Abstract
Background:
Depression is a severe, chronic, and recurring mental health disorder, which prevalence and morbimortality have increased in recent years. Several theories are proposed to elucidate the mechanisms of depression, such as the involvement of inflammation and the release of cytokines. Alternative treatments have been developed to improve outcomes of the commonly used drugs, and the use of Curcuma longa stands out. Its primary compound is named curcumin that exhibits antioxidant and anti-inflammatory effects.
Aims:
Several studies have shown that curcumin may play antidepressant actions and, therefore, this study aimed to perform a systematic review of the antidepressant effects of curcumin to evaluate the impact of this compound in the treatment of this condition.
Methods:
This systematic review has included studies available in MEDLINE–PubMed, EMBASE, and Cochrane databases, and the final selection included 10 randomized clinical trials.
Conclusion:
Curcumin improves depressant and anxiety behavior in humans. It can increase monoamines and brain-derived neurotrophic factor levels and may inhibit the production of pro-inflammatory cytokines and neuronal apoptosis in the brain. Systemically, curcumin enhanced insulin sensitivity, reduced cortisol levels, and reversed metabolic abnormalities. Studies with larger samples and standardized dose and formulation are required to demonstrate the benefits of curcumin in depression treatment since there are many variations in this compound’s use.
Introduction
Depression is a severe chronic and recurring mental health disorder prevalent worldwide. According to the World Health Organization, this condition tends to be, in 2020, the second leading morbidity in public health, second only to cardiovascular disease. It also contributes to the increased risk of morbimortality due to various causes compared to non-depressive individuals (Ali et al., 2020; Fusar-Poli et al., 2020; Lee et al., 2020; Ng et al., 2017).
Several theories are proposed to elucidate the pathophysiological architecture of depression. Monoamines’ hypothesis is based on reducing serotonin and norepinephrine levels and the theory of hypothalamic–pituitary–adrenal (HPA) axis dysfunction that favors the increase of cortisol hormones curbing the negative feedback regulatory mechanism. Other theories postulate the involvement of inflammation and the release of cytokines, neurodegeneration, and neuroplasticity. These hypotheses strengthen the fact that depression is considered a complex condition involving multiple systems, neurotransmitters, and biochemical compounds (Battaglini et al., 2020; Wang et al., 2019).
The neurotrophic factor is known as brain-derived neurotrophic factor (BDNF) that controls neurogenesis in the brain, a process in which pluripotent stem cells can generate new neurons. This protein suffers a decrease in inflammation and HPA axis dysfunction environment and occurs in depression. BDNF is decreased in the brain of people suffering from major depressive disorder, leading to neurodegeneration (Levchuk et al., 2020; Lever-van Milligen et al., 2020; Łoniewski et al., 2021; Malhi and Mann, 2018; Talarowska, 2020; Wang et al., 2019; Yang et al., 2020).
Conventional pharmacological treatment of depression relies on these pathophysiological theories to promote beneficial effects in reducing or remitting symptoms. However, these commonly used drugs do not exert great effectiveness in some patients, as they can generate several harmful adverse effects and lead to non-adherence of the patient (Ng et al., 2019). Some conventional drug treatment has a long onset time, leading to substantial harm in patients with severe depression. Due to these circumstances, alternative and complementary natural treatments such as nutraceuticals have been widely discussed and considered (Fusar-Poli et al., 2020; Karaaslan et al., 2020; Lee et al., 2020; Tizabi et al., 2019; Wang et al., 2019; Zheng et al., 2020).
Considering the treatments with natural products and medicinal plants, Curcuma longa, an herb belonging to the Zingiberaceae family, stands out. Curcumin is the primary compound of this plant and exhibits antioxidant, anti-inflammatory, antitumor, and antimicrobial (Agame-Lagunes et al., 2020; Esmaeili et al., 2020; Faria et al., 2020; Shep et al., 2020). Several studies have shown that curcumin may play antidepressant actions (Asadi et al., 2020; Bhat et al., 2019; Mandal et al., 2020; Wang et al., 2020).
There is a need for new therapeutic approaches for depression. For this reason, this study aimed to review the antidepressant effects of curcumin to evaluate the impact of this compound in the treatment of this condition.
Methods
Focused question
This review was performed to answer the focused question: Can curcumin exert effects on the depression therapeutic approach?
Language
Only studies in English were selected.
Databases
This review has included studies in MEDLINE–PubMed (National Library of Medicine, National Institutes of Health), EMBASE, and Cochrane databases. The MeSH terms that were used were ‘Curcumin or Curcuminoids or Curcuma longa and Depression’. The use of these descriptors helped identify studies related to curcumin and its antidepressant effects. This review’s authors have followed PRISMA (Preferred Reporting Items for a Systematic Review and Meta-Analysis) guidelines (Moher et al., 2009).
Study selection
This review included studies that reported the antidepressive effects of curcumin. The inclusion criteria were randomized clinical trials (RCTs), open-label studies, and primary and interventional studies (we considered studies that included pre- and post-treatment quantifications and comparisons). The exclusion criteria were reviews, studies not in English, editorials, case reports, and poster presentations. Reviews were consulted but not included.
Eligible criteria
This review’s eligible criteria followed the PICO (Population, Intervention, Comparison, and Outcomes) format for RCT. The outcomes were a reduction in depression and anxiety scores. The studies involving patients with depression who were treated with curcumin were included. Only full articles published in the consulted databases were selected.
Data extraction
Two independent reviewers (G.A.S. and S.M.B.) performed the search for the studies to identify the RCT in the databases. The studies’ abstracts were evaluated, and only full-text articles were retrieved to support the decision-making process. Disagreements between the reviewers were evaluated and decided by two other reviewers (E.L.G. and A.C.A.).
The search period for this search included from January 2011 to January 2020, and after reading the identified articles, only 10 of them were devoted to the objective of this review.
Results
The flow diagram (Figure 1) shows the selection of the articles and the inclusion and exclusion criteria. Ten studies were selected to build this review. Among these studies, 1 was an open-label trial and 9 were RCTs. Altogether, 718 individuals were enrolled in the selected studies, 18–65 years old (258 men; 460 women). The included studies participants were Type 2 Diabetes mellitus with peripheral neuropathy, patients with anxiety and obesity, with major depressive episode, and Major Depression Disorder. Curcumin was used alone or associated with piperine or associated with demethoxycurcumin or double demethoxycurcumin. There was a study that associated curcumin with saffron. Curcumin was also associated with standard antidepressants (Fluoxetine, Escitalopram, or Venlafaxine). The doses varied from 280 g to 1500 mg of Curcumin/day or 80 mg of nano curcumin/day (Table 1). The following scales analyzed the evaluation of depression and associated symptoms: BAI (Beck Anxiety Inventory), BDI (Beck Depression Inventory), CGI-I (clinical global impression-improvement), CGI-S (clinical global impression-severity), DASS-21-items (Depression, Anxiety, Stress Scale), DSM-IV (Diagnostic and Statistical Manual of Mental Disorders-IV), HADS (Hospital Anxiety and Depression Scale) HAM-A (Hamilton Anxiety Rating Scale), HDRS-17 (Hamilton Depression Rating Scale 17-item version), IDS-SR30 (Inventory of Depressive Symptomatology self-rated version), MADRS (Montgomery-Asberg Depression Rating Scale), and STAI (Spielberger State-Trait Anxiety Inventory). The time of the treatment varied from 8 days to 16 weeks.
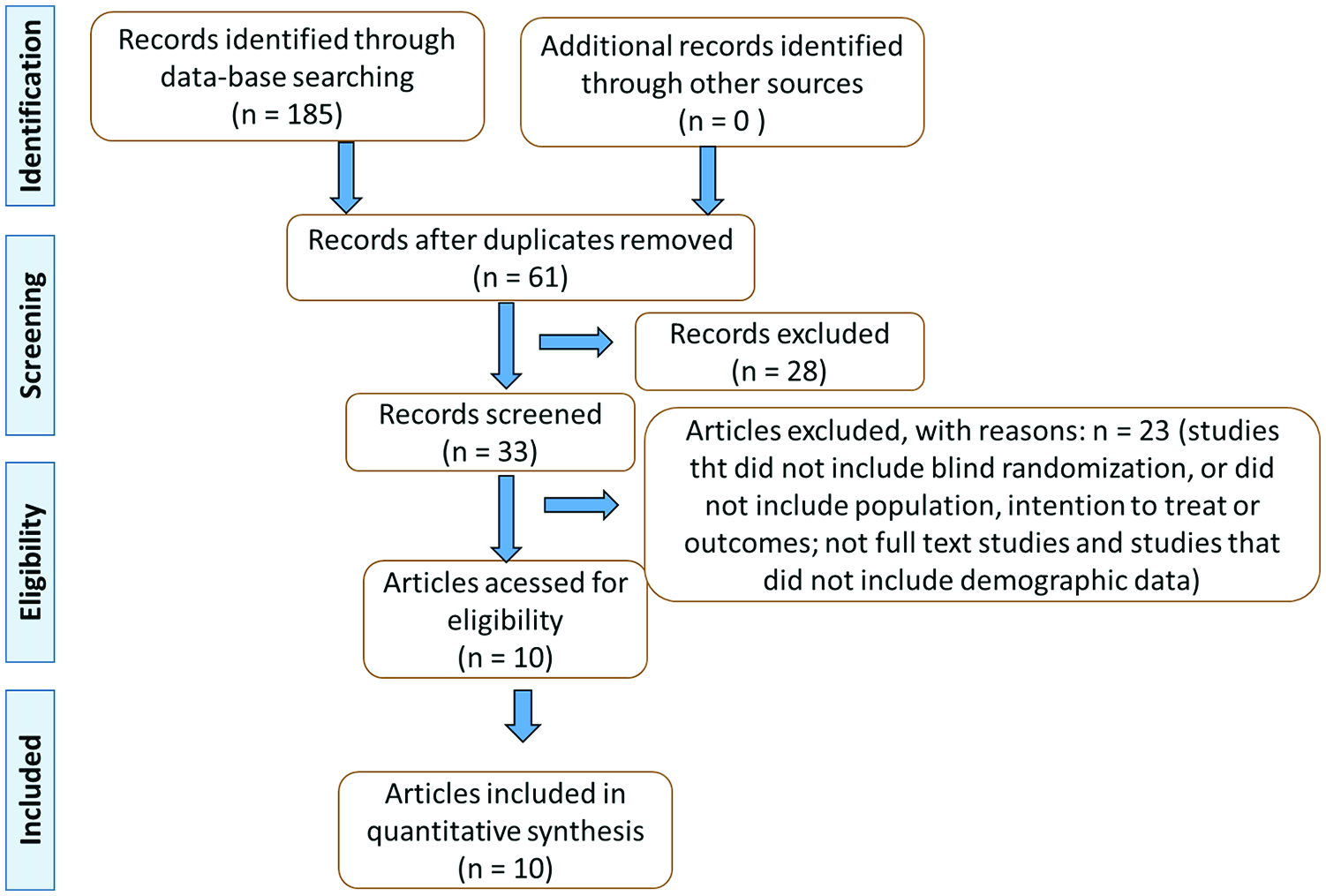
Flow diagram showing the study selection.
Randomized clinical trials that evaluated the effects of curcumin on depression.
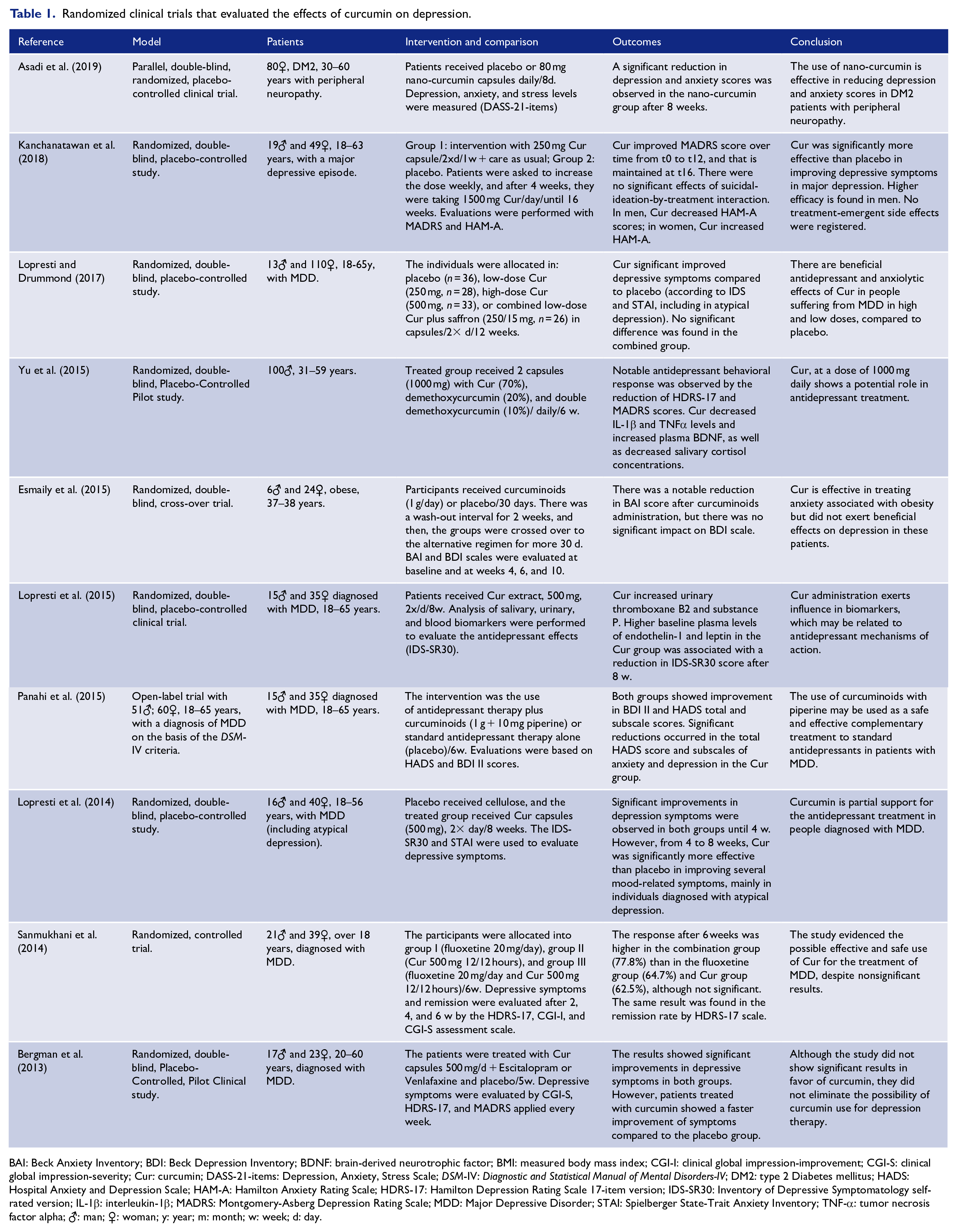
BAI: Beck Anxiety Inventory; BDI: Beck Depression Inventory; BDNF: brain-derived neurotrophic factor; BMI: measured body mass index; CGI-I: clinical global impression-improvement; CGI-S: clinical global impression-severity; Cur: curcumin; DASS-21-items: Depression, Anxiety, Stress Scale; DSM-IV: Diagnostic and Statistical Manual of Mental Disorders-IV; DM2: type 2 Diabetes mellitus; HADS: Hospital Anxiety and Depression Scale; HAM-A: Hamilton Anxiety Rating Scale; HDRS-17: Hamilton Depression Rating Scale 17-item version; IDS-SR30: Inventory of Depressive Symptomatology self-rated version; IL-1β: interleukin-1β; MADRS: Montgomery-Asberg Depression Rating Scale; MDD: Major Depressive Disorder; STAI: Spielberger State-Trait Anxiety Inventory; TNF-α: tumor necrosis factor alpha; ♂: man; ♀: woman; y: year; m: month; w: week; d: day.
Discussion
Depression: pathophysiological mechanisms
Major Depressive Disorder is currently the most common mental illness among the population and is characterized by sadness, hopelessness, loss of energy, and other symptoms. Although the prevalence of depression and its impacts have increased, this condition’s real pathogenesis has not been fully elucidated. There are five theories to explain the mechanism of depression, including the monoamine hypothesis, the HPA axis dysfunction theory, the neural plasticity hypothesis, the inflammatory theory, and the neurodegeneration hypothesis (Figures 2 and 3). The monoamine hypothesis considers that monoamines’ deficiency, especially serotonin and noradrenaline, leads to mood, behavior, motivation, and cognitive disorders. Moreover, high levels of cortisol are often found in these patients, which results from increased activity of the HPA axis and leads to the activation of nuclear factor kappa B (NF-κB). Furthermore, a decrease in brain volume is a common find, especially in the hippocampus, resulting from neuronal insults and BDNF reduction, leading to neurotoxicity, oxidative stress, and loss of neuroplasticity and neurogenesis (Fusar-Poli et al., 2020; Jia et al., 2020; Kaufmann et al., 2016; Wang et al., 2019).
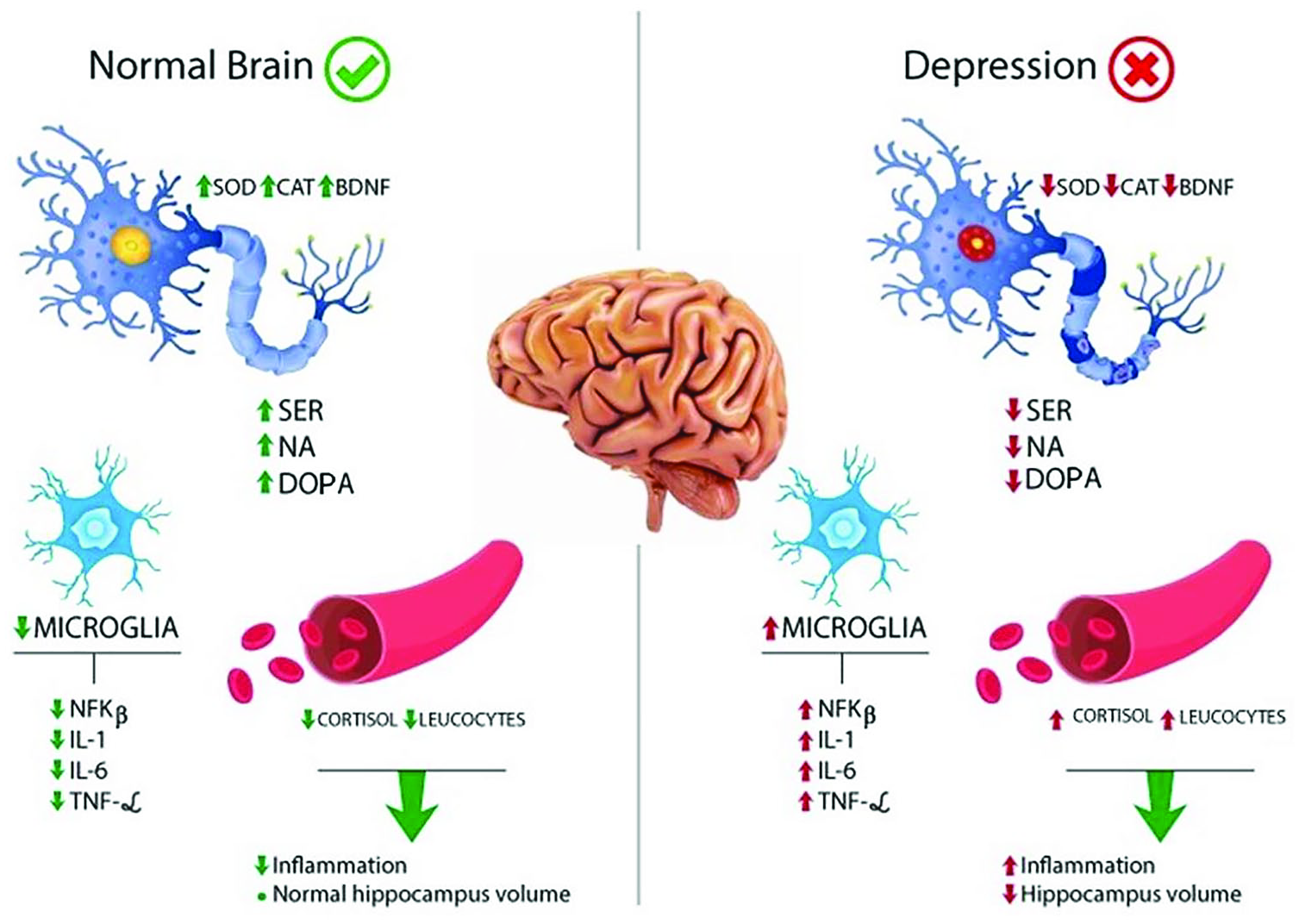
Normal physiology of neurotransmitters and other substances that affect the central nervous system, compared to a secretory, an altered pattern due to the pathophysiological architecture of depression. This comparison covers the central nervous system and serum levels.
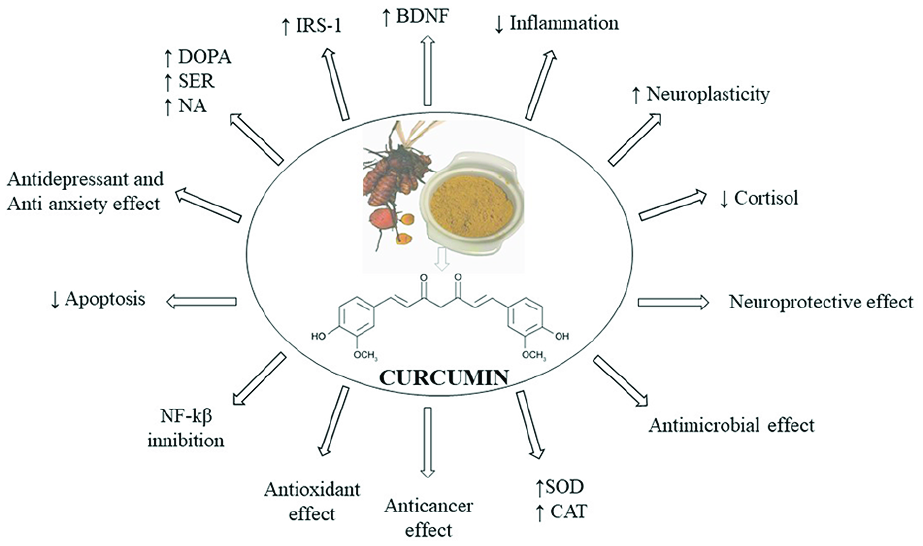
Effects of curcumin in the secretory pattern found in depression and other comorbidities.
One of the theories for the pathogenesis of psychiatric disorders includes increased pro-inflammatory cytokines such as interleukin-1 (IL-1), IL-6, and tumor necrosis factor-α (TNF-α), leading to inflammation and oxidative stress. These cytokines alter neuronal functions by influencing cell survival, metabolism, and neuroplasticity. Also, the endogenous antioxidant defense system is often compromised, which contributes to oxidative stress. Another possibility of involvement of the inflammatory system in depression is the elevated levels of leukocytes found in the patients and the activation of the immune system. These mechanisms have been used to explain the depressive behavior and brought to light the need for medicines that regulate the inflammatory process and immune system behavior (Asadi et al., 2019; Wang et al., 2019).
These theories seem to be related since drugs that increase monoamines’ availability also exert benefits in the pituitary–pituitary–adrenal axis, BDNF, neuroplasticity, and inflammation. The increase in monoamines seems to reduce the HPA axis exacerbation and decreases cortisol levels, which leads to a reduction of NF-κB activation and the release of pro-inflammatory cytokines. The reduction of inflammation restores brain neuroplasticity and neurogenesis by increasing BDNF, mainly in the hippocampus (Wang et al., 2019).
Besides, it is also relevant to mention that the emerging notion that mood disorders lie on a spectrum or a dimensional view. Indeed, major psychiatric diagnostic validity is a controversial and perennial debate (Ng et al., 2020).
Curcumin: general aspects
Curcuma longa is known for its anti-inflammatory effects by modulating NF-κB, decreasing pro-inflammatory cytokines, and showing antioxidant effects (it is related to the increase of serum superoxide dismutase and catalase activities). Beyond that, it also has anti-cancer, antimicrobial, anti-ulcer, and neuroprotective effects (Figures 3 and 4). The main component of Curcuma longa is curcumin, a polyphenolic compound which, in addition to the effects mentioned, also acts modulating monoamine levels such as serotonin and dopamine levels. With this information and considering that two of the depression mechanisms are the monoamine and inflammatory hypothesis, curcumin could be used to treat depressive symptoms. Furthermore, curcumin shows anxiolytic effects that may result from the regulation of monoamines, especially serotonin. Even though the benefits of curcumin, its use is limited by its low oral bioavailability and distribution. One possibility to solve this problem is the curcumin in nanoparticles, liposomes, micelles, and phospholipid complexes formulations (Asadi et al., 2019; Qi et al., 2020; Yohn et al., 2017).
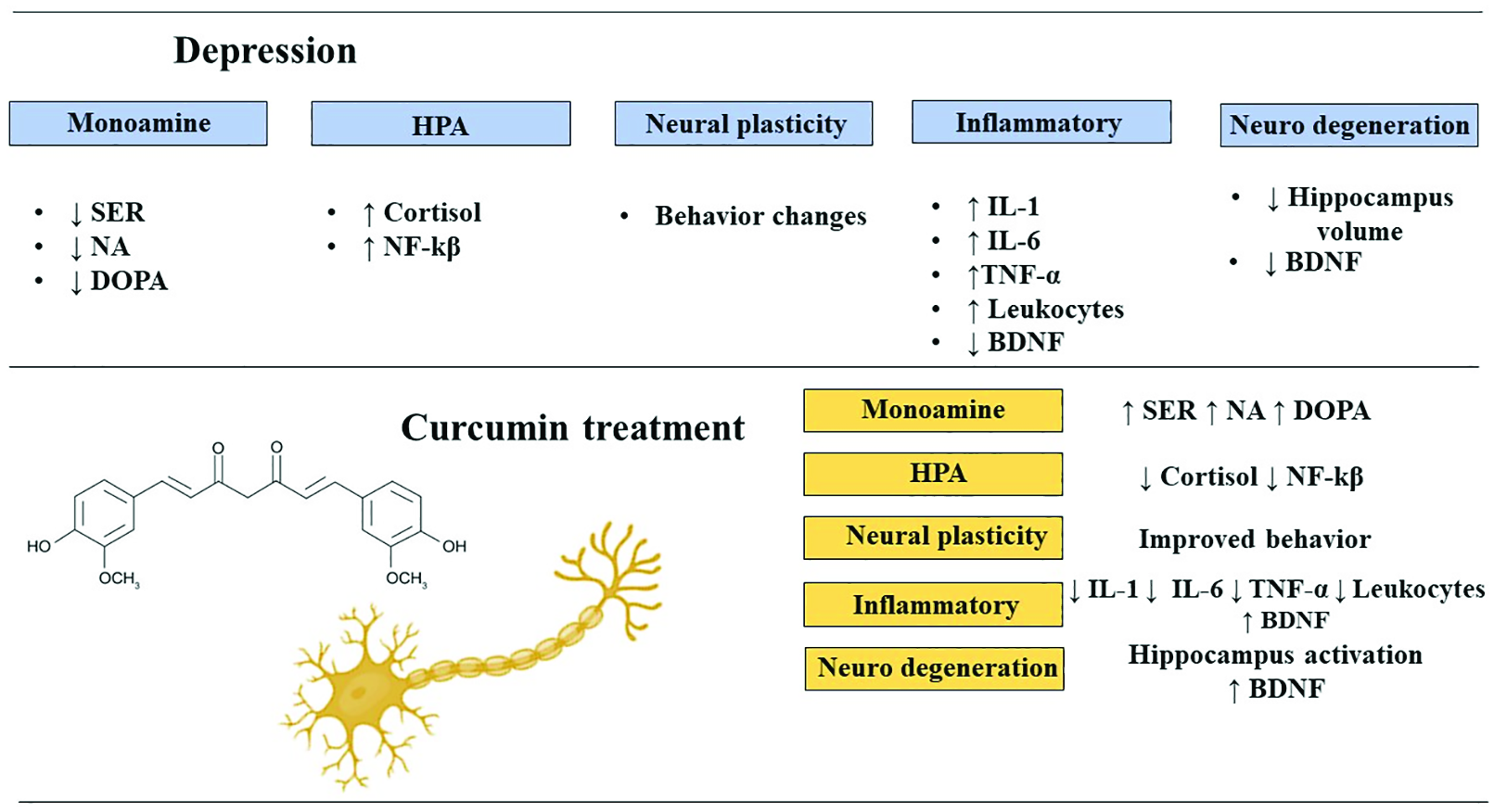
Effects of curcumin in the secretory pattern found in theories to elucidate the pathophysiological architecture of depression and other comorbidities.
Antidepressant effects of curcumin
Clinical trials
Curcumin is commonly consumed in Asia and India as a culinary ingredient in food recipes, but its benefits are also observed in different systems and have been reported in several neurological diseases (Bhat et al., 2019).
According to the RCT performed by Asadi et al. (Table 1), the effects of nano-curcumin for 8 weeks are effective in decreasing depression and anxiety scores in type 2 diabetic patients with peripheral neuropathy. Although the beneficial results are shown in this study, there is a lack of information about the formulation and characterization of nano-curcumin, which excludes the possibility of ensuring effective quality control of the product. Therewithal, the authors requested self-administration of the capsules by patients, which may cause irregularities in the intervention (Asadi et al., 2019).
The study by Kanchanatawan et al. included patients diagnosed with Major Depression (MDD) with a major depressive episode through Mini International Neuropsychiatric Interview 6.0. The curcumin group received curcumi-noids (77% curcumin, 17% demethoxycurcumin, and 6% bisdemethoxycurcumin; Table 1). Evaluations were performed using semi-structured interviews, while the research psychologist scored two measures of effectiveness, the MADRS (Primary Outcome Measure) and the Hamilton Anxiety Rating Scale (HAM-A), 4 weeks after the active phase of the study. The results of this study were positive, considering the significance of the questionnaires and the absence of significant side effects (Kanchanatawan et al., 2018).
Lopresti and Drummond performed a study where the inclusion criteria were a diagnosis of Major Depressive Disorder (MDD) according to the DSM-IV criteria and an Inventory of Depressive Symptomatology self-rated version (IDS-SR30) score ⩾18 (Table 1). The individuals were treated with low-dose curcumin extract (250 mg, n = 28), high-dose curcumin extract (500 mg, n = 33), and a low-dose curcumin extract/saffron combination (250/15 mg, n = 26), while the placebo constituted of cellulose capsules (n = 36). Inventory of Depressive Symptomatology self-rated version (IDS) and Spielberger State-Trait Anxiety Inventory (STAI) were used to evaluate the outcomes. The groups taking curcumin (low dose, high dose, and combine) demonstrated significant improvements in depressive symptoms, showing evidence of antidepressant and anxiolytic effects of curcumin in MDD. Although there was a higher response in the combined group (curcumin/saffron), it was not significant. The study included individuals with atypical depression, and it was verified that the response to combined treatment was higher in this group. For these reasons, it is necessary to consider the effects of curcumin in a larger sample (Lopresti and Drummond, 2017).
Furthermore, Yu et al. demonstrated significant effects of curcumin in treating major depression (Table 1). The results were obtained through behavioral parameters, evaluated through two scores, and laboratory parameters (a measurement of serum cytokines and levels of BDNF in plasma). However, a possible bias could be that the treatment lasted only 6 weeks (Yu et al., 2015).
Esmaily et al. evaluated, through scales, the effect of administering curcumin in patients for 30 days each intervention, separated by an interval of 2 weeks (Table 1). The results showed a reduction in anxiety symptoms after curcuminoids administration. However, there was no impact on depression symptoms. The study was conducted with patients who presented obesity as comorbidity. Besides that, the results of this study showed efficacy only in the treatment of anxiety and not for depression (Esmaily et al., 2015).
Lopresti et al. showed that the use of a daily dose of curcumin promoted antidepressant effects, especially with endothelin-1 and leptin participation. The results correspond to an 8-week treatment, which could not be enough to evaluate curcumin effects (Lopresti et al., 2015).
Panahi et al. investigated the effects of curcumin associated with piperine in patients diagnosed with Major Depression Disorder for 6 weeks. The researchers obtained a satisfactory result regarding the antidepressant effects showing curcumin’s relevance in aspects related to anxiety. The association with piperine leads to the question of whether piperine may interfere with curcumin effects (Panahi et al., 2015).
Another study by Lopresti et al. evaluated the effects of curcumin in patients diagnosed with MDD according to DSM-IV criteria and IDS-SR30 score ⩾14, including individuals with atypical depression. The use of self-reported questionnaires (IDS-SR30 and STAI) was used to evaluate depressive symptoms. Significant improvements in depressive symptoms were observed in the first 4 weeks of treatment in both groups. At the end of the protocol, improvements in depressive symptoms were significant only in the curcumin group (4–8 weeks), with greater efficacy in individuals with atypical depression. A possible bias in this study is that there are a higher number of comorbidities in the placebo group than in the curcumin group, which could interfere with the results (Lopresti et al., 2015).
Sanmukhani et al. investigated the use of curcumin in individuals with MDD according to the Diagnostic and Statistical Manual of Mental Disorders (DSMMD) criteria Fourth Edition and scored more than seven on Hamilton Depression Rating Scale (HDRS-17). The patients were separated into groups that received fluoxetine, curcumin, and fluoxetine plus curcumin for 6 weeks. The results showed a higher response and remission rates in the combination group (77.8%). Nevertheless, the study had some limitations once the participants knew about which group they belonged to. Another limitation was the lack of a placebo group (Sanmukhani et al., 2014).
The first clinical trial that analyzed the effects of curcumin in depression was performed by Bergman et al. in patients diagnosed with major depressive episodes according to the fourth edition of the DSMMD criteria. The patients received curcumin and placebo or curcumin and Escitalopram or Venlafaxine. The CGI-S, HDRS-17, and MADRS scales were applied every week to evaluate the improvement of symptoms, and after 5 weeks of treatment, the authors observed significant improvement in depressive symptoms in both groups. This was a short-term and small sample study that used only one curcumin dose (Bergman et al., 2013).
Experimental studies
The experimental study performed by Fidelis et al. demonstrated the antidepressive effects of nanocapsules of curcumin in mice submitted to neuroinflammation by β-amyloid administration. Animals received an intra-cerebroventricular injection of the amyloid-β peptide associated with neuroinflammation in Alzheimer’s Disease and depression-like behavior. Animals treated with curcumin-loaded nanocapsules (10 mg/mL) for 12 days presented a reduction in depressive behaviors. Nanocapsules of curcumin exerted an antidepressant-like effect upon neurotoxicity induced by β-amyloid peptide and oxidative stress reduction in the prefrontal cortex due to amyloid aggregates. However, free curcumin showed no significant effect on depression-like behavior and oxidative stress (Fidelis et al., 2019).
In another study, the authors administered Curcumin-loaded thermo-sensitive hydrogel by nasal in rats and obtained effectiveness in treating depression. The authors found an increase in the concentration of norepinephrine (NE), dopamine (DA), and 5-Hydroxytryptamine (5-HT) and their metabolites. They concluded that the formulation of curcumin by nasal delivery is useful in the treatment of depression, showing an acceptable form of administration (Qi et al., 2020).
Fan et al. induced depression in male Wistar rats using different models. Neuroapoptosis-associated inflammation and depression-like behaviors were induced by overexpression of IL-1β. The use of curcumin alleviated depression-like behaviors, decreased pro-inflammatory cytokine interleukin-1L-1β (IL-1β), and inhibited neuronal apoptosis within neurons of the prefrontal cortex (Fan et al., 2018b).
In rats, Abd-Rabo et al. evaluated depression-like behaviors after a month of treatment with oral curcumin (100 μg/kg), compared to Fluoxetine and Oestradiol hormone replacement. Curcumin showed good modulation of depression-like behavior, improve serotonin levels in many brain regions by upregulating tryptophan hydroxylase-2 and 5-hydroxytryptamine 1A, 2A receptor mRNA. Also, it showed negative regulation of monoamine oxidase A mRNA in the limbic system, and positive regulation of aromatase, BDNF mRNA, and regulated extracellular protein kinase 1/2 in the limbic system, which is associated with depression. The study showed improvement in depression symptoms, an increase in 5-HT and BDNF in the brain, and a similar effect of curcumin compared to fluoxetine in serotonin modulation (Abd-Rabo et al., 2019).
Zhang et al. (2019) evaluated the effect of curcumin (100 mg/kg) in male Sprague–Dawley rats and showed that this compound improved depressive-like behaviors in stressed rats and decreased mRNA expression of pro-inflammatory cytokines (IL-1β, IL-6, and TNF-α) and suppress NF-βB activation.
Madiha and Haider (2019) investigated depressive-like symptoms in rat models of neurotoxicity induced by rotenone and showed that pre- or post-treatment with curcumin reverted depressive-like behavior and improved neurotransmitters levels.
The experimental study performed by Fan et al. (2018a) showed that curcumin ameliorated the dysregulation of neuronal structural plasticity induced by IL-1β/NF-βB pathway, improving behavior and inflammatory response induced by depression.
In another study, the authors tested curcumin nanoparticles in rats with induced depression, resulting in improved performance of the animals and the levels of 5-HT. The treatment for 15 days also improved FST and monoamine levels (Mohammed et al., 2019).
Lee and Lee demonstrated the effect of curcumin on anxiety and depressive behavior in Sprague-Dawley rats compared to the use of Fluoxetine 10 mg/kg. The treatment with curcumin decreased grooming behavior and reduced the anxiety index (Lee and Lee, 2018).
The experimental study performed by Lian et al. used a turmeric analog J147 in male rats 1, 3, and 24 hours before performing the depression tests. J147 significantly increased 5-HT1A receptor expression after 1 hour at a dose of 10 mg/kg. CAMP, PKA, pCREB, and BDNF levels increased significantly 1 hour after J147 treatment at 5 and 10 mg/kg and regulated pCREB and BDNF levels lasted 3 hours after 10 mg/kg J147 (Lian et al., 2018).
Yohn et al. investigated curcumin’s ability to reverse the low effort bias induced by monoamine storage blocker tetrabenazine (TBZ) in Sprague−Dawley rats. The rats were trained on behavioral procedures and, posteriorly, were treated with (1) TBZ vehicle/10 placebo control pellets; (2) 0.75 mg/kg TBZ/10 placebo control pellets; (3) TBZ/5 control pellets; (4) Curcumin pellets (80 mg/kg), and (5) TBZ/10 Curcumin pellets (160 mg/kg; n = 9). Curcumin pellets were ingested by oral route 2 and 3 hours before the behavioral test, and TBZ was administered ip 90 minutes before testing. Oral ingestion of curcumin 2 hours before the behavioral test was ineffective, while curcumin ingested 3 hours before testing significantly attenuated TBZ-induced shifts in effort-based choice behavior. Also, curcumin administered, icv, was more effective compared to oral ingestion, reverting TBZ-induced shifts in behavior (Yohn et al., 2017).
Shen et al. evaluated the depressive-like behavior and insulin resistance induced by chronic mild stress in Sprague−Dawley rats. They showed that curcumin did not affect food intake but reduced abdominal fat and leptin levels, suggesting that curcumin’s anti-obesity effects are unrelated to appetite. Curcumin upregulated the phosphorylation of insulin receptor substrate (IRS)-1 and protein kinase B (Akt) in the liver, enhanced insulin sensitivity, and reversed the metabolic abnormalities and depressive-like behaviors mentioned above (Shen et al., 2017).
Ceremuga et al. evaluated the anxiolytic and antidepressant effects of curcumin in male Sprague–Dawley rats and compared them with flumazenil and midazolam. Curcumin did not demonstrate significant anxiolytic effects in the experimental model. The FST shows that curcumin did not prevent behavioral despair or anxiolytic effects compared to midazolam (Ceremuga et al., 2017).
Choi et al. performed an experimental study in which 6-week-old male SD rats were exposed to chronic stress such as restraint, water deprivation, food deprivation, foot shock, or isolation once a day over 18 days to induce depressive behavior. After that, rats were treated with curcumin in 50 mg/kg and 100 mg/kg p.o. once a day for 18 consecutive days. The treatment with curcumin in both dosages decreased the immobility time, and COX-2 levels compared to the stressed group and increased BDNF levels, which was more significant in the 100 mg/kg curcumin group, showing a better antidepressant effect with high doses of curcumin (Choi et al., 2017).
Table 2 summarizes the biases of the studies discussed above.
Descriptive table of the biases of the included randomized clinical trials.
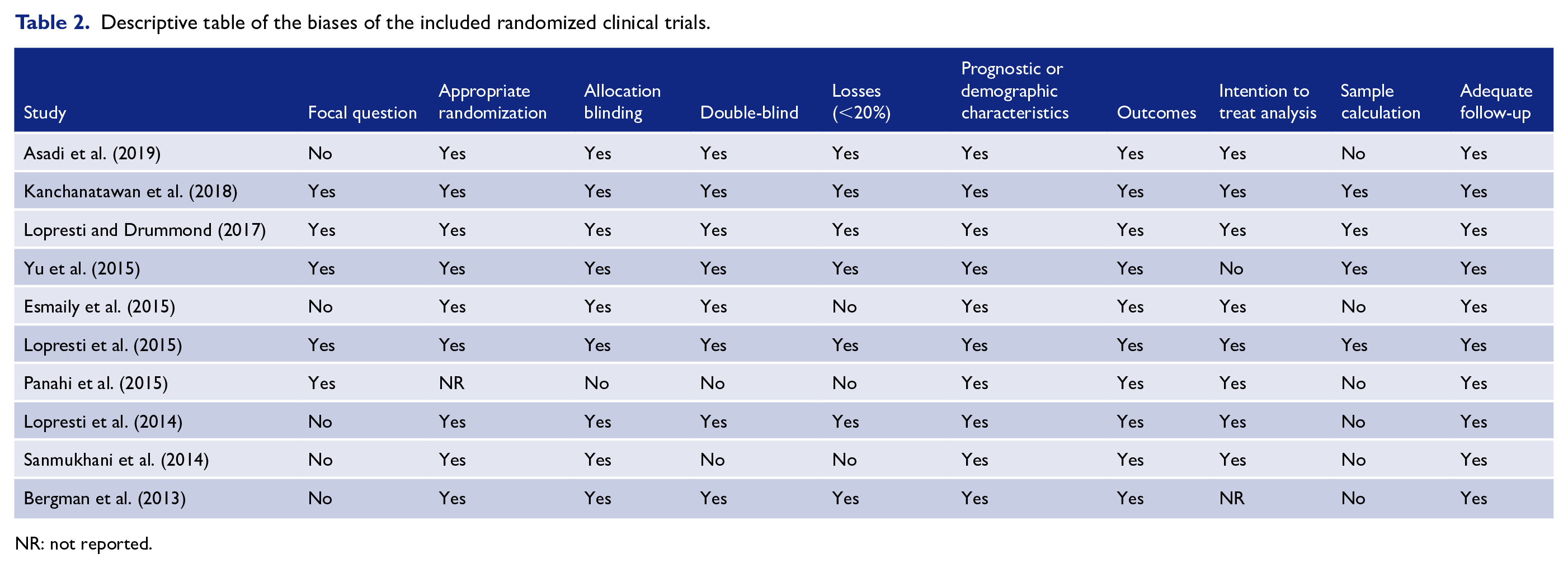
NR: not reported.
Several other animal studies showed the beneficial effects of curcumin in depression (Chang et al., 2016; He et al., 2016; Yusuf et al., 2016) and some showed that the use of piperine could potentiate the effects of this compound (Jangra et al., 2016).
Conclusion
Since inflammation is one of the mechanisms involved in the pathophysiology of depression, curcumin contributes to the prevention and treatment of this condition by decreasing pro-inflammatory cytokines such as IL-1, IL-6, and TNF-α and increasing monoamines and BDNF levels in the brain, mainly in the hippocampus and prefrontal cortex, showing neuroprotective, antidepressant and anxiolytic effects.
Furthermore, the results showed improvement of symptoms using nanoparticle formulation to enrich the distribution of the compound since the disponibility by the oral route is poor. However, the studies used different doses and administration routes, which may interfere with the results and possible comparisons. Therefore, more clinical trials are necessary to determine effective dosages, long-term effects, and better administration routes. We may conclude that curcumin may work as adjuvant therapy for the approach of depression; however, studies with humans are limited, and further controlled clinical trials are needed.
Footnotes
Author Contributions
J.N.M., G.A.S., and S.M.B. involved in conceptualization of this study. Literature search was performed by G.S.S., E.L.G., and A.C.A. S.M.B., E.L.G., G.A.S., and J.N.M. performed writing, reviewing, and editing.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
