Abstract
Objective:
To determine the extent of cardiometabolic monitoring occurring in children/adolescents 18 years and under receiving antipsychotics in Australian general practice: study period 2011 to 2017.
Methods:
Retrospective cohort study using large primary care database, MedicineInsight. Rates of monitoring within 12 months of patients receiving an incident antipsychotic prescription were reported descriptively for weight, body mass index (BMI), waist circumference (WC), glucose, total cholesterol, triglycerides, high-density lipoproteins (HDL), blood pressure, liver function, prolactin, and metabolic syndrome (MetS) defined as weight, blood pressure and glucose. Target trial methodology was used to examine whether monitoring rates were higher in patients prescribed antipsychotics compared to patients not receiving antipsychotics.
Results:
55.9% of patients using antipsychotics were monitored at least once on at least one parameter. Weight was most frequently measured (39.1% patients measured at least once), followed by BMI (31.2%), blood pressure (29.0%), glucose (12.9%), liver function (9.1%), total cholesterol (8.1%), triglycerides (6.7%), HDL (3.1%), WC (1.6%) and prolactin (1.1%). Monitoring for MetS occurred at least once in 1.5% of patients. Antipsychotic users recorded a small but significantly higher rate of monitoring than non-users (1.157 vs 0.691 monitoring visits for any parameter within the 12 month measurement period, difference 0.461; 95% CI [0.271, 0.660], p < 0.001).
Conclusion:
Rates of cardiometabolic monitoring were well below guideline recommendations. Our results suggest the need for education and training to increase awareness of adverse health impacts of antipsychotics and improve adherence to cardiometabolic guideline recommendations. Communication between primary and secondary health care providers also needs to be addressed.
Introduction
In Australia general practitioners (GPs) are increasingly engaged in paediatric antipsychotic prescribing (Klau et al., 2022). A study using data from a large primary care database, MedicineInsight, found prescribing of antipsychotics to children/adolescents attending Australian general practices increased by 62.8% from 2011 to 2018 (Klau et al., 2022).
Children/adolescents taking antipsychotics are especially vulnerable to adverse metabolic effects; they experience more rapid changes in weight and drug-induced lipid and glucose abnormalities compared with adults (Correll et al., 2009). Childhood metabolic syndrome (MetS), defined as being overweight/obese and experiencing abnormal levels of at least two other parameters (i.e., high blood glucose, low levels of high-density lipoproteins [HDLs], high triglycerides, and high blood pressure [BP]) (Zimmet et al., 2007), has been shown to increase significantly within 12 months of children/adolescents initiating second-generation antipsychotics (SGAs) (Sjo et al., 2017). Childhood MetS has also been shown to predict adult MetS from as young as 5 years of age and adult type 2 diabetes and subclinical atherosclerosis from age 8 (Koskinen et al., 2017). This raises concerns that antipsychotics, due to their effect on metabolic parameters, could propel children and adolescents into lifelong health problems and premature mortality.
Concerns about the cardiometabolic effects of antipsychotics have prompted many jurisdictions around the world (McClellan et al., 2013; National Institute for Health and Care Excellence, 2013; Pringsheim et al., 2011; Scottish Intercollegiate Guidelines Network, 2013) and in Australia (Health Education and Training Institute (HETI), 2014; Women’s and Children’s Health Network (WCHN), 2019) to develop protocols for monitoring metabolic health to detect problematic changes early and implement strategies to manage the adverse effects. In Australia, the Women’s and Children’s Health Network (WCHN, 2019), in collaboration with the University of South Australia, developed an evidence-based programme in 2005 (Supplemental Figure 1). Similar to international monitoring algorithms referenced above, this programme includes regular monitoring of weight, BP, glucose levels, lipid profiles, and liver function when commencing antipsychotics in children/adolescents.
Australian studies examining metabolic monitoring of children/adolescents receiving antipsychotics are sparse, with only two including children aged under 15 years: a small pilot study involving 10 doctors (psychiatrists, paediatricians) and 17 patients published in 2008 (Grzeskowiak et al., 2008), and a prospective 3-month audit of patients 0 to 17 years undertaken at the Women’s and Children’s Hospital in Adelaide, South Australia, in 2012 (Ellis and Angley, 2013). No studies to date have examined the extent to which metabolic monitoring is occurring in paediatric patients receiving antipsychotics in Australian primary care.
This study aims to examine rates of cardiometabolic monitoring occurring in children/adolescents who are prescribed antipsychotics in Australian general practice. The results are reported descriptively and differences in monitoring rates between antipsychotic users and non-users analysed using target trial methodology.
Methods
Study design and data source
A retrospective study of de-identified electronic health records was conducted using MedicineInsight, a large longitudinal Australian primary care database. MedicineInsight was established in 2011 with funding from the Australian government to monitor medicine safety and quality use of medicines. In the 2018/2019 financial year, the database included 7% (569) of all Australian general practices and 9% (3255) of all GPs (NPS MedicineWise, 2020). Information on patient demographics, reasons for clinical encounters, medications prescribed, diagnoses, medical tests, and results is uploaded monthly to MedicineInsight using a secure third-party extraction tool. All patients in the database receive a unique identifier, which allows for tracking their data over time and across different data sets. To improve data quality, MedicineInsight only provides researchers with data from general practice sites established for at least 2 years with no interruptions of more than 6 weeks to data provision and a consistent volume of transactions (Busingye et al., 2019).
Study samples
Two samples were used in this study (see Supplemental Table S1 for sample characteristics). Descriptive analyses were initially used to examine monitoring in a subsample of antipsychotic users (3840 patients, 488 practices). While SGAs are known to cause cardiometabolic issues in paediatric patients (Correll et al., 2009; Sjo et al., 2017), first-generation antipsychotics (FGAs) have also been shown to cause significant weight gain (Bak et al., 2014) and have been associated with cardiometabolic adverse events (Falissard et al., 2011) and metabolic syndrome in adults (De Hert et al., 2008). Therefore, we included a sample of all antipsychotics, both first- and second-generation, available in Australia and listed in the Anatomical Therapeutic Chemical (ATC) index (Supplemental Table S2). Patients were included if they received an antipsychotic prescription during the study period and were followed up for 12 months post their incident prescription. Incident prescriptions were identified if there was a minimum of 12 months antipsychotic-free prior to their first recorded prescription. To ensure a 12-month follow-up period was available for all patients, we restricted the sample to patients who had an incident antipsychotic prescribed from 1 January 2011 to 31 December 2017.
For the second analysis using target trial methodology, the sample was restricted to patients aged 3–18 years. The sample of 555,440 (547 practices) included 3829 patients who were prescribed an antipsychotic.
Outcomes
Monitoring parameters were determined by reference to guidelines created by the WCHN in South Australia (WCHN, 2019) and an algorithm provided by the Health Education and Training Institute (HETI, 2014) in New South Wales, Australia. Recommended metabolic measures and monitoring frequencies in both guidelines are presented in Table 1.
Monitoring algorithms for cardiometabolic health in children/adolescents. (a) Health, Education and Training Institute (HETI) – adolescents using psychotropic medications; (b) Women’s and Children’s Health Network (WCHN) – children and adolescents using antipsychotics.
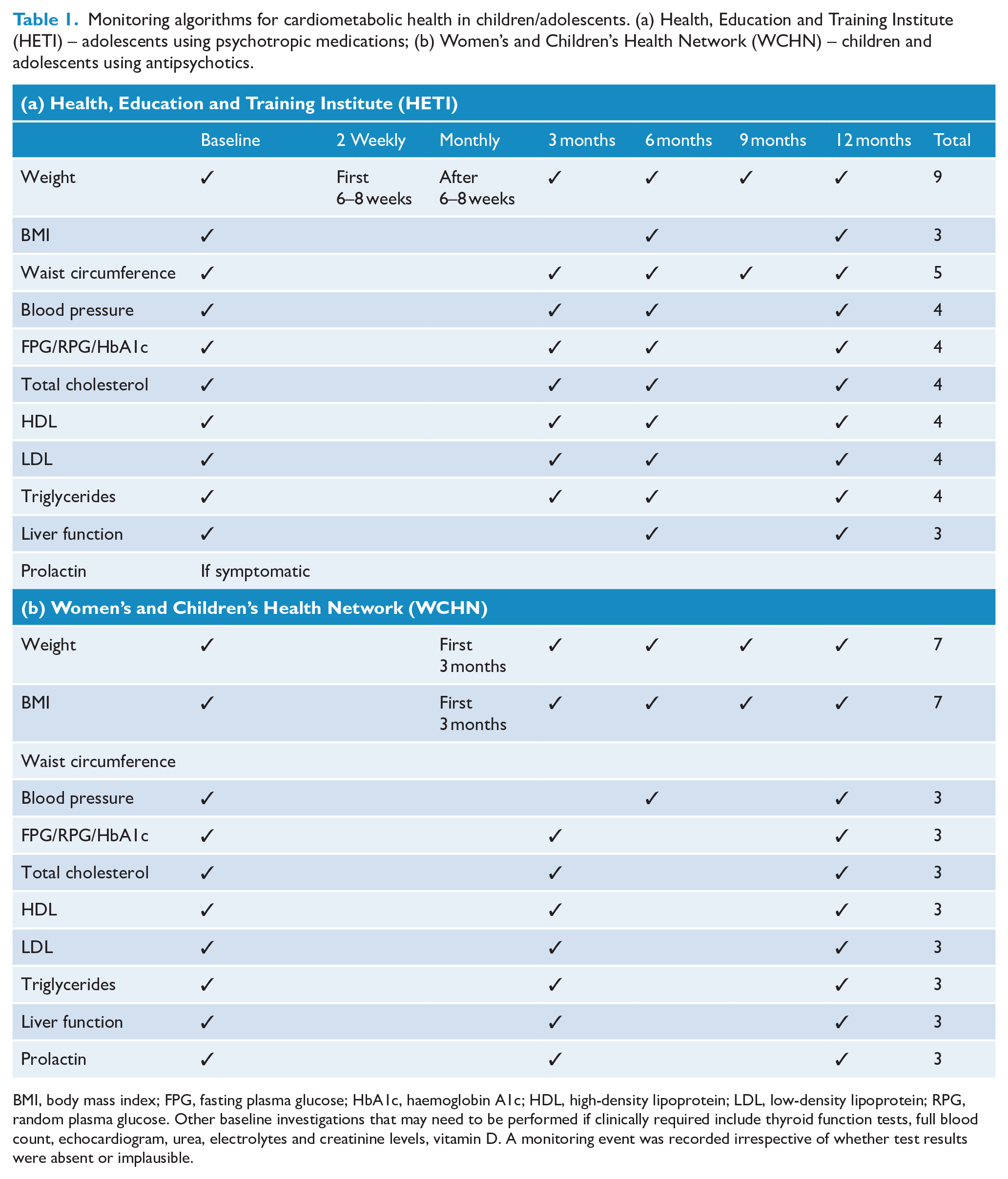
BMI, body mass index; FPG, fasting plasma glucose; HbA1c, haemoglobin A1c; HDL, high-density lipoprotein; LDL, low-density lipoprotein; RPG, random plasma glucose. Other baseline investigations that may need to be performed if clinically required include thyroid function tests, full blood count, echocardiogram, urea, electrolytes and creatinine levels, vitamin D. A monitoring event was recorded irrespective of whether test results were absent or implausible.
Monitoring information was extracted from two sources in the MedicineInsight database: the pathology data set containing records of laboratory tests and the observations data set that contains data on physical examinations. Outcome parameters included weight, BMI, WC, BP, glucose (fasting and random blood glucose, HbA1c), lipids (total cholesterol, HDL, LDL, triglycerides), and prolactin levels. Liver function testing reported measurements of alkaline phosphatase. As we were only interested in determining if monitoring was taking place, we included all monitoring events, even if test results were absent or implausible. We also created a combined variable to determine if any of the parameters were measured, at least once, within the follow-up period. The follow-up period was 12 months after the incident antipsychotic prescription, inclusive of baseline, using date of incident antipsychotic prescription as baseline.
Two variables to examine the rate of measurement of MetS were created. The first measure included all five parameters listed in the International Diabetes Federation (IDF) definition (overweight/obesity, triglycerides, HDLs, BP, glucose) (Zimmet et al., 2007). The second variable consisted of measurements for three parameters: weight plus the two most frequently measured MetS parameters in our data set (glucose, BP). Although the IDF definition specifies WC as the appropriate weight parameter (Zimmet et al., 2007), we substituted ‘weight’ due to very low rates of WC measurement.
Sensitivity analyses
Overweight or obesity at baseline was considered an important covariate to include in target trial analyses but was excluded due to high levels of missing data (89.5%). Therefore, we performed sensitivity analyses on a subsample with weight recorded at baseline and a subsample of patients who were overweight/obese at baseline. We also performed a sensitivity analysis with patients who had at least one GP visit in the 12 months following the incident event (i.e., at least two GP visits during the follow-up period).
Covariates used in target trial analyses
Potential confounding covariates for the target trial analyses were selected based on factors known to be related to the treatment and outcomes, and a directed acyclic graph (DAG) generated using an online tool available at daggity.net (Textor et al., 2016; Williamson et al., 2014). The DAG used in the current analysis is provided in Supplemental Figure S2.
Covariates included in the target trial analyses included age, gender, state, socioeconomic status of the practice and the patient, indigenous status, presence of allergies, smoking status, and health conditions.
Health conditions
The variable for health conditions contained three levels related to the likelihood of receiving an antipsychotic for the condition: (1) conditions for which antipsychotics are approved by the Australian Therapeutic Goods Administration (TGA) for children/adolescents, (2) non-TGA-approved conditions, and (3) no diagnosis on record (Supplemental Table S3 lists diagnoses included in each level). As we were primarily interested in monitoring triggered by the prescription of antipsychotics, we excluded all antipsychotic users who had diagnoses of diabetes, hypertension, or dyslipidaemia on record prior to the incident antipsychotic prescription. Diagnoses were extracted from three fields in the MedicineInsight database (reason for diagnosis, reason for encounter, reason for prescription) using methods that have been validated by Havard et al. (2021). A diagnosis was recorded if present in any of the three fields.
Sociodemographic covariates
Age was recorded in years, and gender recorded as male or female. Socioeconomic status was derived from the Index of Relative Socioeconomic Advantage and Disadvantage (IRSAD) divided into quintiles. IRSAD is an indicator that summarises information derived from census data (Australian Bureau of Statistics, 2016) on individual and household conditions. Rurality was divided into major cities, inner regional and outer regional/remote/very remote areas. Indigenous status was divided into three categories – indigenous, non-indigenous or not stated/recorded. Missing data for all sociodemographic covariates were minimal, ranging from 0% to 0.33% (Supplemental Table S4).
Statistical analyses
Antipsychotic subsample
Using the subsample of patients prescribed antipsychotics, crude descriptive statistics were used to report the percentage of patients who had a given cardiometabolic parameter measured within 12 months, inclusive of the day of incident antipsychotic prescription, reported in categories of once, twice, or three or more times.
Target trial analyses
Target trial methodology (Hernán et al., 2022) was used to determine if rates of monitoring were higher in children/adolescents prescribed antipsychotics compared to those who attended a MedicineInsight practice with no recorded antipsychotic prescription. The incident date for the comparison group was the date of first attending the practice. Target trial methodology emulates a randomised controlled trial using epidemiological data by employing Augmented Inverse Probability Weighting (AIPW) (Funk et al., 2011). AIPW is a doubly robust procedure that estimates causal effects by specifying two regression models: one for the outcome and one for the exposure (Funk et al., 2011). In the doubly robust procedure, only one of the models needs to be correctly specified to produce unbiased results. We used logistic regression for the treatment variable (exposure to antipsychotics) and Poisson regression for the outcome variables.
There were very few children aged under three years receiving antipsychotics (11 patients), so to facilitate the AIPW analyses, we restricted the sample to patients aged 3–18 years.
Stata version 16 (StataCorp, Texas, USA) was used for all analyses.
Ethics approval
This study was approved by the MedicineInsight Data Governance Committee (protocol 2019-029) and exempted from full ethical review by the University of Adelaide Human Research Ethics Committee due to use of non-identifiable data.
Results
Analysis of antipsychotic subsample
The frequencies of cardiometabolic monitoring within 12 months of the incident antipsychotic prescription, inclusive of baseline (the follow-up period), are shown in Figure 1. 55.9% of patients were measured at least once on at least one measure. Weight was the most frequently measured (39.1% at least once during the follow-up period), but only 10.4% of patients were measured on three or more occasions, despite guidelines suggesting 7–9 times (Table 1). Blood pressure was the next most frequently monitored (31.2% at least once during the follow-up period), with 7.1% monitored on three or more occasions. Most measures requiring pathology testing were monitored less frequently, ranging from 1.1% for prolactin to 12.9% for glucose. Monitoring for all five MetS factors occurred once in 0.1% of patients, while monitoring of the three-factor MetS definition (i.e., weight, BP, glucose) occurred once in 1.3% of patients and twice in 0.2% during the follow-up period.
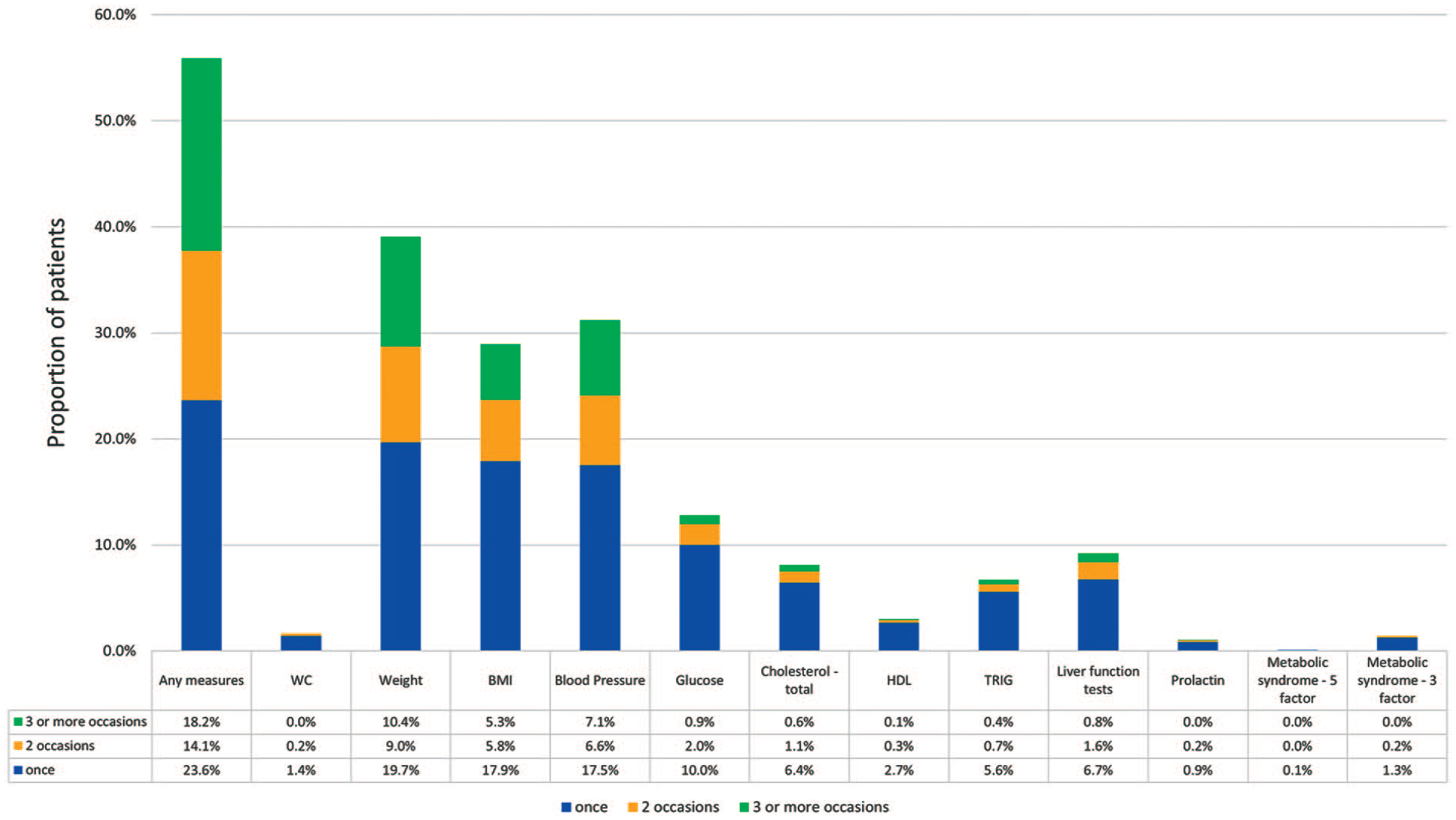
Proportion of patients with cardiometabolic measurements taken within 12 months of incident antipsychotic prescription, by number of occasions.
AIPW analysis of full sample
For the AIPW analysis, aggregated measures were used for weight and lipids. The aggregated weight measure included records for weight, BMI, or WC, while aggregated lipids included total cholesterol, HDL, LDL, or triglycerides.
Antipsychotic users were almost twice as likely as non-users to be measured on any cardiometabolic parameter within the follow-up period (Potential Outcome Mean [POM] = 1.157 with antipsychotic vs POM = 0.691 no antipsychotic; Table 2). Measurement of weight was one-third more likely to occur during the follow-up period for antipsychotic users. The average treatment effect (ATE), defined as the mean outcome difference between the treatment and control groups, was 0.323 for weight. Measurement of BP had the next highest ATE, at one-seventh (ATE = 0.165). All other ATEs were negligible, around 0.050.
Comparison a of the frequency of monitoring events for incident antipsychotic users compared to non-users aged 3–18 years.
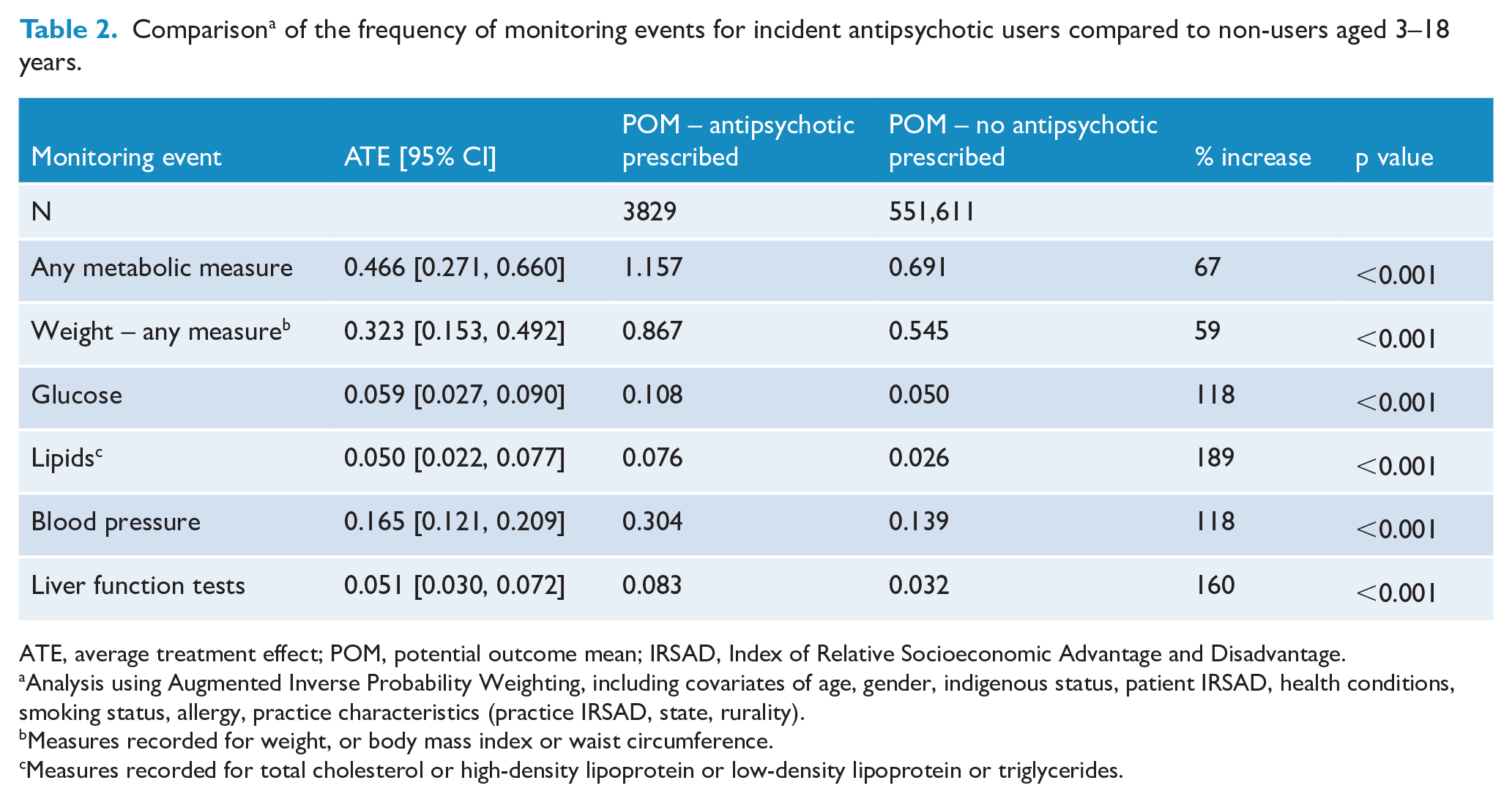
ATE, average treatment effect; POM, potential outcome mean; IRSAD, Index of Relative Socioeconomic Advantage and Disadvantage.
Analysis using Augmented Inverse Probability Weighting, including covariates of age, gender, indigenous status, patient IRSAD, health conditions, smoking status, allergy, practice characteristics (practice IRSAD, state, rurality).
Measures recorded for weight, or body mass index or waist circumference.
Measures recorded for total cholesterol or high-density lipoprotein or low-density lipoprotein or triglycerides.
Sensitivity analyses
The analysis restricted to patients with a record of weight at baseline exhibited almost double the rates of monitoring for any measure for patients prescribed antipsychotics (POM with antipsychotic = 3.282 vs no antipsychotic = 1.735; Table 3(a)). The POMs for all measures analysed were higher for users of antipsychotics, although rates of glucose and lipid monitoring did not differ significantly between users and non-users. POMs for patients receiving antipsychotics ranged from 1.8 times higher for weight assessments to 3.9 times higher for liver function tests. A sample of patients who were overweight/obese at baseline showed similar results (Table 3(b)).
Sensitivity analyses a comparing the frequency of monitoring events for antipsychotic users and non-users aged 3–18 years.
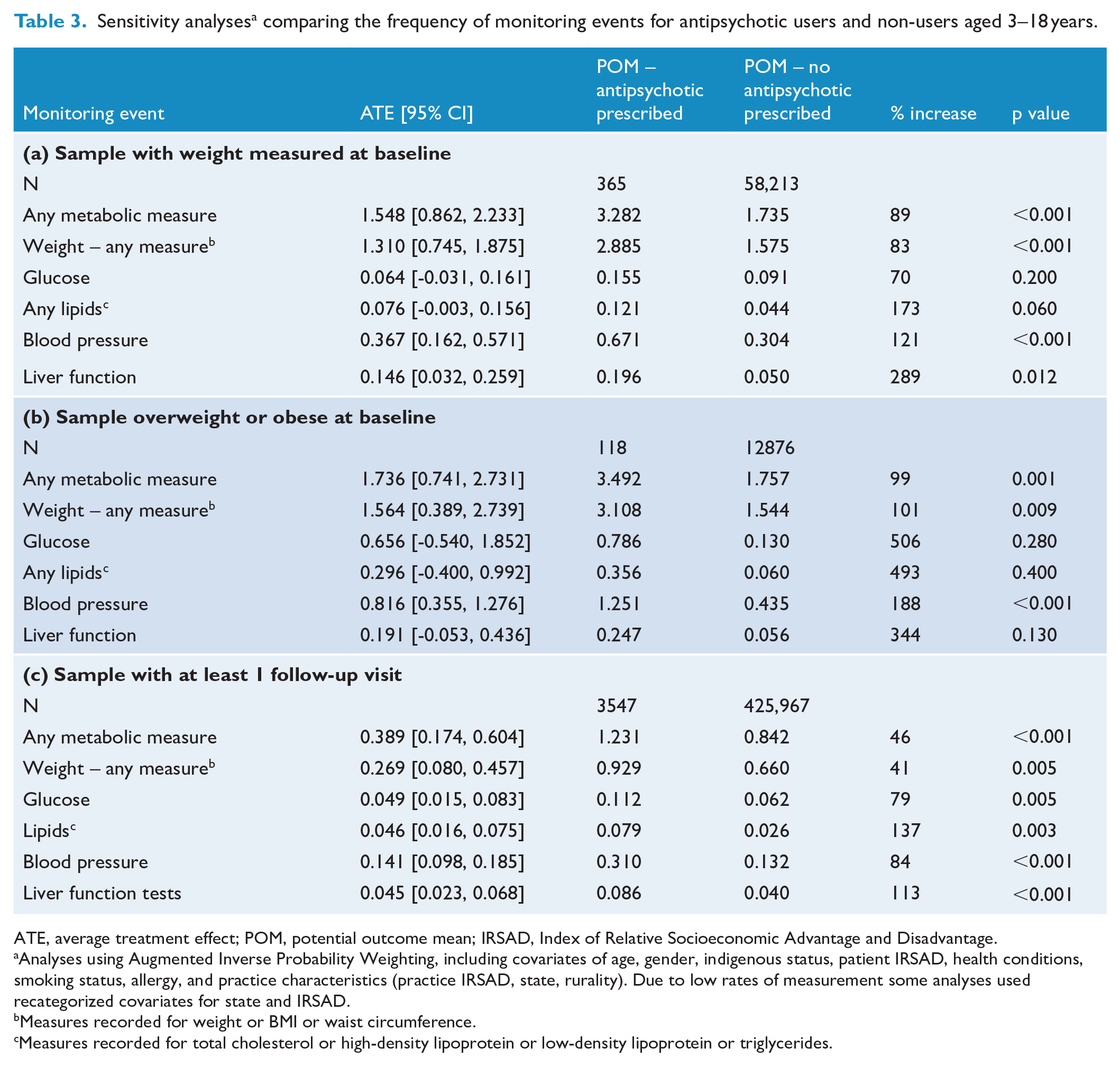
ATE, average treatment effect; POM, potential outcome mean; IRSAD, Index of Relative Socioeconomic Advantage and Disadvantage.
Analyses using Augmented Inverse Probability Weighting, including covariates of age, gender, indigenous status, patient IRSAD, health conditions, smoking status, allergy, and practice characteristics (practice IRSAD, state, rurality). Due to low rates of measurement some analyses used recategorized covariates for state and IRSAD.
Measures recorded for weight or BMI or waist circumference.
Measures recorded for total cholesterol or high-density lipoprotein or low-density lipoprotein or triglycerides.
A sensitivity analysis examining results for those who attended their general practice at least twice during the study period showed similar results, although the ATE for all parameters was lower (Table 3(c)).
Discussion
This study examined rates of monitoring of cardiometabolic parameters in children/adolescents within 12 months of receiving their first antipsychotic prescription using a large primary care database, MedicineInsight. Monitoring rates were well below recommended Australian guidelines. Weight was the most frequently monitored parameter, being measured at least once in 39.1% of patients, inclusive of the day of incident prescription. Glucose and total cholesterol were measured at least once in 12.9% and 8.1% of patients, respectively. Despite these low rates, however, we found limited evidence that patients receiving antipsychotics were more likely to be monitored than patients not receiving antipsychotics.
Monitoring within 12 months of incident antipsychotic prescribing
Rates of monitoring in our study were suboptimal and below those found in international studies. Examining 0–18 year olds using the UK Clinical Practice Research Datalink primary care database (Minjon et al., 2022), researchers found rates of monitoring within 6 months post antipsychotic prescription to be similar to our 12 month figures for glucose (12.0% vs 12.9% our study) and lipid (7.6% vs 8.1%) testing but superior for prolactin (6.2% vs 1.1%). Although we had higher percentages monitored for weight (39.1% vs 22.8%) and BP (31.2% vs 20.9%), this was likely due to the differing study period (6-month vs our 12-month period). Indeed, our longer follow-up period would also suggest our rates of glucose and lipid testing were poorer.
In a Canadian study of almost 7000 children under 19 years (Chen et al., 2018), glucose monitoring rates 3–12 months post antipsychotic prescription (13.0%) matched those found in our study’s 12-month follow-up period (12.9%), but rates for lipid (15% vs 8.1% our study) and prolactin (7.5% vs 1.1%) monitoring were much lower in our data set. Our low rates of prolactin monitoring are particularly concerning. Although some guidelines suggest monitoring prolactin only if patients are symptomatic (American Academy of Child and Adolescent Psychiatry, 2011; HETI, 2014), others recommend regular monitoring (Okumura et al., 2018; Pringsheim et al., 2011; WCHN, 2019). Hyperprolactinaemia is associated with endocrine disturbances and serious adverse effects, including hypogonadism, gynaecomastia, and osteoporosis (Eren et al., 2011), but symptoms do not correlate closely with prolactin levels (Walter et al., 2008). Walter et al. (2008) therefore recommend monitoring prolactin changes from baseline as an essential part of good monitoring practices.
It is also notable that WC was rarely measured despite being included in Australian (HETI, 2014) and international (American Academy of Child and Adolescent Psychiatry, 2011; National Institute for Health and Care Excellence, 2013; Pringsheim et al., 2011) paediatric guidelines and being recommended as a critical measure by the IDF (Zimmet et al., 2007).
Comparing antipsychotic users and non-users
Although crude figures for monitoring were all suboptimal and well below recommended guidelines, we found some evidence that patients receiving antipsychotics were more likely to be monitored during GP visits compared with a comparison group not receiving antipsychotics. In our adjusted AIPW analyses, all parameters were significantly higher in the antipsychotic sample, although this translated into a small half-measurement occasion difference for at least one measurement on at least one parameter within the study period.
This finding is generally consistent with, although much lower than, that observed in a US study of 12–19 year olds by Ghate et al. (2012), using the GE Centricity primary care database and a control group matched on age and gender. The researchers examined monitoring rates 395 days post-prescription (including baseline) of four metabolic parameters: weight, BP, cholesterol, and glucose levels, from 2004 to 2009 (Ghate et al., 2012). The rates of monitoring were low, with less than 1% of all patients exposed to antipsychotics having all four parameters monitored. Similar to our study, the frequency of monitoring of BMI, BP, cholesterol, and blood glucose was higher for patients receiving antipsychotics, but the frequency increase for laboratory parameters (lipids and glucose) was very low. However, our results from the MedicineInsight data set are difficult to compare to the Ghate study, which included specialist prescribers and was restricted to adolescents.
An earlier US study of Medicaid-insured children 6–17 years also compared patients exposed to antipsychotics to a control group (Morrato et al., 2010). Monitoring rates in this study, using patients on albuterol as a comparison group, were much higher than those recorded by Ghate and colleagues and our study. Morrato and colleagues, using a narrower measurement period of 30 days before and 6 months after an incident prescription, found 31.6% of antipsychotic users monitored for glucose and 13.4% for lipids, compared to 12.6% and 3.1% in the albuterol group. However, the prevalence of antipsychotic prescribing is higher in Medicaid-insured youth compared to privately-insured youth in the United States (Hálfdánarson et al., 2017; Rettew et al., 2015; Schmid et al., 2015) and higher than most other countries (Hálfdánarson et al., 2017). There are also programmes aimed at improving the safe and effective use of antipsychotics in Medicaid-insured youth (Mackie et al., 2021; Schmid et al., 2015). This may account for the higher rates of metabolic monitoring found by Morrato and colleagues when compared to our study and the Ghate study.
Sensitivity analyses
In our sensitivity analysis using weight at baseline as a covariate, there was more monitoring for both antipsychotic and control groups, with a significantly higher level of monitoring in patients prescribed antipsychotics. Patients on antipsychotics recorded an additional 1.5 visits in which some form of cardiometabolic monitoring took place within the 12-month follow-up period. The additional analysis of those recorded as overweight/obese at baseline showed similar results, but high levels of missing data for weight mean the results need to be interpreted with caution. The results from this sensitivity analysis suggest being overweight/obese may trigger monitoring of weight and several other metabolic parameters in all children/adolescents, but that GPs recognise a greater need for monitoring if overweight/obese patients are also prescribed antipsychotics. A similar pattern of metabolic monitoring was found by US researchers using the GE Centricity primary care database; the study found an increase in monitoring of BMI and BP if patients were obese at baseline, but no change in monitoring of total cholesterol and glucose (Ghate et al., 2012).
Future directions
As has occurred in many health care settings worldwide (Antoniou et al., 2023; Cotes et al., 2015; Mead et al., 2021), clinician uptake of cardiometabolic monitoring recommendations for paediatric patients receiving antipsychotics in Australian primary care has been suboptimal. Furthermore, a systematic review of metabolic monitoring in child and adolescent mental health services that included several Australian studies found no evidence of an improvement in guideline adherence from 2008 to 2020 (Mead et al., 2021). The reasons for the continued poor adherence to monitoring guidelines are not well understood.
A number of studies have examined strategies to improve rates of metabolic monitoring, such as the provision of educational resources (Cotes et al., 2017; Dhamane et al., 2013; Grzeskowiak et al., 2008; Mead et al., 2021), hands-on training with regular feedback on performance (Cotes et al., 2017; Dhamane et al., 2013; Grzeskowiak et al., 2008) and electronic reminders (Aouira et al., 2022; Dhamane et al., 2013; Minjon et al., 2018). There is some limited evidence that such strategies can improve adherence to cardiometabolic monitoring in paediatric populations (Mead et al., 2021).
However, even in jurisdictions with such guideline implementation strategies in place, rates of metabolic monitoring tend to be low (Antoniou et al., 2023; Cotes et al., 2017; Ronsley et al., 2012). Reasons for the poor rates include refusal by children or their carers due to children’s fear of needles (Lambert et al., 2018; Minjon et al., 2018; Ronsley et al., 2012) or a sense of futility because the drug is considered essential to managing the child’s condition (Chen et al., 2018; Walter et al., 2008). Consequently, some researchers have suggested a more realistic, targeted approach to monitoring whereby blood testing is undertaken only when there are apparent changes in weight or waist circumference (Chen et al., 2018). However, this targeted, stepwise approach to engaging patients in metabolic monitoring may also have limitations (Remington, 2006; Sjo et al., 2017). For example, in a Danish study, two of 15 children met the criterion for MetS at 12 months post initiation of antipsychotic treatment but had registered a normal BMI at baseline and at 12 months follow-up (Sjo et al., 2017). Therefore, using overweight/obesity as a trigger to engage in routine blood monitoring may not detect important metabolic changes and deny patients timely treatment. Research into reasons for poor monitoring practices and strategies to improve guideline adherence is still limited.
In the Australian setting, it is unclear whether there is a lack of awareness of guidelines recommending cardiometabolic monitoring for children/adolescents prescribed antipsychotics, a lack of resources, or a lack of effective communication between health care providers. In a recent survey of 10 child psychiatrists working in Australian Child and Youth Mental Health Services, 40% cited a lack of good communication with GPs as a barrier to implementing cardiometabolic monitoring (Aouira et al., 2022). Inadequate communication between health care providers has also been recognised as a barrier in international jurisdictions (Edelsohn et al., 2015). Further research is needed to determine why inadequate cardiometabolic monitoring is occurring in general practice when antipsychotics are prescribed and which implementation strategies can improve adherence to guideline recommendations.
Limitations
MedicineInsight is a large database considered representative of patients attending Australian general practice, although some states, regions, and IRSAD quintiles are over-represented compared to census data (Busingye et al., 2019). MedicineInsight provides pathology and physical examination records, which can be linked chronologically with medication prescriptions. Although we were unable to determine if the monitoring that was observed was specifically related to the use of antipsychotics, the inclusion of a variable for the presence of a medical condition and the exclusion of patients with pre-existing records of cardiometabolic conditions increased the likelihood that the antipsychotic triggered monitoring. Medications are often initiated by psychiatrists or paediatricians (Kjosavik et al., 2017), so the date of incident prescribing may be inaccurate in some cases. It is possible that monitoring was carried out by paediatricians or psychiatrists in mental health clinics, hospitals, and specialist clinics without being recorded in MedicineInsight, so rates may not represent a complete record of the monitoring being undertaken by the Australian medical community. GP progress notes may also have included some results from clinical encounters that were not available in MedicineInsight records. It is also possible that some important covariates were not included in AIPW analyses.
Conclusion
Rates of cardiometabolic monitoring in children/adolescents prescribed antipsychotics in Australian general practice are well below guideline recommendations. Increasing awareness of the adverse health impacts of antipsychotics using education and training is required, including improved guidelines that clearly delineate the role of psychiatrists, paediatricians, and GPs in paediatric monitoring.
Supplemental Material
sj-docx-1-anp-10.1177_00048674251361696 – Supplemental material for Cardiometabolic monitoring in children and adolescents prescribed antipsychotics in Australian primary care, 2011 to 2017
Supplemental material, sj-docx-1-anp-10.1177_00048674251361696 for Cardiometabolic monitoring in children and adolescents prescribed antipsychotics in Australian primary care, 2011 to 2017 by Julie Klau, David Gonzalez-Chica, Melissa Raven, Natalie Aboustate and Jon Jureidini in Australian & New Zealand Journal of Psychiatry
Footnotes
Acknowledgements
The authors are grateful to the general practices that participated in MedicineInsight and the patients who allowed the use of de-identified information in health research. We also acknowledge the assistance of University of Adelaide staff at the Discipline of General Practice, Dr. Mumtaz Begum from the Division of Medical Specialties, University of Adelaide, and Aline Kunnel from the Biostatistics Unit of the South Australian Health and Medical Research Institute.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a research grant from the Channel 7 Children’s Research Foundation (Project Reference: 20/13437316).
Data availability
The data that support the findings of this study are not openly available due to reasons of sensitivity. Data are located in controlled access data storage at the Discipline of General Practice, University of Adelaide.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
