Abstract
Background:
Predicting the course and complications of perinatal depression through the identification of clinical subtypes has been previously undertaken using the Edinburgh Postnatal Depression Scale and has the potential to improve the precision of care and improve outcomes for women and their children.
Methods:
Edinburgh Postnatal Depression Scale scores were collected twice in pregnancy and twice in the postpartum in a sample of 360 women who met diagnostic criteria for perinatal depression using the Structured Clinical Interview for DSM disorder. These data were used to compare with previous, though conflicting, evidence from cross-sectional studies and extend this by undertaking longitudinal measurement invariance modelling to test the structural validity across the perinatal period. Latent profile and transition modelling was used to identify distinct subtypes of women and assess the utility of these subtypes and transition profiles to predict clinically meaningful outcomes.
Results:
Although our data supported one of the previously reported three-factor Edinburgh Postnatal Depression Scale structures used to compute subfactor totals for depressed mood, anxiety and anhedonia at both early pregnancy and 6 months postpartum, there was little value in using these Edinburgh Postnatal Depression Scale subfactor scores to identify subtypes predictive of clinically meaningful postpartum symptom subtypes, or of general health, pregnancy and neonatal outcomes.
Conclusion:
Our study does not support the use of the Edinburgh Postnatal Depression Scale to distinguish perinatal depressive subtypes for the purposes of predicting course and complications associated with perinatal depression. However, the results give guidance on alternative ways to study the value of personalised management in improved outcomes for women living with or at risk for perinatal depression.
Background
Depression is both highly prevalent and a significant burden of disease for women in their childbearing years and commonly occurs, or recurs, during pregnancy and the postpartum, commonly referred to as the perinatal period (Herrman et al., 2022). There remains a lack of clarity in current research findings as to whether perinatal depression is a distinct depressive disorder and, more recently, whether there are meaningful subtypes and clinical phenotypes (Putnam et al., 2017). Most guidelines define perinatal depression as any depression occurring during pregnancy and up to 12 months postpartum (Howard et al., 2014). However, the current Diagnostic and Statistical Manual of Mental Disorders (5th ed.; DSM-5) provides a specifier of a depressive episode with peripartum onset when occurring during pregnancy or up to 4 weeks postpartum (American Psychiatric Association, 2013). Beyond this specifier, there is no further classification of potential subtypes for perinatal depression in this or other classification systems.
Defining perinatal depressive subtypes could have great value in predicting variations in risk for associated maternal and infant outcomes in pregnancy and the postpartum as well as predicting the course and chronicity of the illness over the perinatal period and beyond. As with studies in adult depression more generally, the definition of subtypes has the potential to substantially progress the endeavour towards personalised management (Maj et al., 2020). Identifying clinically meaningful subtypes of perinatal depression has added value in the context of inconsistent evidence of associations found to date between maternal depression and a range of deleterious pregnancy, infant and longer-term outcomes: this may assist in identifying who is, and is not, at risk.
Studies have used the Edinburgh Postnatal Depression Scale (EPDS) for exploring depression subtypes in perinatal depression. The advantage of using the EPDS is the familiarity, cost-effectiveness and availability of this measure in the perinatal context (Heslin et al., 2022). It requires no training to administer, in contrast to lengthier and more restricted screening measures. Hence, it has been widely used, even though its disadvantages for this purpose are outlined by its creator (Cox, 2019) and its use in cross-cultural contexts subject to critique (Ganjekar et al., 2020).
Two recent studies have explored postpartum depression subtypes using the EPDS. The first study examined 663 women pooled from seven different studies (Putnam et al., 2017). The studies varied considerably in methodology factors including recruitment, selection criteria and measurement variables, and consequently, there was systematic missing data. All used the EPDS, even though at differing time points. A diagnostic measure, to verify depression, was used in only 382 of the 663 women. The analysis reported used one EPDS score collected at a median of 125 days postpartum (IQR: 51–215 days); that is, a score taken anytime between 2 and 7 months postpartum. This study identified a three-factor EPDS structure, and five subtypes derived from clustering methods, interpreted as different subtypes of postpartum depression predicting severity and timing of depression. The five subtypes of perinatal depression that were identified were severe anxious depression, moderate anxious depression, anxious anhedonia, pure anhedonia and resolved depression. The authors described the potential associations between these subtypes, sample demographics and pregnancy complications and outcomes, although further analysis was not undertaken. The second study of 487 postpartum women used a cross-sectional design and aimed to identify depression subtypes using the EPDS (Branquinho et al., 2022) administered at an average timing of 5.32 months postpartum. The study did not include a diagnostic measure; however, only women with an EPDS ⩾ 10 were included. The authors describe an underlying three-factor structure for the EPDS that differs from the factor structure reported in the first study, and three subtypes derived from latent variable modelling interpreted as postpartum depression subtypes. The three subtypes identified were mild anxious-depressive symptom, severe symptom, and moderate anxious-depressive symptoms and were based on EPDS alone as no diagnostic measure was undertaken.
Between these two studies, there is not yet consistency in the reported findings on postpartum depression subtypes using the EPDS with respect to factor structure and depression subtypes. Furthermore, both studies examine factor structure and potential subtypes using postpartum EPDS. Given the increasing emphasis on perinatal depression, including the use of EPDS to screen antenatally and postnatally, it is important to examine whether there is consistency during pregnancy in addition to the postpartum.
In this study, we aim to replicate findings from Putnam et al. (2017) and Branquinho et al. (2022) by examining depressive symptoms reported using the EPDS; however, rather than restricting this to early postpartum, we examine this both in early pregnancy and the postpartum and, in contrast with the previous studies, we use a diagnostic clinical interview administered at a consistent timepoint both in pregnancy and the postpartum to identify women with a depressive disorder, that is at less than 20 weeks gestation and at 6 months postpartum. First, we will confirm a three-factor (depressed mood, anxiety and anhedonia) depressive symptom factor structure in this sample, comparing three-factor structures reported in early postpartum in Putnam et al. and Branquinho et al. in our sample in both pregnancy and the postpartum. Second, we will identify distinct subtypes of women in this sample using the three subfactors of depressive symptoms in pregnancy and assess patterns of transition into the postpartum. Finally, we will analyse the resulting subtypes for consistency with clinical subtypes characteristics reported in Putnam et al. and Branquinho et al.
Methods
Sample
The sample analysed in this paper is 360 perinatal women who met criteria for a depressive disorder drawn from a prospective pregnancy cohort, the Mercy Pregnancy and Emotional Well-being Study (MPEWS). Women were recruited in early pregnancy (<20 weeks gestation) between September 2012 and February 2020 via antenatal clinics at Mercy Hospital for Women in Heidelberg, Victoria, and at several hospitals in Perth and regional WA. Women considered suitable for recruitment were identified by clinic managers and approached by the project’s research co-ordinators. Further details of the study are described in the published study protocol (Galbally et al., 2017). Mercy Health Human Research Ethics Committee and the WA South Metropolitan Health Service Human Research Ethics Committee both approved this study and all participants provided informed, written consent.
Measures
Depressive disorder and mental health outcomes
The Structured Clinical Interview for DSM-IV (SCID-IV) or the SCID DSM-5 (SCID-5) Mood disorders schedule (First et al., 1997) was administered by at recruitment and again at 6 months postpartum; administration of the SCID-5 commenced in June 2017 when recruitment of the WA cohort commenced. Depressive symptoms were measured using the EPDS (Cox et al., 1987) at recruitment, third trimester, and 6 and 12 months postpartum. State anxiety symptoms were measured using the state subscale of the State-Trait Anxiety Inventory (STAI) (Spielberger et al., 1983), and stressful life events were assessed using an adapted Stressful Life Events Scale, which includes 24 common stressful life events (Brown et al., 2011).
Maternal general health and well-being outcomes
Sleep quality was measured using the Pittsburgh Sleep Quality Index at early pregnancy, third trimester, and 6 and 12 months postpartum (Buysse et al., 1989). Social support from a partner was measured using the Social Support Effectiveness scale (Rini et al., 2006) and childhood trauma using the Childhood Trauma Questionnaire (CTQ) (Bernstein et al., 2003).
Sociodemographic and pregnancy and neonatal outcomes
Maternal age, ethnicity, parity, relationship status, education, body mass index, and smoking and alcohol consumption were collected at recruitment in early pregnancy. Antidepressant use was self-reported by women and obtained from hospital records at delivery. Data were collected from hospital delivery records for pregnancy complications and birth outcomes. Gestational age at delivery and birthweight were used to determine small-for-gestational age (SGA) based on Australian birthweight percentiles (Dobbins et al., 2012) (SGA, < 10th centile). Continuous Apgar scores at both 1 and 5 minutes were dichotomised into < 7 and ⩾ 7, respectively.
Statistical analyses
Confirmatory factor analyses (CFAs) of the EPDS factor structures reported in Putnam et al. and Branquinho et al., and longitudinal measurement invariance testing of the best fitting of the EPDS subfactor models across the perinatal period was conducted using methods for invariance testing of ordinal items outlined by Millsap and Yun-Tien (2004). See Supplementary Material for a detailed description of the CFA modelling and longitudinal measurement invariance testing.
Following measurement invariance testing and using the best-fitting confirmed factor structure, EPDS items were summed to create subfactor totals, depressed mood (Items 6, 7, 8, 9 and 10), anxiety (Items 3, 4 and 5) and anhedonia (Items 1 and 2) for both early pregnancy and 6 months postpartum. These EPDS subfactor totals were then used for finite mixture modelling, where a series of candidate latent transition analysis (LTA) models were fit using a robust maximum likelihood estimator (MLR) to identify distinct underlying subtypes of women in early pregnancy and at 6 months postpartum and quantify their transition profiles between the two time points. As per Wang and Wang (2012), latent profile analysis (LPA) models should be conducted on each time point separately first as part of the LTA model building process (see Supplementary Material for a detailed description of LPA methods). Following LPA modelling, candidate LTA models were fit and compared with groups sizes at each time point ranging from two- through five-subtype solutions. Model selection between the different LTA subtype sizes was guided using model fit indices (i.e. lower AIC, BIC and aBIC). Furthermore, within LTA models with differing subtype sizes, measurement invariance was tested using a Δχ2 test based on MLR-estimated loglikelihood values and scaling correction factors (TRd), comparing a non-invariant LTA model (i.e. EPDS subfactor means to estimate freely between time points for each latent subtype) to an invariant LTA model (i.e. EPDS subfactor means constrained to equality between time points for each latent subtype). A significant TRd suggests the invariant model fits significantly worse the non-invariant models; that is, an LTA model that allows subtypes (i.e. EPDS subfactor means) between time points to vary fits the data better than an LTA model that assumes equality in latent subtypes between time points. All factor and finite mixture models were conducted using Mplus 8 (Muthén and Muthén, 1998–2017).
Following selection of the best-fitting candidate LTA model, early pregnancy and 6-month postpartum latent subtypes and transition memberships and probabilities between subtypes (i.e. transition profiles) were imported into SPSS 24 (IBM Corp, 2016) for association analyses with subtypes and transition profiles. Chi-square tests of association and F tests were used to compare the early pregnancy latent subtypes across maternal categorical and continuous variables, respectively. Where omnibus tests are significant, post hoc tests were conducted with Bonferroni p-value correction to control for multiplicity. The model-estimated probabilities of the latent transitions were then presented to describe patterns of EPDS subfactor symptom transitions between early pregnancy subtypes and 6 months postpartum subtypes. Finally, to identify factors that are associated with either maintenance or transition profiles between early pregnancy and 6 months postpartum subtypes, we conducted exploratory analyses using non-parametric correlations (Spearman’s rho) to measure the association between model-estimated transition profile probabilities with maternal variables from both early pregnancy and 6 months postpartum.
Results
Cohort sociodemographic characteristics
Table 1 displays the sociodemographic characteristics of the sample at recruitment during early pregnancy. In total, 317 women met diagnostic criteria for a depressive episode (past or current) during early pregnancy using either the SCID-IV or the SCID-5. The remaining 43 women developed a depressive episode between early pregnancy and 6 months postpartum, assessed at 6 months postpartum.
Sample sociodemographic characteristics and other key variables at recruitment (N = 360).
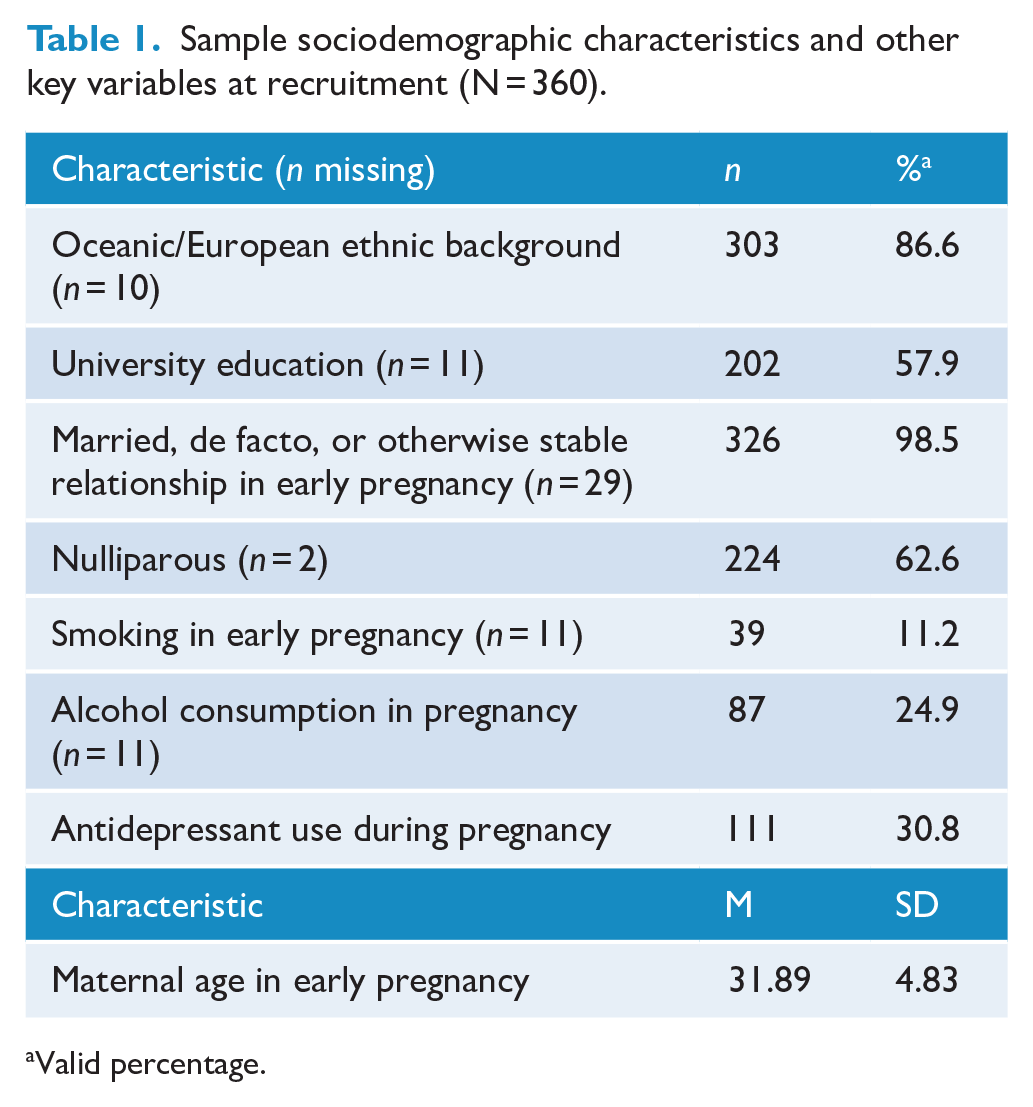
Valid percentage.
Latent transition subtypes underlying EPDS subfactors
A full description and presentation of the individual confirmatory factor models for the EPDS items and invariance testing are presented in the Supplementary Material. Briefly, results from the CFA modelling and longitudinal invariance testing support the three-factor EPDS structure reported in Branquinho et al. Hence, we have used this factor solution to compute EPDS subfactor totals for depressed mood, anxiety and anhedonia at both early pregnancy and 6 months postpartum for inclusion in the LTA models. Prior to the LTA modelling, we first conducted LPA modelling for early pregnancy and separately for 6 months postpartum. At both time points, a three-subtype solution appeared to both fit the data best and provide both significantly different and clinically relevant EPDS subtypes. See the Supplementary Material for a complete description and presentation of the LPA modelling.
Table 2 presents the results of the candidate LTA models with two-, three- and four-subtype solutions and chi-square difference tests using model loglikelihood values testing for measurement invariance within subtype size solutions. The fit of both the two-subtype (TRd = 35.83, d.f. = 6, p < 0.001) and three-subtype (TRd = 46.41, d.f. = 9, p < 0.001) LTA non-invariant models significantly worsened when subfactor means were constrained to equality (i.e. invariant models) on latent variables between early pregnancy and 6 months postpartum, suggesting that the EPDS subfactor subtypes were significantly different at each timepoint. Although the four-profile LTA fits the data best and the invariant model was not significantly worse compared to the non-invariant model (TRd = 19.88, d.f. = 12, p = .069), examination of EPDS subfactor scores from the four-subtype invariant LTA model suggested similar issues discovered from the individual timepoint four-subtype LPA models: EPDS subfactor means did not differ significantly between all subtype groups, suggesting no qualitative differences in subfactor scores between some of the groups. As such, we selected the three-subtype non-invariant LTA model to further examine and analyse the latent transition subtypes given lower AIC, BIC and aBIC values relative to the two-subtype LTA model.
Comparison of model fit and subtype invariance of candidate latent transition models between early pregnancy and 6 months postpartum (N = 360).
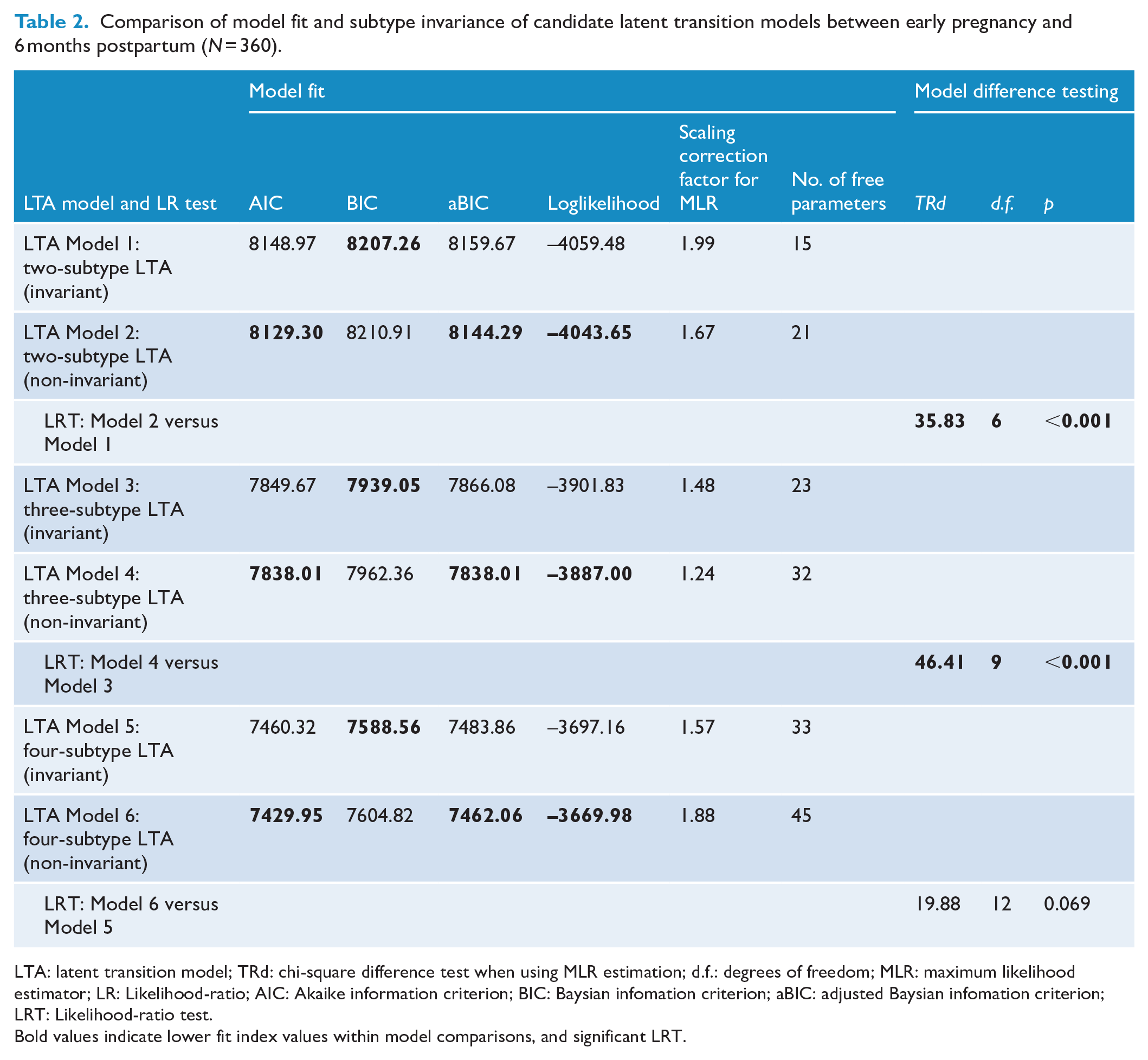
LTA: latent transition model; TRd: chi-square difference test when using MLR estimation; d.f.: degrees of freedom; MLR: maximum likelihood estimator; LR: Likelihood-ratio; AIC: Akaike information criterion; BIC: Baysian infomation criterion; aBIC: adjusted Baysian infomation criterion; LRT: Likelihood-ratio test.
Bold values indicate lower fit index values within model comparisons, and significant LRT.
Latent subtype descriptions and labelling
Figure 1A displays the EPDS subfactor subtypes at early pregnancy derived from the three-subtype non-invariant LTA model. Given the variability in the number of items for each subfactor total score, the ratio of subfactor total score divided by subfactor maximum score is presented. The first early pregnancy subtype, which comprises the largest proportion of women in the sample (n = 273, 75.8%), is characterised by relatively low scores across the anhedonia and depressed mood subfactors despite relatively mild anxiety scores; we have labelled this subtype Mild Anxious Symptoms. The second early pregnancy subtype comprised the second largest proportion of women in the sample (n = 76, 21.1%) and is characterised by moderate scores across EPDS subfactors; we labelled this subtype Moderate Symptoms. The third subtype comprised the fewest women in the sample (n = 11, 3.1%) and is characterised by the highest scores across EPDS subfactors, with relatively elevated anhedonia scores; we labelled this subtype Severe Symptoms with Anhedonia.
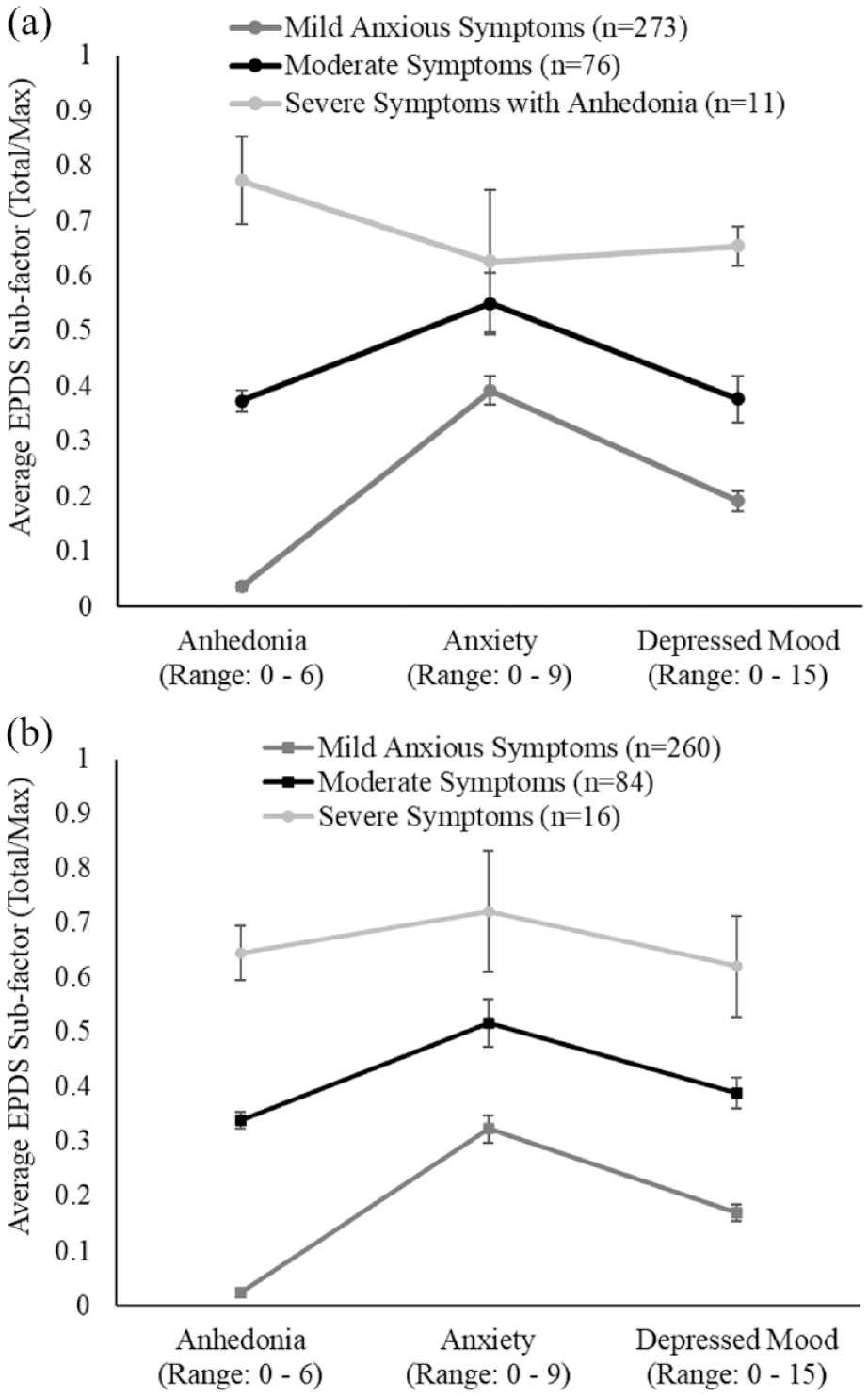
EPDS subfactor plot for latent subtypes derived from the three-subtype non-invariant LTA at (A) early pregnancy and (B) 6 months postpartum. Scores represent the class average EPDS subfactor score divided the maximum subfactor score (minimum and maximum range displayed for each EPDS subfactor on x-axis).
Figure 1B displays the EPDS subfactor subtypes at 6 months postpartum derived from the three-subtype non-invariant LTA model. At 6 months postpartum, Mild Anxious Symptoms and Moderate Symptoms are characterised by relatively similar patterns of EPDS subfactor scores when compared to their respective early pregnancy subtype counterparts. At 6 months postpartum, the third subtype was again the least prevalent, characterised by the highest scores across all three EPDS subfactors. However, the anhedonia score is a less defining characteristic for this subtype compared to the Severe Symptoms with Anhedonia subtype during early pregnancy; as such, we labelled this subtype Severe Symptoms. For EPDS subfactor total scores across subtypes, see Table 3.
Comparative analysis of EPDS subfactor latent subtypes in early pregnancy (N = 360).
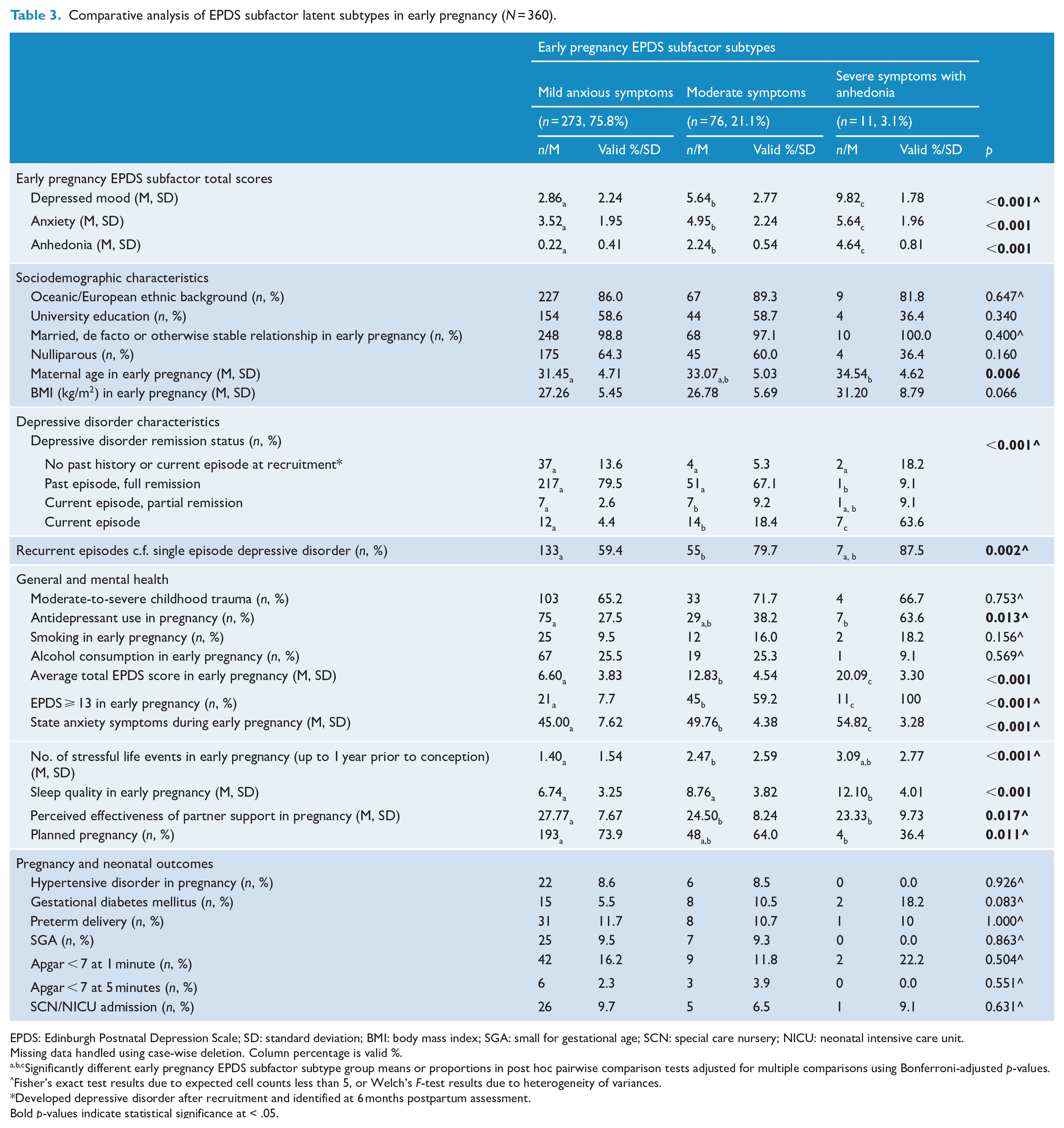
EPDS: Edinburgh Postnatal Depression Scale; SD: standard deviation; BMI: body mass index; SGA: small for gestational age; SCN: special care nursery; NICU: neonatal intensive care unit.
Missing data handled using case-wise deletion. Column percentage is valid %.
a,b,cSignificantly different early pregnancy EPDS subfactor subtype group means or proportions in post hoc pairwise comparison tests adjusted for multiple comparisons using Bonferroni-adjusted p-values.
Fisher’s exact test results due to expected cell counts less than 5, or Welch’s F-test results due to heterogeneity of variances.
Developed depressive disorder after recruitment and identified at 6 months postpartum assessment.Bold p-values indicate statistical significance at < .05.
Analysis of early pregnancy EPDS subfactor subtype characteristics
Table 3 displays observed early pregnancy subtype frequencies and means across a range of characteristics and outcomes. Maternal age differed significantly between subtypes, such that women with Severe Symptoms with Anhedonia were significantly older than women with Mild Anxious Symptoms only.
More women with Severe Symptoms with Anhedonia met diagnostic criteria for a current depressive episode (63.6%) compared to women in the Mild Anxious Symptoms and Moderate Symptoms subtypes, with more women with Moderate Symptoms (18.4%) also meeting diagnostic criteria for current depressive episode compared to women with Mild Anxious Symptoms (4.4%). Depressive episodes were more likely to be recurrent compared to single episodes in Moderate Symptoms (79.7%) compared to Mild Anxious Symptoms (59.4%).
Antidepressant use was more prevalent in the Severe Symptoms with Anhedonia (63.6%) and Moderate Symptoms (38.2%) subtypes compared to the Mild Anxious Symptoms subtype (27.5%). All 11 women (100%) with Severe Symptoms with Anhedonia scored an EPDS of ⩾ 13 during early pregnancy, compared with 7.7% of women with Mild Anxious Symptoms and 59.2% of women with Moderate Symptoms, with all three pairwise comparisons significant. State anxiety was significantly higher in women with Severe Symptoms with Anhedonia compared to both women with Mild Anxious Symptoms and Moderate Symptoms, and significantly higher in women with Moderate Symptoms compared to women with Mild Anxious Symptoms. Sleep quality was significantly poorer in women with Severe Symptoms with Anhedonia compared with both Mild Anxious Symptoms and Moderate Symptoms subtypes, whereas perceived effectiveness of partner support was significantly lower for women with both Moderate Symptoms and Severe Symptoms with Anhedonia compared to women with Mild Anxious Symptoms. Significantly fewer women with Severe Symptoms with Anhedonia (36.4%) reported that their pregnancy was planned compared to women with Mild Anxious Symptoms only (73.9%). There were no differences between early pregnancy subtypes across the pregnancy and neonatal outcomes measured.
Latent transition subtypes
Table 3 presents the latent transition probability matrix estimated by the three-subtype non-invariant LTA model. Women with Mild Anxious Symptoms in early pregnancy had a strong probability of remaining in the Mild Anxious Symptoms subtype at 6 months postpartum (LTP1 = 0.81), a relatively moderate probability of transitioning into the Moderate Symptoms subtype (LTP2 = 0.16) and a relatively low probability of transitioning into the Severe Symptoms subtype (LTP3 = 0.03). Women with Moderate Symptoms in early pregnancy had a relatively higher probability of transitioning into the Mild Anxious Symptoms subtype at 6 months postpartum (LTP4 = 0.48) than remaining in the Moderate Symptoms subtype (LTP5 = 0.42). The probability was relatively low for women with Moderate Symptoms in early pregnancy to transition into the Severe Symptoms subtype at 6 months postpartum (LTP6 = 0.10). Interestingly, the model-estimated probability of women with Severe Symptoms with Anhedonia at early pregnancy remaining in the Severe Symptoms subtype at 6 months postpartum was zero (i.e. LTP9); this indicates that none of the women in our sample who were had Severe Symptoms with Anhedonia in early pregnancy scored similarly high patterns on the EPDS subfactors to remain in the Severe Symptoms subtype at 6 months postpartum. Rather, the probability was very high for women who were had Severe Symptoms with Anhedonia in early pregnancy to transition to the Moderate Symptoms profile at 6 months postpartum (LTP8 = 0.92), with a relatively low probability of transitioning to the Mild Anxious Symptoms subtype at 6 months postpartum (LTP7 = 0.08).
Supplementary Table 3 presents bivariate measures of association (Spearman’s rho) between all model-estimated latent transition probabilities with several sociodemographic characteristics, and pregnancy and neonatal outcomes, and general and mental health measures at 6 months postpartum. Higher state anxiety scores and poorer sleep quality both at 6 months postpartum were significantly associated with higher probability estimates in all three deteriorating latent transition profiles (LTPs) (i.e. LTP2, LTP3 and LTP6 in Table 4; ps < 0.05). Interestingly, other than higher state anxiety scores in early pregnancy being associated with significantly higher probability estimates in all three improving LTPs (i.e. LTP4, LTP7 and LTP8 in Table 4; ps < 0.05), none of the other variables were associated with the probability of being in an improving LTP.
Latent transition probability matrix between early pregnancy and 6 months postpartum subtypes, nominal reference and characterisation of LTPs as maintaining, improving and deteriorating depressive symptoms.
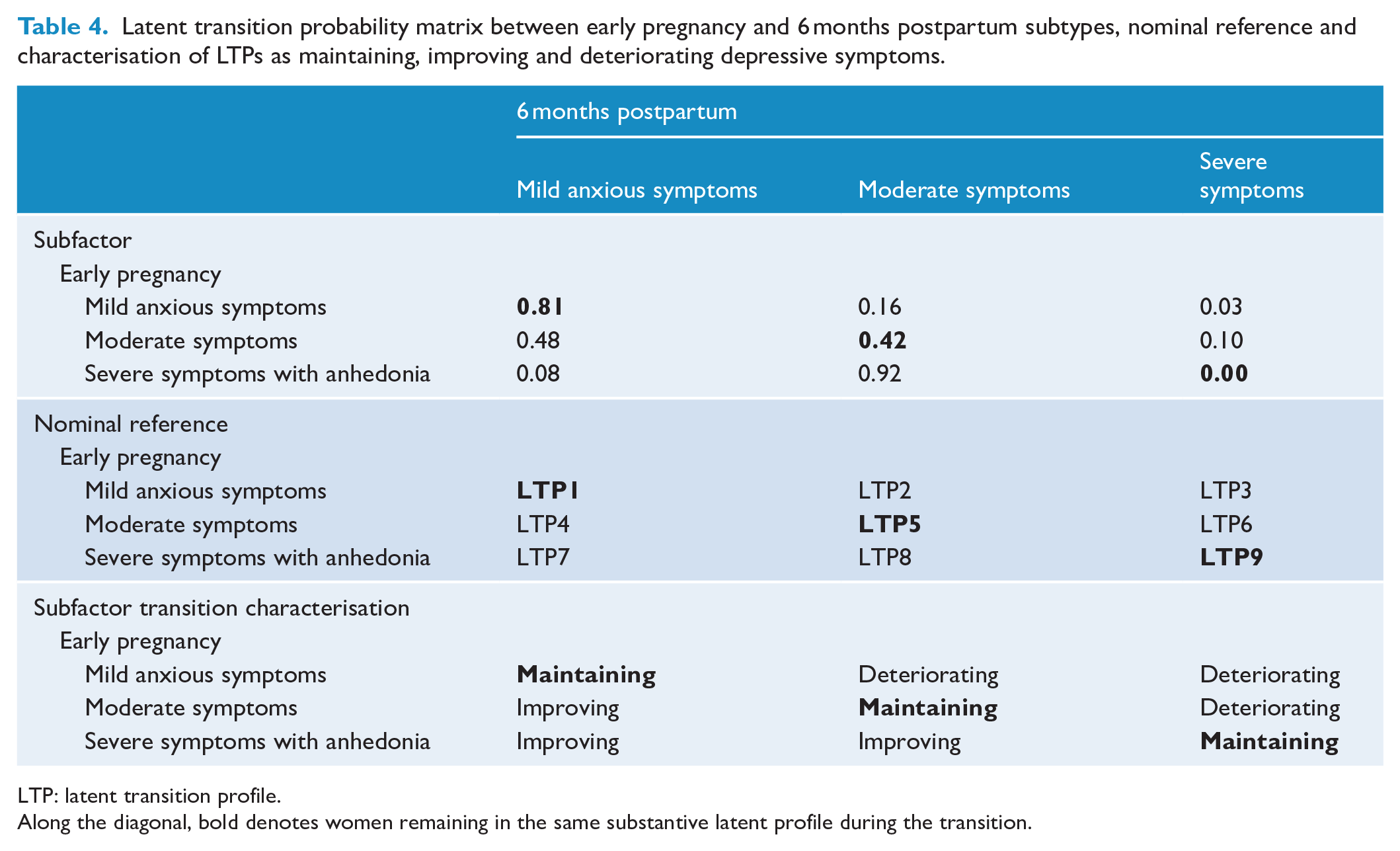
LTP: latent transition profile.
Along the diagonal, bold denotes women remaining in the same substantive latent profile during the transition.
Discussion
Our study set out to replicate prior use of the EPDS to define subtypes of perinatal depression and both expand and improve on previous methodology using a consistent protocol. We administered a diagnostic measure across the whole sample in early pregnancy at recruitment and repeated at 6 months postpartum, and administered the EPDS twice in pregnancy and at 6 and 12 months postpartum, as defined by the term ‘perinatal’ depression. Our data did not support the EPDS factor structure reported by Putnam et al.; however, both our early pregnancy and 6-month postpartum data confirmed the three-factor EPDS structure reported in Branquinho et al. for depressed mood, anxiety and anhedonia subfactors (Branquinho et al., 2022; Putnam et al., 2017). Using these subfactors, we identified three latent subfactors in early pregnancy and three in the postpartum. When examining subtype stability across the perinatal period, the Mild Anxious Symptoms subtype was found to be most stable. Women in the Moderate Symptoms subtype and those in the Severe Symptoms with Anhedonia subtype both improved. The subtypes did not predict any associated clinical risk factors or outcomes in the perinatal period for mothers or infants. A curious finding was higher state anxiety that was associated with an increased probability of improving, likely due to being correlated with the improving transition profiles. However, this finding of the role of anxiety symptoms as predictors of subtypes of depression in the perinatal period warrants further investigation. Furthermore, there were no clear patterns of association identified between the specific broader characteristics and the transition profiles estimated to describe the change in EPDS subfactor profiles between early pregnancy and the postpartum subtypes. Overall, this would suggest that using the EPDS screening measure to create subtypes for perinatal depression, although possible, is limited in clinical utility or significance. However, it is worth reiterating that techniques for clustering patients based on data, require subjective decision-making by the researchers in the pursuit of a meaningful and clinically valid solution. As such, the impetus is on the researchers to document and present in as much detail the method and rationale for their decisions, particularly given there are as many solutions for the number of classes as there are participants in the sample. Ultimately, these decisions will have impacted on the magnitude of associations between class and transition probabilities and participant characteristics and clinical variables.
In considering the use of the EPDS to define clinical subtypes, it is worth remembering that this is a measure developed as a screening tool for use in clinical settings for identifying women potentially at risk of having perinatal depression (Cox et al., 1987). This screening measure is not a diagnostic measure of depressive disorders and although it is a well performing screening tool that is widely used within clinical services, rates of sensitivity, specificity and predictive validity vary (Gibson et al., 2009). Indeed, a recent commentary by the original author of this scale emphasised that this was not designed as a diagnostic tool, outcome measure or measure of clinical change. Rather, it is a measure designed to assist with universal or targeted screening programmes within the community (Cox, 2019). Despite these ongoing cautions, the EPDS is one of the most common measures used to establish perinatal depression within many areas of related research and frequently is relied on as the only measure of the disorder (Bell et al., 2015; Dachew et al., 2021; Viuff et al., 2018). The EPDS is a scale that produces a continuous score of symptom severity rather than being able to classify the presence/absence (e.g. psychomotor change/retardation) and differential presentation (e.g. increased or decreased appetite or early or late insomnia) of symptoms, which are core to the developments of depression subtypes outside of the perinatal period. As such, it is not surprising that our findings suggest it may be problematic to rely on the EPDS alone to examine important pathophysiological mechanisms underlying perinatal depression or to predict outcomes from exposure of depression on offspring. Indeed, previous studies in adult depression examining subtypes have all relied on symptoms identified through diagnostic measures and administered clinical diagnostic and observer-rated measures (Lamers et al., 2016). They have not used a measure designed to be used as an initial screening tool for clinical settings, such as maternity or community health.
Theoretical debates on the classification of depression are long-standing with ongoing debates on whether diagnosis is best supported by a unitarian, binary or other approaches to classification and the place for identification of clinical subtypes, as documented in a recent Lancet Commission (Herrman et al., 2022). The unitarian approach taken in contemporary classification systems has demonstrated limitations when it comes to progress in understanding underlying etiological factors, such as genetics, co-morbidities (e.g. cardiovascular disease risk) and response to treatment (Parker, 2000; Penninx, 2017). In his seminal paper in 2000, Parker highlighted the flaws in many of the approaches where prevalence of subtypes may vary in samples, the ‘white noise’ of other overlapping disorders and the requirement ideally for unique rather than overrepresented symptoms to clarify potentially different depressive disorders (Parker, 2000). He proposed that psychotic symptoms and observed psychomotor agitation/retardation did allow meaningful different disorders to be distinguished. Recently, Penninx and colleagues have also highlighted the difficulty in progress in understanding the relationship between depression and risks for common physical health disorders, such as cardiovascular disease, without understanding better the subtypes of depression (Penninx, 2017). They have also identified using longitudinal data differences between ‘atypical’ depression and ‘melancholic’ depression, with the former associated with immune-metabolic risks over time including obesity, cardiovascular disease and raised inflammatory profiles, whereas the latter does not make this potential distinction important for clinical care (Lamers et al., 2016). In a recent paper by Maj et al. (2020), the challenges in distinguishing clinical subtypes, severity, symptom profile and clinical staging are outlined in detail. What is clear from this comprehensive overview and discussion is the complexity and the need for careful characterisation if progress in personalisation and identification of meaningful subtypes of depression is to occur (Maj et al., 2020). In perinatal depression, this has also great theoretical potential given the lack of clarity as to factors that predict when depression is and is not associated with impacts on pregnancy and parenting outcomes. Our findings suggest EPDS is unlikely to be a measure that can meaningfully explore subtypes and their relationship to these important associations with perinatal depression; however, future research could draw on the methodology in other areas of depression subtype research and examine this in the specific context of the perinatal period and associated outcomes.
This study has many strengths including the use of a repeat diagnostic measure, the inclusion of only those who meet criteria for a depressive disorder, and repeated and consistent use of EPDS at two time points in pregnancy and two time points in the postpartum, and the consistent collection of a wide range of other relevant variables and expert statistical modelling. The consistent use of a diagnostic measure to identify a clinically depressed sample and in repeat measurement of EPDS across pregnancy and the postpartum meant this study was not merely a replication of the previous two as neither were similar in these approaches. A limitation was our study did not examine subtypes or different types of depressive disorders using symptoms observed or collected using the diagnostic clinical measure as this study’s focus was based on the use of the EPDS. Another limitation is the sample only included those who speak English with little cultural and linguistic diversity in the sample. Our sample and study lacked cultural diversity with the majority from Oceanic/European background, and for findings to be generalisable and clinically meaningful, it is important to collect data, replicate in diverse samples and utilise instruments designed for use in various cultures and languages. Furthermore, a common limitation in longitudinal research and, indeed, in this study, is missing data and how to minimise the impact of missing data on the results. Although we chose to impute missing EPDS scores because we could draw on four repeat measurements characterised by strong correlations, multiple imputation procedures can introduce bias and this should be a consideration during the interpretation of the results.
There is no doubt as to the impact of depression on women and their children globally. Ensuring a prevention focus, early identification through screening, and access for women to affordable and locally available support and treatment is as important a quest as ever. However, as we seek answers to this significant, increasingly well recognised, and growing area of health need, it is important we approach this with rigour and replicate and build on findings. In many areas of science and, in particular, in mental health, we face a replication crisis with the not infrequent concern that many new findings may be subject to the ‘winner’s curse’ (van IJzendoorn and Bakermans-Kranenburg, 2021). Our study does not support the use of the EPDS to distinguish and classify perinatal depressive types. However, it does provide direction for future research to pursue understanding personalised management of perinatal depression and this is through the inclusion and use of symptoms collected through diagnostic measures of depression.
Supplemental Material
sj-docx-1-anp-10.1177_00048674231193640 – Supplemental material for Perinatal depression: The use of the Edinburgh Postnatal Depression Scale to derive clinical subtypes
Supplemental material, sj-docx-1-anp-10.1177_00048674231193640 for Perinatal depression: The use of the Edinburgh Postnatal Depression Scale to derive clinical subtypes by Megan Galbally, Stuart J Watson, Philip Boyce, Louise Howard and Helen Herrman in Australian & New Zealand Journal of Psychiatry
Footnotes
Acknowledgements
The authors thank those who have both supported the development of MPEWS, including Michael Permezel, Andrew Lewis and Marinus van IJzendoorn, and the Spinnaker Foundation, WA Country Health Service and WA Children’s Research Fund. The authors also thank the staff and students on the study and research coordinators for their contribution to MPEWS. They also thank the study participants who have contributed a substantial amount of time to participating in this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This study was supported through the 2012 National Priority Funding Round of Beyond blue in a 3-year research grant (ID 519240) and a 2015 National Health and Medical Research Council (NHMRC) project grant for 5 years (APP1106823).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
