Abstract
Objective:
Harmonized tools are essential for reliable data sharing and accurate identification of relevant factors in mental health research. The primary objective of this study was to create a harmonized questionnaire to collect demographic, clinical and behavioral data in diverse clinical trials in adult psychiatry.
Methods:
We conducted a literature review and examined 24 questionnaires used in previously published randomized controlled trials in psychiatry, identifying a total of 27 domains previously explored. Using a Delphi-method process, a task force team comprising experts in psychiatry, epidemiology and statistics selected 15 essential domains for inclusion in the final questionnaire.
Results:
The final selection resulted in a concise set of 22 questions. These questions cover factors such as age, sex, gender, ancestry, education, living arrangement, employment status, home location, relationship status, and history of medical and mental illness. Behavioral factors like physical activity, diet, smoking, alcohol and illicit drug use were also included, along with one question addressing family history of mental illness. Income was excluded due to high confounding and redundancy, while language was included as a measure of migration status.
Conclusion:
The recommendation and adoption of this harmonized tool for the assessment of demographic, clinical and behavioral data in mental health research can enhance data consistency and enable comparability across clinical trials.
Keywords
Introduction
Sociodemographic, clinical and behavioral factors play a crucial role in shaping mental health outcomes and can significantly impact the course, prognosis and response to treatment in mental disorders (Cai et al., 2022). Despite their importance, these factors are often inadequately described and inconsistently assessed across clinical trials. A major issue is the usage of non-standard sociodemographic questionnaires across studies, hindering data comparison and compilation (Jeste and Pender, 2022).
Randomized clinical trials (RCTs) in psychiatry have struggled with replicability, often due to small and heterogeneous samples which limit the statistical power (Dean and Walker, 2023). Although meta-analyses can be used to combine multiple datasets, heterogeneity and inconsistent data collection remain an obstacle (Imrey, 2020). To address this challenge, the Common Measures in Mental Health Science (CMMHS) Initiative was launched by a consortium of mental health research funders and journals, including the Lancet Psychiatry and the Journal of the American Medical Association (JAMA) Psychiatry. In its initial framework, the initiative recommends that funders and journals require, rather than recommend, standardized tools for assessment in funded clinical trials and publications. The goal is to facilitate the harmonization of clinical and phenotypical data collected across contexts (Farber et al., 2023).
The first requirements and data collection instruments endorsed by the CMMHS include specific tools for clinical assessment (Diagnosis and Statistical Manual of Mental Disorders-5 crosscutting assessment [DSM-5 crosscutting]), outcomes for depression (Patient Health Questionnaire-9 [PHQ-9]) and anxiety (General Anxiety Disorder [GAD-7]) in adults and in younger populations (Revised Children’s Anxiety and Depression Scale [RCADS-25]), as well as an endorsed tool for assessment of impairment (WHO Disability Assessment Schedule 2.0 [WHODAS 2.0]). Only two demographic items were included in this initial list, namely age and sex at birth (Farber et al., 2023).
Building on this foundational work, there is a need to extend the scope of standardized measures to encompass a broader range of sociodemographic, clinical and behavioral factors relevant to mental health research. To enable large-scale analyses of how unique sociodemographic and clinical factors may interact with treatment outcomes or mediate therapeutic responses, the field would benefit from a harmonized tool for assessment (Jeste and Pender, 2022). The creation and adoption of such a tool can maximize participant contributions, enhance research quality, reduce bias, and enable effective data compilation and result comparison across studies.
Here, we describe the development of a harmonized sociodemographic, clinical and behavioral questionnaire that can be recommended as a complement to the existing requirements of the CMMHS, filling the data gap by consistently capturing critical information. We present the rationale and methodology behind its creation and provide guidance on its implementation. The primary objective of this international consensus study was to provide an initial framework for the development of a freely available harmonized tool for the collection of sociodemographic, clinical and behavioral data in mental health research.
Methods
The Harmonized Demographics Questionnaire (HDQ) is based on a literature review that aimed to identify the most common domains and items explored in the sociodemographic assessment of well-designed clinical trials in psychiatry.
Search strategy
A literature search was conducted using PubMed to identify RCTs published between January 2015 and March 2020 in the top five journals in psychiatry, based on Scientific Journal Rankings (SJR) scores published by the SCImago Journal Index in 2018. The selected journals were JAMA Psychiatry (SJR = 7.867), World Psychiatry (SJR = 7.559), The Lancet Psychiatry (SJR = 6.145), Molecular Psychiatry (SJR = 5.987) and The American Journal of Psychiatry (SJR = 5.807).
In the first stage, we included studies that used ‘randomized controlled trial’ or ‘randomized clinical trial’ as search terms. Titles and abstracts of the identified papers were reviewed, and RCTs that did not use a sociodemographic questionnaire were excluded. We then sent an invitation via email to the corresponding authors of the remaining publications, requesting their demographic questionnaire. We reviewed the received questionnaires and excluded those that were duplicates or not available in English.
Domain and item selection
The domains of the available questionnaires were extracted by 2 researchers (with the involvement of a third if any disagreements occurred) and presented to a taskforce team of 11 researchers with expertise in psychiatry, psychology, statistics and study design, with extensive experience in the conduct of clinical trials in mental health. Supplementary file 1 lists the taskforce members and their relevant expertise. The selected team has a long history of collaboration and was selected based on this ground. Prof Michael Berk is the leading researcher and was responsible for this selection. The team was invited via email. The panel built on the results of the International Alliance of Mental Health Research Funders (IAMHRF) survey, which included 136 researchers collecting demographic data across the world, to decide on which specific questions and answering formats were preferred by researchers.
We then utilized the Delphi method to gather the panel’s opinions on which social, clinical and behavioral factors to include in the final version of the harmonized questionnaire. The Delphi method is commonly used in mental health research to create and refine research criteria, and to establish guidelines and protocols (Jorm, 2015; Sforzini et al., 2022). The method involves repeatedly surveying a panel of experts, either agreeing or disagreeing in an iterative manner until a consensus is reached. This study follows the recommendations of the ACCORD (Accurate Consensus Reporting Document) for consensus methods in biomedicine (Gattrell et al., 2023).
The panel members were provided with a summary of the items within each domain and how frequently each item appeared in the collected questionnaires (Supplement Table 1). The team thoroughly discussed each item after filling up 2 individual surveys and meeting 11 times to decide on the specific questions and answer options to be included in the final version of the questionnaire. The panel also discussed adding domains that were not present in any of the reviewed questionnaires but were considered valid and meaningful constructs for mental health research across cultural contexts. A preliminary version of the questionnaire was circulated to the HDQ investigator group, which includes the authors of each RCT included in this study and every item was revised according to their input. These are experts in the conduction of clinical trials and were able to provide important insight on specific challenges faced in different cultural backgrounds, including those experienced in low- and mid-income countries. A list of the HDQ investigator group is available in Supplement 2.
Consensus was defined as reaching agreement by all panelist members. The co-authors (i.e. associate panelists) also reviewed the developed questionnaire. The questionnaire was then revised accordingly to reach full agreement between the core panelist members and the associated co-authors feedback. Some items that satisfied the predetermined consensus criteria were subject to additional voting rounds. This was particularly the case for domains deemed sensitive, such as those pertaining to sex or ethnicity. Multiple voting iterations were conducted to ensure comprehensive consideration of these areas. The initiation of a re-voting process occurred when either a co-author or a panel member identified previously neglected facets of a domain under discussion. The steering committee managing consensus also had voting rights.
The study protocol was not registered in any platform and participants did not receive any financial incentives to participate in this study.
Results
A total of 129 randomized clinical trials (RCTs) were identified in the literature review. Of these, 24 authors (18.6%) accepted the invitation to participate and sent their questionnaires for review (Figure 1). Through reviewing the questionnaires, the taskforce identified a total of 27 previously explored domains. Supplement Table 1 summarizes the RCT titles and the identified domains in each questionnaire. All panel members completed the surveys and participated in the majority of meetings. Should a panelist have been unable to attend a meeting, comprehensive minutes and the decisions reached were circulated to the absent member for review and commentary, thereby maintaining consensus among all members on the decisions made. There was no deviation from the initial protocol.
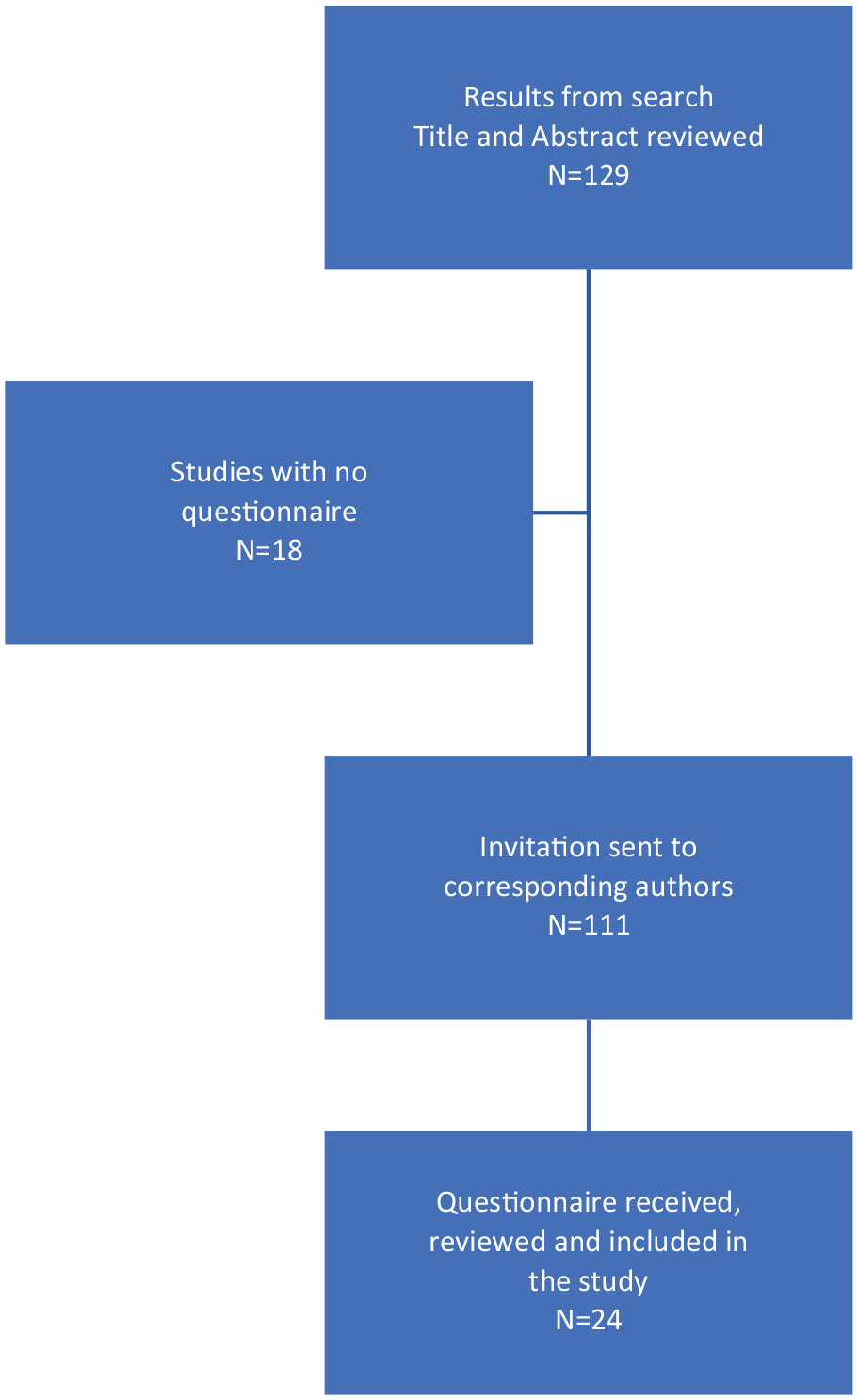
Study flowchart.
The following domains were discussed by the panel: age, date of birth, sex, gender, ethnicity, race, ancestry, education, living, family arrangement, language, occupation, employment, home location, homelessness, marital, relationship status, behavioral factors (diet, physical activity, smoking, alcohol, illicit drug use), history of medical and mental illness, family history of mental illness and reproductive status (World Health Organization, 2014).
The taskforce selected 13 out of the 27 identified domains to be included. Table 1 shows the frequency of each domain identified in the literature review and the taskforce team’s decisions on whether to include or exclude them from the HDQ, along with the justifications.
Identified domains from the 24 questionnaires included and panel consensus.
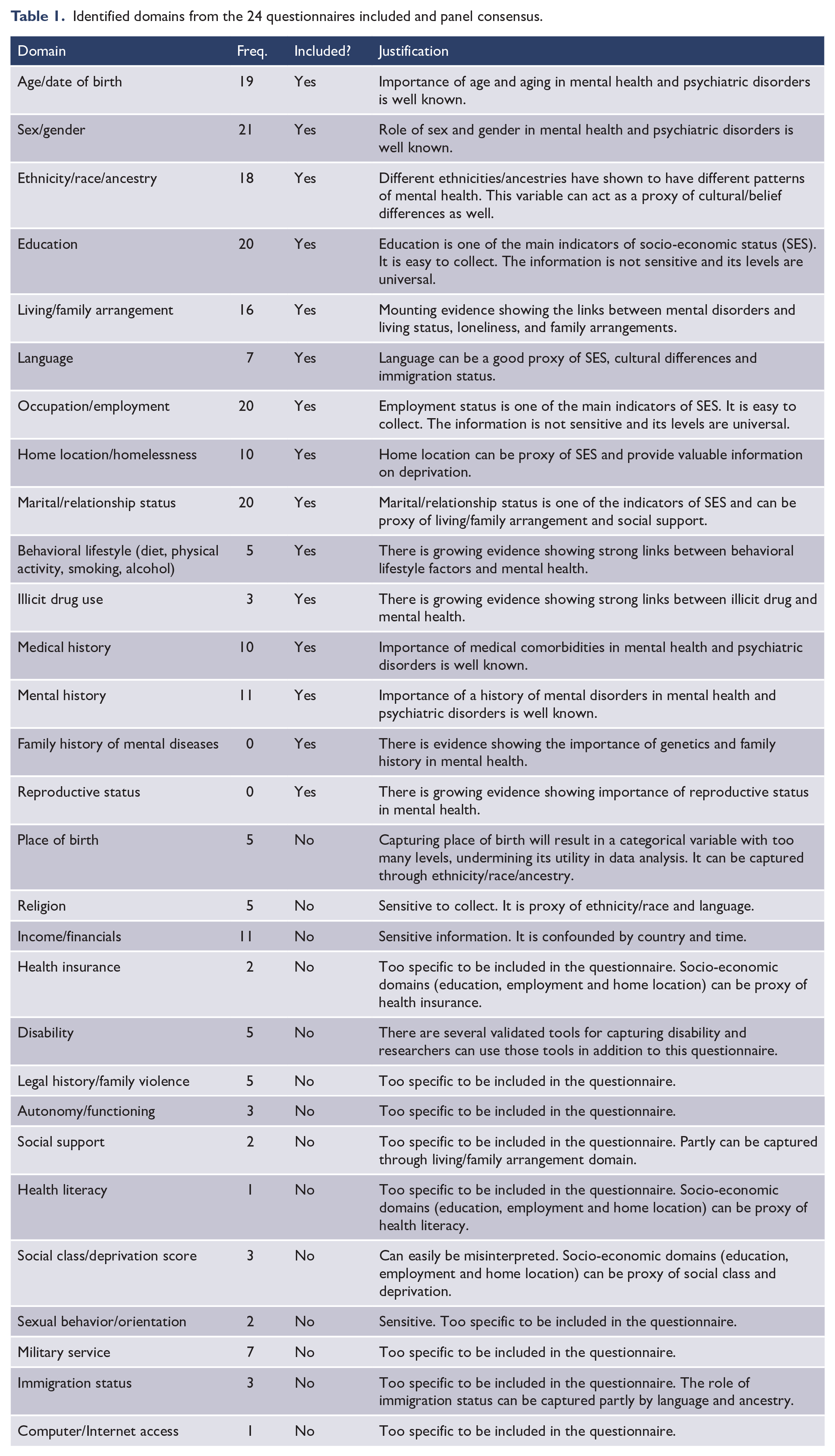
The panel decided to exclude 14 domains previously explored in RCTs; only one of these (income) was found in more than 10 of the previous questionnaires. The panel decided that the assessment of income is highly confounded by setting and country of origin and can be better reflected using other measures, such as education, employment and home location. Place of birth was not included as it was agreed that capturing it would result in a categorical variable with too many levels, undermining its utility in data analysis. Rather, ethnicity/race/ancestry was included as a proxy for place of birth. Sexual behavior/orientation and religion were excluded due to the sensitive nature of these questions. The disability and autonomy/functioning domains were excluded as these are already covered in the CMMHS initial requirement of using the WHO-DAS 2.0 scale. Social class and deprivation were excluded as it was agreed that these could be misinterpreted, and other socio-economic related variables, such as education, employment and home location, could serve as a proxy. Finally, the taskforce found the following domains too narrow to be included in an initial assessment: health insurance, legal history/family violence, social support, health literacy, military service, immigration status and computer/Internet access.
The task force reached a consensus on the items to be included in the final version of the questionnaire. Table 2 shows the selected items, questions and allowable answers for each domain. The panel selected age in years to reduce the risk of re-identification of participants. The team also included two separate questions for the collection of sex and gender. The response options for ethnicity/race and education domains were primarily derived from the World Health Organization (WHO) geographic super-regions and the International Standard Classification of Education (ISCED) (Classification ISCED, 1975), respectively, and subsequently revised by the panel. For living arrangement, home location, employment and marital/relationship status, the taskforce evaluated the items in the collected questionnaires and selected the most appropriate.
The Harmonized Demographic Questionnaire (HDQ).
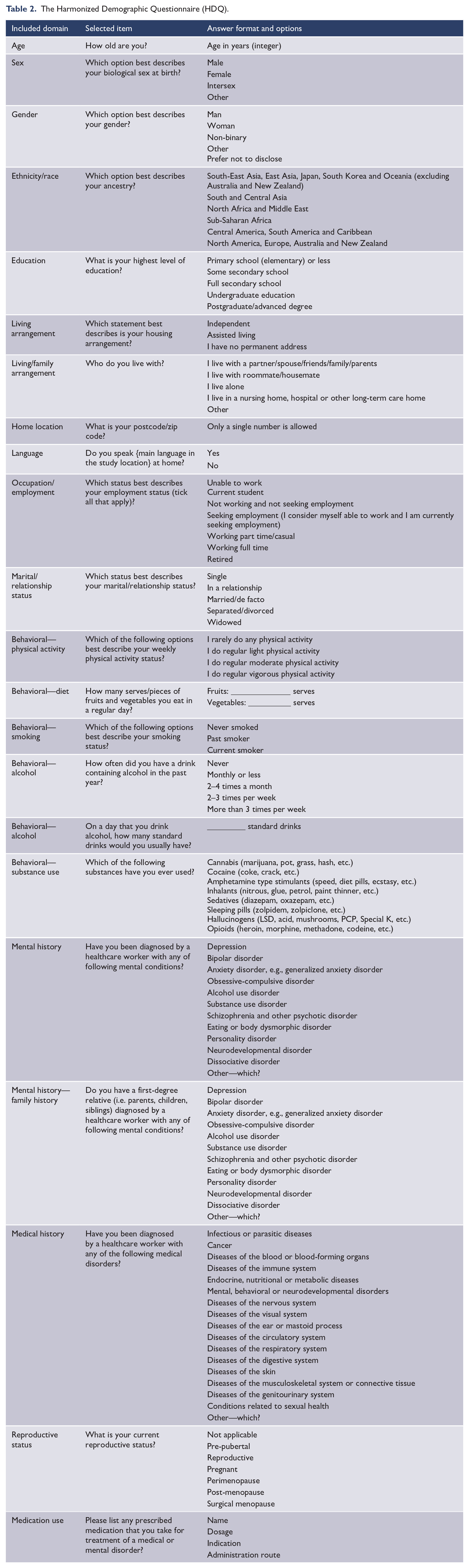
The panel decided to include six questions to assess behavioral and lifestyle factors. These include physical activity, fruit and vegetables intake, smoking status, alcohol consumption and illicit drug use. Clinical factors, such as concurrent medical conditions and a personal and family history of mental disorders, were also included. The list of mental and medical conditions was derived from the International Classification of Diseases, 10th Revision (World Health Organization, 2019). The options for reproductive health status were selected after seeking expert opinions on potential answers. The final 22-question version of the HDQ questionnaire is available in Supplement 3.
Discussion
The primary objective of this study was to create a concise harmonized questionnaire to be used as a standard tool for the collection of demographic, clinical and behavioral data in mental health research. Based on a Delphi-method process, the task force team selected 11 sociodemographic, 6 behavioral and 5 clinical domains to be explored, resulting in a concise set of 22 questions in the final version of the harmonized questionnaire. By incorporating these variables, the questionnaire aims to further facilitate data sharing and enable more effective cross-study comparisons. The recommendation and adoption of this tool as a basic assessment of demographic, clinical and behavioral data can enhance data consistency and adds to the effort of finding common measures in mental health research.
Of the 22 items included in the final version of the HDQ, 10 questions explored factors that were most frequently asked in previous RCTs, including age, sex, gender, ancestry, education, living arrangement, employment status, home location, relationship status, and history of medical and mental illness. Questions enquiring about substance use, nutrition and physical activity levels, language spoken and family history of mental illness were also deemed relevant and included in the final version due to their likely role in the course and outcomes associated with mental illness, and possible roles as mediators of treatment response. For instance, a recent trial of mitochondrial agents for bipolar depression found the intervention to be protective in those who practiced some kind of physical activity, a possible moderator of the effect of treatment (Ashton et al., 2020). Diet quality is also consistently linked to mental illness and deserves further exploration in light of recent research (Marx et al., 2021). Family history of mental illness was included as a possible indicator of genetic vulnerability, and language spoken at home was included as a proxy of immigration status, known to affect risk and prognosis for mental illness (Alegría et al., 2017). The items excluded by the panel were better represented in previous questions and/or were considered too sensitive for participants and/or highly disorder specific. The panel suggests that these domains can be included individually according to a particular RCT needs, design and location.
Although certain variables like ‘religion’, ‘sexual behavior’ and ‘sleep’ were not included in the final version of the questionnaire, it is important to acknowledge their recognized impact in mental health. This omission was a result of careful consideration, considering factors such as time, sensitivity and general relevance. Researchers focused on these variables are advised to include further questions and explore specialized instruments for their studies. Notably, validated tools for assessing sleep quality are available and can be considered for future adaptations of the HDQ to specific research contexts (Fabbri et al., 2021).
Consistent characterization of participants can enable large-scale analysis and individualized approaches. While biological psychiatry has made significant progress in classifying participants based on shared characteristics, such as symptomatic clusters and specific biomarkers, the role of demographic and behavioral aspects is frequently neglected in the construction of clinical phenotypes of course and treatment response (Chekroud et al., 2017; Raison et al., 2013). Psychosocial factors can differentially impact stress-related biological systems and activate specific components of the immune response (Agustini and Berk, 2020; Segerstrom and Miller, 2004). Understanding the social context in relation to biology may help to refine therapeutic targets and support precision medicine (Fernandes et al., 2017).
Harmonized questionnaires promote statistical homogeneity and increase the consistency and accuracy of results, a basic premise of the CMMHS initiative. Here, we provide an initial framework to be recommended as a complementary tool, subject to constant re-evaluation and continuously evolving as data sharing allows us the power to identify the most relevant factors to explore in mental health research, akin to the methods employed by the CMMHS initiative. The HDQ is designed to align with the CMMHS initiative’s existing framework, ensuring seamless integration and adoption. This standardized questionnaire will be employed in all clinical trials involved in The Mental Health Australia General Clinical Trial Network (MAGNET) in Australia, a collaborative effort that aims to standardize the assessment of participants and facilitate multi-site collaborations nationally and globally. By enabling more reliable data sharing and the compilation of multiple data sources, these variables can be easily integrated and explored cross-culturally using machine-learning and sophisticated statistical models that can adequately controlled for a myriad of confounders (Chekroud et al., 2021). With this approach, one can examine the associations of specific social factors with the possible course, prognosis and treatment response of patients exposed to unique environmental conditions (Rutledge et al., 2019; Toyokawa et al., 2012). Furthermore, combining this information with biological measures such as brain imaging and biochemical profiling can provide an opportunity to better explore the underlying biopsychosocial mechanisms involved, a premise of the Research Domain Criteria (RDoC) (Nielsen et al., 2020; Ross et al., 2017; Tai et al., 2019).
This study has several strengths. First, our team conducted a comprehensive literature review to identify the most frequently explored factors in previous RCTs in mental health research. The inclusion of the most prominent journals in the field in the literature review ensured the quality and representativeness of the identified RCTs. Second, we utilized the Delphi method, a widely recognized and rigorous methodology for clinical consensus in research. This allowed us to capture opinions from experts in the field with a long track record in the conduct of clinical trials, resulting in more informed and practical results. Third, our team was diverse and able to discuss individual factors through various paradigms, aiming to find the best balance between thoroughness and feasibility when applying the questionnaire to real-life participants.
Despite these strengths, the questionnaire has some limitations. First, we have made every effort to ensure the relevance and comprehensiveness of the selected items to be included in the final version of the questionnaire, based on our joint research and clinical research experience. However, the HDQ has not yet been road-tested and is recommended as a proof-of-concept approach. The next step is to evaluate its validity and reliability in different cultural contexts. Second, most of our expert panel members were from English-speaking high-income countries, which may limit the generalizability of our findings. The feedback from other members of the HDQ investigator group, especially those working in low- and middle-income countries, was crucial to limit this bias. The social determinants of mental health may differ significantly across countries and cultures, and further research is needed to ensure the relevance of our questionnaire in different settings (Maselko, 2017). In addition, while we included a broad range of sociodemographic, behavioral and clinical factors, there may be other relevant factors that were not included in our questionnaire. The questionnaire was designed mindful that specific study needs may have to be explored using different instruments and additional questions and we aimed to provide a minimum dataset that researchers can build on. Some variables were difficult to operationalize to meet the specific needs of individual populations. Gender, for instance, may require more flexible response choices. While it is important to customize questionnaire items to include diverse identities and experiences, these elements were deemed too specific for inclusion in a basic assessment. RCTs designed for distinct samples, exposed to unique cultural and environmental influences, are encouraged to include additional questions according to their individual aims and needs.
An additional consideration arises from the different requirements used by distinct funding bodies. For example, the National Institutes of Health (NIH) has specific standards for compiling race/ethnicity information (Collins et al., 2021). It is recommended that researchers operating within different funding bodies to evaluate the alignment of the HDQ measure (ancestry) with existing guidelines. Researchers may find it necessary to incorporate a separate question to the HDQ measure to align with country-specific requirements.
In conclusion, this study provides the rationale for implementation of a harmonized questionnaire to collect demographic, behavioral and clinical data in mental health research. The HDQ was built to serve as a complementary standardized tool for assessment in clinical research adding to the efforts for data harmonization in mental health research. The recommendation and adoption of this harmonized tool as a basic assessment can further promote consistency in data collection and facilitate the comparison and synthesis of data across clinical studies in psychiatry.
Supplemental Material
sj-docx-1-anp-10.1177_00048674241253452 – Supplemental material for Development of a harmonized sociodemographic and clinical questionnaire for mental health research: A Delphi-method-based consensus recommendation
Supplemental material, sj-docx-1-anp-10.1177_00048674241253452 for Development of a harmonized sociodemographic and clinical questionnaire for mental health research: A Delphi-method-based consensus recommendation by Mojtaba Lotfaliany, Bruno Agustini, Adam J Walker, Alyna Turner, Anna L Wrobel, Lana J Williams, Olivia M Dean, Stephanie Miles, Susan L Rossell, Michael Berk and Mohammadreza Mohebbi, the HDQ Investigator Group in Australian & New Zealand Journal of Psychiatry
Supplemental Material
sj-docx-2-anp-10.1177_00048674241253452 – Supplemental material for Development of a harmonized sociodemographic and clinical questionnaire for mental health research: A Delphi-method-based consensus recommendation
Supplemental material, sj-docx-2-anp-10.1177_00048674241253452 for Development of a harmonized sociodemographic and clinical questionnaire for mental health research: A Delphi-method-based consensus recommendation by Mojtaba Lotfaliany, Bruno Agustini, Adam J Walker, Alyna Turner, Anna L Wrobel, Lana J Williams, Olivia M Dean, Stephanie Miles, Susan L Rossell, Michael Berk and Mohammadreza Mohebbi, the HDQ Investigator Group in Australian & New Zealand Journal of Psychiatry
Footnotes
Acknowledgements
The authors would like to thank the HDQ investigator group for their valuable input in the final stages of developing this questionnaire.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: M.B. is supported by a National Health and Medical Research Council (NHMRC) Senior Principal Research Fellowship and Leadership 3 Investigator grant (1156072 and 2017131). M.B.: Grant/Research Support: MRFF, NHMRC, Congressionally Directed Medical Research Programs (CDMRP) USA, AEDRTC Australian Eating Disorders Research and Translation Centre, Patient-Centered Outcomes Research Institute (PCORI), Baszucki Brain Research Fund, Danmarks Frie Forskningsfond, Psykiatrisk Center Kobenhavn, Stanley Medical Research Institute, Victorian Government Department of Jobs, Precincts and Regions, Wellcome Trust, Victorian Medical Research Acceleration Fund, Controversias Psiquiatria Barcelona, CRE, Victorian COVID-19 Research Fund; Consultancies: Lundbeck, Sandoz, Servier, Medisquire, HealthEd, ANZJP, EPA, Janssen, Medplan, RANZCP, Abbott India, ASCP, International Society of Bipolar Disorder, Precision Psychiatry, Penn State College of Medicine, Shanghai Mental Health Centre (last 3 years)—all unrelated to this work. O.M.D. is Associate Professor (Research) Deakin University and has received grant support from the Brain and Behaviour Foundation, Simons Autism Foundation, Stanley Medical Research Institute, Deakin University, Lilly, NHMRC and ASBDD/Servier. She has also received in-kind support from BioMedica Nutraceuticals, NutritionCare and Bioceuticals. L.J.W. is supported by a NHMRC Emerging Leader Fellowship (1174060). A.J.W. is supported by a Trisno Family Fellowship, funded in part by an NHMRC CRE (1153607). He has previously received grant/fellowship support from the Trisno Family Gift and Deakin University. M.B., S.L.R., A.T., O.M.D. and A.J.W. are named investigators on the Medical Research Futures Fund 2020 Million Minds Mission—MAGNET: Mental Health Australia General Clinical Trial Network (2006296).
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Contributors
M.L., M.M. and M.B. were involved in the project conception. M.L. performed the data collection and analysis. O.M.D., M.L., M.M. and M.B. carried out questionnaire design. All authors participated in the Delphi discussion. B.A. wrote the first draft of the manuscript, with manuscript preparation handled by B.A., M.L., M.M., O.M.D. and A.J.W. M.B., A.J.W., L.J.W., M.M., S.M. and O.M.D. revised and edited the manuscript. All authors had full access to available data and approved the final version of the manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
