered the development of learning sequences in measurement through their research on geometric measurement concepts. They developed a ‘Learning Framework in Measurement’ based on three stages: (1) identification of the attribute, (2) informal measurement and (3) unit structure. Later, the same conceptual milestones and principles were applied to young children measuring mass. However, because the units of mass are unstructured, the ‘unit structure’ stage was omitted and the extended use of informal units of mass prior to using standardised units was found to be unnecessary (Cheeseman et al., 2014).
The Learning Framework in Measurement formed the basis of the sequence of levels in the sub-element UuM within the NNLP (ACARA, 2021a). This sequence interweaves the measurement of mass, angle, temperature, length, area, volume and capacity, capacity meaning the internal volume of a container (ACARA, 2021b). The capacity of a container, like the attributes of mass and weight, is not measured geometrically. The sub-element UuM begins with students describing attributes of objects (Level UuM1) and at the next level (Level UuM2) students use direct comparison to order objects according to these attributes. They then learn to estimate and measure attributes using informal before formal units. This leads to the application of formulas for finding areas and volumes. Only geometric measurement proceeds beyond Level UuM6 of this ten-level sequence except that for all attributes, conversion between metric units is at Level UuM8.
The elements of the sub-element ‘Understanding units of Measurement’ that require students to understand the attributes being measured.

Note: UuM: understanding units of measurement.
Learning progressions are considered by Suh and Seshaiyer (2015), and Confrey (2019) to be a means of enabling teachers to anticipate misconceptions or alternative conceptions so they can be explored, thereby developing thinking and reasoning. The exploratory research presented in this article raises questions as to whether the omission of science concepts from the sub-element UuM may instead be inadvertently reinforcing alternative conceptions.
Mass and volume concepts in the Australian curriculum
Outcomes of the Australian Curriculum: Science and the Australian Curriculum: Mathematics (F-6) that involve ‘mass’, ‘volume’ and ‘properties of materials’.
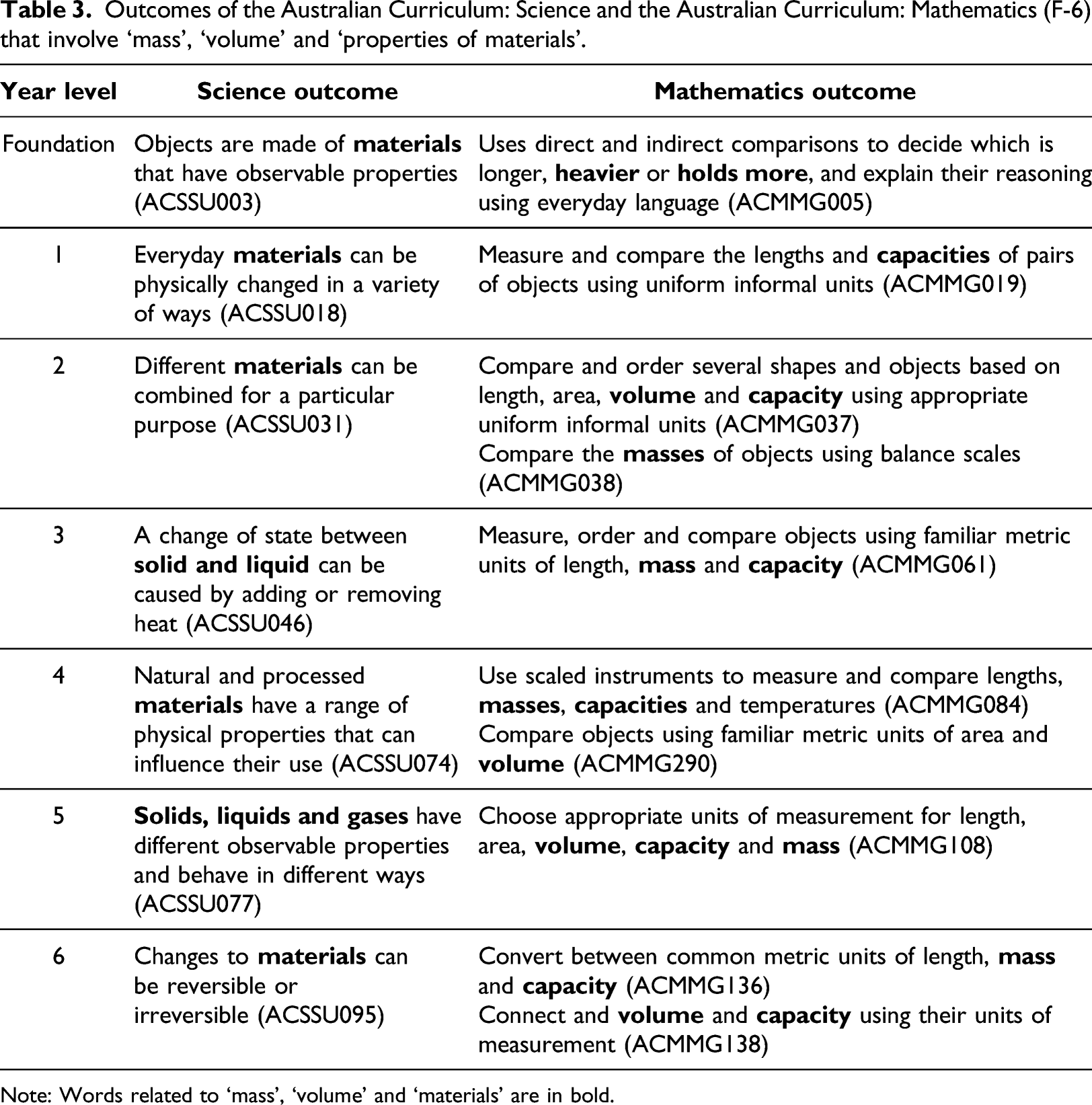
Note: Words related to ‘mass’, ‘volume’ and ‘materials’ are in bold.
The concept of ‘volume’ is understood differently across the two domains. In the AC: M and the NNLP, two different aspects of volume are measured. Volume is reserved to mean ‘the space (cm3) occupied’ and in the primary years (Foundation to Year 6), ‘volume’ is only used when referring to solid objects. The glossary of the AC: M defines capacity as “a term that describes how much a container will hold” and states that capacity “is used in reference to the volume of fluids or gases and is measured in units such as litres or millilitres” (ACARA, 2021e). For example, the amount of water (mL) required to fill a fish tank is its capacity (ACARA, 2021a). The aspect of volume called 'capacity' is introduced to students before the volume of solid objects because this aspect is easier to measure using informal units (e.g., cups) and can be read on a single scale. In teaching young children Mathematics, capacity is measured by ‘filling’ (meaning filling a container with a pourable material) whereas volume is measured by ‘packing’ (meaning packing cubes in rows, columns and layers) (Outhred et al., 2003). Following on from this, volume is understood abstractly as something that can be calculated using formulas based on length measurements. In Mathematics, students understand volume as being conserved so they can calculate the volume of an object by splitting it into two or more objects with known formulas for volume. In Science, students are introduced to the fundamental idea that all matter ‘takes up space’. This matter can be in different states and may not be just in the form of an incompressible solid. The word ‘volume’ is not used in the AC: S (Foundation to Year 6) so students may not think that ‘taking up space’ is synonymous with ‘having volume’.
The AC: M glossary defines mass as ‘the measure of how much matter is in a person, object, or substance’ but apart from this instance, the word ‘matter’ is not mentioned (ACARA, 2021e). Mass, being the measure of matter, is fundamental to Science but the first mention of mass in the AC: S is not until Year 5 where the elaboration to an outcome in the Science Understanding strand (Chemical Sciences – ACSSU077) is “observing that gases have mass and take up space, demonstrated by using balloons or bubbles” (ACARA, 2021d). This is also the first instance when the Science curriculum uses the words ‘take up space’.
Neither the AC: M nor the AC: S mention density in the primary school years. The density of an object (the ratio of its mass to its volume) depends on the material the object is made from. Density is not formally introduced in the AC: S until Year 8 (‘Science Understanding’ strand), where students are required to explain density in terms of a simple particle model (ACARA, 2021d). A key issue for Science educators is whether the particle model should be introduced in primary school (Skamp, 2020a). Comprehending the particle nature of matter is difficult because it is at a microscopic level of conception, but this notion is key to understanding all changes in matter. Skamp (2020b) supposes that students have fewer learning issues if the model is introduced early, albeit using a non-traditional approach such as that used in The Inquiry Project, where it is introduced in Grade 5 (TERC, 2021).
Theoretical framework and methodology
The two studies reported in this article were based on the Variation Theory of Learning (VT), a theory developed from phenomenography which is commonly employed in the design of mathematics and science instruction (Åkerlind, 2008). The underlying principle of VT is that by keeping one aspect constant, students’ attention is drawn to the critical aspects that vary (Kullberg et al., 2017). To integrate two different aspects, the learner must experience variations in both aspects simultaneously (Marton, 2014).
The purpose of using VT in our research was to highlight children’s understanding of mass and volume using patterns of invariance. In the first study, children were shown the same mass of two different materials and expected to notice that they have different volumes. In the second study, children were expected to notice that the mass of materials can be conserved while their volume changes under different conditions. This is because their density changes. Half the children in the second study were also shown a macroscopic model of the particle nature of matter, so we could evaluate its effectiveness in augmenting students’ understanding of a change in density at the microscopic level.
Semi-structured task-based interviews underpin the theoretical approach of our research, as the student-centred and practical nature of data collection is reflective of a constructivist view of learning (Stofflett & Stoddart, 1994). Task-based interviews were conducted with individual children in both studies. This type of interview provides opportunities for them to express, discuss and consolidate their ideas and thought processes within a structured and controlled environment (Goldin, 2000). In developing the LPM, Smith and Wiser (2013) utilised data from a large number of studies involving structured task-based interviews with children. In our second study, participants were also asked to express their understanding through drawing, enabling us to gain an accurate and comprehensive understanding of their thinking surrounding the concept of ‘density’, without introducing this word. Task-based interviews and children’s drawings are widely considered to provide highly valid information about student understanding (Hadenfeldt et al., 2016).
Participants and tasks
Year 5–6 children’s descriptions of why different solid and liquid materials floated or sank.
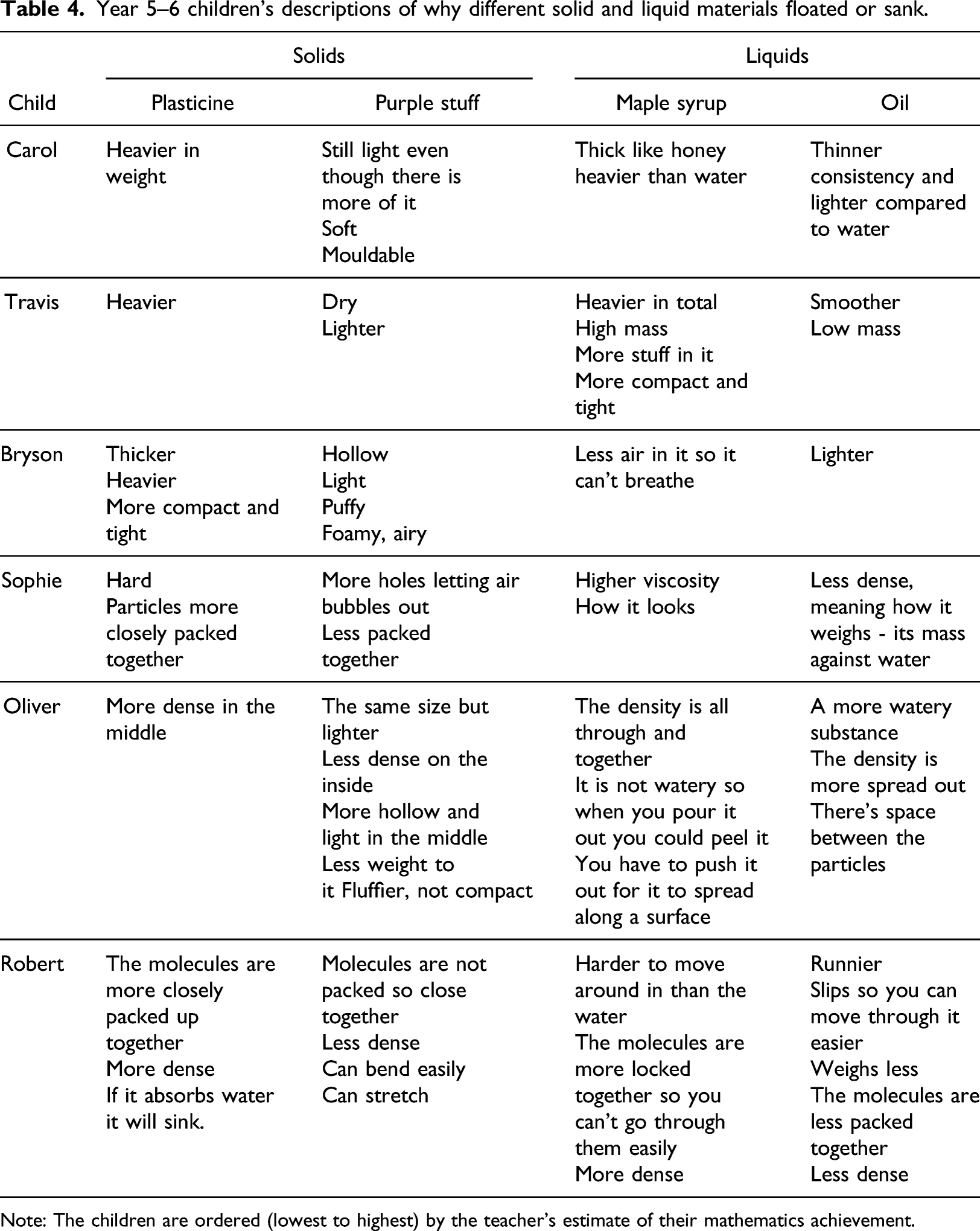
Note: The children are ordered (lowest to highest) by the teacher’s estimate of their mathematics achievement.
Study 1
The six children in this study (pseudonyms: Carol, Travis, Bryson, Sophie, Oliver and Robert) were all at the same independent school in an upper-middle socio-economic area of Sydney as measured by the school’s Index of Community Socio-Educational Advantage. They were interviewed by their teacher who prior to the study, indicated their level of mathematics achievement as ranging from lower-middle (Carol) to very high (Robert). They were interviewed at school in a relaxed environment and were familiar with the type of questioning used in the interview.
The tasks involved a comparison of two liquids and two mouldable solid materials when different quantities were placed in transparent containers, partially filled with water. Prior to this, they had compared their masses using an equal-arm balance and predicted whether the materials would float or sink. They were asked to describe any changes in their thinking and reasoning based on what they observed. The solid materials were plasticine which sank, and foam play-dough (called purple stuff) which floated. The liquid materials were syrup which sank, and oil which floated. For each pair of materials, the children were given a set of parallel tasks. Initially they were given equal volumes of the materials. Next, they were given equal masses and after that, they were given a much larger mass of the less dense material compared with the denser material. They conducted these tasks with the solid materials before conducting them with the liquids.
Study 2
The six children in this study were at two different schools. Four (pseudonyms: Harry, Nick, Sam and Tony) were at a comprehensive public school in a medium socio-economic area of Sydney and two (pseudonyms: York and Evan) were in an opportunity class for gifted students at a school located nearby. They were interviewed at a place their parents had selected so they felt relaxed. They all knew the researcher who interviewed them.
There were two tasks. Task 1 involved describing and drawing changes in kinetic sand when it was compressed in a box. Task 2 involved describing and drawing what they thought was happening to invisible particles of air in a balloon as it expanded and contracted due to a change in temperature. The balloon was attached to the top of a bottle that was moved between a hot and a cold water-bath.
Before conducting these tasks, the children were asked whether the materials had weight and volume and to predict what would happen. Only three of the children (Group 1: Harry, Nick and York) were then given Task 1 because the researchers wanted to see whether the kinetic sand served as a useful macroscopic model for the particle nature of matter. This kinetic sand model was found by Senzamici and McMaster (2019) to make density salient. The remaining children (Group 2: Sam, Tony and Evan) did Task 2 only. The two gifted children in an opportunity class were in different groups: York was in Group 1 and Evan was in Group 2.
Data collection and analysis
Interview data were collected via video recordings and transcribed by two of the authors: Perivollaris transcribed the data from Study 1 and Wang transcribed the data from Study 2. All video data were viewed by three of the authors to ensure the accuracy of transcripts, noting gestures and salient responses. The data were analysed using a phenomenographic method (Åkerlind, 2005) as follows: (1) the interview transcripts were coded according to matter-related concepts, (2) three researchers read the transcripts multiple times looking for key qualitative similarities and differences between the concepts held by the participants in each study, (3) shared understandings were established between three researchers who met weekly over an eight-week period and (4) The participants’ matter-related understandings were summarised and tabulated.
Results
Study 1
Understandings of mass and volume
This study revealed differences between the children in their understanding of mass and volume that matched quite closely with their teacher’s estimation of their mathematics achievement levels (Table 4). When Robert (the highest achiever) was asked why, when the syrup and the oil have the same mass, the oil has a greater volume, he referred to the syrup being denser than the oil and he gave the same explanation for the two solids. Conversely, Travis (an average achiever) experienced difficulties distinguishing between mass and volume. He consistently referred to ‘different amounts’ of the materials, this being a phrase that could suggest either mass or volume, and he spoke about a material being ‘lighter’ because it had less volume.
The other children were better able to differentiate mass from volume, though they failed at first to do so, thinking that for the equal-arm balance to be ‘even’ (meaning balanced) there must be an equal volume of the different liquids in the containers on opposite sides of the balance. This indicates a belief that either mass and volume are correlated, or that mass is measured in millilitres. Oliver (a high achiever) quickly self-corrected.
Understandings of the relationship between mass and volume
Providing the children with two materials with different densities helped them in their thinking about this relationship. Specifically, when Bryson was exploring liquids and questioned why oil with the same mass as the syrup, floated, he was able to explain the inverse relationship between the heaviness of a liquid (meaning its density) and the amount of it needed to give the same mass: “That one [syrup] is heavier which means it needs less to equal that one [oil] which is lighter”. Understanding of this inverse relationship was also mirrored in Sophie’s response. When the syrup was balanced against the oil on the equal-arm balance, she stated: “The smaller volume was heavier, and the oil had to be about 150 mL to get close to balancing them both out”.
Table 4 summarises the descriptions each child used to describe why, for each pair of materials, one floated on water and the other sunk. The lower achieving children (Carol, Travis and Bryson) all employed the terms ‘heavier’ or ‘lighter’ to describe the materials. Travis confused density with mass, describing the denser liquid as a material with ‘high mass’. Sophie used the words ‘less dense’ to describe the less dense liquid but referred to density as mass when she used the word ‘heavy’ (Table 4). Oliver used the word ‘dense’ but thought of the density of solids as something ‘on the inside’ or ‘in the middle’ of a solid material. Robert also used the word ‘dense’. He knew that the density of the two liquids caused them to float or sink and also recognised that density was not the only factor determining whether a solid material floated or sank.
Carol, Travis and Bryson all correctly predicted that the less dense material would float and the denser material would sink when their mass was the same, but were not confident in making the same prediction when the less dense material had a much greater mass. Conversely, the higher achieving children (Sophie, Oliver and Robert) were confident that increasing the mass of the less dense materials would not change their floating/sinking behaviour. Sophie predicted that the purple stuff would still float ‘because it is still the same substance’.
The higher achieving children also provided a microscopic level description of the materials, namely the arrangement of particles within materials (Table 4). Sophie referred to particles of the solids. Oliver referred to particles of the liquids. Robert referred to molecules of solids and liquids. The lower achieving children presented only a macroscopic description of the materials, making reference purely to their observable physical appearance. Although not referring to particles, Bryson and Travis described a denser material as being ‘more compact’ and ‘tight’, revealing some understanding that the matter of a denser material takes up less space.
Study 2
Understandings of mass and volume
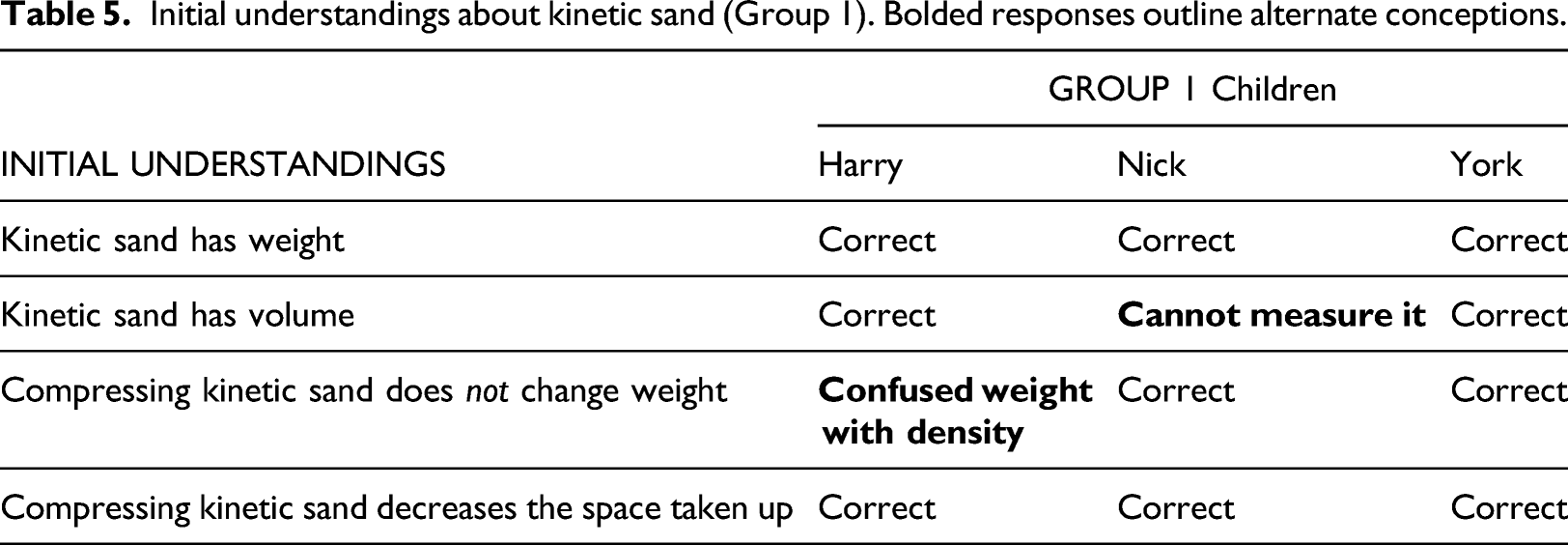
Initial understandings about kinetic sand (Group 1). Bolded responses outline alternate conceptions.
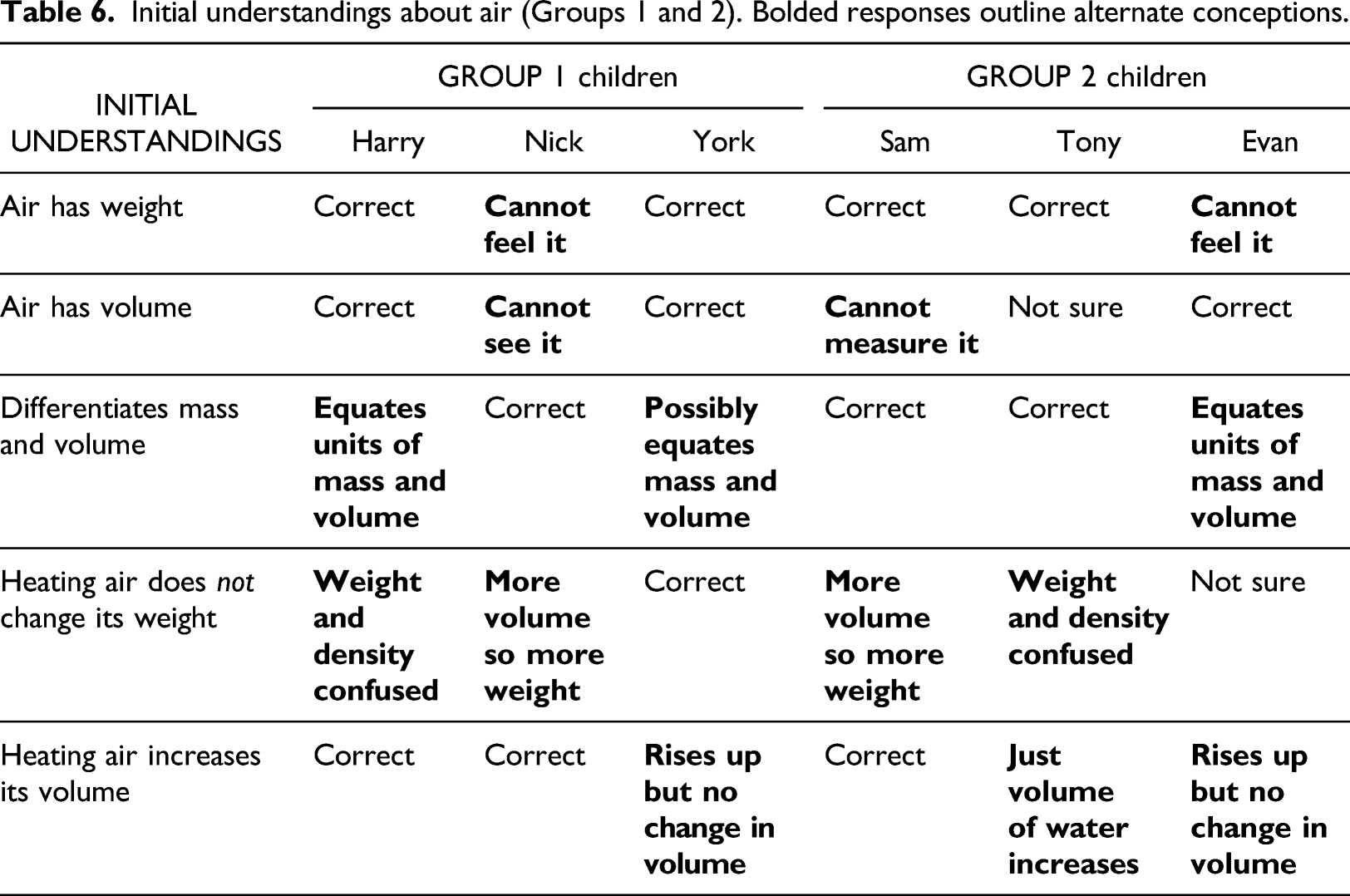
Initial understandings about air (Groups 1 and 2). Bolded responses outline alternate conceptions.
All the children thought sand has weight, but not all thought it has volume (see Table 5). Nick thought sand did not have volume because it cannot be calculated using length measurements. In his thinking, sand only ‘has volume’ when it is in a box so he can use the formula, Volume = Length × Width × Height. When compressing the sand, however, they all thought its density would increase.
Nick thought air has no weight because it cannot be felt and it has no volume because it cannot be seen (see Table 6). Evan also thought air has no weight because it cannot be felt but has volume because a balloon gets bigger when inflated with air. Sam thought the opposite of Evan. He thought air has weight because you can feel it: “It's like when you skydive or something you can feel air against you”, but air has no volume because “there’s too much air in the world and you can’t really measure the volume of it”.
Two and possibly three children of the six children equated mass and volume. Evan and Harry equated their units. Evan thought a number of grams was a measure of volume. Harry said “one gram is like one centimetre squared, I mean, um cubic centimetres or something”. York thought he could work out the volume of sand from its weight but did not know how to do this: “You would find how heavy it is and then yeah...”
Understandings of the relationship between mass and volume
The children’s understandings of the relationship between mass and volume were revealed by their verbal responses coupled with their drawings showing the materials before and after a change in volume. When they compressed the sand, they all attended primarily to the particles being closer together, supporting the findings of Senzamici and McMaster (2019). They only thought about changes in mass and volume when the interviewer questioned them about these attributes.
Nick and York knew the weight of the sand would be conserved because no sand had been added to the box. Although they noticed more space at the top of the box, they were not convinced that the volume of the sand had really changed. Nick said compression just made the kinetic sand ‘seem smaller’. York said ‘the volume that the sand takes up’ was less. He did not say that the volume of sand itself had changed. He then added height to his drawing of the box to show the sand was ‘using up less space’ (Figure 1). Harry had a different view. He predicted that compressing the sand would add weight and thought his prediction was confirmed when measurement error resulted in the compressed sand having one gram of additional weight. Harry showed more weight by drawing more particles in his diagram after the sand had been compressed, but he changed his mind about the weight changing when the interviewer drew attention to the fact that the amount of matter was the same. Children’s drawings of Kinetic sand when loosened and when compressed.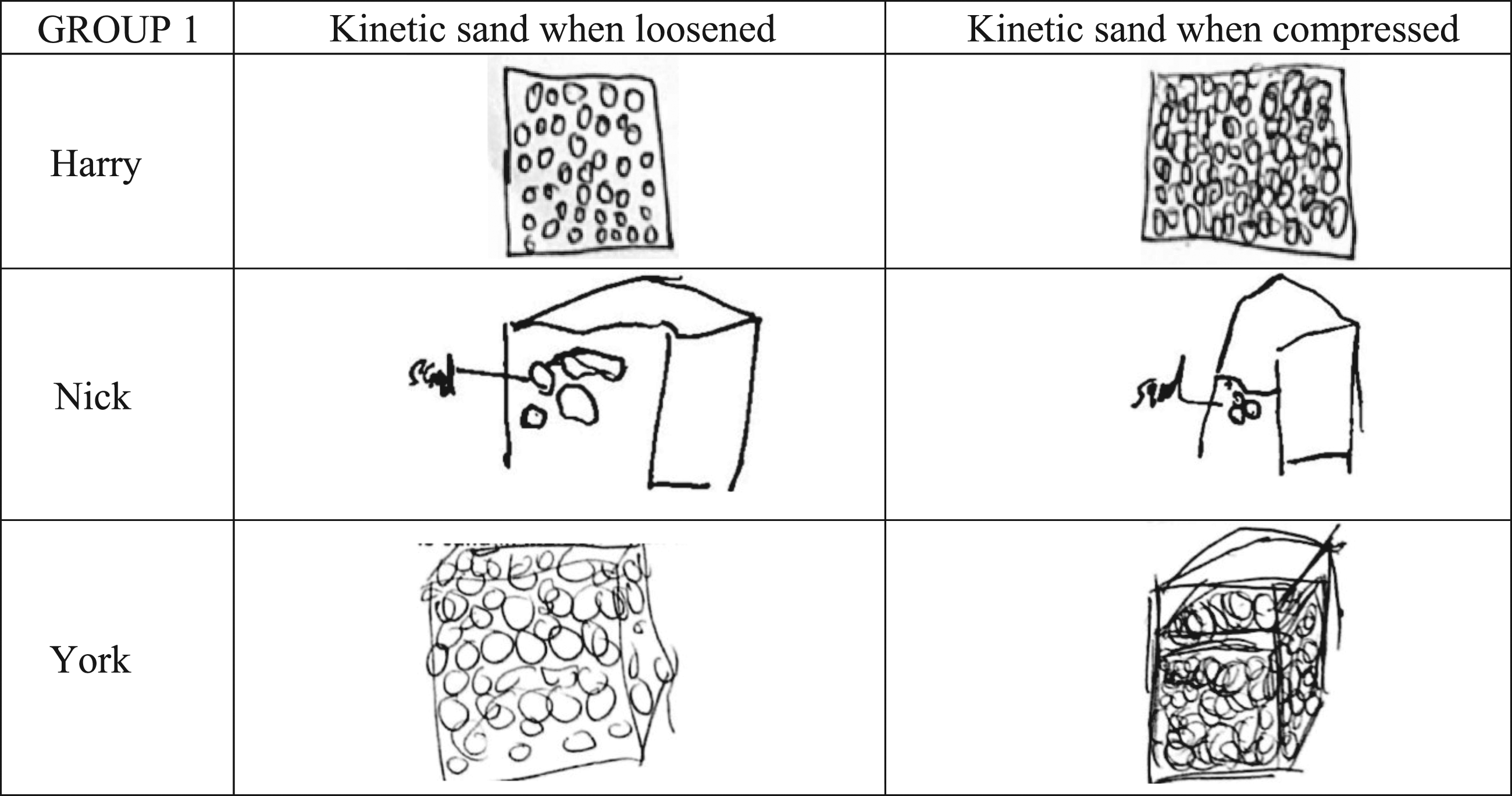
All the children did Task 2 during which they were asked to predict how the mass/weight of air in a balloon attached to a bottle would change, when this apparatus was heated and cooled. Nick and Sam predicted the weight would change simultaneously with the volume of air in the balloon as it expanded and contracted (see Table 6, row 4). Harry and Tony imagined weight as density, so they expected it to change with volume. Evan was unsure if air had weight. He thought the air would rise with heating but not change its volume. York confidently thought neither its weight nor volume would change because ‘there’s no opening…you can’t make something out of nothing’.
When the apparatus was weighed after heating and the scales showed that the mass/weight had not changed, all students thought that the volume must not have really changed. This resulted in them making various hypotheses, which they illustrated with their drawings (Figures 2 and 3). Task 2 drawings by students who had done Task 1. Task 2 drawings by students who had not done Task 1.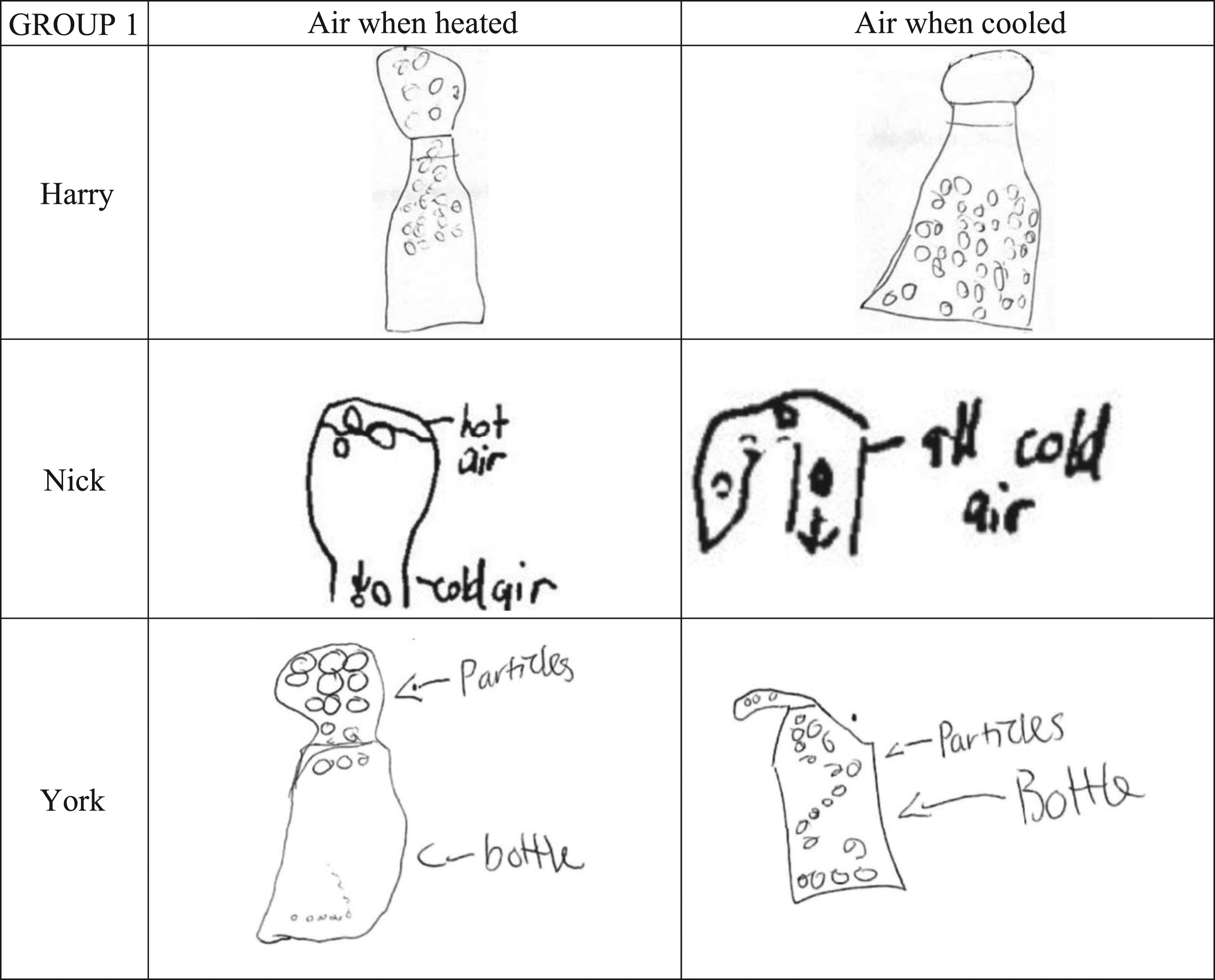
The Group 1 children’s explanations as to why the weight did not increase when the balloon expanded were as follows. Harry thought the particles rose up from the bottle into the balloon because they became lighter. He used the analogy of a hot air balloon: ‘Um, I think it’s lighter because that’s how hot air balloons get inflated’. Nick thought the volume of the balloon increased because the hotter particles moved up while the colder ones moved down. He said: ‘because like cold air goes to the bottom, it will grow bigger’. York thought that although air was taking up more space in the balloon, this did not mean it had a greater volume. He described the space without volume as ‘empty space’. Previously, he had called the volume of the sand, the space that the sand particles ‘take up’. He did not think space taken up, meant the same as ‘volume’. In other words, he thought the gaps between the sand particles were not part of the volume of the sand and the gaps between air particles were not part of the volume of the air.
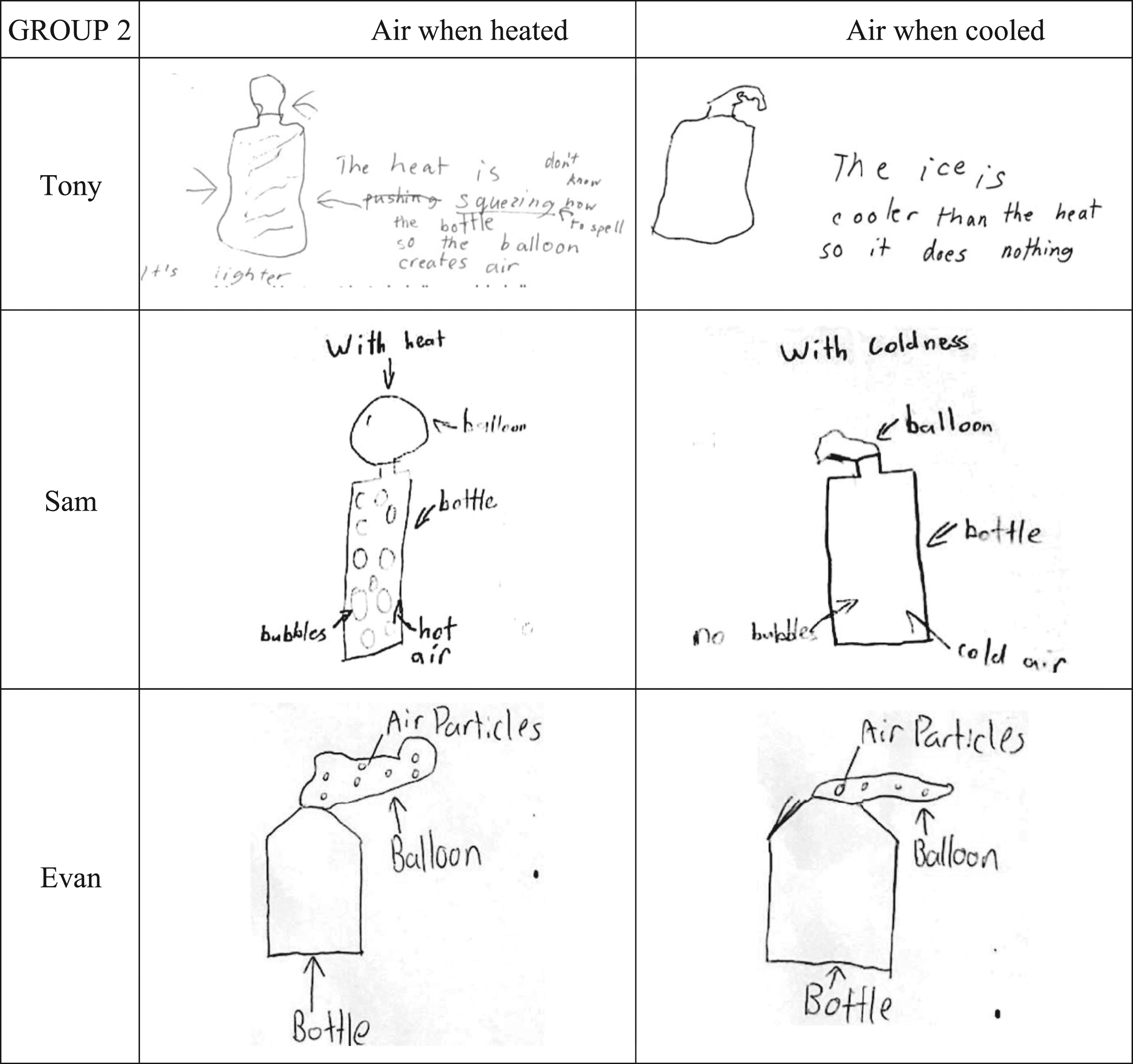
In Group 2, Tony thought heating the bottle and balloon by putting it in a hot water-bath would result in the bottle being squeezed by the water ‘pushing the bottle so it creates more air in the balloon’. Sam thought that heating resulted in bubbles of nothing inside the air. Evan was the only person in Group 2 to draw air particles, but he showed the increase in the size of the balloon as an increase in the number of particles inside it. He did not change the distance between particles.
Discussion
Understandings of mass/weight and volume
Both studies revealed a high proportion of students who were confused about mass or weight. The lower achieving students in Study 1 and Harry in Study 2 confounded mass or weight with density, seemingly because ‘heaviness’ describes both attributes: the mass or weight of an object and the density of a material. McMaster (2019) attributed this difficulty to the separation of ‘object’ in the AC: M with ‘material’ in the AC: S (Table 3). This distinction needs to be made more explicit. One suggestion for teaching is to regularly use the adjective ‘heavy’ together with the attribute, so talk about a ‘heavy object’ or a ‘heavy material’ rather than something being heavy.
Children’s alternative conceptions about ‘volume’ in Science also appeared to stem from learning in Mathematics. This was the belief that (1) matter has no volume if a formula cannot be applied to its shape or if it has no clearly defined boundary and (2) volume is always conserved because mass is always conserved. These ideas are consistent with the AC: M’s focus on the geometric measurement of non-compressible objects. In Science, when children learn about space being ‘taken up’, they might not be understanding this as meaning ‘volume’.
Four children in Study 1 and three in Study 2 initially equated mass or weight with volume. Teaching the measurement of ‘capacity’ in Mathematics by filling containers with water and the approximation of a millilitre with a gram of water, could be one source this of confusion (McMaster, 2019). Equating a millilitre with a cubic centimetre can lead students into believing that a cubic centimetre is also equated with a gram. The use of VT in Study 1 appeared to be a helpful strategy for helping children differentiate between these two attributes.
Understandings of the relationship between mass and volume
In Study 1, every child had some conceptual understanding of density, though density is not mentioned in the AC: M or AC: S in primary school. Even without knowing the word ‘density’, young children are quite capable of understanding that an object is heavy or light ‘for its size’ (Smith et al., 2006), so the concept could reasonably be included in the early years using a task with same-sized objects made from materials like plasticine and foam play-dough. An understanding of density at a macroscopic level could lead to an introduction of the particle model of matter using a concrete model similar to the model of kinetic sand used in Study 2.
Though not having been taught the particulate theory of matter, the three higher achieving children in Study 1 all referred to particles. Given that two of the remaining three children described the denser materials as being ‘more compact’ and ‘tight’ (see Table 4), it seems likely that they would consent to the existence of all matter being composed of particles that are too small to see, if they were provided with this model in earlier years as recommended by Smith et al. (2006) and others (Skamp, 2020a).
In Study 2, the value of the particle model was evident in the drawings made by the Group 1 compared to the Group 2 children. When asked to draw what happened to the invisible air particles, all of Group 1 but only Evan in Group 2 drew particles. Evan’s drawing inferred that the density of the air didn’t change when heated because the particles remained the same distance apart. He would be placed at Level 1 of the LPM because having not been given any prior experience with the kinetic sand model, his drawing showed an increase in the number of particles (Hadenfeldt et al., 2016). The children in Group 2 showed that the number of particles remains the same for the same amount of material but thought that the properties of the particles changed. Harry thought the particles became lighter, Nick thought they became hotter and York thought they became larger when heated. This would place them all at Level 2 of the LPM (Hadenfeldt et al., 2016).
If students cannot correctly relate mass or weight to volume, they cannot understand expansion by heating, where volume increases but mass and weight stay the same. An understanding of density could lead to an introduction of the particle model of matter using a concrete model similar to the model of kinetic sand used in Study 2. All the Group 1 children knew the number of particles did not change, but did not see the change in the amount of space occupied as being a change in volume or a change in density. Foremost in their thinking was that air rises when it’s heated. A better appreciation of density could help them appreciate that air rises because when heated, it expands and becomes less dense. If students cannot correctly relate mass or weight to volume, they cannot understand that density changes when volume increases or decreases but mass and weight stay the same.
Conclusion
One of the objectives of learning progressions is that they provide curriculum designers, assessment developers and teachers with “a framework for scaffolding students’ learning about core concepts” (Hadenfeldt et al., 2016, p. 684). The core concept of ‘measuring matter’ has long been advocated as one that should span the primary school mathematics and science curricula (Smith et al., 2006). According to Smith (2007), understandings of matter and understandings of measure “not only reinforce each other, but also provide a powerful foundation for later science teaching” (p. 395).
Although the results reported here are from two small exploratory studies, they align with those of the large-scale, long term studies that informed the LPM (Hadenfeldt et al., 2016; Smith & Wiser, 2013). We posit that the sub-element UuM be better coordinated with the AC: S. For example, in UuM1 where the learning is “uses everyday language to describe attributes in absolute terms that can be measured” (ACARA, 2021a, p. 43), the distinction could be made between materials and objects, and when discussing the properties of objects, the concepts of ‘size’ and ‘heaviness’ could be combined with discussion about ‘material type’ and whether an object is heavy or light ‘for its size’. The foundational concept that ‘all matter has mass and volume’ should be included within the progression and students made aware that ‘volume’ is any three-dimensional space. To bridge Mathematics and Science, students also need to appreciate that the volume of compressible materials is not conserved.
Alternatively, an additional sub-element ‘Matter Measurement’ could be developed. If this sub-element is added and includes temperature measurement within it, the sub-element ‘UuM in the NNLP’ could be re-named ‘Geometric Measurement’ as it would be restricted to the concepts of length, area, geometric volume and angle. A ‘Matter Measurement’ sub-element could be developed for the NNLP based on the well-established LPM (Hadenfeldt et al., 2016; Smith et al., 2006; Wiser et al., 2012) in which the measurement of mass/weight and volume is interwoven with the concepts of material type, density and the particle theory of matter.
More generally, our research highlights the need for teachers to be made aware of apparent conflicts between key learning areas when students learn them simultaneously. The Australian NNLP could provide an opportunity to do this.
Footnotes
Acknowledgments
The authors would like to thank Dr. Gabrielle Oslington, a mathematics teacher who assisted by interviewing some of the students who participated in this research.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
