Abstract
Objectives:
The Australian government recently rescheduled psilocybin and 3,4-methylenedioxymethamphetamine for limited clinical uses. This change has raised various regulatory concerns and challenges for the field of psychedelic-assisted therapy. To provide clarity, we aimed to comprehensively catalogue the matters relating to psychedelic-assisted therapy that are or could be regulated.
Methods:
We conducted a desktop review of the literature and current regulatory sources, semi-structured interviews with professionals who had expertise in fields relating to psychedelic-assisted therapy and a framework analysis to generate a taxonomy of relevant regulatory matters. In relation to each matter, we further identified what type of regulation (if any) currently applies to that matter, any uncertainty as to how the matter should be addressed in clinical practice in the context of current regulation and whether there are conflicting views as to how the matter could or should be further regulated.
Results:
The taxonomy is structured into six main regulatory domains, three of which have a substantial proportion of matters with uncertainty or conflicting views: Service Establishment, Practitioner, and Treatment Delivery. Key examples of such matters include the location of services and facilities required, which professionals are eligible to become psychedelic therapists, and with what qualifications and experience. Matters in the remaining three domains, Patient Evaluation, Drug Supply and Service Oversight, appear by comparison relatively settled, with regulation either well-established or thought unnecessary.
Conclusions:
The taxonomy provides a roadmap for health services establishing and implementing a psychedelic-assisted therapy program, or for government and other policymakers when determining areas that may require further regulation.
Keywords
On 1 July 2023, the Therapeutic Goods Administration (TGA) in Australia reclassified psilocybin and 3,4-methylenedioxymethamphetamine (MDMA) as schedule 8 substances, for the sole indications of treatment-resistant depression and post-traumatic stress disorder, respectively (TGA, 2023c). The rescheduling applies exclusively to authorised prescribers and only when delivered as part of an approved treatment protocol (TGA, 2023c). The decision comes on the back of emerging but limited clinical evidence as to the efficacy and safety of these substances (Kisely et al., 2021; Smith et al., 2022; van Amsterdam and van den Brink, 2022). Structured protocols are commonly referred to as psychedelic-assisted therapy (PAT) and generally involve a course of psychotherapeutic supports before, during and after the administration of a ‘classic’ serotonergic psychedelic (such as psilocybin, lysergic acid diethylamide [LSD] or 5-methoxy-N,N-dimethyltryptamine) or the empathogen MDMA. While there are notable differences in the subjective effects of psilocybin and MDMA, and the clinical protocols accompanying them, we believe the regulatory issues are sufficiently comparable to consider them together.
The rescheduling of psilocybin and MDMA makes Australia the first national jurisdiction in the 21st century to formally recognise these substances as medicines. Elsewhere, regulatory frameworks exist that functionally allow for wellness or spiritual psychedelic use, including in the United States, the Netherlands, Peru, Jamaica, Costa Rica and several other localities, as evidenced by the robust private psychedelic retreat industry (Retreat Guru, n.d.). Special access pathways also exist for medical use under narrowly defined circumstances in (at least) Canada, Switzerland and Israel (Mocanu et al., 2022). Psychedelics were also historically legal for use in psychiatric settings in several countries, including LSD in Australia until the mid-1970s under an authorised prescriber scheme (Lomax, 2017).
The 2023 Federal regulatory changes introduced a similar authorised prescriber model, with the additional oversight of a Human Research Ethics Committee (HREC) and, in some jurisdictions, additional state- or territory-based requirements. Individual psychiatrists will need to apply for Authorised Prescriber (AP) status by submitting the proposed treatment protocol to a HREC. If the application receives approval from the HREC, it will then be submitted for final approval by the TGA. The TGA has published a guidance document that provides an overview of what details are expected to be addressed in an AP application, including details of the protocol, information about the specific substance to be administered, and the measures used to address safety, efficacy and outcomes reporting (TGA, 2023a). However, the TGA has not specified any particular standards or protocols for the psychotherapeutic supports, noting this is outside of their regulatory purview (Skerritt and Langham, 2023).
PAT sits within a broad milieu of complex supply, clinical, professional, governance and financial systems that intersect with existing regulation and/or have the potential to be regulated further (Andrews and Wright, 2022). Although Australian psychiatrists appear to be cautiously in support of the introduction of PAT, many are uncertain and somewhat concerned as to how it will operate in practice (Berger and Fitzgerald, 2023; Kunstler et al., 2023). Given the breadth of potentially relevant matters it is not hard to see why.
To better appreciate these regulatory complexities, we aimed to comprehensively catalogue the matters relating to PAT that are or could be regulated. We defined ‘regulated’ broadly as any way in which the establishment and implementation of a PAT program may be standardised or constrained. We assumed regulation to include both external government regulation (i.e. legislation, rules and policy), and instruments of professional self-regulation such as standardisation documents produced by professional bodies (guidelines, codes, standards, recommendations, statements, memorandum, etc.).
Method
Ethics
The study was approved by the University of Sydney’s Human Research and Ethics Committee (Approval No. 2023/236).
Study design
The study comprised two data collection and analysis phases: (1) a desktop review of the literature, to generate a list of potential regulatory matters and arrange these into a draft taxonomy; and (2) semi-structured interviews with purposively selected experts, to obtain feedback and further input into the taxonomy.
Desktop review
A search was conducted in the Ovid MEDLINE database using the following keywords and operators: (psychedelic OR psychedelic-assisted OR hallucinogen) AND (therap* OR psychotherap* OR medic*) AND (legal OR legislat* OR regulat* OR code OR guideline OR polic* OR ethic*). A total of 226 abstracts were reviewed, and papers were selected for further analysis based on an assessment of whether there was likely to be a discussion of matters relevant to PAT regulation, policy, ethics or program design. Information was also sourced directly from relevant government websites including the TGA, the Office of Drug Control (ODC), State health departments, the US Food and Drug Administration (FDA) and from relevant professional association websites including the Royal Australian and New Zealand College of Psychiatrists (RANZCP), the Australian Psychological Society (APS), the newly formed Australian Multidisciplinary Association for Psychedelic Practitioners (AMAPP) and the US-based Multidisciplinary Association for Psychedelic Studies (MAPS). A total of 48 academic papers and 21 organisational documents were eligible.
Data and framework
The theoretical and operational basis of our data collection and analysis was an adapted version of the framework analysis methodology (Goldsmith, 2021). The key elements of framework analysis are: (1) initial familiarisation with the data; (2) generation of a thematic framework; (3) indexing the data points; (4) charting the data according to the framework; and (5) mapping and interpretation. Framework analysis is intended to be flexible and iterative, allowing for refinement of the framework and subsequent mapping of the data according to themes and patterns that emerge, and as such is well-suited to applied policy research (Goldsmith, 2021).
We defined our data points as any matters relating to PAT that are or could be regulated. A brief survey of the literature was conducted to become familiarised with the types of matters and to construct a plausible framework of top-level domains and categories. The literature was then examined in-depth in order to index all potential regulatory matters, which were then charted within the framework. Specific information associated with each matter was also noted, such as current regulation, unresolved issues and suggestions for future regulation.
Matters were excluded if they were not sufficiently relevant or specific to PAT, e.g. regulatory requirements that apply to healthcare generally. Furthermore, we did not index matters relating only to the administration of below- or threshold-perceptual doses (‘microdosing’) or multiple medium doses administered during psychotherapy sessions (‘psycholytic therapy’), given these have not to date generated a sufficient evidence-base and are thus currently unlikely to be approved for clinical practice.
Based on the relative distribution of matters, the categories and domains themselves were refined and rearranged within the framework. The draft taxonomy was then provided to participants for the interview phase.
Participants
To best address our research aims, it was necessary to consult professionals with experience and knowledge relevant to PAT. Given Australia’s limited pool of such professionals, purposive sampling was the most suitable recruitment method. The sample included people known professionally to the research team, authors of relevant academic papers, experts referenced in media reports and people working in PAT research and development. Participation was sought from a range of professional fields to ensure a broad spectrum of perspectives, including psychiatry, psychology, the pharmaceutical industry, research, professional bodies and government departments. A total of 30 potential participants were contacted by email and, where appropriate, a follow-up phone call. Participants self-selected by indicating their willingness to participate and provided written informed consent.
Interviews
Participants were asked for feedback on both the framework structure and the matters themselves, including any not yet identified matters. A semi-structured interview guide was developed consisting of questions on the scope and structure of the taxonomy framework. It was not intended that participants would systematically address all aspects of the taxonomy during the interviews, but rather only areas where they had expertise. Interviews ranging between 31 and 59 minutes were conducted via video conference and audio-recorded. As the interviews were intended to elicit further matters not yet identified in the literature search, participants were encouraged to speak at length about areas of interest. Interview notes were taken both contemporaneously and during review of the recordings.
The same framework analysis methodology was applied to the interview notes. Specifically, additional matters identified were indexed and charted, along with associated information such as unresolved issues and suggestions for future regulation. Participant feedback also led to the removal or merging of some matters that had appeared in the draft taxonomy, largely because these matters were found to be inconsequential and/or not specific to PAT. A total of 102 matters appeared in the final taxonomy. Finally, based on participant feedback, the top-level domains were further amended, and the categories re-sorted, to better reflect the order in which they would likely be considered by a health service implementing PAT.
Analysis
‘Key Notes’ were written for each of the matters, based on a synthesis of the participants’ discussions around that matter together with the associated information derived from the literature. From this process, three factors emerged that we determined could be applied to all matters (i.e. the final mapping stage of framework analysis): (1) whether the matter was subject to current regulation (being Federal/State legislation or policy, or professional guidelines); (2) whether there was uncertainty as to how a health service would address the matter in the context of current regulation; and (3) whether there were conflicting views (between participants and/or in the literature) as to how the matter should or could be further regulated. Accordingly, we mapped the data by coding the matters on these factors.
Results
Eleven experts participated in the interviews and provided feedback on the draft taxonomy. The participant characteristics are presented in Table 1. The final taxonomy is presented in two formats. The Taxonomy Overview (Table 2) presents a one-page summary of all domains, categories and matters. The Detailed Taxonomy (Table 3) also describes the Key Notes and coding for each matter, along with a numerical identifier of which participant(s) discussed that matter.
Participant characteristics.
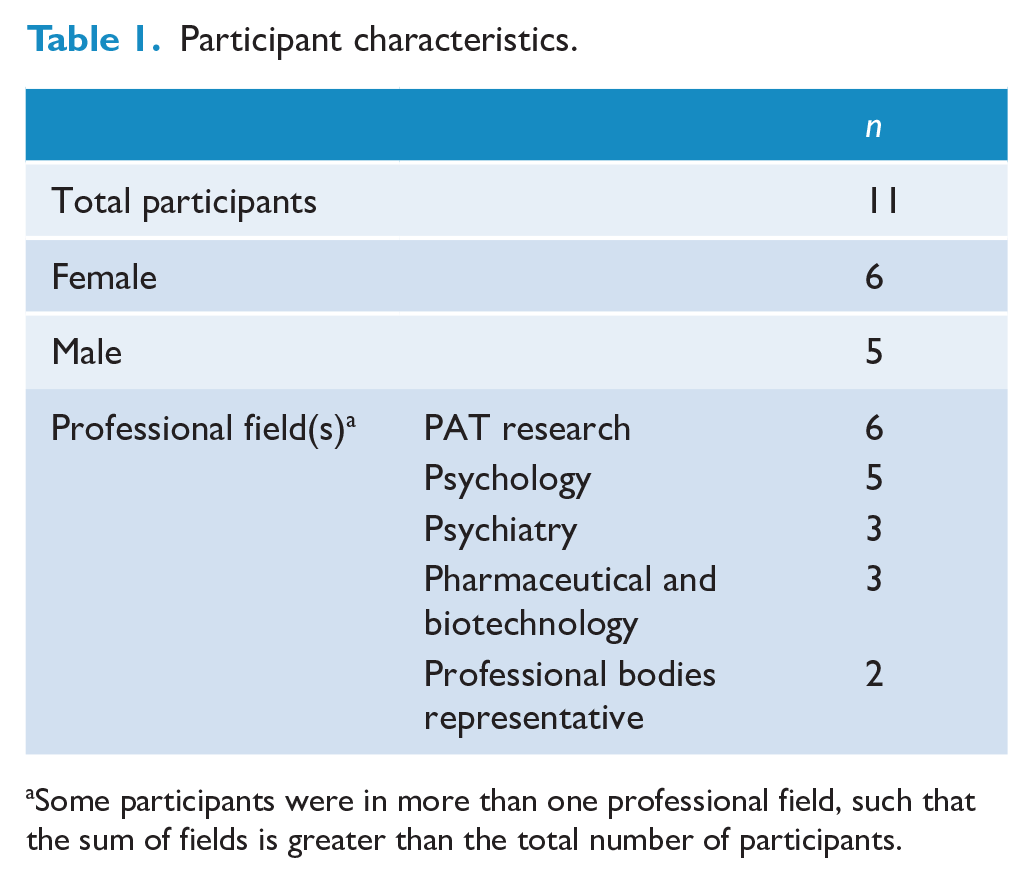
Some participants were in more than one professional field, such that the sum of fields is greater than the total number of participants.
Taxonomy overview.
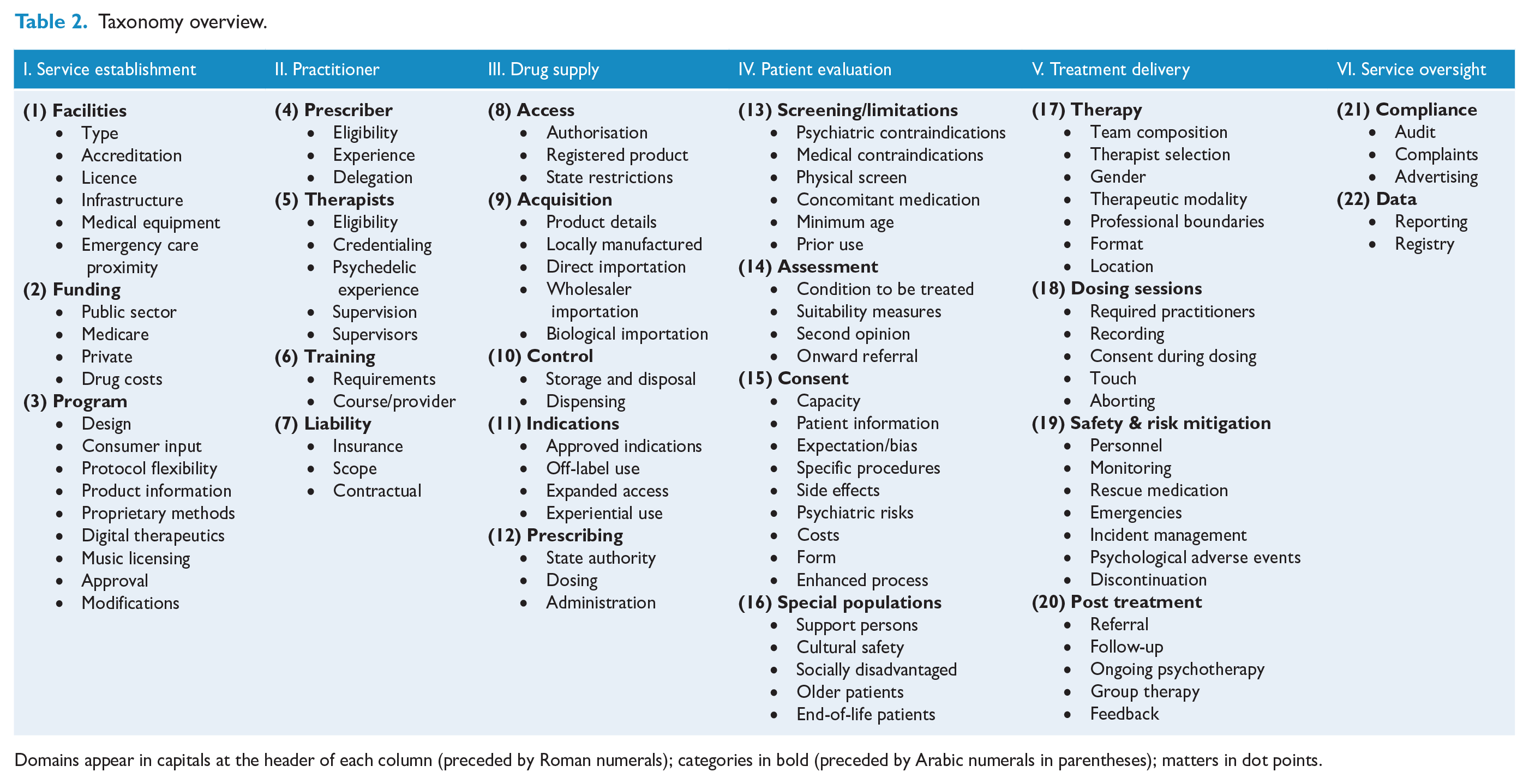
Domains appear in capitals at the header of each column (preceded by Roman numerals); categories in bold (preceded by Arabic numerals in parentheses); matters in dot points.
Detailed taxonomy.
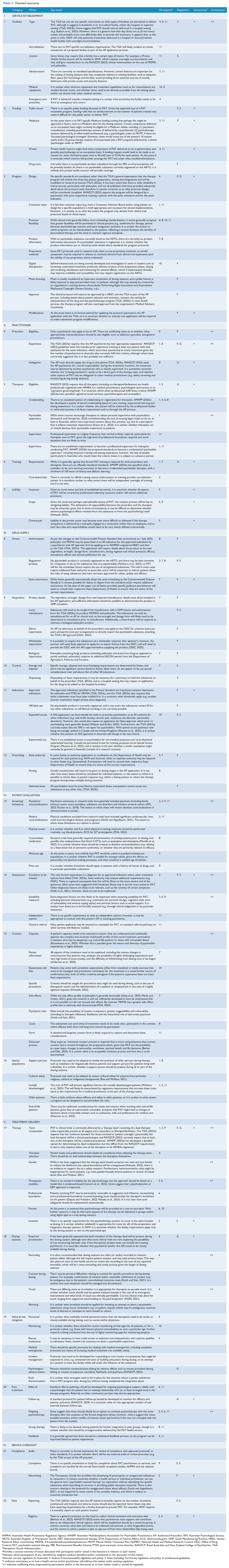
AHPRA: Australian Health Practitioner Regulation Agency; AMAPP: Australian Multidisciplinary Association for Psychedelic Practitioners; AP: Authorised Prescriber; APS: Australian Psychological Society; ARTG: Australian Register of Therapeutic Goods; DBT: dialectical behavioural therapy; DVA: Department of Veteran Affairs; ECG: electrocardiogram; GMP: Good Manufacturing Practice; HREC: Human Research Ethics Committees; MDMA: 3,4-methylenedioxymethamphetamine; MHTP: Mental Health Treatment Plan; NHMRC: National Health and Medical Research Council; ODC: Office of Drug Control; PAT: psychedelic-assisted therapy; PBS: Pharmaceutical Benefits Scheme; PTSD: post-traumatic stress disorder; RANZCP: Royal Australian and New Zealand College of Psychiatrists; TGA: Therapeutic Goods Administration.
Provides a concise summary of issues discussed with participants and/or raised in the literature in relation to each matter.
Indicates which participant/s (if any, identified by assigned number) identified and/or contributed to the discussion of this matter.
Denotes current regulation (in Australia): F: Federal (Commonwealth) legislation and policy; S: State (including Territories) legislation and policy; G: professional guidelines.
+ indicates uncertainty as to how a health service and/or practitioner will address this matter under existing regulation.
++ indicates conflicting views (expressed by participants and/or in the literature) as to how matter could or should be regulated.
International readers should note that Australia has a federal (two-tier) system of government regulation, with Federal regulation applying across the country and State regulation applying only within that jurisdiction. In the coding of the taxonomy, (F) is used to designate that Federal regulation applies to that matter and (S) is used to designate that State regulation applies. These include both formal legislative instruments and published departmental policies and procedures that in practice direct or constrain the operations of a health service and/or practitioner. (G) refers to guidelines and other standardisation documents produced by non-government professional bodies.
The final taxonomy domains were Service Establishment, Practitioner, Drug Supply, Patient Evaluation, Treatment Delivery and Service Oversight. A description of each domain follows, together with a brief outline of what were considered the most pressing or contentious matters in each domain.
Service establishment
This domain contained matters that would likely be prominent during establishment of a new PAT program within a health service. There were matters with relative regulatory uncertainty across almost the entire domain. A key example was the facility: although psychedelics are considered relatively safe (Evans et al., 2023; cf. Schlag et al., 2022), there is uncertainty and a lack of consensus as to the determination and regulation of appropriate facility types and the accessibility of emergency care in clinics operating outside of a hospital environment. The TGA noted that administration and monitoring of treatment should be in an ‘accredited facility, either day hospital or inpatient setting [our emphasis]’ (TGA, 2023b). However, one jurisdiction in Australia (NSW) has issued regulations requiring PAT to be provided in a mental health facility with overnight accommodation, a regulation not required elsewhere. This will have direct implications for cost and accessibility, and there were conflicting views between participants as to whether this requirement was unnecessarily restrictive or necessary from a risk perspective.
Funding presented some uncertainty, with a course of PAT expected to cost between AU$25,000 and AU$35,000 (Chrysanthos, 2023). It seems unlikely that PAT will be government-funded under current reimbursement mechanisms, and review of private health insurance reimbursement, public hospital activity-based funding and Medicare items (e.g. if the treating psychiatrist must be present for the entire treatment duration) will be required if PAT is to be at all accessible.
Practitioner
This domain refers to the regulation of professionals who will administer and/or oversee the treatment, such as qualifications and training. Substantial uncertainties were identified, reflecting the fact that there is no established clinical framework for managing what will likely be a multidisciplinary practice. For example, the TGA guidance is nonspecific as to which professions are eligible to provide therapy. RANZCP guidelines state that therapists must be registered with the Australian Health Practitioner Regulation Agency (AHPRA) or an equivalent governing body, but unhelpfully do not specify what these other ‘governing bodies’ are. Furthermore, there are no consistently accepted standards or mechanisms for assessing what training would qualify a therapist to provide PAT, which may have implications for liability and insurance. Participants held conflicting views as to which professionals should be eligible to provide PAT, with what experience, the amount of training and ongoing professional development required and how accreditation should operate.
Drug supply
This domain refers to mechanisms for obtaining and supplying the actual compounds. There was greater certainty in this domain, as the existing regulatory systems for Schedule 8 substance are relatively defined (albeit with some complexity). There are notable differences between States in terms of additional requirements to prescribe the substances, with some States requiring authorisation (NSW) or notification (Victoria) on a per patient basis, and other States (such as Queensland) not imposing any further requirements. However, there was no real contention among participants about these matters, as most viewed them as fixed and ultimately workable (if at times cumbersome) administrative requirements.
Patient evaluation
This domain refers to those matters that would arise in the process of evaluating and accepting a patient for a course of PAT. This domain is relatively unregulated and, for the most part, participants did not hold strongly conflicting views about the need for further regulation. It would seem participants felt this was an area where existing clinical guidelines and professional judgement was sufficient to enable evaluation of patients for the indicated and approved conditions, albeit there was some practical uncertainty in relation to the screening of patients.
Treatment delivery
This domain contains matters that would arise during the provision of the treatment itself. There was substantial uncertainty along with conflicting participant views in relation to matters in this domain, perhaps unsurprisingly given PAT has to date only been delivered in clinical trial settings. For instance, there is some confusion as to the allowable composition of the treating team, with the TGA (2023a) suggesting a minimum qualification of clinical psychologist for those with ‘patient oversight’, and the RANZCP (2023) guidelines requiring that at least one of the therapists is a medical practitioner. AMAPP’s (2023a) proposed composition requires at least one of the treating therapists to hold AHPRA registration (from any profession) combined with PAT-specific training. There are also several as-yet unregulated matters where conflicting views were expressed as to whether and how they should be regulated, such as whether a therapist should be able to continue providing psychotherapy to a patient following cessation of the formal integration sessions.
Service oversight
This domain includes administrative or compliance matters where regulation imposes ongoing requirements not relating to the actual treatment of a specific patient. Participants identified some uncertainty in this domain, such as whether a health service or practitioner can use general terms like ‘psychedelic therapy’ without directly naming the substances when describing its services or providing public-facing materials. Relevantly, the Therapeutic Goods Act 1989 prohibits direct advertising of prescription-only medicines, and there have been a number of recent proceedings and convictions of medicinal cannabis providers for breach of these regulation (TGA, n.d.).
Discussion
We developed a taxonomy of 102 matters relevant to PAT that are or could be regulated, structured into six broad domains and a number of categories. Our analysis revealed uncertainty and contention to varying degrees across the domains. We emphasise that this was not a quantitative process, so we have not made any quantitative comparisons between the domains. We also emphasise that, while the domains were intended to structure the matters in a sequence that a health service might logically follow, in practice the domains are not in fact independent, and any number of the uncertainties identified might be concurrently operating. Readers are encouraged to consult the full taxonomy for an appreciation of all matters that may be relevant to them. Nonetheless, we felt there were some particularly important or pressing matters that warrant further discussion.
Current regulation stipulates that only psychiatrists may apply for AP status, with an expectation that the AP will ‘provide ongoing psychotherapeutic management’ (TGA, 2023a) rather than simply prescribing and then delegating the psychotherapy aspects of the treatment entirely to others. The wisdom of this requirement was questioned by some participants, who pointed out that psychiatry is already undersubscribed and overburdened. They suggested that other professionals may be equally placed to perform these responsibilities, such as psychologists, or other medical practitioners like addiction medicine specialists or general practitioners with mental health credentials. Relatedly, the requirement that at least one of the therapists is a medical practitioner (RANZCP, 2023) may add substantial cost to a treatment course and so potentially contribute to further inequities in treatment access. We are not aware of any research suggesting that patients of medical practitioners have more positive outcomes relative to other mental health professionals in the psychotherapeutic management of PAT.
It was observed by some participants that, while it may seem self-evident that regulation is required to appropriately manage the risks of PAT – and to protect the public from unscrupulous operators – it is also arguable that some matters may be best left unregulated, particularly while the treatment is still largely experimental and while many questions remain to be addressed through further research. For example, while it might seem desirable to regulate some kind of minimum standards in terms of psychotherapeutic support, overregulation at this point could have the effect of stifling further exploration of therapeutic models, with the potential for undesirable consequences in terms of reduced treatment efficacy and increased cost. Some participants (who were health professionals themselves) also raised the danger of ‘overmedicalisation’ of PAT whereby, due to inflexible regulatory standardisation, the therapy becomes only available in ‘one-size-fits-all’ protocols administered by doctors ill-suited to deal with the wide range of experiences these substances elicit. Given the profound, often spiritual, frequently challenging and highly personal nature of these experiences (Kelly et al., 2021) together with their tendency to increase suggestibility and vulnerability to influence including from those who administer the therapy (Cavarra et al., 2022; Palitsky et al., 2023), the medical model itself may carry a risk of harm if these issues are not appropriately considered.
On the other hand, and perhaps unsurprisingly given the novelty of the treatment and the speed at which the scheduling changes were implemented by the TGA (Kisely, 2023), participants also identified the risks of under-regulation of certain matters. A key example is the matter of training and credentialing: while both RANZCP (2023) and APS (2023) concur that appropriate training is required for all therapists, at the time of publication there is no clear direction as to what constitutes appropriate training, nor are there regulatory systems for evaluating the growing number of PAT training programs available in Australia and online. Furthermore, neither RANZCP nor APS (nor any other preexisting body) may be well-positioned for this task, given it would need to cover practitioners from a range of professions outside their formal remit. AMAPP (2023a) has proposed that it acts as an independent cross-disciplinary credentialing body; notably, an analogous system of trans-professional credentialing is being established for practitioners who have training and experience in treating eating disorders (Hurst et al., 2022).
Several limitations to this study warrant consideration. First, it is possible that the sampling pool was overly represented by those in clinical research and drug development, which may have led to a bias that precluded the identification of other relevant matters. However, given the unexpectedly speedy down-scheduling by the TGA together with the peculiarities of implementing PAT into clinical practice, people closely involved in the current clinical research space were arguably those who could provide the most relevant data. Relatedly, a more in-depth thematic analysis might have uncovered matters not obviously apparent in the data we reviewed. However, most matters were relatively straightforward, at least in terms of identifying and defining them, and appeared across multiple sources. This suggests other approaches would have been unlikely to reveal substantial additional matters. Accordingly, we are confident the approach taken was sufficiently robust, particularly in the context of the need for a timely review in a rapidly evolving policy environment.
In addition, substantial components of the psychotherapeutic components of modern PAT clinical trials derive from so-called ‘underground’ uses in the era of prohibition, in conjunction with methods developed in the 1950s–1970s when LSD was widely researched in psychiatric use. There are likely experiences and expertise from underground and historic use that are relevant to developing contemporary systems of regulation that we did not obtain through our sampling. Notably, in Australia abuses involving LSD at the Newhaven Hospital in the late 1960s and early 1970s occurred under an authorised prescriber scheme that had the semblance of stringent oversight but lacked substantive authority (Lomax, 2017). Examining gaps in regulation and implementation in historic cases may be vital to building a safer system for patients in the contemporary era.
Future directions might include a study to systematically acquire feedback on the desirability of further regulation for those matters where there is currently uncertainty and/or conflicting views, e.g. via written surveys using numerical scales that are distributed to a wider range and number of health professionals. In addition, the taxonomy itself will likely become quickly out-of-date, given the speed at which this field is developing. Accordingly, it may be useful to periodically review and update the taxonomy.
To our knowledge, our study is the first to systematically identify and consolidate the regulatory matters relevant to PAT. The specificity of our taxonomy to the Australian context means it may be useful for Australian health services to scope and cost the establishment and ongoing delivery of a regulatory-compliant PAT program. It may be a convenient roadmap for a psychiatrist preparing an AP application and/or during the implementation phase of a program. Regulatory bodies may find use in the taxonomy as a starting point for determining whether there are any regulatory or policy gaps, and private insurers may find it useful to orient themselves to the regulatory requirements when determining the scope of coverage they may provide. Finally, professional bodies may make use of the taxonomy to review and update their guidelines.
In conclusion, we were able to identify a comprehensive set of regulatory matters relating to PAT, and structure these in a format that enables both a quick survey of the field and more detailed analyses. Crucially, the project demonstrated that there are many uncertainties, and in some cases conflicting views, as to how certain matters could or should be regulated. The taxonomy thus remains a work in progress, mirroring the field itself. Regulation may be somewhat indiscriminate, but it is one of the few tools available to ensure patient safety and, hopefully, equity in the medical use of psychedelics. The taxonomy in this paper is intended to bring the regulatory considerations and possibilities into greater focus, in the hope that this will ultimately improve outcomes for patients receiving psychedelic treatments.
Footnotes
Acknowledgements
The authors thank the expert contributors who participated in the study by providing feedback and input into the taxonomy.
Author’s Note
Kayla Greenstien is also affiliated to Students for Sensible Drug Policy, Sydney, NSW, Australia.
Author contributions
S.P.H. designed the project, conducted the desktop review, interviews, analysis and interpretation, and drafted the paper. N.L.R.T. provided input into the conduct of the project, analysis and the manuscript. K.G. provided input into the literature review, analysis and the manuscript. N.G. co-designed and supervised the conduct of the project, and provided input into the analysis and the manuscript.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: N.L.R.T. has provided consulting services and served on an expert advisory panel for Janssen. In the past 36 months, N.G. has received speaker’s bureau honoraria from Servier Laboratories, Janssen and Lundbeck and served as an Advisor to Esia, Seqirus, Atai and Lundbeck. N.G. also works in clinical services providing ketamine treatment and has received NHMRC and MRFF funding in this area. N.G.’s employer is funded to undertake a clinical trial of psychedelics by Beckley. He is the chair of a Data Safety Monitoring Board for a psychedelic trial.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This research was supported partially by the Australian Government through the Australian Research Council’s Centre of Excellence for Children and Families over the Life Course (Project ID CE200100025).
