Abstract
Australia’s reclassification of psilocybin as a Schedule 8 substance for treatment-resistant depression represents a significant shift in psychiatric policy. While this regulatory change positions Australia as a global leader in psychedelic medicine, its implementation has revealed substantial challenges. This article critically examines the regulatory, ethical and operational complexities surrounding the provision of psilocybin-assisted therapy in clinical practice. Key issues include limited prescriber access, absence of Australian Register of Therapeutic Goods-listed products, lack of standardised training pathways and significant cost barriers. Ethical considerations such as informed consent, cultural safety and therapeutic fidelity are also discussed, particularly in the context of trauma-informed care. This article proposes a series of structural recommendations to support safe and equitable deployment, including national training accreditation and fidelity monitoring tools. In addition, to maximise the efficacy of psilocybin-assisted therapy, we recommend that research explores the potential of neurobiologically informed stratification models to assist with treatment recommendations. These recommendations aim to enhance clinical integrity through evidence-based patient selection, improved safety, and to ensure that emerging psychedelic treatments are integrated responsibly within Australia’s mental health system. By addressing these foundational gaps, Australia can move beyond regulatory novelty ensuring the therapeutic potential of these products is realised in a manner which is scientifically sound and upholds the integrity of psychiatric practice.
Introduction
The therapeutic potential of psilocybin (a serotonergic tryptamine acting primarily via 5-HT2A receptor agonism) has moved from experimental interest to a subject of serious clinical investigation. Early-phase randomised controlled trials have demonstrated rapid and sustained effects in individuals with treatment-resistant depression (TRD), with preliminary evidence suggesting possible advantages over conventional pharmacological treatments when administered alongside psychological support (Erritzoe et al., 2024; Goodwin et al., 2022; Goodwin et al., 2023). Psilocybin has also shown promise in phase 2 trials (Carhart-Harris et al., 2021; Kittur et al., 2025; Raison et al., 2023). These results may be due to psilocybin’s ability to induce powerful perceptual changes, which include enhanced environmental sensitivity, along with associated enhancements to neural plasticity and psychological receptivity, which together may offer a unique opportunity to modify existing beliefs and remodel neural circuitry (Aday et al., 2023; Moliner et al., 2023).
In response to the promise shown by psilocybin in these clinical trials, the Therapeutic Goods Administration (TGA) reclassified psilocybin as a Schedule 8 substance for TRD in 2023, positioning Australia as a global leader in psychedelic regulation and policy reform, and attracting international attention (TGA, 2023). However, the TGA’s decision was a ‘delegate-only’ action which bypassed the conventional regulatory pathway, including the TGA’s own expert Advisory Committee on Medicines Scheduling, and this regulatory advancement has unfolded in the context of limited clinical infrastructure and readiness (Kisely, 2023). Critical foundations for ethical and scalable implementation, such as clinician training, equitable access and preservation of therapeutic fidelity, remain underdeveloped. This exposes structural vulnerabilities and ethical challenges.
Therapeutic deployment remains encumbered by significant policy, operational and ethical barriers. We examine these limitations and propose solutions informed by evidence-based practice, trauma-informed principles and integrated service models.
Australia’s regulatory landscape
Australia’s 2023 reclassification of psilocybin from Schedule 9 (Prohibited Substance) to Schedule 8 (Controlled Drug) for TRD was unprecedented globally. This landmark decision signalled a regulatory shift towards the use of therapeutic psychedelics in Australia. However, the accompanying framework remains narrow, cautious and structurally complex.
Controlled access via the authorised prescriber scheme
Psilocybin is currently not listed on the Australian Register of Therapeutic Goods (ARTG) and is therefore considered an unapproved therapeutic good. Access is restricted to the TGA’s Authorised Prescriber Scheme (TGA, 2023), which involves multiple layers of regulatory oversight (Figure 1).
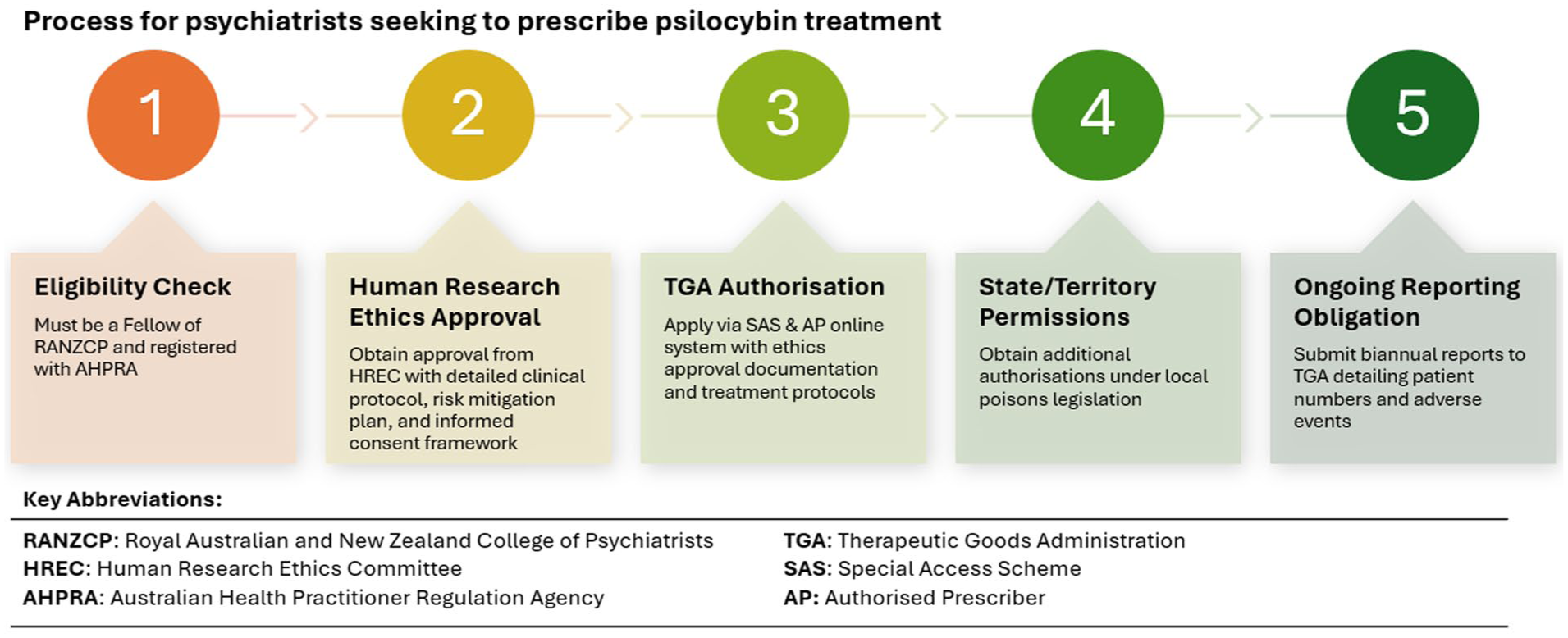
The regulatory pathway for psilocybin-assisted therapy authorisation in Australia – a multi-tiered approval process that is required for psychiatrists to prescribe psilocybin for treatment-resistant depression under the Therapeutic Goods Administration’s Special Access Scheme. This framework represents the current (2025) regulatory landscape for psychedelic-assisted therapy implementation in Australian clinical practice, highlighting the substantial administrative burden that may limit patient access to potentially transformative treatments for treatment-resistant depression.
There are several limiting factors within this multi-step regulatory framework that both ensure patient safety is protected and accountability is maintained, but also creates access bottlenecks:
Eligibility: Only psychiatrists who are Fellows of the Royal Australian and New Zealand College of Psychiatrists (RANZCP) and registered with the Australian Health Practitioner Regulation Agency (AHPRA) are eligible to apply.
Human Research Ethics Approval: Applicants must first obtain approval from a Human Research Ethics Committee (HREC), providing a detailed clinical protocol, risk mitigation plan and informed consent framework.
TGA Authorisation: Once HREC approval is secured, psychiatrists must apply to the TGA via the Special Access Scheme (SAS) & Authorised Provider (AP) online system, providing documentation of ethics approval and treatment protocols.
State/Territory Permissions: Additional authorisations may be required under local poisons legislation (e.g. NSW Section 29 authority for Type A drugs of addiction).
Reporting Obligations: Prescribers are required to submit biannual reports to the TGA detailing patient numbers and adverse events.
As of late 2024, only a small number of psychiatrists nationwide had been authorised to prescribe psilocybin (Nutt et al., 2024) and as of August 2025 only 12 were listed with the self-nominated RANZCP directory. Furthermore, the layered approval requirements may inadvertently centralise prescribing capacity within major urban academic institutions, amplifying geographic and socioeconomic disparities. While early rollout indicates private psychiatrists and clinics have shown interest in becoming authorised prescribers (Nutt et al., 2024), not-for-profit charity and lobby group Mind Medicine Australia report 13 Authorised Prescribers across 5 Australian States as of September 2025.
Compounding these challenges is the absence of any psilocybin products listed on the ARTG. At present clinics rely on imported formulations accessed under Section 19A exemptions, a pathway that raises concerns about product consistency and supply reliability.
As a result of the current regulatory framework and lack of psilocybin products listed on the ARTG, current limitations for clinical application of psilocybin include a narrow prescriber pool, regional access inequities, the absence of national formularies or standardised dosing protocols and absence of centralised oversight within current approval pathways. Concerningly, the absence of regulatory integration with existing mental health service frameworks also risks creating fragmented care pathways and oversight gaps. Without alignment between psychedelic therapy protocols and established clinical governance structures, such as referral systems, multidisciplinary care models and outcome monitoring, patients may experience discontinuity in care, reduced therapeutic coherence and limited access to follow-up support. Addressing these issues will require the urgent development of national evidence-based guidelines alongside robust accountability mechanisms.
Quality standards and product availability
New Therapeutic Goods Orders (TGOs) were established in 2024, and came into effect in January 2025, setting minimum quality standards for psilocybin products, including both active pharmaceutical ingredients (API) and finished formulations (TGA, 2024). These TGOs prescribe requirements for:
Purity: Psilocybin must meet 80–120% assay limits, with strict impurity thresholds.
Good Manufacturing Practice (GMP) Compliance: All products must be tested in Australian GMP-licenced laboratories, even if manufactured overseas.
Exemptions: These standards do not apply to clinical trial products, which follow separate regulatory pathways.
Despite these standards, no psilocybin product is currently listed on the ARTG, and domestic manufacturing capacity remains limited (Hatfield et al., 2024). Currently, most clinics rely on imported formulations, which require both Office of Drug Control permits and TGA exemptions. This dual-layer approval process further compounds administrative delays and can interrupt continuity of care in the case of supply chain disruptions.
Regulatory tensions and systemic challenges
Australia’s framework reflects a cautious adoption of psychedelic medicine, seeking to balance therapeutic potential with public health safeguards. Nonetheless, several challenges remain (see Table 1).
Constraints limiting the clinical utility of psilocybin in Australia.

These constraints have led to low patient uptake, despite legal access. By late 2024, only 10 psychiatrists nationwide had TGA authorisation to prescribe psilocybin, a concerning contrast compared to the scale of unmet need (Nutt et al., 2024) and this may reflect the broader clinical and ethical landscape of psychedelic treatments in Australia.
Therapist training and model fidelity: the missing pillars
Unlike ketamine or transcranial magnetic stimulation (TMS), which are now supported by established nationally recognised training ecosystems, psilocybin-assisted therapy currently lacks a nationally endorsed professional training framework. Existing training options are fragmented, delivered primarily by private providers, unregulated and often out of step with public sector standards. Programmes such as those offered by Mind Medicine Australia (Certificate in Psychedelic-Assisted Therapies) and the Psychedelic Institute Australia are promising in intent, but lack national endorsement, independent oversight, and integration with existing clinical accreditation pathways.
Fidelity risks in psychedelic-assisted therapy (PAT) extend far beyond the technique utilised. In PAT, the therapist’s ontological stance including their worldview, bias and capacity to provide the non-intrusive psychological support that is helpful for patients in altered states becomes central to therapeutic outcomes (Sloshower et al., 2020). In the absence of standardised competencies, structured supervision and clear ethical frameworks, risks include deviation from established protocols, breaches of professional boundaries and misinterpretation of patient experiences (Phelps and Henry, 2021).
While foundational training programmes are now on offer in Australia, there is no centralised body responsible for overseeing therapist competencies. The RANZCP has initiated consultation on minimum competencies, emphasising experiential learning, ongoing professional development and trauma-informed care principles (Hatfield et al., 2024).
Thus, there is an urgent need for a nationally endorsed therapeutic framework, ideally modelled on evidence-based programmes used in clinical trials, such as the EMBARK model (Brennan and Belser, 2022) or the Compass Psychological Support Model (Kirlić et al., 2024) while integrating with trauma-informed care principles and safeguards against psychological overreach. In PAT contexts, where patients are vulnerable to identity disruption, the presence of trained and credentialed clinicians must be non-negotiable.
However, fidelity risks remain, including therapeutic drift and boundary violations (Johnson, 2020), the misapplication of psychedelic therapy to unsuitable populations and the phenomena of spiritual bypassing (the use of or reference to spiritual experiences to avoid addressing distressing emotional content) and identity destabilisation (Greń et al., 2024). While several therapeutic frameworks have been proposed including the EMBARK model and Compass Psychological Support Model, there is currently no consensus on a single best approach (Hatfield et al., 2024). A national framework need not imply rigid standardisation, but rather a set of minimum competencies, ethical safeguards and fidelity tools that can be adapted across therapeutic models. Hatfield et al. (2024) highlight the need for regulatory clarity and service delivery alignment, suggesting that a flexible but accountable structure is both necessary and feasible (Hatfield et al., 2024). As the evidence base evolves, it is likely that multiple frameworks will coexist, provided they meet agreed standards of safety, efficacy and professional oversight.
Economic barriers: equity and access in emerging psychedelic therapies
Preliminary cost modelling estimates for a full psilocybin-assisted therapy protocol in Australia is upwards of $20,000 per patient (Mihalopoulos et al., 2023). This figure includes psychiatrist assessment, preparatory psychotherapy, medical screening, dosing sessions, and integration therapy, none of which are covered under the Pharmaceutical Benefits Scheme (PBS), though Medicare rebates for psychiatry or psychology, may be applicable for some services. Thus, without public funding mechanisms, access to psychedelic therapy will remain largely limited to individuals with substantial financial means. This risks creating a two-tiered mental health system, where those most likely to benefit, such as people with severe, treatment-resistant conditions, are least likely to afford treatment. Concerningly, prohibitive costs combined with increasing media attention may result in those unable to afford legitimate treatment seeking out unregulated or non-medically prescribed treatment, putting them at substantial risk.
Nutt et al. (2024) report comprehensive psilocybin treatment outcome monitoring and data analysis mechanisms within the Australian clinical ecosystem; however, this highlights an additional risk. The absence of equity-focused funding models not only limits access to psilocybin-assisted therapy but also threatens the generalisability of treatment outcome findings. When treatment is accessible primarily to individuals with financial means, data reporting and analysis may become skewed towards socioeconomically advantaged groups. This reduces the external validity of data focused on clinical outcomes, particularly in conditions like TRD, which disproportionately affect individuals facing economic hardship, trauma and systemic disadvantage. Without inclusive funding mechanisms, such as public reimbursement, subsidised care pathways or value-based models there is a risk of overlooking the very populations most in need of innovative interventions. Ensuring equitable access is therefore not only a matter of social justice but also a prerequisite for generating clinically meaningful and generalisable evidence. Integrating public-private partnerships and exploring value-based care models such as outcomes-linked funding or bundled mental health packages may offer scalable solutions. Early general population evidence also supports the feasibility of appropriately supported group dosing (Fitzgerald et al., 2025), which may reduce treatment monitoring costs; however, this requires replication in clinical populations.
Thus, financial models should be designed to support equitable access and population-level benefit, complementing Australia’s regulatory leadership. Co-development of cost-effective models and reimbursement pathways through public–private partnerships could enhance access and system sustainability, safeguarding the integrity of the emerging psychedelic treatment ecosystem.
Ethical terrain: consent, identity and cultural safety
Psychedelic therapies uniquely challenge traditional psychiatric ethics. The consent process is complicated by the ineffability of psychedelic experience and resulting enhanced safety requirements, including the patient being unable to withdraw consent during a dosing session, after medicine administration. Informed consent must therefore address the unpredictable and subjective nature of the psychedelic experiences to mitigate risks to patient safety. Moreover, psilocybin’s potential to alter personality structure, shift core beliefs and reshape meaning-making processes challenges traditional boundaries of therapeutic neutrality (Smith and Sisti, 2021). Patients may leave treatment with profoundly altered life perspectives, spiritual orientations or social priorities (Lutkajtis and Evans, 2023). While such shifts are not inherently pathological, they can be destabilising without adequate support, especially when intersecting with trauma histories or cultural identities. They also pose consent complexities regarding properly informing patients about the potential for their personality or worldview to change as a result of these therapies. Navigating post-treatment meaning-making and addressing potential existential distress through psychological therapy engagement (post-treatment integration) is therefore pivotal to ensuring therapeutic gains are sustained and the experience is integrated in a way that supports long-term well-being (Gashi et al., 2021). Research has highlighted the importance of a supportive psychotherapeutic context and inclusion of integration sessions, as the provision of psilocybin treatment on a time-limited basis without integration into longer term therapy may further contribute to destabilisation, fragmented care and suboptimal treatment outcomes (Carhart-Harris et al., 2018). However, the necessity of these integration sessions increases the cost barriers if funding support is not provided.
Culturally responsive care must be embedded into every stage of psychedelic therapy with safety measures extending beyond tokenistic gestures. Without genuine therapeutic integration of the psychedelic experience into a patient’s ongoing life, and appropriate safeguards, marginalised populations may experience heightened risk of social, cultural and psychological harm. Indigenous frameworks of healing, community and spirituality offer profound wisdom. Engagement with Indigenous healing frameworks must be conducted respectfully and collaboratively to avoid misrepresentation or cultural harm (Brittain et al., 2025). Addressing these concerns requires embedded trauma-informed care as a foundational, not supplementary, element of practice.
Treatment processes should emphasise screening for vulnerability factors, relational safety, clinician experience and robust post-treatment support, including peer-led integration and the availability of post-dosing day supervision, after leaving the clinic. Spriggs et al. (2023) provide a timely example of a collaborative approach to the development of ethical guidelines.
Stratified psychiatry
The emergence of psychedelic-assisted therapies presents an opportunity to continue to advance a more neurobiologically informed approach to psychiatric care. Traditional diagnostic, and hence treatment, frameworks often assume homogeneity within mental health disorders, yet accumulating evidence demonstrates substantial neurophysiological variation both within and across diagnostic categories (Segal et al., 2023; Song et al., 2024). This heterogeneity has significant implications for the suitability of specific treatments for individuals within a diagnostic category (suggesting some treatments may not be suitable for some individuals, despite positive evidence from studies examining group average responses). This underscores the need for personalised approaches that move beyond group-level averages.
Psilocybin is not universally effective; individual variability in response degree and duration is well-documented (Kisely et al., 2023). Incorporating biomarkers into clinical decision-making may enhance therapeutic precision by enabling stratification of patients to treatments based on neurobiological profiles (Arns et al., 2022; Song et al., 2024). As data accumulates nationally and internationally, probabilistic models may emerge to estimate treatment response likelihood – supporting a personalised, data-driven approach to care. Even when biomarkers serve primarily to identify likely non-responders, this information holds clinical and ethical value by facilitating timely exploration of alternative treatments and reducing unnecessary exposure to ineffective interventions (as well as unnecessary exposure to the associated risks of interventions that are predicted to be ineffective).
In research contexts, biomarker-informed psychiatry is already influencing treatment selection for major depressive disorder and TRD, including applications in antidepressant therapy (Korgaonkar et al., 2020), TMS (Tozzi et al., 2024) and ketamine (Ionescu et al., 2018). Emerging evidence suggests similar relevance for psilocybin, with individual differences in functional brain connectivity predicting subjective experience – a key determinant of therapeutic outcome (Goodwin et al., 2025; Siegel et al., 2024; Yaden and Griffiths, 2021).
Biomarkers may be derived from various modalities; however, neuroimaging techniques are particularly promising, given their capacity to capture dynamic brain network activity. A focus on neuroimaging aligns with contemporary models of psychiatric illness as disturbances in network-level functioning rather than isolated regional deficits (Fornito and Harrison, 2012). Neuroimaging approaches that assess individual variation in receptor expression and topology – particularly within the serotonergic system – may offer valuable insights into how psilocybin modulates neural circuits and influences treatment trajectories (Palomero-Gallagher and Zilles, 2019; Singleton et al., 2022). Evidence-based biomarker treatment selection approaches are still developing, including for psilocybin where models have currently only been tested retrospectively in small sample sizes. Nevertheless, once effective models have been successful demonstrated in large sample sizes using sufficiently robust methods, the application of biomarker-informed treatment selection methods is likely to improve response rates to psychedelic therapy (as well as response rates to other treatments).
Designing services for the real world: proposals for structural system integrity
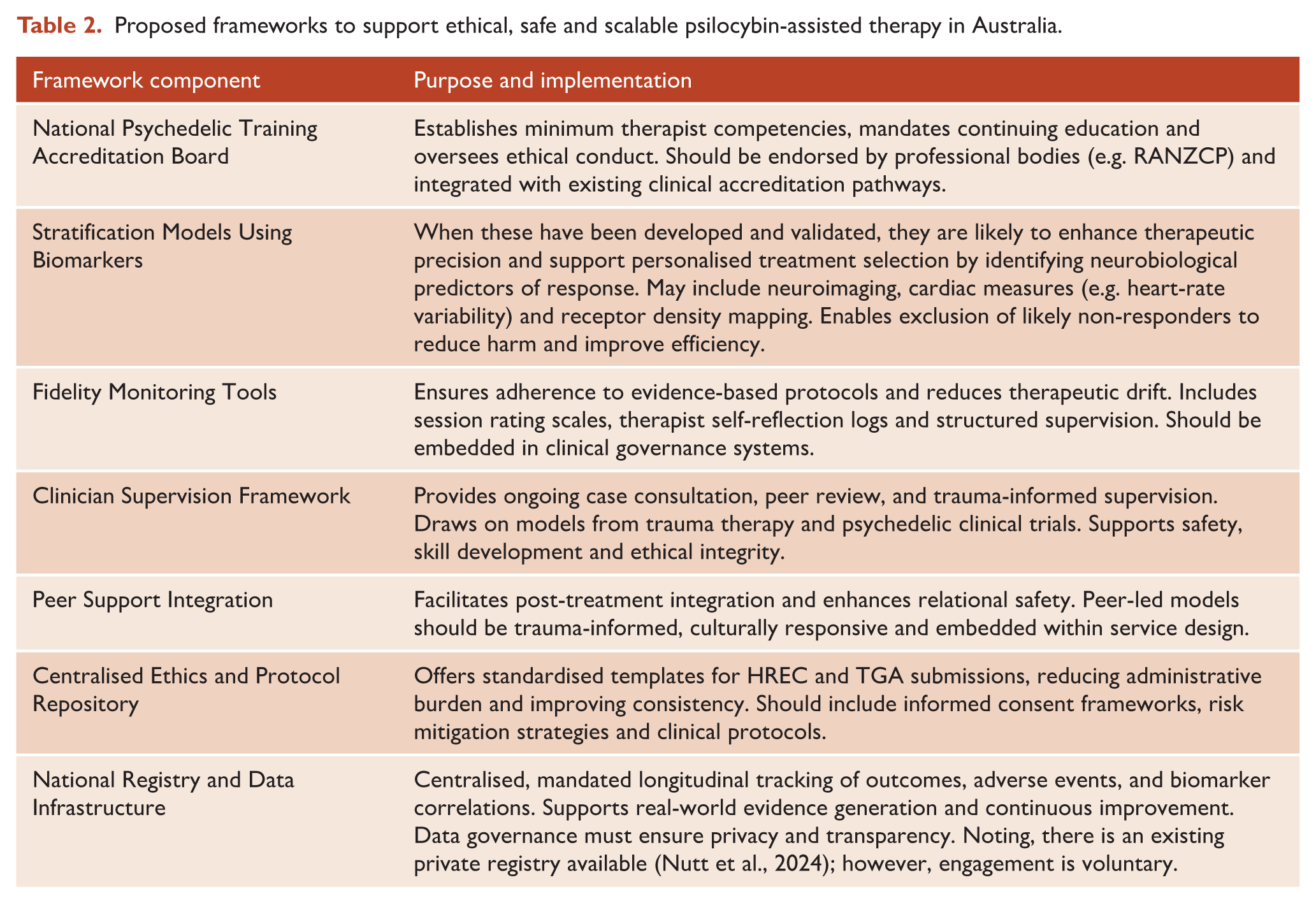
Psilocybin-assisted therapy must be delivered within systems that ensure accountability, patient stratification and ongoing clinician skill development. To minimise translational risks and advance ethical standards, we propose the development of a comprehensive governance framework (see Table 2) to guide, regulate and improve service provision.
Proposed frameworks to support ethical, safe and scalable psilocybin-assisted therapy in Australia.
Conclusion: complexity as the pathway to legitimacy
Australia’s regulatory leadership in psychedelic medicine presents a unique opportunity to establish a clinically rigorous and ethically grounded framework for psilocybin-assisted therapy. The reclassification of psilocybin for TRD has sparked significant interest, yet its successful integration into psychiatric practice requires more than regulatory reform. It calls for a coordinated strategy spanning clinical training, therapeutic fidelity, product availability and equitable access.
To support improved access, prescriber eligibility should extend beyond psychiatrists to other trained prescribers under robust supervision. Expanded eligibility could include mental health-trained practitioners with PAT supervision experience. Unlike medical cannabis, which can be used outside therapy, psilocybin is administered only within PAT, requiring strict safeguards. Australia’s medical cannabis rollout shows the risks of commercialised prescribing without proper clinical governance (Australian Health Practitioner Regulation Agency, 2025). Streamlining approval processes through centralised and nationally consistent protocols, alongside fast-tracked ARTG registration of psilocybin products and strengthened domestic manufacturing would help reduce operational delays and improve consistency of care. While limited commercial interest due to small market size may contribute to supply challenges, regulatory clarity and product registration remain essential to ensure reliable clinical access. Addressing economic barriers through public reimbursement mechanisms, such as PBS listing, Medicare coverage, National Disability Insurance Scheme (NDIS), or value-based funding models is essential to prevent socioeconomic exclusion and support population-level impact.
Equally important is the establishment of a national therapeutic framework that embeds trauma-informed care, cultural safety and structured pathways for ongoing professional development. Ethical considerations, particularly concerning consent, the potential for identity transformation and post-treatment integration, must be addressed through clinician education, clear professional standards and peer-supported models of care. Finally, future research should focus on long-term outcomes, biomarkers for patient stratification and culturally responsive models of care. Together, implementation of these recommendations can move psilocybin-assisted therapy from regulatory novelty towards a credible, ethical and accessible component of psychiatric practice. Australia has the opportunity to lead not only in regulatory innovation but also in the creation of a robust and evidence-based framework for clinical implementation. By embracing complexity and prioritising accountability, inclusivity and clinical integrity, the nation can ensure that psilocybin’s therapeutic potential is realised in a manner that meets the needs of diverse patient populations, is scientifically sound and upholds the integrity of psychiatric practice.
Footnotes
Acknowledgements
Large Language Model AI was used to optimise organisation and clarity.
Author contributions
M.D. conceptualisation wrote first draft; P.S., J.M., P.H., N.B., P.F., J.L., A.C. contributed to manuscript development, review and editing.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This research was internally funded by Thompson Brain Mind Healthcare. P.B.F. is supported by an NHMRC Leadership award (1193596).
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: In the last 3 years, P.B.F. has received reimbursement for educational activities from Otsuka Australia Pharmaceutical Pty Ltd and equipment for research from Brainsway Ltd. He is a founder of TMS Clinics Australia. The other authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
No data were produced in this research.
