Abstract
Background:
Subjective cognitive complaints represent a leading reason for suboptimal prescription of electroconvulsive therapy, the most acutely effective treatment for depression. However, research findings regarding their presentation are conflicting.
Objective:
To describe, quantify and explain the variability in subjective cognitive complaints and subjective cognition following electroconvulsive therapy for depression.
Methods:
We searched systematically PubMed, Embase, PsycARTICLES, CINAHL and relevant reviews for research published from 1 January 1972 to 31 August 2020. We included all independent studies of patients whose subjective cognition was assessed following brief-pulse electroconvulsive therapy for depression. Three meta-analyses were conducted to quantify: (1) the prevalence of patients presenting with cognitive complaints following electroconvulsive therapy; (2) the pre–post electroconvulsive therapy change in subjective cognition and (3) the association between change in depressive symptoms and change in cognitive complaints. Effect sizes were calculated using random-effects models. Eight prespecified demographic and clinical moderators of between-study variability were assessed using sub-group analyses and meta-regressions.
Results:
Data from 72 studies including 5699 participants were meta-analysed. Weighted mean prevalence of patients with cognitive complaints was 48.1% (95% confidence interval = [42.3%, 53.9%]). The type of subjective measure used significantly impacted the pre–post electroconvulsive therapy change in subjective cognition, showing, respectively, large improvement (g = 0.81; 95% confidence interval = [0.59, 1.03]) after electroconvulsive therapy with the Squire Self-Rating Scale of Memory and small worsening (g = –0.48; 95% confidence interval = [–0.70, –0.26]) with 1-item memory scales. The strongest predictors of improved post-electroconvulsive therapy subjective cognition were increased age and larger post-electroconvulsive therapy decrease in depressive symptoms. The weighted mean correlation between pre–post electroconvulsive therapy decrease in depressive symptoms and pre–post electroconvulsive therapy decrease in cognitive complaints was 0.46 (95% confidence interval = [0.39, 0.53]).
Conclusion:
Variability in subjective cognition following electroconvulsive therapy is mostly explained by the degree of post-treatment persisting depression and by the subjective assessment used. Scales measuring exclusively subjective worsening present limited clinical utility and instruments allowing the detection of both improvement and worsening in subjective cognition should be preferred.
Keywords
Introduction
Electroconvulsive therapy (ECT) is the most acutely effective treatment for depression, presenting with the highest response and remission rates among existing antidepressant treatments (Semkovska, 2021; UK ECT Review Group, 2003). However, concerns relative to cognitive side effects, especially long-term deficits, curtail its use (Leiknes et al., 2012). Several systematic reviews and meta-analyses have shown decreases in objective cognitive function to be short-lived with modern ECT (Semkovska, 2021). Objective cognitive function refers to cognitive abilities as measured with standardised neuropsychological tests where the patient’s performance is compared to normative values. Conversely, subjective cognition refers to the patient’s own evaluation of his or her own cognitive functioning, and subjective complaints – to the patient’s own perception of worsened cognition (e.g. in memory or concentration). Throughout ECT’s history, techniques evolved with the aim to minimise these objective cognitive effects, including the move from sinewave electrical stimulus to more efficient brief-pulse (~1 ms) stimuli, the preferential use of unilateral over bilateral electrode placement, and the adjustment of intensity to the patient’s seizure threshold (i.e. the minimum charge required to induce a generalised seizure as needed for therapeutic effect) (Sackeim et al., 2007; Semkovska et al., 2011). More recently, ultra-brief pulse (<0.5 ms) ECT was developed with a similar aim. However, given meta-analytical evidence of its lower efficacy for depression relative to brief-pulse stimulus (Tor et al., 2015), current research efforts are focused on establishing ultra-brief ECT’s parameters of effectiveness (Brunner and Grozinger, 2018; Tor et al., 2015). These advances, while associated with better cognitive outcomes, have not necessarily improved ECT’s acceptability (Chen et al., 2017; Rose et al., 2003). In fact, subjective cognitive complaints have been evoked as one of the main reasons for suboptimal prescription of ECT (Kellner, 2012).
While improvements in ECT techniques have led to less-pronounced objective cognitive deficits, findings on subjective complaints are much less consistent. Specifically, the effect of modern brief-pulse ECT on subjective cognitive complaints has been found to be – deleterious (Brus et al., 2017; Hughes et al., 1981), non-significant (Frith et al., 1983; Semkovska et al., 2016), or advantageous (Coleman et al., 1996; Sienaert et al., 2010) by different research groups. Similarly, while it is established that unilateral ECT is overall associated with less objective cognitive side effects than bitemporal ECT (Sackeim et al., 2007; Semkovska et al., 2011), research on subjective complaints has not lead to consistent results. Indeed, both early and contemporaneous brief-pulse studies have found that bitemporal ECT could lead to more complaints than unilateral ECT (Fleminger et al., 1970; Semkovska et al., 2016), or could be associated with equivalent subjective complaints rates (Brus et al., 2017; Coleman et al., 1996).
Seminal ECT research found post-treatment memory complaints to be associated with persistent depressive symptoms (Cronholm and Ottosson, 1963; Fleminger et al., 1970). Since, these findings have been replicated several times. Specifically, post-ECT depression severity was associated with subjectively reported memory impairment, with larger improvements in mood leading to lower memory complaints (e.g. Berman et al., 2008; Coleman et al., 1996; Sienaert et al., 2010). Nevertheless, this finding is also not unanimous, with some research groups failing to detect a significant association between change in memory complaints and improvement in depressive symptoms post-ECT (Anderson et al., 2017; Calev et al., 1991).
Besides consistent evidence that some patients complain of cognitive difficulties after receiving ECT, over 70 years of research on such subjective reports has done little to inform clinical predictions. The variety of methodologies (including diverse treatment parameters), time elapsed between the end of ECT course and the subjective assessment, and levels of persisting depressive symptoms, may explain the heterogeneity of above-described results. However, to date, no systematic research has attempted to summarise the existing subjective data or assess the explanatory power of potential sources of heterogeneity. Thus, the present review, using a meta-analytical approach, had the following four aims: (1) estimate the prevalence of patients presenting cognitive complaints attributable to treatment following ECT for depression; (2) estimate the degree of pre–post ECT change in cognitive complaints; (3) evaluate the strength of the association between the pre–post ECT change in depressive symptoms and the pre–post ECT change in cognitive complaints and (4) examine the contribution of prespecified demographic, clinical and therapeutical moderators to observed results.
Methods
Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines for conducting and reporting systematic reviews were followed (Moher et al., 2009). See protocol in Supplemental material.
Search strategy and selection criteria
The electronic databases PubMed, Embase, PsycArticles, PsycINFO and CINAHL were searched from the year 1972 to August 2020. The year 1972 was selected as the year of publication of the Research Diagnostic Criteria (RDC; Feighner et al., 1972) for depression, since which the diagnostic criteria for major depression have remained broadly unchanged in predominant classifications (Uher et al., 2014). For each database, the following search string was used: [‘electroconvulsive therapy’ OR ECT] AND [depress* OR MDD] AND [‘cognitive side effects’ OR side effects OR complain* OR subjective OR self-report or patient-report* OR self-rat*]. Relevant reviews and the reference lists of studies meeting the inclusion criteria were also manually searched.
Inclusion criteria for the meta-analysis were studies of (1) an adult (age ⩾ 18) population; (2) treated with brief-pulse ECT for a major depressive episode (or primary depression) according to DSM-III, DSM-III-R, DSM-IV, DSM-IV-TR, DSM-5, ICD-9, ICD-10, or RDC, and where (3) data on subjective cognitive complaints were available as either: (a) prevalence rates (proportion or %) of patients reporting perceived ECT-associated cognitive deficits or (b) change in subjective cognition following ECT within a longitudinal (pre/post-ECT) design. Exclusion criteria were prescription was for maintenance ECT, cognitive effects of ECT only assessed with objective measures, severe psychiatric comorbidity (e.g. schizophrenia), neurological comorbidity (e.g. Parkinson disorder), redundant reports (for multiple publications, the most recent report with the largest sample size was included), and case series.
Data extraction and coding procedures
Citations generated from each database were imported into the reference managing software Endnote X9.2 where duplicates were identified and removed. Pairs among authors independently screened titles and abstracts of articles for potential inclusion. Disagreements were resolved through consensus discussions with the first author. Retained articles of potentially eligible studies were then assessed independently against the inclusion and exclusion criteria by pairs among the authors. Again, disagreements were resolved through consensus discussions with the first author. Data extracted by each individual author were then cross-checked against the original publication by a second author.
From each study (n) who met the inclusion criteria, the following variables were coded for each included sample (k): (1) number of participants per sample; (2) method of assessment of ECT-related cognitive complaints (prevalence rates of patients complaining vs use of self-report scale of subjective complaints); (3) for studies reporting rates of patients complaining: % patients from the studied sample who responded ‘yes’ to the question if ECT has affected negatively their cognition; (4) for studies reporting rates of patients complaining: type of cognitive complaint (e.g. memory, concentration); (5) for studies using self-report: the subjective scale used; and (6) for studies using self-report: mean (M) and standard deviation (SD) of pre- and post-scores (or pre–post ECT change) on the subjective scale. The distinction between samples (k) and studies (n) was made as the majority of studies provided data on more than one sample: e.g., if the study included four samples, each treated with a different type of ECT (e.g. Sackeim et al., 1987, 2008), that study (n = 1) provided data for four samples (k = 4).
Where available, data on eight prespecified moderators were also extracted. These were time interval (days) between end of ECT course and assessment of subjective cognition, age (years), gender (% women in sample), electrode placement, mean number of ECT sessions, mean electrical charge dose; improvement in depression (pre–post ECT change in depression severity), and objective cognitive effects. To be coded as moderator for a given sample, depression severity had to be assessed using a standard scale (e.g. the Montgomery–Asberg Depression Rating Scale [MADRS], Beck Depression Inventory [BDI]) both before and at the same time point following ECT as the corresponding subjective cognitive report. All depression severity scores were transformed to the Hamilton Rating Scale for Depression, using standard transformation formulae (Heo et al., 2007 for the MADRS; Vittengl et al., 2005 for the BDI-II). Positive values of the pre–post ECT change in depressive symptoms indicated better post-ECT function. To be coded as moderator for a given sample, objective cognition had to be assessed with at least one validated neuropsychological test (Semkovska et al., 2019) administered both before and at the same time point following ECT as the corresponding subjective report. Then, objective cognitive effects were calculated using Lipsey and Wilson’s (2001) composite score of pre–post ECT change in performance on all neuropsychological tests used. Thus, only one objective composite score per sample was used in order to avoid weighing individual studies relative to the number of cognitive tests used. Positive values of the pre–post ECT objective cognitive change indicated better post-ECT function.
Statistical analyses
Three sets of meta-analyses were conducted to provide a quantitative estimate of: (1) prevalence of patients presenting with cognitive complaints following ECT; (2) pre–post ECT change in subjective cognition and (3) strength of the association between change in depressive symptoms and change in cognitive complaints. The three corresponding main outcomes were defined as follows. The mean estimated prevalence equated the weighed by study sample size pooled mean percentage of patients reporting ECT-related cognitive complaints. The pre–post ECT change in subjective cognition corresponded to the standardised mean difference between pre-ECT and post-ECT score on a scale measuring subjective complaints. Effect sizes were calculated for each individual sample using Cohen’s d index of individual effects: dk = (Mkpre-ECT-Mkpost-ECT)/SDpk, where d is the effect size, k: the individual sample, Mkpre-ECT: the pre-treatment mean on the scale, Mkpost-ECT: the post-treatment mean and SDpk is the pooled standard deviation. Effect sizes were adjusted for sample size using Hedges’ g (1985). Given the diversity of possible scoring methods identified, the corresponding subjective cognition variables for each were coded so a positive effect size indicates less-subjective cognitive complaints (better subjective cognition) after ECT relative to pre-treatment and a negative effect size – more cognitive complaints post-ECT (worse subjective cognition) than before treatment. Weighted average effect sizes were interpreted according to Cohen’s (1988) recommended cut-offs of 0.80, 0.50 and 0.20 for large, medium and small effect size, respectively. The mean strength of the association between change in depressive symptoms and change in cognitive complaints corresponded to the Fisher’s r-to-z transformation of the observed in each sample correlation between these two variables. The correlation coefficients were then combined into a single estimate of the population correlation by averaging them weighted by sample size (Hedges and Olkin, 1985). The significance of all results was tested with the z statistic and level of significance set at α = 0.05.
Heterogeneity of observed rates, effect sizes and correlation coefficients was assessed with the I2 statistic, which allows comparisons across meta-analyses on different sample sizes, unbiased by the sample size (Borenstein et al., 2009). When significant heterogeneity was found (I2 ⩾ 25%), we investigated the pre-specified moderators’ capacity to explain variability for each of the main outcomes (Borenstein et al., 2009). For the electrode placement moderator, a categorical variable, sub-group mixed-effects analyses were used when at least two samples were available per moderator level. For the remaining, continuous moderators (e.g. age and number of ECT sessions) random-effects, method of moments, meta-regressions were conducted when ⩾10 samples contained data on the corresponding moderator variable. All analyses were conducted with the Comprehensive Meta-Analysis (v.2.2; Biostat, Englewood, USA) software.
Risk of bias
When evaluating risk of bias in observational studies, methodologists recommend against the use of scales and summary scores to assess the quality of studies. Indeed, applying a quality scale creates an additional study bias through their intrinsically subjective nature (Herbison et al., 2006) and prevents the examination of heterogeneity sources (for a full systematic scoping review of recommendations, see Mueller et al., 2018). Instead, expert consensus advises that moderator analyses should be conducted to examine variability between studies, giving particular consideration to the topic area (Mueller et al., 2018; Shamliyan et al., 2012). The prespecified moderators represent variables with established influence on cognitive outcomes in ECT research analyses (see review of meta-analyses by Semkovska, 2021), thus addressing the expert recommendation of quality assessment.
The results of any meta-analysis are at risk of publication bias because studies with non-significant results are less likely to be published than those with significant results. We assessed publication bias in two ways: through Egger’s test of significance of bias and a fail-safe N file-drawer analysis (Orwin, 1983) to determine how many unpublished studies with null findings would negate the significant findings derived from published studies.
Results
After deleting duplicates, the search strategy identified 5454 citations from which 251 were assessed for eligibility and 72, including data from 5699 patients, were meta-analysed. Figure 1 presents the review process. Tables S1–S3 in the Supplemental Material include all extracted and coded variables from each study and each included sample.
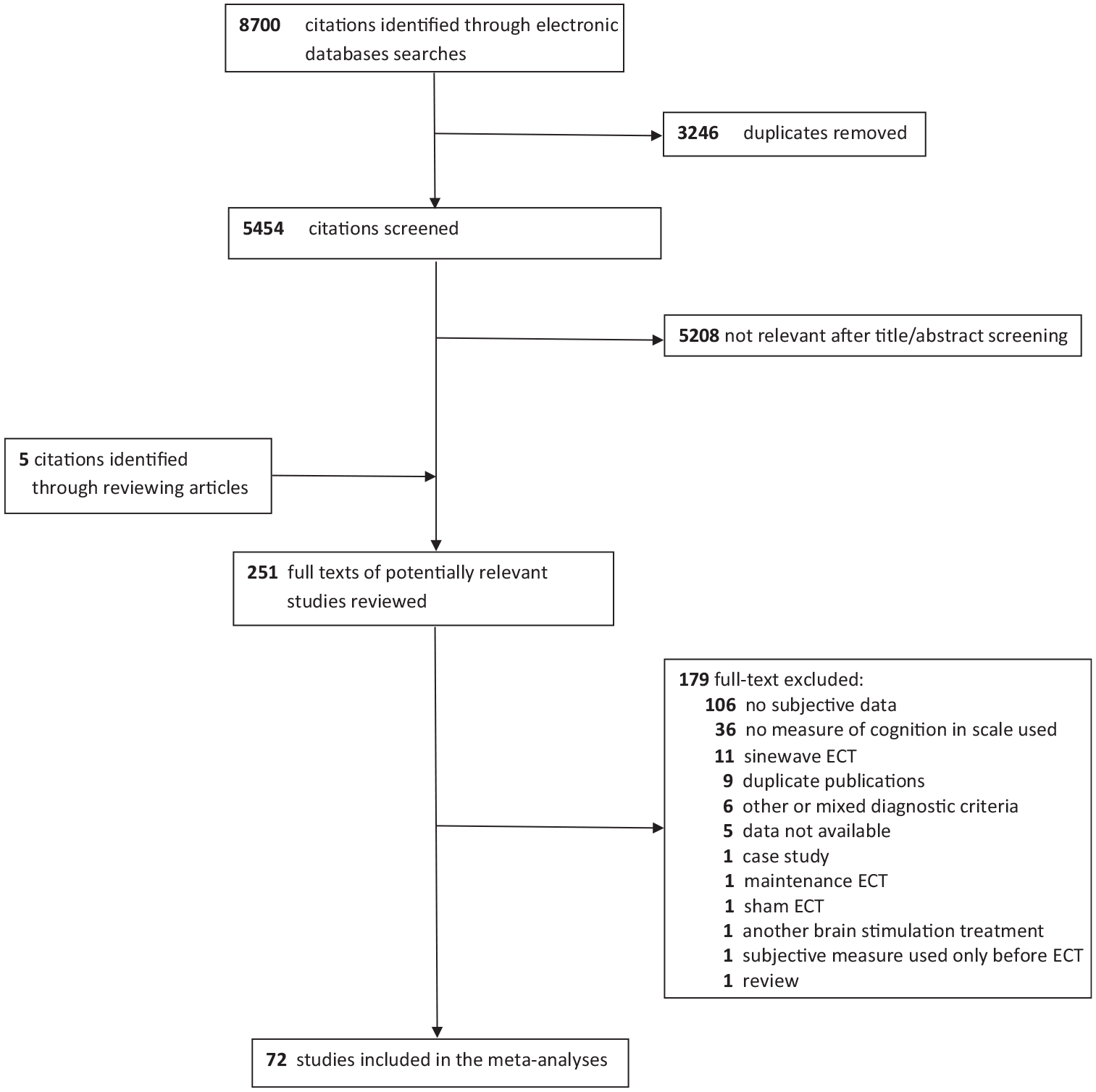
PRISMA flow chart of studies’ selection.
Prevalence rates of patients with cognitive complaints attributed to ECT
Among the selected studies, 27 provided data for overall 59 prevalence rates (k) of perceived ECT-associated cognitive complaints, collected from 2625 patients. Samples sizes ranged 6–732, with an average of 58 patients. Among the recorded types of ECT-associated complaints, ‘memory’ difficulties were reported in 96% (26/27) of the meta-analysed studies, followed by reports of ‘confusion’ in 15% (4/27) of the studies, and feelings of being ‘slowed down’ – 7% (2/27). Problems with ‘general cognition’, ‘concentration’ and ‘thinking’ were the least frequently reported with 4% each (1/27). The weighted mean prevalence rate of patients with cognitive complaints was 48.1% (95% confidence interval [CI] = [42.3%, 53.9%]), with an Egger’s regression intercept of –3.58, p = 0.25, suggesting no evidence of publication bias. The distribution of prevalence rates was highly heterogeneous with I2 = 93%, p < 0.001, thus the planned moderator analyses were conducted (Table 1). Effects of electrode placement, age and number of ECT sessions contributed significantly to between-samples rates’ variability. Significantly higher prevalence of patients complaining of cognitive effects were observed following bitemporal ECT than after right unilateral (RUL) ECT. Furthermore, the higher the patients’ age, the lower the prevalence of cognitive complaints after ECT and the higher the number of sessions received, the higher the prevalence of complaints. The effect of the remaining moderators was not significant.
Moderators’ effects on the prevalence rates of patients with cognitive complaints attributed to ECT.
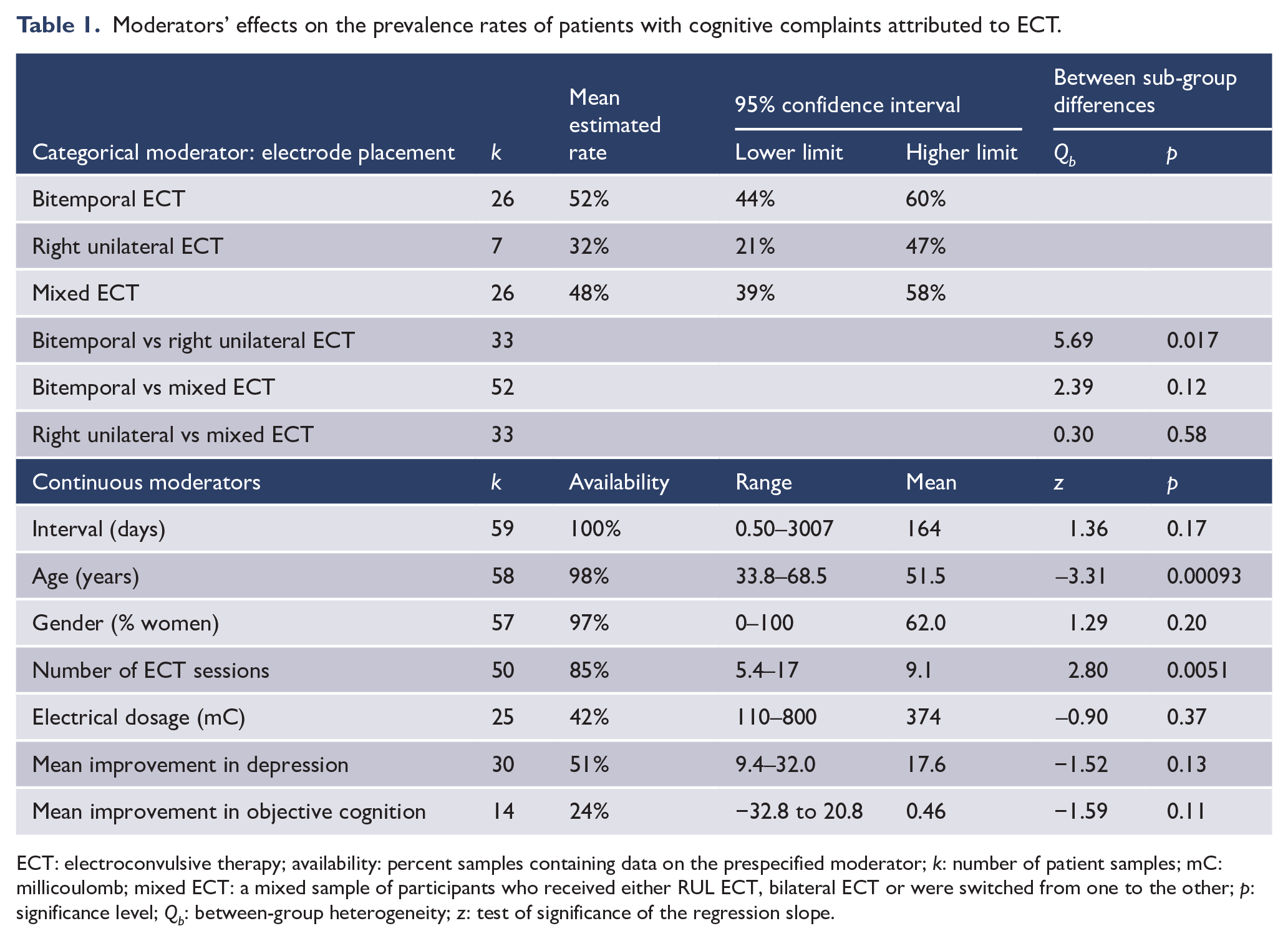
ECT: electroconvulsive therapy; availability: percent samples containing data on the prespecified moderator; k: number of patient samples; mC: millicoulomb; mixed ECT: a mixed sample of participants who received either RUL ECT, bilateral ECT or were switched from one to the other; p: significance level; Qb: between-group heterogeneity; z: test of significance of the regression slope.
Change in subjective cognition following ECT
Data from 56 studies, 146 samples, totalling 3897 patients reporting on their change in subjective cognition following ECT relative to pre-ECT levels, were meta-analysed. Sample sizes ranged 10–1018, with an average of 46 patients. The weighted mean effect size of change in subjective cognition was 0.18 (95% CI = [0.05, 0.31]), z = 2.74, p = 0.006. However, the scales used by the various meta-analysed studies were notably heterogeneous in terms of number and type of items included, with some scales offering the possibility for the patient to choose an improved cognition relative to pre-treatment levels, such as the Squire Self-Rating Scale of Memory (SRSM) function (Squire et al., 1979), while others only allowed for comparable or worse than pre-treatment levels reports of subjective effects, such as the cognitive component of the Columbia Subjective Side Effects Schedule (CSSES; Sackeim et al., 1987). Thus, we classified each sample providing change in subjective cognition data within one of five categories, depending on the scale used (Table 2). Then, moderator analyses were conducted to assess the effect of scale used, which was overall significant: Qb = 67.6, p < 0.0001. Samples using a 1-item memory problems severity measure found a small, but significant increase in subjective memory complaints following ECT relative to pre-treatment levels. On the contrary, samples using the SRSM showed a large decrease in memory complaints following ECT relative to pre-treatment levels. Fail-safe analyses showed the need for, respectively, 2164 (1-item memory scales) and 4646 (SRSM) unpublished studies to negate these significant results. The remaining scale categories did not show a significant pre–post ECT change in subjective cognition (Table 2). Large heterogeneity remained within each scale (all I2 > 75%). Given the observed between-scales differences, all pre-specified moderator analyses were thus completed independently on each scale category, if enough samples were available to conduct them. See Tables 3 and 4 for the results of, respectively, the sub-group mixed-effects analyses and the random-effects meta-regressions.
Type of scales used to measure pre–post ECT change in subjective cognition with associated mean effect sizes and mixed-effects between-scales comparisons.
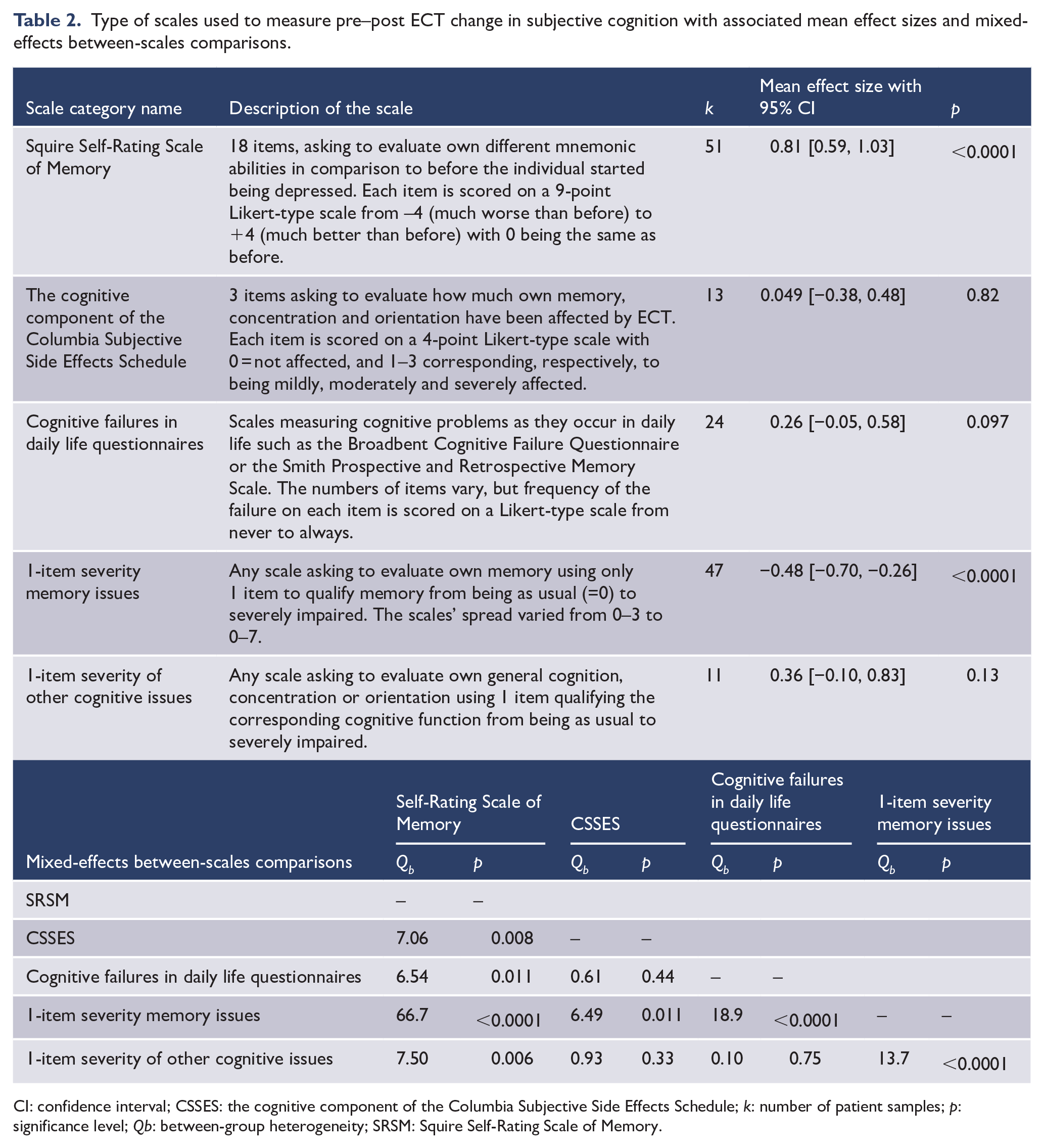
CI: confidence interval; CSSES: the cognitive component of the Columbia Subjective Side Effects Schedule; k: number of patient samples; p: significance level; Qb: between-group heterogeneity; SRSM: Squire Self-Rating Scale of Memory.
Moderator’s effects of electrode placement on the pre–post ECT change in cognitive complaints’ severity according to the subjective measure used.
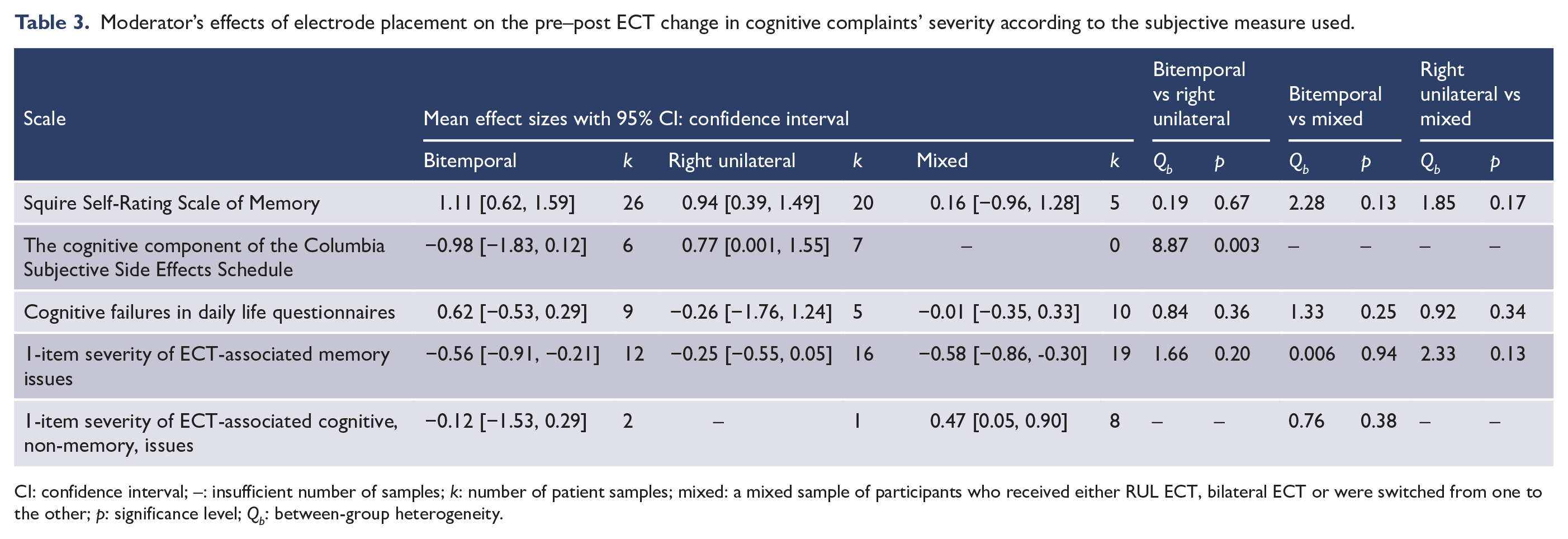
CI: confidence interval; –: insufficient number of samples; k: number of patient samples; mixed: a mixed sample of participants who received either RUL ECT, bilateral ECT or were switched from one to the other; p: significance level; Qb: between-group heterogeneity.
Continuous moderators’ effects on pre–post ECT change in cognitive complaints’ severity according to the subjective measure used.
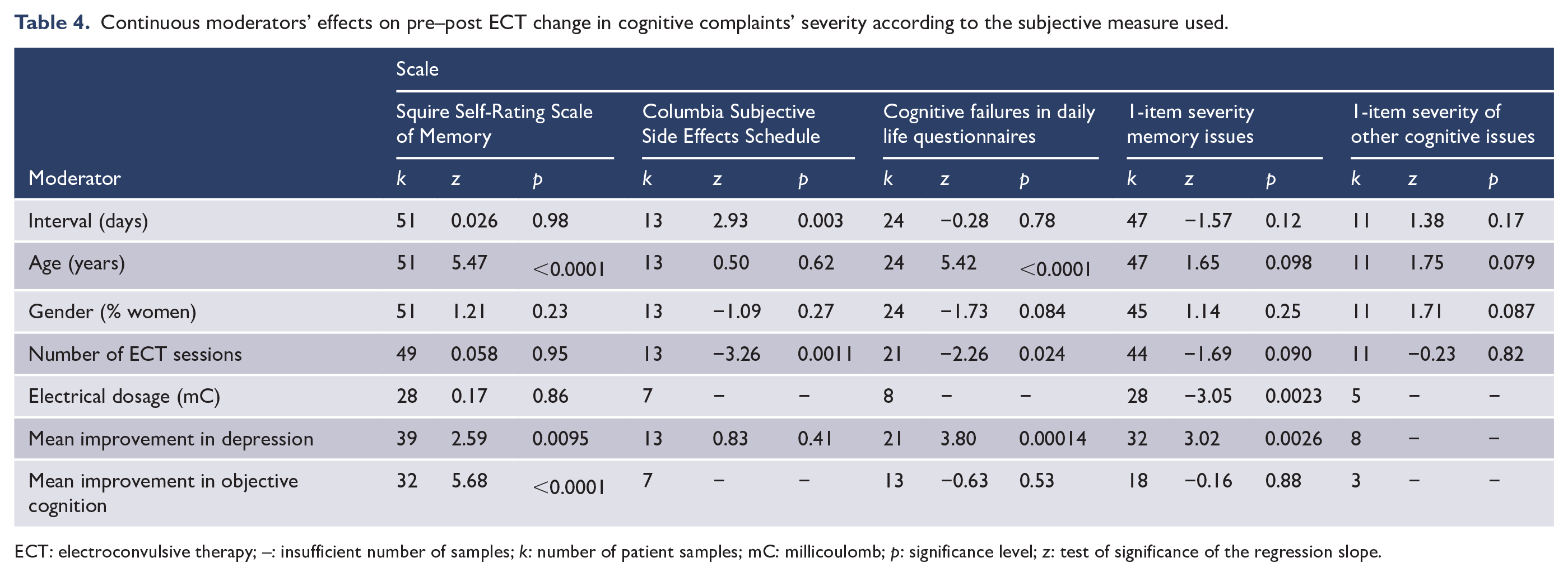
ECT: electroconvulsive therapy; –: insufficient number of samples; k: number of patient samples; mC: millicoulomb; p: significance level; z: test of significance of the regression slope.
Mean improvement in depression moderated significantly the pre–post ECT change in subjective cognition for three of the four meta-analysed scales, all significant regressions coefficients having small to medium effect sizes (0.20 for the SRSM, 0.69 for the cognitive failure in daily life questionnaires and 0.29 for the 1-item scales of severity of memory problems). The larger the improvement in depressive symptoms, the larger the improvement in subjective cognition following ECT. Older age was associated with less cognitive complaints after ECT if these were measured with the SRSM (medium association effect size = 0.59) or a cognitive failure in daily life questionnaires (very large association effect size = 1.22). Number of ECT sessions received was associated with higher cognitive complaints after ECT if these were measured with the CSSES (large association effect size = 0.82) or with a cognitive failure in daily life questionnaire (small association effect size = 0.24). Increasing interval between end of ECT and subjective assessment predicted better cognitive function following ECT, only if the CSSES was used (medium association effect size = 0.66). The same was true for the effect of electrode placement on the change in cognitive complaints: only studies using the CSSES found it to be significant, i.e. patients who received RUL ECT showed a medium subjective improvement post-ECT where patients receiving bitemporal ECT showed a large subjective worsening post-ECT. Increased electrical dosage predicted worse subjective cognitive function following ECT, only if a 1-item scale of severity of memory problems was used (small association effect size = 0.33). Improved objective cognitive function predicted better subjective cognition following ECT, only if the SRSM was used (very large association effect size = 1.01). Gender was not a significant moderator of the pre–post ECT change in subjective cognition, independently from type of scale used. Fail-safe analyses showed no evidence of publication bias for the moderator analyses (see Supplemental Table S4).
Relationship between change in depressive symptoms and change in cognitive complaints
Correlations between pre–post ECT change in depressive symptoms and pre–post change in cognitive complaints were reported for 40 samples from 22 studies, totalling 2010 patients. Samples sizes ranged 9 to 1018, with an average of 91 patients, while delays between the last ECT session and the post-ECT assessment varied from 1 day to 2 years (mean of 70 days). The weighted mean correlation was 0.46 (95% CI = [0.39, 0.53]), z = 11.3, p < 0.0001. Thus, the larger the improvement in depressive symptoms, the larger the improvement in subjective cognition relative to pre-ECT levels. The correlations’ effect size indicated that a significant proportion of the variance in post-ECT change in subjective cognition (21.4%) was explained by the post-ECT change in mood. Fail-safe analyses showed the need for 5577 unpublished studies with nil results to negate this significant finding. Correlations were homogeneous with I2 < 0.001, thus moderator analyses were not performed.
Post hoc moderator analyses on the effect of ECT pulse width on subjective outcomes
The systematic review identified four studies where ultra-brief pulse ECT was used, including six ultra-brief samples totalling 118 patients. All 6 samples provided data on change in cognitive complaints’ severity following ECT. Given evidence for lower objective cognitive effects following ultra-brief pulse ECT relative to brief-pulse ECT (Sackeim et al., 2008), post hoc moderator analyses were conducted to assess the contribution of pulse width to the pre–post change in subjective cognitive complaints’ severity following ECT. No significant effect of pulse width was found with all p values > 0.13. See full results in Supplemental Table S5.
Retrospective power analyses on the moderators’ testing
Moderators tests in meta-analyses are at higher risk to be underpowered than the main outcome effects, making the use of retrospective power analyses especially important (Hedges and Pigott, 2004; Valentine et al., 2010). Indeed, researchers may fail to find a significant moderator effect in meta-analyses and perhaps erroneously conclude on the absence of such effects (Valentine et al., 2010). Therefore, we conducted, for each moderator’s effects testing, a retrospective power analysis, based on Hedges and Pigott (2004) formulae for mixed- and random-effects meta-analyses of moderators. These calculations consider: the observed average number of participants per sample (58 for the prevalence of patients with cognitive complaints outcome, and 46 for the change in subjective cognition within a pre–post ECT design); the observed number (k) of effect sizes per moderator (see, Table 1, 3 and 4 for k for each moderator analysis conducted); and the degree of heterogeneity (which was high for both sets of meta-analyses where moderators’ testing was performed).
First, we conducted all the retrospective power analyses to determine the observed power of the moderator analyses to detect a small effect size (δ= 0.2). See Supplemental Table S6 for full results. Six of the eight prespecified moderator analyses of the prevalence rates of patients with cognitive complaints attributable to ECT showed a power > 0.80 to detect a small effect, with 5 of them (electrode placement, interval, age, gender and number of ECT sessions) showing a power >0.95. The observed power for the mean improvement in depression was 0.84. The moderator analyses of electrical dosage and mean improvement in objective cognition were insufficiently powered to detect a small effect of these variables on the prevalence rates of patients with subjective cognitive complaints, with values of 0.78 and 0.52, respectively.
A similar pattern was observed with the moderator analyses of the Squire SRSM pre–post ECT change: the same above-listed six moderators showed a power > 0.80 to detect a small effect. Again, electrical dosage and mean improvement in objective cognition were insufficiently powered to detect a small effect of these variables on the pre–post ECT change in the SRSM, with values of 0.72 and 0.77, respectively.
Five of the eight pre-specified moderator analyses of the 1-item severity memory issues pre–post ECT change showed a power > 0.80 to detect a small effect: electrode placement, interval, age, gender, and number of ECT sessions. The moderator analyses of electrical dosage, mean improvement in depression and mean improvement in objective cognition were insufficiently powered to detect a small effect of these variables on the pre–post ECT change in the 1-item severity of memory issues, with values of 0.72, 0.77 and 0.52 respectively.
The moderator analyses conducted on the remaining scales showed insufficient power to detect a small moderator effect on the pre–post ECT change in subjective cognition. Specifically, that power was <0.41 for all moderator analyses using the CSSES; <0.65 for all moderator analyses using cognitive failures in daily life questionnaires; and <0.36 for all moderator analyses using 1-item severity scale of other than memory cognitive issues associated with ECT.
Second, we calculated the power of all conducted moderator analyses to detect a medium effect size (δ = 0.5) and then, third – their power to detect a large effect size (δ = 0.8). All conducted moderator analyses showed a power > 0.97 to detect a medium or large moderator effect size on the studied subjective outcomes. See full results in Supplemental Table S6.
Discussion
Using three types of published data, we quantified the presentation of subjective cognitive complaints attributed to ECT and estimated the contribution of demographic, clinical and treatment technique variables to these complaints. We found that close to half of patients reported experiencing ECT-associated cognitive side effects. However, how much cognitive complaints changed following ECT relative to their pre-treatment level depended on the type of measure used, with one instrument actually showing a significant large improvement in subjective memory following ECT. Improvement in depressive symptoms emerged as the strongest predictor of improvement in subjective cognitive function following ECT. Together, these results support and expand on previous reviews’ conclusions (Fraser et al., 2008; Prudic et al., 2000; Vann Jones and McCollum, 2019).
To our knowledge, to date, no systematic review or meta-analysis has sought to estimate the prevalence of patients treated for depression who would report that ECT has impacted negatively their cognition. In addition to determining this to be the case for every second patient, we also found prevalence rates of patients with complaints to be affected by electrode placement (more patients with complaints after bilateral ECT than after RUL ECT), by the number of ECT sessions (receiving more sessions was associated with higher rates of cognitive complaints) and by the patients’ age (older patients complained less than younger patients). Increased age was also a strong predictor of improved post-ECT subjective cognition, when assessed with the Squire SRSM or with questionnaires measuring cognitive failures as they occur in daily life. This effect of age on SRSM has already been described by one previous review of eight studies on subjective memory complaints following ECT (Vann Jones and McCollum, 2019). We replicated the finding through a comprehensive analysis of 51 samples assessed with the SRSM and extended it to other cognitive complaints (e.g. attention and planning difficulties during daily activities).
Unlike objective cognitive ECT effects (Semkovska & McLoughlin, 2010; Blomberg et al., 2020), subjective cognitive complaints did not decrease with the passage of time following the end of treatment. Indeed, objective cognition progressively improves after ECT for up to 6 months following the end of treatment. However, delay following ECT completion was not found to affect significantly neither the prevalence of subjective complaints, nor their pre–post ECT change in severity, except when measured with the CSSES. Specifically, the moderator analyses on the interval variable were well powered to detect even small effects on the following main outcomes: prevalence rates of patients with cognitive complaints, pre–post ECT change on the SRSM and pre–post ECT change on the 1-item severity of memory issues scales. All moderator analyses on the interval variable were also well powered to detect medium and large effects on the main outcomes. Thus, when present, subjective cognitive complaints remained the same, whether they were measured 1 day, 3 months or 2 years after ECT’s end, except if the CSSES was used (8% of all studied samples), where a decrease in cognitive complaints could be observed with the passage of time.
The relationship between subjective and objective cognitive assessments is notoriously poor in depression (Petersen et al., 2019; Semkovska, 2019; Srisurapanont et al., 2017). Thus, consistent with non-ECT research in depression, our meta-analysis also did not find improvement in objective cognitive function to be a significant predictor of neither complaints’ rates nor pre–post ECT change in subjective effects, with one exception. Interestingly, post-ECT improvement on the SRSM was associated with improved objective cognition, with a corresponding very large and robust against publication bias effect size (i.e. needing 8567 unpublished studies with a non-significant association between the two to be unvalidated). While underpowered to detect small effects, this moderator analysis was also well powered to detect medium and large effects. The observed positive association could be explained by the specific characteristics of the scale – the SRSM allows for subjective memory to be rated impaired as well as improved relative to pre-ECT levels of functioning, whereas all other meta-analysed subjective measures only quantify degree of cognitive worsening.
The type of subjective measure used significantly impacted the pre–post ECT change in subjective cognition. Specifically, samples using a 1-item memory problems severity measure differed significantly from all other subjective measures and showed collectively a small worsening in subjective memory complaints following ECT relative to pre-treatment levels. Samples using the SRSM also differed significantly from all other measures and showed a large post-ECT improvement in subjective memory relative to pre-treatment levels. The latter result, based on 51 samples, supports and extends observations from previous reviews on smaller number of studies (six by Prudic et al., 2000, to eight by Vann Jones and McCollum, 2019). The remaining subjective scales did not differ from each other and did not show a significant pre–post ECT change in subjective cognitive complaints. Their common characteristic was that they all assessed different types of cognitive slips, which either included memory as one type of slip among several others (cognitive failures in daily life questionnaires; CSSES) or did not assess memory at all (1-item severity of other than memory cognitive issues). Each of the five scale categories was associated with a distinct moderators’ profile. However, the positive effect of improved mood on subjective cognition was the most common cross-scale moderator’s result.
Indeed, the association of better mood with better post-ECT subjective cognition emerged as the strongest research finding, supported by several analyses. In addition to the above-described significant moderating effect of mood on subjective cognition, our meta-analysis showed a homogeneous, moderate positive correlation between the two variables. This robust to publication bias finding has important clinical implications. Specifically, it suggests that subjective cognitive complaints do not reflect persistent, ECT-associated, objective cognitive effects, but rather point towards a persistent post-ECT depressive symptomatology. Importantly, this association does not imply that patients mistake symptoms of depression for cognitive problems as it has been previously suggested (Prudic et al., 2000), but probably indicates the patients’ ability to detect cognitive issues associated with their persistent depression. The observed association between SRSM and improved objective cognition supports that hypothesis. Furthermore, our findings relative to the association between mood and subjective cognition mirror observations of both the general population and patients presenting with depression not treated with ECT. Indeed, two large studies conducted two decades apart in healthy individuals showed that subjective forgetfulness is associated with the presence of subsyndromal depressive mood symptoms (in the Netherlands: N = 2000, age range: 24–86 [Ponds et al., 1997]; in the United Kingdom: N = 2544, age range: 18–87 [Schweizer et al., 2018]). Similarly, the association between subjective cognitive complaints and depressive symptoms in major depression is well established (see two recent systematic reviews of Chakrabarty et al., 2016; Hill et al., 2016).
Clinical implications
Clearly, the literature on subjective cognitive complaints is quite heterogeneous as previously recognised by narrative reviews (Prudic et al., 2000). Nevertheless, the pattern of our results, including the significant amount of variability in effect sizes explained by both the pre-specified moderators and the scale moderator variable permit the proposal of some recommendations. The Squire SRSM should be used preferentially to measure subjective cognition in relation to ECT. Our meta-analysis shows it to be not only the most researched, but also the most valid instrument. Indeed, performance on the scale is moderated by the most common predictors of subjective levels of complaint we found through the meta-analyses, i.e. levels of depressive symptoms and patient’s age. It was also the only measure that correlated significantly with objective indicators of cognitive improvement and the only that permits to quantify the occurrence of improvement in subjective cognition following ECT. The questionnaire asks how memory functioning in different situations compares to the patients’ pre-morbid functioning. Such method requires a higher level of cognitive processing (Schwarz, 2009) and thus engagement than a 1-item scale asking the individual to quantify degree of cognitive worsening because of ECT. Consequently, the SRSM could be useful for both psychoeducational purposes and to tackle cognitive recovery. Objective cognitive effects of ECT are well established, while cognitive tests are time-consuming and intrinsically challenging for patients. The relatively short SRSM could be used routinely as a valid and reliable alternative to gauge levels of post-ECT memory issues, ensuring patient engagement by placing them at the centre of assessing this core treatment side effect. Comprehensive cognitive assessments could be kept for severe presentations of side effects (e.g. prolonged disorientation) or severe cognitive complaints detected on the SRSM.
Use of the CSSES cognitive component should be limited to research on new ECT techniques. Indeed, the CSSES cognitive component appeared to be sensitive to the ECT’s technique where enough samples were available for the corresponding moderator analyses (i.e. electrode placements and number of ECT sessions). However, unlike most other measures of subjective cognition, the CSSES variance in effect sizes was not explained by individual (e.g. age) or clinical (e.g. mood) variables; thus highlighting the instrument’s limited clinical utility.
Finally, results from our systematic review and meta-analysis suggest that 1-item scales of subjective severity memory issues (e.g. GSE-My) offer relatively limited clinical or research value. These scales represent the second-most researched group after the SRSM, but do not allow the detection of possible memory improvement and are not sensitive to ECT technique (except for electrical dose). While by nature the quickest to administer (which may explain their relatively wide use), such 1-item scales do not reliably capture the patient’s experience and contributed little to our understanding of subjective cognition after ECT. The results of a recent, large study on long-term subjective memory after ECT for any clinical indication where 77.9% of the 277 participants were treated for unipolar depression (Sigström et al., 2020) supports this view. Indeed, using the GSE-My, the authors found that pre-treatment negative expectations relative to ECT’s effects were associated with increased subjective memory worsening post-ECT.
Strengths and limitations
Strengths of the meta-analyses were broad scope with comprehensive cover of all subjective cognitive measures; using three types of data; evaluation of heterogeneity through analyses of prespecified moderators; robustness of significant findings against publication bias assessment; and the relatively large number of studies included. Availability of data was over 95% for most moderators, with no missing data for time interval, electrode placement and, for the pre–post ECT meta-analysis, age. However, only 50% of studies provided data on both subjective and objective cognition. Thus, apart from analyses of effects on SRSM where enough samples were available, observed results concerning the association between these two types of cognitions should be interpreted with caution. Similarly, due to an insufficient number of samples, the effect of electrical dosage could only be tested for half of the subjective outcomes. In addition, while a substantial portion of between-study variability in subjective cognition could be explained by the pre–post ECT change in depressive symptoms, over half of the variance remained unaccounted for by the pre-specified moderators, suggesting that sources of unexplained heterogeneity in subjective cognition remained. The effect of other potential moderators, such as previous number of depressive episodes, duration of untreated depression, frequency of ECT (twice vs thrice a week), or pre-ECT expectations relative to cognitive side effects, could be assessed by future research. Finally, a significant proportion of studies collected subjective data on mixed samples of participants who received either RUL ECT, bilateral ECT or were switched from one to the other. Thus, while testing the effect of electrode placement was possible for all outcomes, analyses on the mixed samples contributed little to explaining heterogeneity. Future reports should provide distinct subjective complaints’ results for distinct ECT techniques.
Assessment of subjective cognition following ECT using a reliable and valid instrument should be prioritised in routine clinical care. Such assessment would ideally occur about 10 days after the end of ECT course to allow for sufficient recovery of objective cognitive functions, as systematic reviews and meta-analyses show that, on average, these recover to pre-treatment levels between 4 and 14 days following end of treatment (see reviews of Porter et al., 2020; and Semkovska, 2021). The SRSM could be used as a screening tool where the detection of worsening cognition could indicate the need of a comprehensive neuropsychological evaluation. Considering the present meta-analytical findings, subjective cognitive assessment could also be used as a psychoeducation tool, that highlights the contribution of persisting depression (intrinsically linked to persisting cognitive issues) and age to subjectively experienced cognitive issues.
Supplemental Material
sj-pdf-1-anp-10.1177_00048674221089231 – Supplemental material for Subjective cognitive complaints and subjective cognition following electroconvulsive therapy for depression: A systematic review and meta-analysis
Supplemental material, sj-pdf-1-anp-10.1177_00048674221089231 for Subjective cognitive complaints and subjective cognition following electroconvulsive therapy for depression: A systematic review and meta-analysis by Maria Semkovska, Hannah Knittle, Janet Leahy and Johanne Raasø Rasmussen in Australian & New Zealand Journal of Psychiatry
Supplemental Material
sj-pdf-2-anp-10.1177_00048674221089231 – Supplemental material for Subjective cognitive complaints and subjective cognition following electroconvulsive therapy for depression: A systematic review and meta-analysis
Supplemental material, sj-pdf-2-anp-10.1177_00048674221089231 for Subjective cognitive complaints and subjective cognition following electroconvulsive therapy for depression: A systematic review and meta-analysis by Maria Semkovska, Hannah Knittle, Janet Leahy and Johanne Raasø Rasmussen in Australian & New Zealand Journal of Psychiatry
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
