Abstract
Objective:
Stress system dysregulation is considered to have an important role in the aetiology of paediatric functional neurological (conversion) disorder. This study examined salivary cortisol and α-amylase awakening responses in children with functional neurological disorder to determine activation patterns of the hypothalamic–pituitary–adrenal axis and sympathetic system. A healthy cortisol awakening response involves a robust increase in cortisol within 30 minutes of awakening. Alpha-amylase awakening response is variable in children.
Methods:
Cortisol and α-amylase were measured in saliva from 32 patients with functional neurological disorder (26 girls and 6 boys, aged 11.3−16.1 years) and 31 healthy controls (23 girls and 8 boys, aged 8.6–17.7 years). Saliva samples were collected using a Salivette sampling device at two time points – upon awakening and 30 minutes after awakening.
Results:
Patients with functional neurological disorder showed a decrease in cortisol awakening response (–4 nmol.min/L) and controls showed an increase (107 nmol.min/L), t(55) = –.4.6, p < 0.001. Within the functional neurological disorder group, 57% showed an attenuated cortisol awakening response and 43% showed an obliterated/reversed cortisol awakening response: Cortisol awakening response was negatively correlated with adverse childhood experiences, r(58) = –0.6, p = 0.002, and subjective distress (total Depression Anxiety and Stress Scales score), r(58) = –0.4, p = 0.050. In controls, cortisol awakening response showed no correlation with adverse childhood experiences and a positive correlation with subjective distress, r(56) = 0.4, p = 0.023. Total cortisol remained similar between the functional neurological disorder and control group. No significant differences were observed between the functional neurological disorder and control group in any of the α-amylase analyses.
Discussion:
The results suggest dysregulation of the hypothalamic–pituitary–adrenal axis in children with functional neurological disorder. Hypothalamic–pituitary–adrenal dysregulation in children with functional neurological disorder may contribute to comorbid symptoms of fatigue, sleep disturbance and subjective loss of well-being because circadian rhythms and energy metabolism are disrupted. Hypothalamic–pituitary–adrenal dysregulation – and changes in glucocorticoid (cortisol) signalling at the molecular level – may also contribute to increased vulnerability for functional neurological disorder symptoms because of epigenetically mediated changes to neural networks implicated in functional neurological disorder.
Keywords
Introduction
Functional neurological (conversion) disorder (FND) is a multi-network brain disorder that involves complex interactions between the brain regions underpinning motor and sensory function and those underpinning attention, salience detection, arousal, emotional states, motivational processes, beliefs and expectations (Perez et al., 2021). Children (including adolescents) with FND present with a broad range of symptoms: motor symptoms that affect the limbs, voice production or the digestive track; functional seizures; loss of sensory function; dizziness and balance difficulties and changes in cognitive function. Comorbid pain and non-specific functional symptoms – fatigue, dizziness, nausea, disrupted sleep – are common (Ani et al., 2013), as are anxiety or depression, which are present in 22–80% of cases (Vassilopoulos et al., 2022). Within paediatrics, the current understanding of FND is that stress system activation and dysregulation play a central role in generating these symptoms (Apazoglou et al., 2017; Kozlowska, 2017; Kozlowska et al., 2020; Sharma and Szaflarski, 2021). Of particular relevance in this context are the stress system components that are embedded within the brain and that modulate neural network function (Agorastos et al., 2019; Andersen, 2022; Apazoglou et al., 2017; Frank et al., 2016). Accordingly, in the Mind-Body Program at The Children’s Hospital at Westmead, we use a stress system model of FND to inform our clinical practice and guide our treatment of patients (Kozlowska, 2017; Kozlowska et al., 2020). In the current study, we examined cortisol and α-amylase responses upon awakening to determine activation patterns of the hypothalamic–pituitary–adrenal (HPA) axis and sympathetic system in children (including adolescents) with FND.
The stress system – encompassing the circadian clock, autonomic nervous system, inflammatory/immune system and HPA axis – plays an important role in homeostatic regulation, brain–body responses to threat and the biological embedding of experience (Agorastos et al., 2019; Pervanidou and Chrousos, 2018). Leading researchers in the field contend that while time-limited stress system activation is advantageous for adaptation and survival, chronic activation or dysregulation is disadvantageous, contributes to a subjective experience of ill health and increases the risk of developing a host of disease states, including functional somatic symptoms (Agorastos et al., 2019; McEwen, 1998; McEwen et al., 2015; Pervanidou and Chrousos, 2018; Zannas and Chrousos, 2017).
The HPA axis – whose end products are glucocorticoid hormones (including cortisol) – is a complicated system that has both proximal and distal effects on health and well-being. Proximally, the HPA axis regulates energy metabolism throughout the body (basal and stress-related homeostasis) and it influences many functions of the central nervous system (arousal, cognition, mood, sleep and subjective sense of well-being) (Agorastos et al., 2019; McEwen, 1998; McEwen et al., 2015; Pervanidou and Chrousos, 2018; Zannas and Chrousos, 2017). More distally, ‘glucocorticoid secretion in response to stress, as well as genes involved in the glucocorticoid signalling pathway, play a fundamental role in shaping a form of epigenetic memory through which stressful experiences become biologically embedded’ (Mourtzi et al., 2021: 1–2; Zannas and Chrousos, 2017). A growing number of studies suggest that indirect epigenetic processes, modulated by glucocorticoids (including cortisol), may have a central role, especially in the longer term, in promoting changes in all body tissues, including the brain. The potential implication, when read together with recent studies of patients with FND (Diez et al., 2020; Kozlowska et al., 2017a; Spagnolo et al., 2020) or chronic fatigue (Nguyen et al., 2017) (see Table 1), is that the resulting changes in the brain may increase the risk of developing functional neurological symptoms (Diez et al., 2020; Kozlowska et al., 2017a; Nguyen et al., 2017; Spagnolo et al., 2020).
FND studies examining changes in gene expression, Gene × Environment interactions, and plasticity changes (all presumably mediated by glucocorticoid-mediated epigenetic changes).
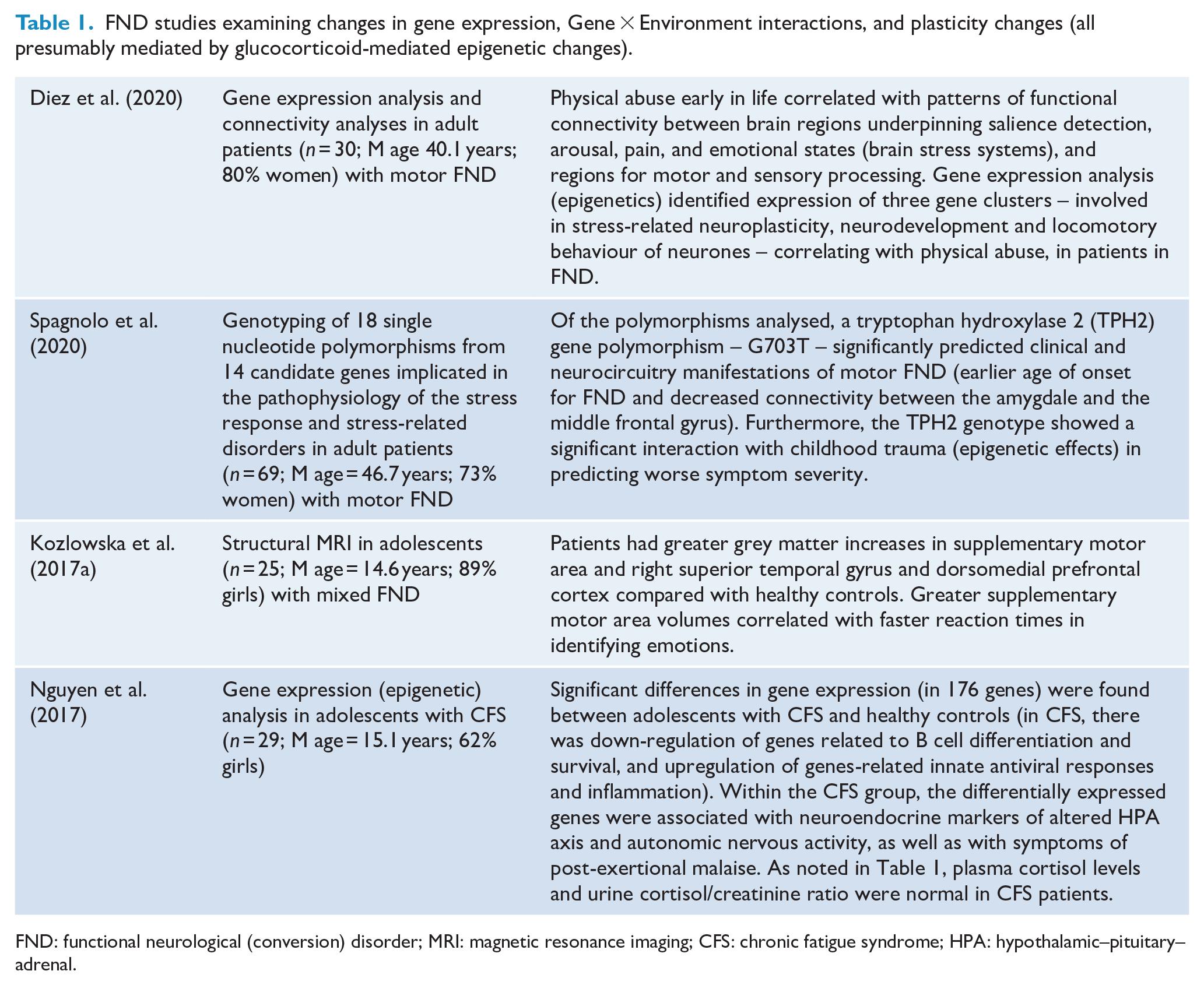
FND: functional neurological (conversion) disorder; MRI: magnetic resonance imaging; CFS: chronic fatigue syndrome; HPA: hypothalamic–pituitary–adrenal.
In the broader literature, three patterns of HPA dysregulation have been identified in patients with functional somatic symptoms (see Table 2) (Agorastos et al., 2019; Tsigos et al., 2015):
Summary of studies looking at HPA function in children with functional somatic symptoms and adult patients with FND (no child studies available).
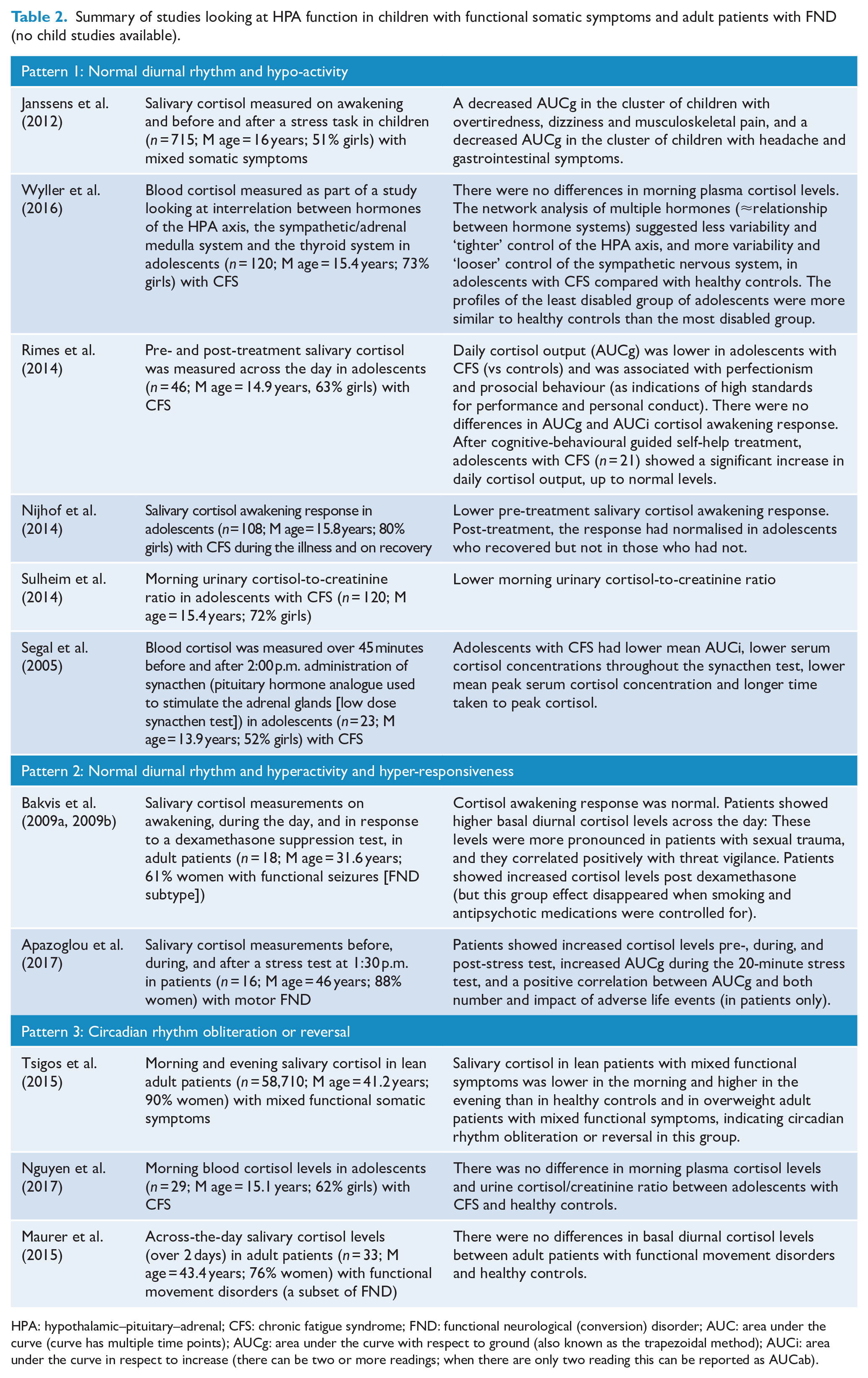
HPA: hypothalamic–pituitary–adrenal; CFS: chronic fatigue syndrome; FND: functional neurological (conversion) disorder; AUC: area under the curve (curve has multiple time points); AUCg: area under the curve with respect to ground (also known as the trapezoidal method); AUCi: area under the curve in respect to increase (there can be two or more readings; when there are only two reading this can be reported as AUCab).
Pattern 1 is characterised by a normal diurnal rhythm and hypo-activity: lower cortisol levels, attenuated diurnal variation of cortisol, enhanced negative feedback within the HPA axis loop, changed glucocorticoid receptor sensitivity to cortisol and blunted HPA axis responsiveness.
Pattern 2 is characterised by a normal diurnal rhythm and hyperactivity and hyper-responsiveness: higher cortisol levels, enhanced cortisol awakening response (CAR), increased adrenocorticotropic hormone, insensitive negative glucocorticoid feedback of the HPA axis loop and increased cortisol responses to psychosocial stress or endocrine challenges.
Pattern 3 involves circadian rhythm obliteration or reversal.
The autonomic nervous system – another component of the stress system – also appears to play a crucial role both in the biological stress response and in the manifestation and maintenance of functional somatic symptoms and disease states (see Online Supplement 6.1 in Kozlowska et al., 2020). Studies using cardiac markers derived from the electroencephalogram indicate that children with FND and children with other functional somatic symptoms more broadly show a pattern of sustained autonomic arousal characterised by a shift in parasympathetic–sympathetic balance. This shift in balance involves withdrawal of the restorative parasympathetic activity ± sympathetic activation (see Table 3).
Activation of autonomous nervous system.

Measurements of salivary cortisol and α-amylase offer non-invasive means of looking, respectively, at HPA axis and sympathetic system function on awakening. Salivary cortisol reflects unbound serum cortisol and therefore HPA axis function (Kirschbaum and Hellhammer, 1994). In contrast, α-amylase – an enzyme involved in digesting carbohydrates and modulated by the autonomic system – is a proxy measure, not a direct measure, of sympathetic arousal (Ali and Nater, 2020; Nater and Rohleder, 2009).
Cortisol levels reach peak values within approximately 30−45 minutes of wakening (Clow et al., 2010) and then gradually decline over the day and night, reaching the lowest levels in the early morning hours. The stability of the CAR is well established in healthy children and adolescents (Baumler et al., 2013; Platje et al., 2013). Exposure to acute stress is generally associated with an increase in CAR, and chronic, prolonged, uncontrollable, or traumatic stress is associated with an attenuation of CAR (Filaire et al., 2013).
In adults, salivary α-amylase levels drop in the first 30 minutes after awakening and then rise into the evening, dipping again during the night (Ali and Nater, 2020; Nater et al., 2007). In adolescents, the direction of the α-amylase awakening response (AAR) is more variable (Adam et al., 2011; Filaire et al., 2013).
In individuals who have experienced chronic stress, either short term (as in athletes during periods of intensive training), or long term (as in sexual abuse), the relationship between the HPA axis and the autonomic nervous system can change over time, leading to a loss of healthy synchrony between the systems (Baumeister et al., 2016; Bertone-Johnson et al., 2012; Filaire et al., 2013; Trickett et al., 2010).
The present study utilised a Salivette sampling device to collect saliva at two time points – upon awakening and 30 minutes after awakening – in children with FND and healthy controls. The primary goal was to ascertain whether the HPA axis was dysregulated in children with FND. The secondary goal was to ascertain whether sympathetic system activation on awakening was a feature of autonomic system dysregulation in children with FND.
Methods
Forty-one children admitted for treatment of FND to the inpatient Mind-Body Program during the period October 2018–October 2020 agreed to participate in the current study. All children had undergone a comprehensive neurology assessment and had been given a Diagnostic and Statistical Manual of Mental Disorders, 5th edition (DSM-5) diagnosis of FND by a paediatric neurologist (American Psychiatric Association, 2013). All had participated in a biopsychosocial assessment with the mind-body team: a structured interview with the child and family documenting the child’s developmental history (including adverse childhood experiences [ACEs]), history of the presenting symptoms (including comorbid non-specific symptoms), and functional disability rating on the Global Assessment of Functioning (GAF) scale (see Table 4).
Summary of the measures used in the study.

DSM-IV-TR: Diagnostic and statistical manual of mental disorders, 4th edition, text revision.
On self-report, the children completed the Depression Anxiety and Stress Scales (DASS-21) and the Early Life Stress Questionnaire (ELSQ) (see Table 4).
Thirty-seven healthy controls were recruited from the same age bracket and geographical catchment area. Control participants were screened for the absence of the following: mental health disorders, history of head injury, family history of mental health disorders and chronic health concerns. All controls completed the DASS-21 and ELSQ and were rated with the GAF.
The study was approved by the Sydney Children’s Hospital Network Ethics Committee. Participants and their legal guardians provided written informed consent.
Saliva acquisition
Cortisol and α-amylase were measured in saliva collected using the Salivette sampling device (Salimetrics, Carlsbad, CA, USA). The children with FND collected the samples while admitted into the Mind-Body Program, and controls collected the samples at home. All children collected samples immediately on waking up – according to their normal schedule (including attendance at hospital or census school) – and were expected to stay in bed, reading or engaging in some quiet activity, until they took the second sample half an hour later. They were also asked to refrain from going to the toilet, eating any breakfast, or brushing their teeth until both samples had been taken. Saliva samples were stored at –40°C before assaying. Samples collected during the hospital stay were placed in storage immediately on collection, and those collected at home were placed in a home refrigerator and dropped to the hospital on the day of collection or the day after.
Unusable samples excluded from the analysis
Fifteen participants (9 patients and 6 controls) were excluded from the analysis because one or both of their saliva samples were insufficient to run the analysis or because the sample had been mistakenly compromised (e.g. by addition of acid by pathology staff).
Salivary cortisol analysis
Salivary cortisol levels were determined with the Salimetrics Salivary Cortisol Enzyme Immunoassay Kit (Salimetrics, Carlsbad, CA, USA). The assay is an enzyme-linked competitive immunoassay, whereby cortisol in patient samples competes with cortisol conjugated to horseradish peroxidase, which catalyses a colour-change reaction. Enzyme activity is determined by measurement of the optical density at 450 nm and is inversely proportional to the concentration of cortisol in the samples. The assay has a coefficient of variation of 3.6% at 27.8 nmol/L and of 3.2% at 3.1 nmol/L. All analyses were performed in duplicate to minimise analytical imprecision.
CAR was calculated as the area of the Cortisol × Time curve above the baseline (AUCab) (Pruessner et al., 2003). The total amount of cortisol (AUCg) was calculated as the area of the trapezoid under the Cortisol × Time curve (Pruessner et al., 2003).
Alpha-amylase analysis
Alpha-amylase in the saliva samples was measured on the Ortho Vitros 5600 automated analyser (Ortho-Clinical Diagnostics, Rochester, NY, USA), which utilises a dry slide method. Amylase in the saliva sample reacts with a dyed substrate to form a coloured product, the concentration of which is determined by reflectance spectrophotometry. Due to the high levels of α-amylase present in saliva, samples exceeding the assay measurement range were manually diluted with manufacturer-provided special diluent prior to analysis. The assay has a coefficient of variation of 4.0% at 244 U/L.
Analogous to cortisol, AAR was calculated as the area of the α-Amylase × Time curve above the baseline (AUCab), with the total amount of α-amylase (AUCg) calculated from the area of the trapezoid under the α-Amylase × Time curve.
Analysis of self-report data
Chi-square analyses and independent t tests were used to calculate differences between the FND and control groups on categorical and continuous variables, respectively.
Other post hoc analyses
Pearson’s correlations could potentially be used to explore the relationship (with a Bonferroni-corrected p level of 0.025) of ACEs (documented on the ELSQ) and the child’s level of overall distress (documented by the total DASS score) to the pattern of HPA axis/sympathetic activity (if found).
Results
Participant characteristics: age, sex and wake-up findings
The final study groups comprised 32 children and adolescents (26 girls and 6 boys) with FND aged 11.3–16.1 years (M = 14.1, SD = 1.4, median = 14.2) and 31 healthy controls (23 girls and 8 boys, aged 8.6–17.7 years; M = 13.7, SD = 2.5, median = 14.2). The groups were well matched for sex (χ2 = 0.5, p = 0.50) and age, t(47.3) = 0.6, p = 0.53. The mean time of waking was 7:53 a.m. in the FND group and 7:34 a.m. in the healthy group, which was not significantly different, t(44.6) = 1.2, p = 0.26.
Missing data
Mandatory shutdowns during COVID-19 disrupted the analysis process, resulting in missing cortisol data for 4/32 patients with FND and 2/31 controls, and missing α-amylase data for 1/32 patients with FND and 1/31 controls.
Participant characteristics: the clinical presentations of the children with FND
Participants with FND had been ill for 2 weeks to 36 months (M = 6.6 months, median = 4 months). In clinical terms, approximately half the cohort (n = 18; 56%) had acute presentations of less than 6 months, and half (n = 14; 44%) had chronic presentations of 6 months or more. Motor symptoms were most common (n = 27; 85%), followed by sensory symptoms (n = 14; 44%) and functional seizures (n = 13; 41%) (see Table 5). Multiple FND symptoms were the norm (n = 28; 88%; range = 1–10, M = 3.8, median = 3.0). The majority of children (n = 30; 94%) also reported non-specific somatic symptoms (see Table 5). Twenty children (81%) suffered from comorbid pain (see Table 5). Twenty-six children met criteria and were treated for comorbid mental health diagnoses (see Table 5).
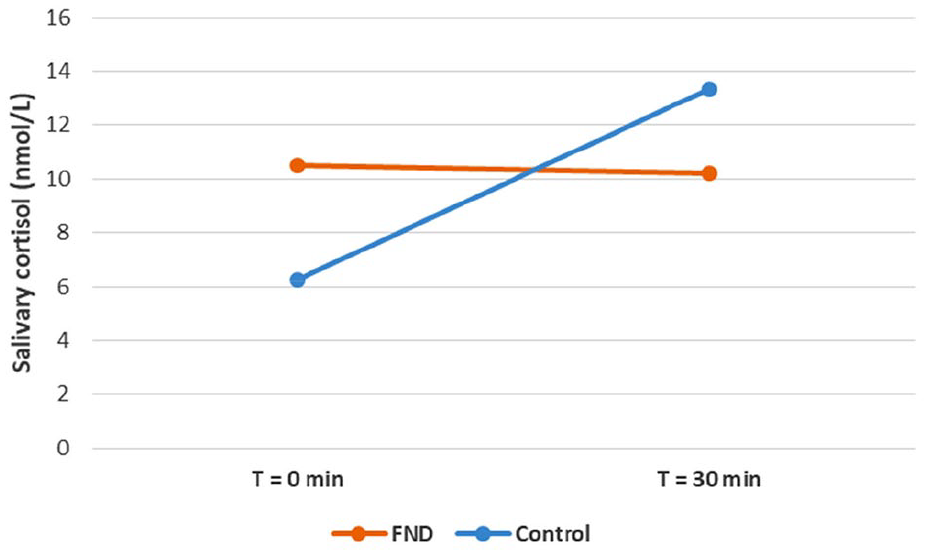
Average salivary cortisol responses in FND vs controls (whole-group analysis).
Clinical and demographic information about participants with FND from clinical assessment.
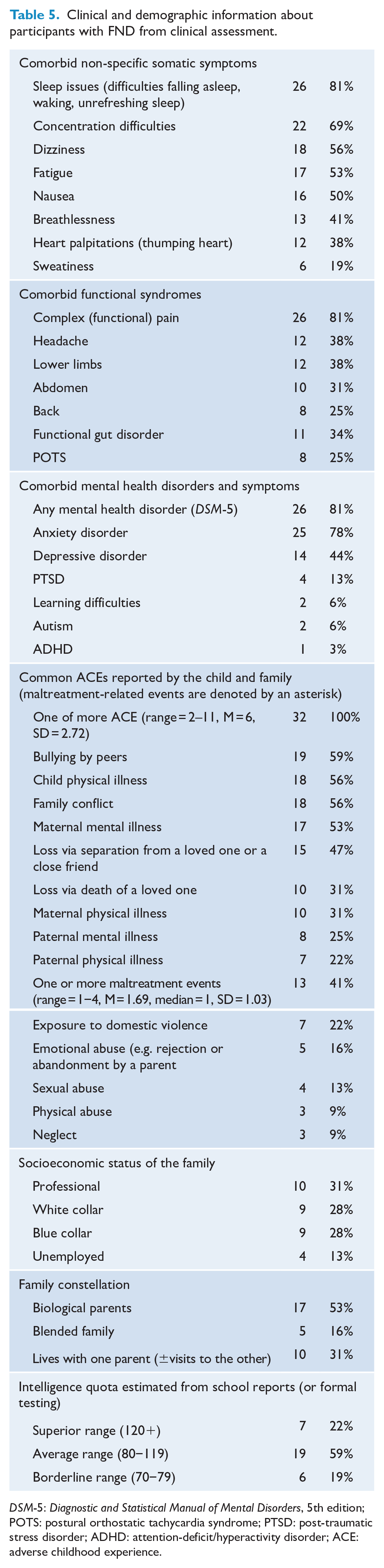
DSM-5: Diagnostic and Statistical Manual of Mental Disorders, 5th edition; POTS: postural orthostatic tachycardia syndrome; PTSD: post-traumatic stress disorder; ADHD: attention-deficit/hyperactivity disorder; ACE: adverse childhood experience.
Levels of functional impairment were high: 18 (56%) mobilised with a wheelchair, 3 (9%) mobilised with crutches and 5 (16%) were bed bound (see also GAF scores in Table 6). Relative to controls, patients with FND had significantly lower GAF scores and significantly higher scores on the DASS and ELSQ (see Table 6). Other clinical characteristics are documented in Table 5.
Comparisons between FND and healthy control groups on GAF, family constellation, DASS-21 and ELSQ.
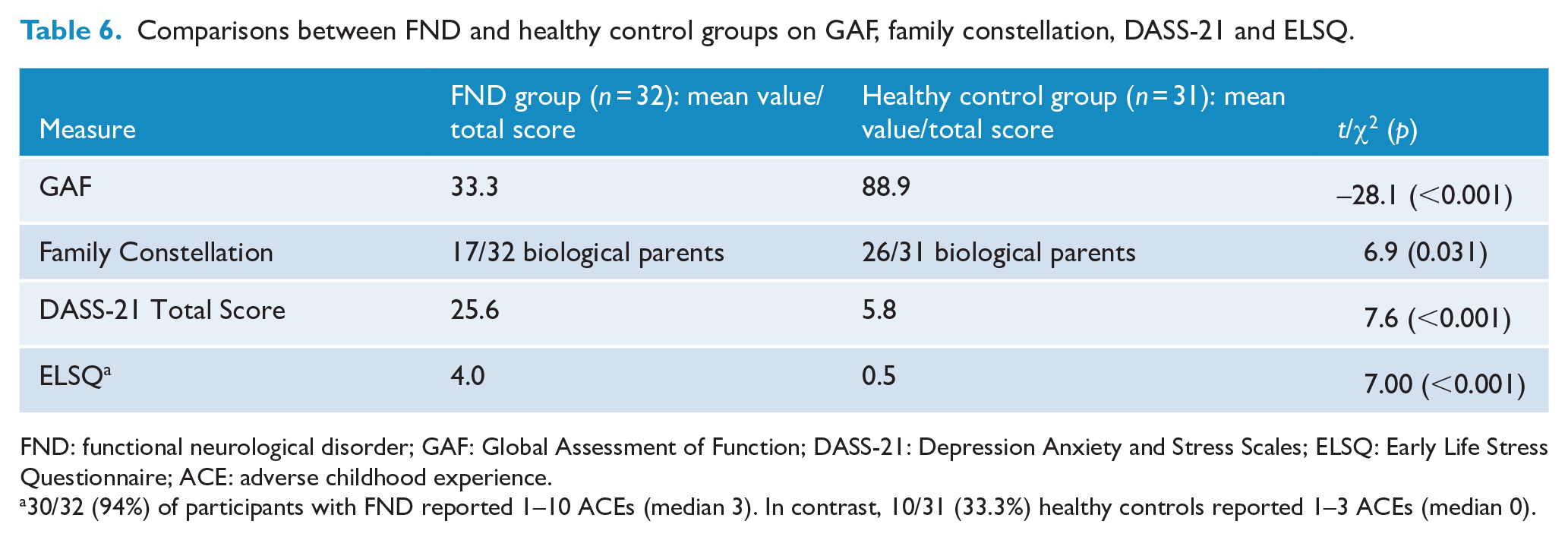
FND: functional neurological disorder; GAF: Global Assessment of Function; DASS-21: Depression Anxiety and Stress Scales; ELSQ: Early Life Stress Questionnaire; ACE: adverse childhood experience.
30/32 (94%) of participants with FND reported 1–10 ACEs (median 3). In contrast, 10/31 (33.3%) healthy controls reported 1–3 ACEs (median 0).
Cortisol findings
There were 57 paired samples suitable for cortisol analysis, comprising 28 patients with FND and 29 controls.
In the whole group analyses (all participants in the FND group and all participants in the control group), cortisol levels in the 30 minutes after awakening decreased in the FND group (Δcortisol = –0.3 nmol/L, SD = 6.1 nmol/L) and increased in the control group (Δcortisol = +7.1 nmol/L, SD = 6.0 nmol/L), t(55) = –4.634, p < 0.001 (see Table 7 and Figure 1). Unexpectedly, 12/28 FND group participants and 2/29 control participants had a 30-minute salivary cortisol level lower than the awakening cortisol level. In line with these findings, the CAR (AUCab) was negative in the FND group (−4 nmol.min/L) and positive in the healthy controls (107 nmol.min/L), t(55) = –.4.6, p < 0.001. There was no difference between the FND and control groups in total cortisol (AUCg), t(55) = 0.347, p = 0.730 (see Figure 2).
Whole group comparisons between FND and healthy control groups for cortisol data.
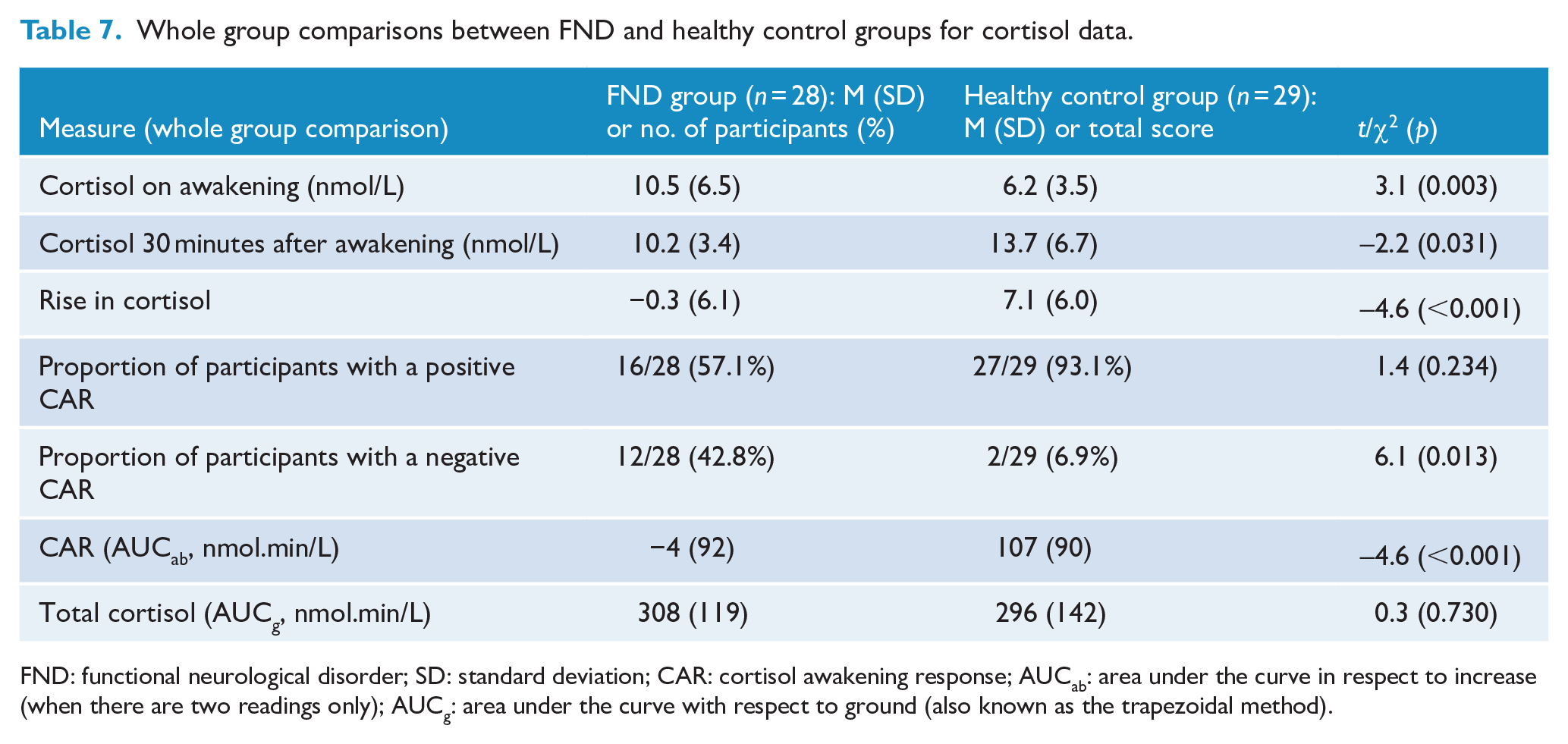
FND: functional neurological disorder; SD: standard deviation; CAR: cortisol awakening response; AUCab: area under the curve in respect to increase (when there are two readings only); AUCg: area under the curve with respect to ground (also known as the trapezoidal method).
A subanalysis was performed in the FND group and control group participants who demonstrated a positive CAR (see Table 8, Figure 3, and Figure 4). The participants in the FND group had a smaller rise in cortisol than participants in the control group, M = 3.8 vs 7.7 nmol/L, t(41) = –2.543, p = 0.015, and an attenuated CAR(AUCab), M = 57 vs 116 nmol.min/L, t(41) = –2.549, p = 0.015 (see Table 8 and Figure 3). There was no difference between the FND and control groups in total cortisol (AUCg), M = 271 vs 300 nmol.min/L, t(40.217) = –0.937, p = 0.354 (see Figure 3).
Subgroup–group comparisons – for participants in the FND group and healthy control group who showed an increase in cortisol on awakening.
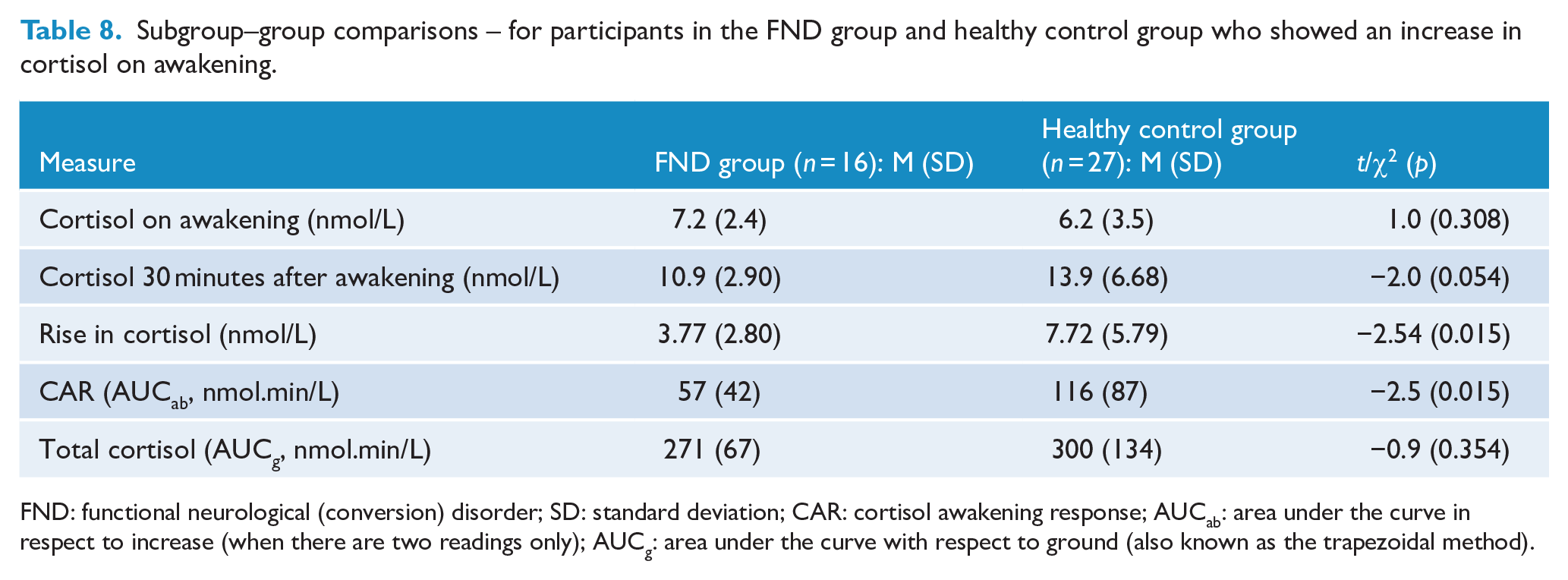
FND: functional neurological (conversion) disorder; SD: standard deviation; CAR: cortisol awakening response; AUCab: area under the curve in respect to increase (when there are two readings only); AUCg: area under the curve with respect to ground (also known as the trapezoidal method).
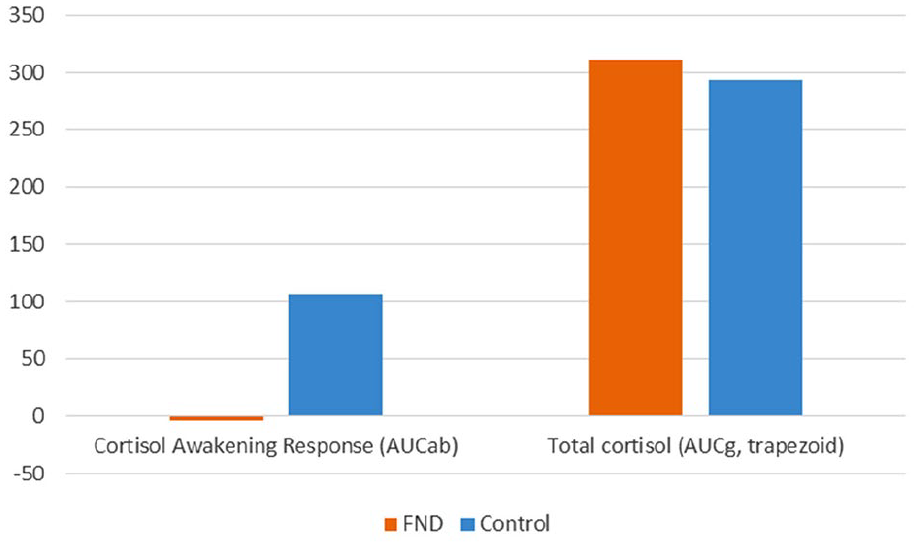
Cortisol awakening response and total cortisol in FND vs controls (whole group analysis).
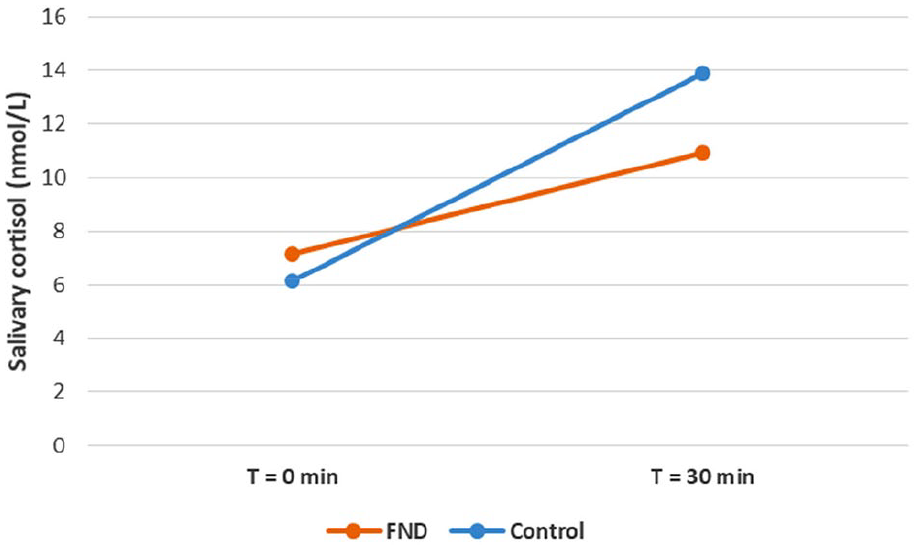
Average salivary cortisol responses in FND vs controls (subgroup analysis in participants who had a positive cortisol awakening response).
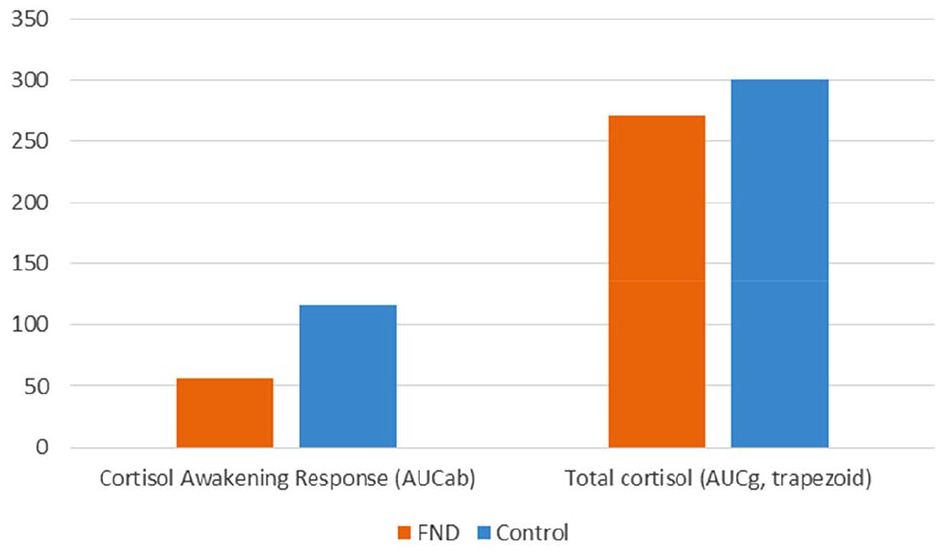
Cortisol awakening response and total cortisol in FND vs controls (subgroup analysis in participants with a positive cortisol awakening response).
Alpha amylase findings
There were n = 61 paired samples suitable for α-amylase analysis, comprising 31 patients with FND and 30 controls. Both patients and control participants showed a mixed pattern of results, with α-amylase decreasing in some participants and increasing in others (see Table 9). In this context, all analyses pertaining to amylase were conducted as whole group analyses that included all participants in the FND and controls group. There were no differences between the FND and control groups in Δα-amylase in the 30 minutes after awakening, in AAR (AUCab) and in total amylase (AUCg) (see Table 9).
Whole group comparisons between FND and healthy control groups for amylase data.
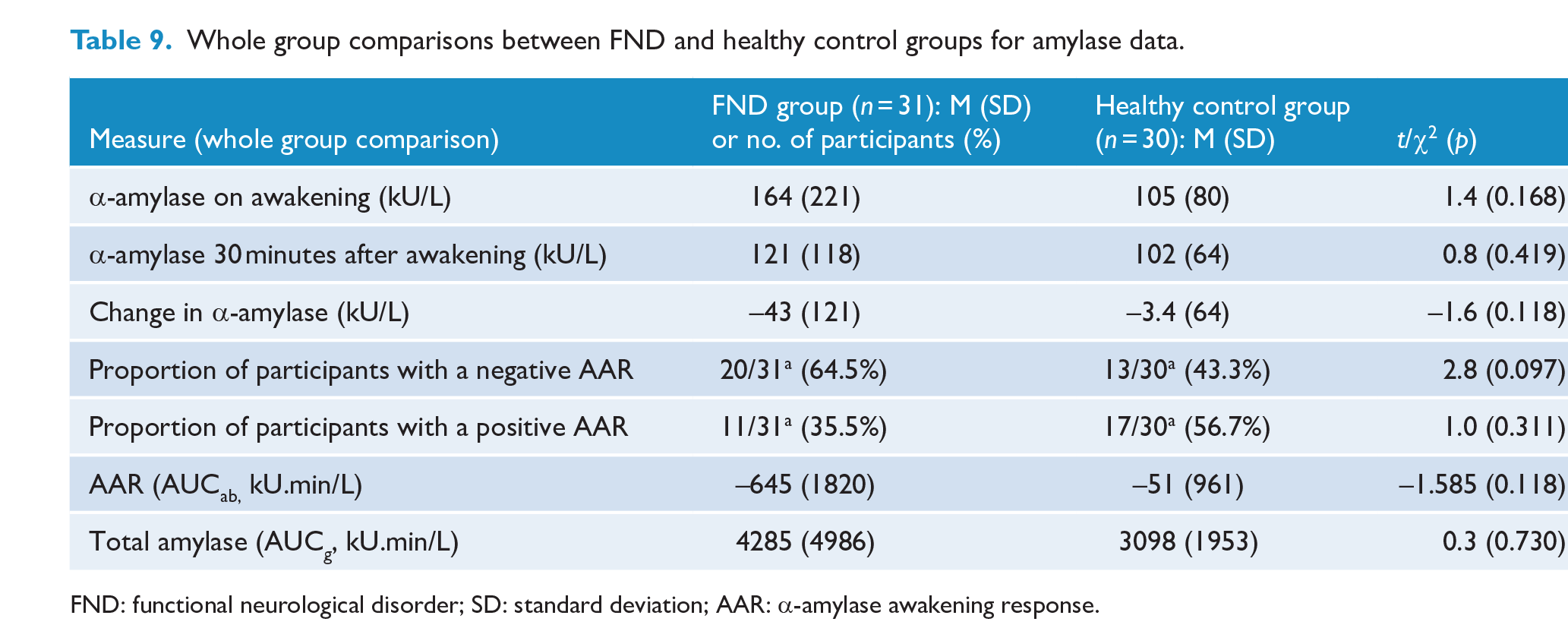
FND: functional neurological disorder; SD: standard deviation; AAR: α-amylase awakening response.
Correlations
There was a negative correlation between CAR and ACEs (total ELSQ score) in the FND group, r(58) = –0.6, p = 0.002 (the greater the number of ACEs, the smaller the rise in cortisol or decrease in cortisol level). A cross-correlation table identified key ACEs driving this correlation (see Table 10). There was no correlation between ACEs and CAR in the control group.
Pearson Correlations pertaining to ACEs that drove the correlation between CAR and ACEs.
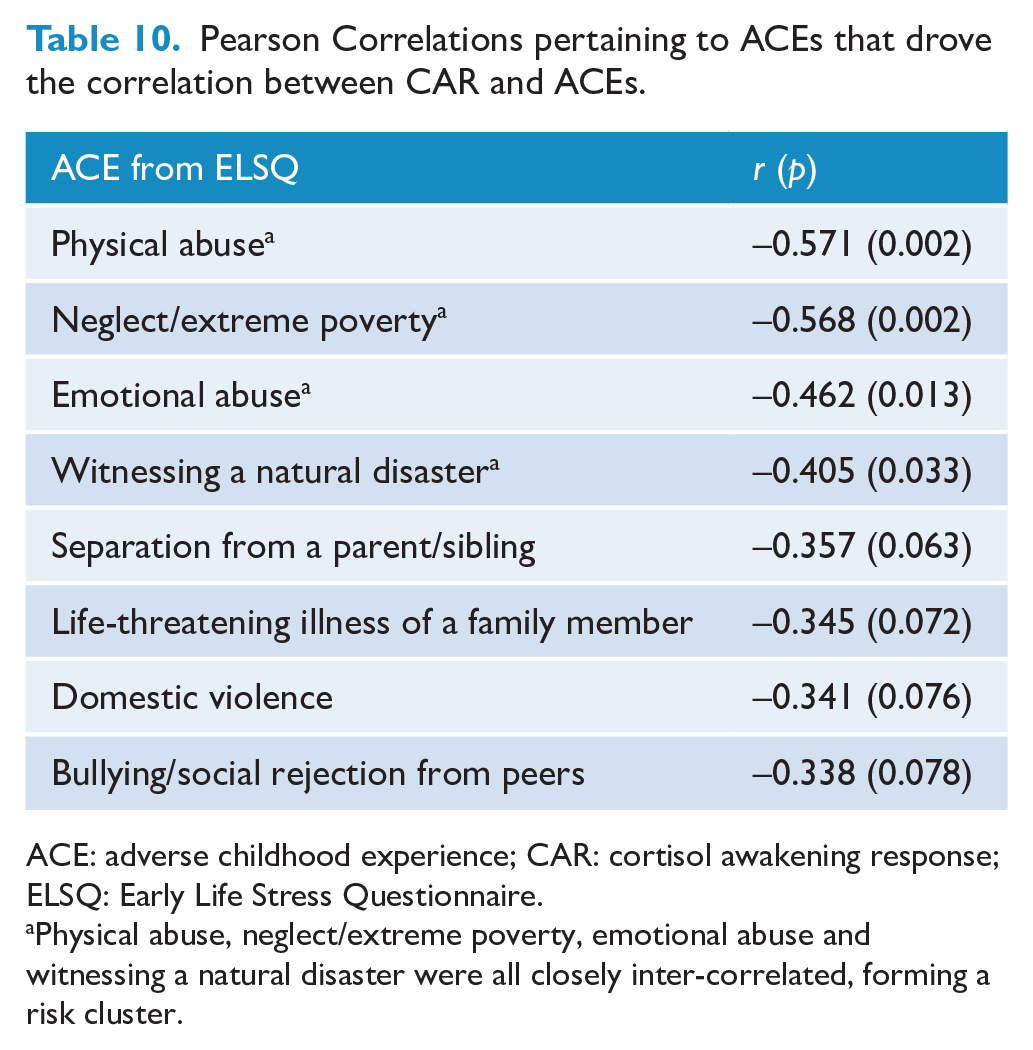
ACE: adverse childhood experience; CAR: cortisol awakening response; ELSQ: Early Life Stress Questionnaire.
Physical abuse, neglect/extreme poverty, emotional abuse and witnessing a natural disaster were all closely inter-correlated, forming a risk cluster.
There was also a negative trend-level correlation at the Bonferroni-corrected p value (p < 0.025) between CAR and subjective distress (total DASS score) in the FND group, r(58) = –0.4, p = 0.050. The greater the subjective distress, the greater the degree of HPA axis dysregulation (smaller rise in cortisol or decrease in cortisol level). A cross-correlation table showed that the depression subscale drove this negative correlation, r(54) = –0.5, p = 0.010. There was likewise a moderate negative correlation between a DSM-5 diagnosis of depression and CAR, r(54) = –0.5, p = 0.010.
Healthy controls showed the opposite pattern of findings. There was no correlation between CAR and ACEs, r(58) = 0.2, p = 0.356, and a positive correlation between CAR and subjective distress (total DASS score), r(56) = 0.4, p = 0.023. A cross-correlation table showed that the stress subscale, r(60) = 0.6, p = 0.001, and the depression subscale, r(60) = 0.513, p = 0.004, drove this correlation in controls. Within the FND group, other post hoc analyses found no correlation between CAR and any of the following: a DSM-5 diagnosis of anxiety, self-report of disrupted sleep, the acute vs chronic nature of the clinical presentation or the child’s level of function (GAF score).
Discussion
This study used saliva measurements of cortisol and α-amylase to assess the state of the HPA axis and sympathetic component of the autonomic nervous system in children with FND (vs healthy controls) at waking and 30 minutes later. Children with FND were found to have two patterns of HPA axis dysregulation. One subgroup (16/28; 57%) showed an attenuated CAR (vs healthy controls) in the 30 minutes after waking (see pattern 1 in Table 2). This is consistent with previous findings in children with mixed somatic symptoms and in children with fatigue (see pattern 1 in Table 2). The second subgroup (12/28; 43%) showed a negative CAR in the 30 minutes after waking, suggesting circadian rhythm obliteration, reversal or temporal shift (to the left), where the rise in cortisol may have occurred during the early hours of the morning rather than at waking (see pattern 3 in Table 2). Despite the attenuated/reversed CARs, the total amount of cortisol remained similar between groups. Within the FND group, CAR correlated negatively with number of ACEs and with subjective distress (total DASS score). In the control group, CAR and ACEs were not correlated, but CAR and subjective distress (total DASS score) were positively correlated; that is, healthy controls had an increased CAR in the context of higher levels of subjective distress. No differences were found between the FND and control group in any of the α-amylase analyses.
The finding that almost half of our patients with FND had a reversed CAR was unexpected. Notwithstanding, this finding does cohere with the clinical and research experience of our multidisciplinary team. As corroborated in our research studies, patients with FND who are admitted for treatment to our Mind-Body Program present in a state of high arousal and neurophysiological dysregulation (see summary of studies in chapter 4) (Kozlowska et al., 2020). For example, when these patients (vs healthy controls) attempt to utilise slow-paced breathing to down-regulate autonomic arousal, their autonomic measures show a paradoxical increase in arousal (a further decrease in heart rate variability) and not the expected increase (Chudleigh et al., 2019). Likewise, hyperventilation – with breathing rates of 30+ breaths per minute – is common, and in the subset of children with functional seizures, hyperventilation increases cortical arousal and may trigger functional seizures (Kozlowska et al., 2017b). In a study looking at the immune-inflammatory component of the stress system, we found that children with FND (vs healthy controls) had elevated high-sensitivity C-reactive protein (hsCRP), suggesting a systemic shift of balance towards an inflammatory body state.
A similar pattern of findings has been reported in the adult literature. In a large adult sample (n = 58,710) of lean adults with multiple mixed functional somatic symptoms (vs lean controls), Tsigos et al. (2015) found that salivary cortisol was lower in the morning and higher in the evening – indicating circadian rhythm obliteration or reversal, and that patients also had increased hsCRP. These levels of morning and evening salivary cortisol and of serum hsCRP were associated with the presence of functional somatic symptoms.
Taken together, these data for children and adults suggest that our findings of attenuated or reversed CAR reflect a severe degree of HPA axis dysregulation and that this dysregulation is just one small piece of a much larger puzzle – dysregulation both within and between multiple components of the stress system. This system is involved in helping the body mount a protective response in the face of stress/threat and that is also involved in the daily tasks of homeostasis (including arousal and energy regulation) – both of which are problematic for children with FND.
All the patients in our study reported one of more antecedent stressors (see Table 5) and the majority also reported a history of cumulative stress (see Table 6). Our finding of a negative correlation between CAR and ACEs in the FND group only, as well as our findings of a negative correlation between CAR and total DASS score (vs a positive correlation between CAR and total DASS score in healthy controls), coheres with the broader paediatric literature. In a study of adolescents (n = 241; 14–17 years), Starr et al. (2017) showed that ACEs moderated the influence of proximal episodic stress on CAR and depressive symptoms. In adolescents with low ACEs, episodic stress predicted an increased CAR but did not predict depression. In adolescents with high ACEs, episodic stress predicted both an attenuated CAR and depression. In a study of adolescent girls with a history of sexual abuse (n = 24; 12–17 years), Keeshin et al. (2014) found an attenuated CAR, which was more pronounced in girls with high ACEs prior to the sexual abuse and in those with comorbid post-traumatic stress disorder (PTSD) (high levels of intrusive symptoms and hyperarousal). Taken together, these findings suggest that ACEs moderate the association between current/recent stress, CAR and depressive (or PTSD) symptoms.
While DASS subscales are not validated in children and adolescents (Patrick et al., 2010) (see Table 4), it was interesting to see that the depression subscale (as well as a DSM-5 diagnosis of depression) appeared to contribute – but did not explain – the attenuated/reversed CAR. It was also interesting to see that the stress subscale and depression subscale contributed to subjective distress in controls but were associated with a normal, healthy increase in CAR, rather than with a decrease as seen in the FND group. In essence, controls, when they were stressed, were able to mount a robust HPA response (increased CAR), whereas patients with FND were not.
It is also helpful to consider our findings in the context of current advances in neuroscience, molecular biology and genomic/epigenetic research. An emerging body of work suggests that recurrent activation of the stress system in relation to early life stress – particularly when the stress is cumulative or exceeds the child’s adaptive capacity – may over- or under-sensitise neuroendocrine responses to stress, leading to a dysregulated stress system over the life span (Agorastos et al., 2019; McEwen et al., 2015; Zannas and Chrousos, 2017) and to an increased risk of both physical and mental health disorders later in life (Felitti et al., 1998). Importantly, children vary in their sensitivity to early life stress and this variation is influenced by interactions among genetic/epigenetic factors, family, community and school environments and developmental timing (Boyce et al., 2021). To make things even more complex, the pattern of stress system dysregulation can shift across development so that the patterns of dysregulation early and later in life can differ (Baumeister et al., 2016; Trickett et al., 2010). Along these lines, we highlight that while the current study showed an attenuated or obliterated/reversed CAR in children with FND, studies with adults with FND show a normal CAR (Bakvis et al., 2009a; Maurer et al., 2015). In addition, studies with adults also show higher baseline cortisol levels in the morning and afternoon (Apazoglou et al., 2017; Bakvis et al., 2009a) and positive correlations between afternoon total cortisol (AUCg) and number of adverse life events (Apazoglou et al., 2017) and also between increased basal diurnal cortisol levels and a history of sexual abuse (Bakvis et al., 2009a).
An important theme emerging from these FND studies – including our own – is a patterned relationship between HPA axis dysregulation and adverse life experiences. At the molecular level, the effects of HPA axis activation and glucocorticoid signalling are largely mediated by the glucocorticoid receptor that drives the genomic actions of glucocorticoids, mediating the biological embedding of experience via plasticity changes in every tissue of the body (including the brain) (McEwen et al., 2015; Zannas and Chrousos, 2017). Because stress activates the HPA axis, stress exposure influences cortisol secretion and signalling, and can result in lasting epigenetic and plasticity modifications that persist throughout life and even across generations (Boyce et al., 2021; Zannas and Chrousos, 2017). Of particular interest in the field of FND is whether adverse experiences play a pivotal role in activating a biological cascade of changes that lead to the neural network dysregulation – the patterns of aberrant changes in functional connectivity – that underpins FND. Looking at this very question, in a recent study with 30 adult patients with motor FND, Diez et al. (2020) found that experience-dependent changes in gene expression were coupled with aberrant changes in functional connectivity – between neural networks underpinning motor and sensory function and those underpinning attention, salience detection, arousal and emotional states. Of note, increased activation in brain regions mediating salience detection, arousal, and emotional states, as well as increased connectivity of these regions with motor-processing and sensory-processing regions, reflects the recurring pattern of neural network changes seen in studies of FND. Figure 5 outlines the interconnections between adverse life experiences and FND.
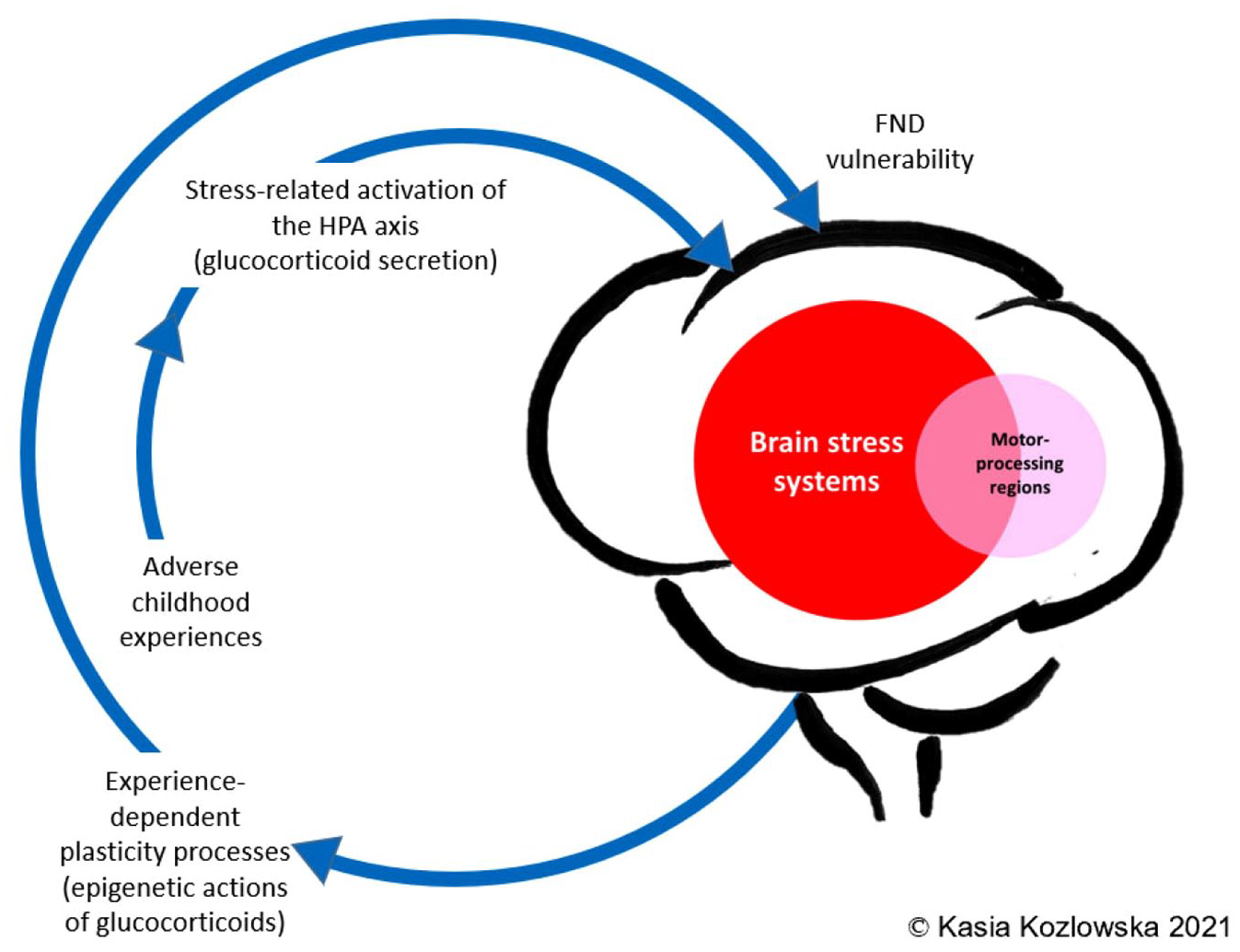
Visual representation linking adverse life experiences, stress system activation, and epigenetic/plasticity processes that increase vulnerability for FND.
Of particular interest with regard to aberrant neural network function is the growing understanding that ACEs interact with inflammation – the immune-inflammatory component of the stress system – to alter brain maturation and to increase vulnerability for a broad range of symptoms and psychiatric disorders (Andersen, 2022). In the field of FND, ACE-related neuroinflammatory changes are hypothesised to contribute to neural network dysregulation (Radmanesh et al., 2020; Sharma and Szaflarski, 2021). It is likely that the role of neuroinflammation in FND will be an important topic of research in the decades to come.
Another important issue raised by this study concerns the high rate of comorbid non-specific functional symptoms in children with FND: difficulties falling asleep, waking at night and unrefreshing sleep, concentration difficulties, dizziness, fatigue, nausea, breathlessness and so on (see Table 5). In the clinical setting, paediatricians find such symptoms are difficult to explain because they find little wrong on standard clinical measures. The current finding of HPA axis dysregulation, taken together with previous findings of autonomic system activation, immune-inflammatory system activation, and increased cortical activation/arousal (see summary of studies in chapter 4) (Kozlowska et al., 2020), suggests that non-specific functional symptoms may reflect activation/dysregulation of stress system components coupled with a loss of synchrony between components (including loss of circadian rhythm).
The concept of stress system dysregulation in children with FND has important implications for clinical practice and for the conceptualisation and treatment of FND and other functional disorders (Kozlowska et al., 2020). The stress system perspective is concerned with the continuous flow of homeostatic processes and rhythms, and with the quality of the relationship between these processes and rhythms. A healthy biological system is one whose subsystems function in a synchronised way, enabling and maintaining the efficient utilisation of energy, the capacity to respond to environmental demands and the processes of tissue regeneration and repair (McCraty and Childre, 2010). An unhealthy biological system is associated with a loss of normal flows, rhythms and patterns of connection between systems. In consequence, regulation strategies – both bottom-up and top-down Kozlowska et al., 2020; Myers et al., 2021; Velani and Gledhill, 2021) – that facilitate regulation within the stress system should be considered be a core element of biopsychosocial interventions for children with FND. These strategies aim to facilitate healthy sleep, down-regulate the autonomic system and down-regulate inflammatory processes (e.g. via regular exercise; Scheffer and Latini, 2020), with an overarching aim of reinstating normal circadian rhythms, healthy synchrony between stress system components (including the HPA axis) and neuroprotective mechanisms. Regulation within these systems may provide the necessary backdrop in which dysregulated neural networks – the aberrant connectivity changes that underpin FND – can more easily revert back to normal function. Interestingly, the process of integrating such interventions into paediatric (and adult) treatment programmes has already begun (Kozlowska et al., 2020; Myers et al., 2021; Sawchuk et al., 2020a, 2020b; Vassilopoulos et al., 2022; Velani and Gledhill, 2021).
Finally, in paediatric practice, children with FND (and their families) commonly report a broad range of adverse experiences that are part of the child’s developmental story. In the current study, the key adverse events driving the negative correlation between CAR and ACEs included a broad range of factors (see Table 10). This range of ACEs is broader than those typically discussed in the adult literature, where maltreatment events – in particular, physical, sexual and emotional abuse – are the predominant focus of attention. In the current Australian context, witnessing a natural disaster came up high on the list of ACEs. The importance of, and effect from, cumulative natural disasters associated with climate change (Intergovernmental Panel on Climate Change [IPCC], 2021) – severe bushfires, drought, flooding, and the COVID-19 pandemic – cannot be underestimated. For example, on the eastern coast of Australia, where the current study took place, approximately 80% of the population was affected by the 2019−2020 bushfires in some way (Nguyen et al., 2021). COVID-19, with its mandatory lockdowns, has led worldwide to an increase in domestic violence. Along the same lines, in the current study, physical abuse, neglect/extreme poverty, emotional abuse and witnessing a natural disaster were all inter-correlated, forming a risk cluster. As climate change continues, and as natural disasters become more frequent and severe, we can expect far-ranging psychosocial consequences for communities, families and individuals, along with an increase in stress-related health problems, both physical and psychological (Hickman et al., 2021). For example, in the context of the current COVID-19 pandemic, our tertiary care children’s hospital, as well as other clinical services around the world (Pringsheim et al., 2021), has witnessed a sharp increase in adolescent girls presenting with functional tics (Han et al., 2022).
Limitations and conclusion
The current study provides information about cortisol and amylase awaking responses in children with FND admitted for treatment to an inpatient Mind-Body Program. The study has a number of limitations. First, because the regulation of CAR is relatively distinct from cortisol secretion across the rest of the day (Clow et al., 2010), the current study is insufficient to provide information about the cortisol secretion curve across the 24-hour circadian cycle or about the robustness of cortisol secretion in response to stress (e.g. as assessed via the Trier Social Stress Test). Second, amylase is a proxy measure of sympathetic arousal and its direction of change appears to vary between different cohorts of adolescents (Doane and Van Lenten, 2014; Filaire et al., 2013). In future studies, it may potentially be more useful to obtain electrocardiogram data from a portable device. Third, it would have been helpful to collect soluble urokinase plasminogen activator receptor (suPAR) as a potentially better biomarker of stress-related inflammatory burden (Rasmussen et al., 2019). Finally, the children in this study presented with substantial functional impairment and were therefore offered a place in the Mind-Body Program, an inpatient intervention. Presumably, children with lesser degrees of impairment and those who are treated in community settings will show a lesser degree of HPA axis dysregulation.
Footnotes
Acknowledgements
We would like to thank all participants who took part in this study. We would like to acknowledge the Department of Clinical Biochemistry and Pharmacokinetics Laboratory at The Children’s Hospital at Westmead for the use of laboratory instrumentation and storage space, and we thank Dr. Christa Nath for her guidance and support. We also thank our registrars from Psychological Medicine – Yogesh Khachane, Owen Wraight, Kelvin Chan, Rachel Kour, Kai Chua, Seung Yeon Lee, Nadia Rajabalee and Yu-Na Kim – who managed saliva sample collection during their terms.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: We are very grateful to the family – who prefer to remain anonymous – whose generous donation enabled us to purchase the Salivette sampling device from Salimetrics, Carlsbad, CA, USA – an essential instrument for conducting the current study.
