Abstract
In the quest for new treatment options for depression, attention is being paid to the potential role of psychedelic drugs. Psilocybin is of particular interest given its mechanism of action, its benefits in early trials and its relatively low side effects burden. This viewpoint outlines a number of key issues that remain to be elucidated about its potential use in the clinical environment, including clarification of the profile of people most likely to benefit and those who might experience adverse effects, longer-term outcomes and the role of psychotherapeutic input alongside the drug itself. There are also opportunities to understand better, the neurobiology underpinning its effects.
Keywords
There is no doubt that we need new treatment options for mental illnesses, with novel mechanisms of action. In depression, we have been flogging the monoamine reuptake inhibition horse for decades now. Agomelatine has arguably been the only ‘novel’ antidepressant to come to market in many years (Rouillon, 2006). But now we are seeing renewed interest in ‘old’ drugs – licit and illicit – being repurposed to treat various mental illnesses. Medicinal cannabis is being explored for a number of indications (Hill, 2015); ketamine is all the rage (Krystal et al., 2019), with studies of intravenous and oral ketamine, and now intranasal esketamine (Canuso et al., 2018) demonstrating rapid improvements in depression and suicidality; 3,4-methylenedioxymethamphetamine (MDMA) has been usefully employed as an adjunct to psychotherapy in post-traumatic stress disorder (PTSD) (Mithoefer et al., 2019); and we can now add psilocybin to this list, which in conjunction with psychotherapy has shown benefit for end-of-life depression and anxiety, with emerging evidence of usefulness in addressing treatment-resistant depression. Indeed, the US Food and Drug Administration has designated psilocybin-assisted psychotherapy as a Breakthrough Therapy in the treatment of depression, expediting its development towards a prescription medication (see Williams and Warner, 2019).
Relevant to this discourse, three recent articles in The Australian and New Zealand Journal of Psychiatry serve as a ‘call to arms’ for Australian psychiatry, encouraging serious consideration of, and contribution to, the growing body of research into psychedelic therapies for maladies of the mind (Inserra, 2019; Puspanathan, 2017; Strauss et al., 2016). Here, we consider the next steps that Australian psychiatry might take in meeting this challenge. We focus specifically on psilocybin, as the first modern psilocybin trial in Australia is soon to commence (psilocybin-assisted psychotherapy to treat end-of-life anxiety and depression) and an Australian study of medicinal psilocybin for treatment-resistant depression is in advanced planning. Also, a recent study ranking drugs of abuse in Australia according to their potential to do harm has confirmed a very low harm rank for psilocybin (Bonomo et al., 2019).
Psilocybin is a naturally occurring molecule found in a number of mushroom species. Recreational use of psilocybin-containing ‘magic mushrooms’ for their ability to occasion an altered state of consciousness is widespread in Australia and globally, and users have long attested to beneficial mood effects (Carhart-Harris and Nutt, 2010). Indeed, through the 1950s and 1960s, tens of thousands of individuals participated in psychedelic research, mostly using lysergic acid diethylamide (LSD; a molecule close in structure and effect to psilocybin), with many of them showing positive mood and clinical improvements (Baumeister et al., 2014; Nichols, 2016). While few of these early studies stand up to current methodological standards, over the past decade, psilocybin’s ability to alleviate affective and addictive disorders has been tested in leading labs by highly credentialed researchers and with modern scientific rigour (see Williams and Warner, 2019).
Two of these studies – one from New York University and one from Johns Hopkins – focussed on end-of-life depression and anxiety in palliative care settings. The New York study (Ross et al., 2016) involved 29 patients in a randomised double-blind active placebo-controlled cross-over design. Antidepressant response was recorded in 83% of the psilocybin group, and the effect was very rapid (within a day). Only 14% of those in the control condition had an antidepressant response. Following only nine psychotherapy sessions, one psilocybin session, and one placebo session, positive effects found post-intervention were for the most part sustained at 6-month follow-up.
The Johns Hopkins study involved 56 individuals with end-stage cancer and used a similar crossover design to the study by Ross et al. (2016). Over 80% of those randomised to the therapeutic dose of psilocybin (‘placebo’ in this trial was low-dose psilocybin) showed improvements in mood, anxiety and quality of life, and again these effects largely endured at 6-month follow-up (Griffiths et al., 2016). As with all clinical psilocybin trials over the past decade, side effects were mild and transient.
With respect to protracted major depressive disorder, the Imperial College (London, UK) group of Carhart-Harris et al. (2018) performed a pilot open-label study of psilocybin in 20 people with treatment-resistant depression. Again, and noting the very small sample, results were encouraging. Low-dose (‘placebo’: 10 mg) psilocybin had no demonstrable mood effects, but an ‘active’ dose (25 mg), administered 7 days later, showed a significant and substantial reduction in depressive symptoms at 1 week, largely maintained at 6 months. Both anxiety and anhedonia symptoms were also reduced. Side effects were minimal and typically took the form of transient anxiety.
These results are encouraging. Rapid responses in people who have failed conventional antidepressants are clearly something both patients and physicians sorely need. With this kind of potential, we need to allocate considerable effort and resources to add to the knowledge-base to provide the field with a clearer sense of the place of this approach in the treatment armamentarium. Obviously, much larger samples are required in carefully powered studies. A key problem with extant trials is the selection process for participants. Participants are typically self-referred in clinical psychedelic trials, with many having prior experiences with psychedelics, begging questions about generalisability as well as anticipation bias. The Imperial College study authors acknowledge that psychedelics ‘promote suggestibility, which might have further enhanced positive outcomes’ (Carhart-Harris et al., 2016a: 626). These issues need to be assessed in future studies and factored into analyses where possible.
Furthermore, open-label studies need to be advanced to placebo-controlled experimental designs. However, psychedelics pose a particular challenge to blinding, as psychedelic experiences are difficult to mistake by either participant or therapist. Low-dose psilocybin and niacin have been used as ‘placebo’ in some studies. Another approach is to use a drug that mimics some of the psychedelic effects of psilocybin, but through a different mechanism of action. One of the complexities for the field is that there is evidence to suggest that certain psychedelic experiences are important for therapeutic benefit – an adequate active placebo would therefore need to produce large subjective alterations (e.g. perceptual distortions or hallucinations) which do not resemble features of the psychedelic experience that are likely to be clinically important. Given these difficulties in anticipation bias and blinding, the addition of objective outcome measures to future trials is crucial.
We also need to know how psilocybin stacks up against ‘conventional’ antidepressants. The above-mentioned Imperial College group have in train a randomised trial comparing psilocybin to the selective serotonin reuptake inhibitor escitalopram (ClinicalTrials.gov, number NCT03429075). This will be an influential study, but again, patient blinding will be difficult in both acute and continuation phases.
Studies to date have been very reassuring in terms of safety parameters, with mild (e.g. anxiety) and short-term (typically no more than a few hours) side effects being noted. But there needs to be ongoing scrutiny of safety and careful screening out of people who might be particularly vulnerable to an adverse psychiatric response to the drug. Most studies to date have excluded people with bipolar disorder, a psychotic illness, or family history of psychosis. Given the established psychotomimetic aspects of classical psychedelic effects, this might be refined further, potentially using a dimensional measure of ‘psychosis proneness’ for screening.
We need to understand better, dosing and dose strategies over the longer term. Some trials have employed a fixed dose (e.g. 25 mg in Carhart-Harris et al., 2018), others a weight-dependent dose (e.g. 0.3 mg/kg in Ross et al., 2016). Variation in dose–response curves is common among antidepressants, and it is unlikely that there will be a simple ‘one size fits all’ with psilocybin. To note, it has generally been shown that 10 mg is sub-therapeutic and 30 mg is too strong (Carhart-Harris et al., 2018; Griffiths et al., 2016).
Regarding longer-term outcomes, Carhart-Harris et al. (2018) reported that some of their patients had sustained mood improvement out to 6-month follow-up, but others showed a recurrence of depression, almost back to their baseline. We are aware of some people who find that self-medicating with ‘magic mushrooms’ has an acute beneficial effect on mood, but that the effects wane and require repeat exposure every 3–4 weeks. Understanding these different patterns of response and persistence of response over longer time periods is critical. In addition, given the central importance of various non-chemical factors in this therapeutic approach, more systematic exploration of the psychotherapeutic, procedural and contextual factors in which psilocybin is delivered may highlight methods to optimise sustained clinical outcomes.
Another area in which more clarity and depth of understanding is required is the mechanism of action of psilocybin. Psilocybin is a prodrug of psilocin, which is a partial agonist at serotonin 5HT2A and 5HT2C receptors, thus directly impacting the serotonergic system. Ly et al. (2018) showed that several psychedelics increase brain-derived neurotrophic factor (BDNF), with effects on neural plasticity. A number of transient neurobiological changes in functional connectivity are also associated with psilocybin. The Imperial College group (Carhart-Harris et al., 2017) showed acute decreases in functional connectivity within the default mode network (DMN) on exposure to psilocybin: a network that is associated with introspection, self-reference and autobiographical memory. This change is then reversed post-psilocybin, with an increase in DMN connectivity (Carhart-Harris et al., 2017). Taken together, these findings have been interpreted as a potential ‘reset’ mechanism. At the same time, psychedelics increase synchrony between higher-order modules (e.g. DMN) and lower-order, task-positive networks, structures that are usually anti-correlated (Carhart-Harris et al., 2016b). Psychedelic compounds have also been shown to reduce the constraints imposed on lower-order regions by higher-order modules, transiently reversing normal hierarchical dynamics (Alonso et al., 2015). These changes may correspond to a rebalancing of predictive processing, a reduction in excessive top-down processing leading to a reduction in cognitive and affective biases. Indeed, more realistic forecasting of future life events has been observed after psilocybin-assisted psychotherapy in patients with depression (Lyons and Carhart-Harris, 2018). These alterations may also relate to decreases in self-focus and increases in perceptual coupling, potentially interrupting abstract self-oriented rumination that is characteristic of depressive thinking. This story needs further explication and interrogation, both in short- and longer-term studies.
Conventional antidepressants have also been shown to increase BDNF (Björkholm and Monteggia, 2016; Castrén, 2014) and resolve biases (Harmer et al., 2009), indicating they may have partially overlapping mechanisms. However, the potency and timescale at which these compounds induce such effects may be a crucial difference. While conventional antidepressants induce changes gradually, psilocybin appears to produce an intense acute state of increased neuroplasticity and altered functional connectivity. This state may offer an invaluable opportunity for psychotherapy to guide and solidify these changes. This concept of pharmacotherapy and psychotherapy working synergistically is gaining traction within psychiatry (see Wilkinson et al., 2018) and the properties of psilocybin may place it at the forefront of this. However, whether there are synergistic or simply additive effects of psilocybin and therapy is yet to be tested.
Despite these suggestions that psychedelics may ‘open up the mind’ to psychotherapeutic input, therapies employed in research remain largely non-directive and unstructured. The Imperial College group describe the ‘psychological support’ in their psilocybin study as ‘allowing the patient to experience a mostly uninterrupted ‘inner journey’’ (Carhart-Harris et al., 2016a: 624). While the usefulness of certain common aspects of psychedelic psychotherapy is patent (e.g. developing the therapeutic alliance, non-avoidance coaching), and the clinical approach (compared to recreational, for example) is considered important for clinical outcomes, what works and what is worth improving is not known. Relatedly, it is not known whether more structured psychotherapeutic interventions would be more or less effective in this context. In the end-of-life studies, components from a number of existential modalities were utilised – including meaning-centred psychotherapy (Breitbart and Heller, 2003), dignity therapy (Chochinov et al., 2004) and elements of psychodynamic psychotherapy – to facilitate processing and meaning-making of emerging symbolic content (Ross et al., 2016). But arguably the psychological techniques appropriate to the terminal stage of life might not translate to depression at other life stages. In terms of learnings from the PTSD psychedelics literature, of the six Phase 2 trials testing MDMA-assisted psychotherapy for PTSD, all found consistently favourable outcomes, and all but one were conducted by clinicians trained through the Multidisciplinary Association for Psychedelic Studies (MAPS) who followed a specific manualised therapy (Mithoefer et al., 2019). The MAPS treatment protocol incorporates aspects of ‘sensorimotor psychotherapy’ (trauma-based psychotherapy), and as such is quite specific to the PTSD/complex PTSD population. Perhaps more directly applicable to people with major depression, there are effective mindfulness-based emotion regulation interventions/strategies that could be applied in preparatory, dose and integration phases of the psychedelic experience, where chronically depressed patients experience anxiety upon accessing avoided affects during therapy. At the very least, we suggest that future psilocybin studies provide detailed descriptions of what ‘psychotherapy’ actually entails. Questions of interest include the possible mechanistic role of schema change (especially self-related schema), emotional breakthrough and acceptance. A nuanced understanding of patient’s subjective experience while under the influence of the drug will also be informative, as certain types of experience might be more (or less) predictive of response (or failure of response). Although the inherent ineffability of the psychedelic experience makes it difficult to capture, the development of new questionnaires and measures may help facilitate this.
The ability of psilocybin to induce neuroplasticity and changes in functional connectivity suggests it may be useful not only in treating depression but also in a range of mental health disorders in which there are deeply entrenched maladaptive patterns of thoughts, feelings and behaviours. Indeed, psilocybin has shown promising results in obsessive-compulsive disorder (Moreno et al., 2006) and substance use disorders (Rao and Lydall, 2019). Other potential indications that would be predicted to benefit from these changes, including anorexia nervosa and body dysmorphic disorder, remain untested, though a study of anorexia has begun at John Hopkins University (ClinicalTrials.gov, number NCT04052568). For a summary of ongoing research using classical psychedelics for therapeutic purposes, see Table 1. Investigation of a wider range of indications may help uncover the therapeutic mechanisms of psilocybin, as well as common underlying pathologies of disorders.
Ongoing studies of classic psychedelics for various clinical indications.
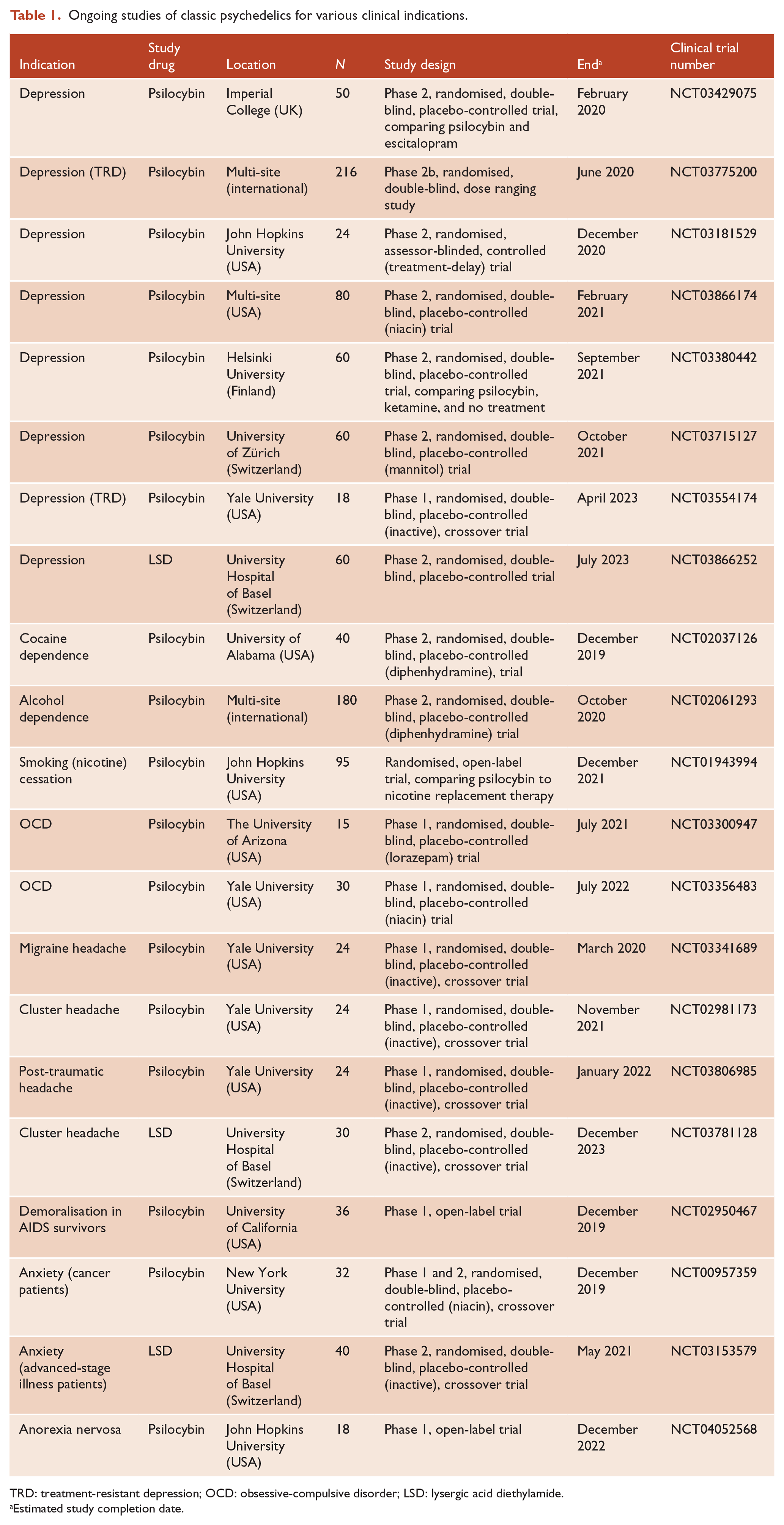
TRD: treatment-resistant depression; OCD: obsessive-compulsive disorder; LSD: lysergic acid diethylamide.
Estimated study completion date.
Psilocybin will not be approved as a stand-alone therapy, and researchers will need to work with authorities to develop regulatory frameworks for psilocybin-assisted therapy. Drugs for mental illnesses are regulated, psychosocial therapies are not, and regulators are just beginning to tackle the questions raised by such hybrid interventions (see, for example, Bain et al., 2017).
In sum, the potential reintroduction of psychedelics such as psilocybin into our psychiatric armamentarium is exciting for the field as well as for those many people who have enduring psychiatric symptoms despite current medications. Australia should be at the forefront of future research into this potentially disruptive intervention. We suggest that in taking the next steps, the field needs to seek to refine therapeutic ‘best practice’ as well as use the opportunity to advance our understanding of the psychology and neurobiology of disorders of the mind.
Footnotes
Declaration of Conflicting Interests
D.J.C. has received grant monies for research from Eli Lilly, Janssen Cilag, Roche, Allergen, Bristol-Myers Squibb, Pfizer, Lundbeck, Astra Zeneca, Hospira; Travel Support and Honoraria for Talks and Consultancy from Eli Lilly, Bristol-Myers Squibb, Astra Zeneca, Lundbeck, Janssen Cilag, Pfizer, Organon, Sanofi-Aventis, Wyeth, Hospira, Servier, Seqirus, and is a current Advisory Board Member for Lu AA21004: Lundbeck; Varenicline: Pfizer; Asenapine: Lundbeck; Aripiprazole LAI: Lundbeck; Lisdexamfetamine: Shire; Lurasidone: Servier; Brexpiprazole: Lund-beck; Treatment Resistant Depression: LivaNova. He is founder of the Optimal Health Program, currently operating as Optimal Wellness. He is on the board of Mind Medicine Australia. He does not knowingly have stocks or shares in any pharmaceutical company.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
