Abstract
Objectives:
To evaluate the utilisation and persistence of antipsychotics for the treatment of schizophrenia in Australia.
Methods:
A retrospective study using the Australian Pharmaceutical Benefits Scheme database of a representative 10% sample. All adults with schizophrenia who were dispensed three or more supplies of oral (including clozapine) or long-acting injectable antipsychotics between 1 June 2015 and 31 May 2020 were included. Persistence time in treatment was evaluated using survival analysis and Cox hazard ratios.
Results:
In all, 26,847 adults with schizophrenia were studied. Oral second-generation antipsychotics were more frequently dispensed than the other antipsychotic groups studied. Median treatment persistence times were 18.3 months for second-generation antipsychotic long-acting injectables, 10.7 months for oral second-generation antipsychotics and were significantly lower for both formulations of first-generation antipsychotics at 5.2 months (long-acting injectables) and 3.7 months (oral). The median persistence time for clozapine was significantly longer than all other antipsychotics groups.
Conclusions:
Oral second-generation antipsychotics and second-generation antipsychotic long-acting injectables accounted for over 75% and 13% of all antipsychotics in Australia, respectively. Concerns over medication adherence and subsequent relapse have not translated into increased long-acting injectable usage despite their significantly longer persistence. Clozapine, the single most ‘persistent’ antipsychotic, was only used in 9% of people, although up to a third of all cases are likely to be treatment-resistant. Our data suggest clinicians should give consideration to the earlier use of second-generation antipsychotic long-acting injectables and clozapine, to ameliorate prognosis in schizophrenia.
Keywords
Introduction
Relapse in schizophrenia is frequently seen in clinical practice (Kahn et al., 2015; Taylor and Jauhar, 2019) and can worsen prognosis, including suicide (Hor and Taylor, 2010). In Australia, psychotic disorders such as schizophrenia have a point prevalence of 0.47% and have significant social and personal costs (Jablensky et al., 2000). Antipsychotic medication remains the cornerstone of treatment for schizophrenia given it reduces relapse (Taylor and Jauhar, 2019), with most guidelines recommending maintenance antipsychotics for at least a year after a first episode of psychosis (FEP) and for over 2 years after a relapse (Galletly et al., 2016; Scottish Intercollegiate Guidelines Network, 2013).
Medication adherence is defined by the World Health Organization (Brown and Bussell, 2011) as ‘the degree to which the person’s behaviour corresponds with the agreed recommendations from a healthcare provider’. Limited adherence with medication is known to be a problem with many chronic diseases including schizophrenia (Brown and Bussell, 2011; McCutcheon, 2020; Yaegashi et al., 2020). The reasons for both cessation of medication and partial or non-adherence include the following: adverse medication effects (Young et al., 2015), forgetfulness, stigma, lack of efficacy (Yaegashi et al., 2020) and poor service delivery. Importantly, poor adherence is the main cause of relapse in schizophrenia (McCutcheon, 2020; Yaegashi et al., 2020).
Medication persistence is related to medication adherence and refers to the duration of supply of antipsychotic medication, i.e., from initiation to cessation. Medication persistence has been less well studied than adherence, but time to all-cause discontinuation of antipsychotic medication is similar to persistence and has been used in the seminal CATIE and EUFEST studies as a measure of effectiveness (Kahn et al., 2008; Stroup et al., 2003). Using the Australian national Pharmaceutical Benefits Scheme (PBS) database of a representative 10% sample of all prescriptions, we aimed to describe the national patterns of antipsychotic utilisation between 2015 and 2020, as well as examine which types of antipsychotics were associated with the longest persistence in the ‘real world’.
Methods
Data source
This was a retrospective Australian national pharmaceutical claims database study, using a random 10% sample of the national PBS, which is representative of the whole database. Australia’s PBS subsidises prescriptions for all Australian citizens and permanent residents. This dataset contained all PBS supplies (new and repeat prescriptions) for all individuals in the 10% random sample with a date of supply between 1 June 2015 and 31 May 2020. The 10% PBS sample represents 10% of all patients accessing the PBS in Australia, and the sample was selected by Services Australia. Approval to use the PBS 10% sample for this analysis was provided by the Government’s External Requests Evaluation Committee on 21 July 2020.
Study population
The main study population included all patients who had three or more (monthly) supplies of antipsychotics with an indication of schizophrenia at any time between 1 June 2015 and 31 May 2020 and were at least 18 years old at the date of supply in the study period. The requirement for three or more supplies was intended to exclude off-label use. The index date was the date of the first antipsychotic supply in the observation period.
The ‘de novo’ study population was derived from the main study population and was limited to patients with an initial antipsychotic supply for the treatment of schizophrenia occurring between 1 January 2016 and 31 May 2019. A 6-month ‘look back’ period from 1 June 2015 until 31 December 2015 was implemented to ensure these patients were not being treated. This set was used to assess treatment persistence in patients beginning their treatment in the study period.
An indication of schizophrenia was ascertained for each supply from the diagnosis-specific Authority codes for schizophrenia, which clinicians are required to provide for a patient to receive a PBS subsidy for any second-generation antipsychotic (SGA). Oral first-generation antipsychotics (FGA) do not require a PBS Authority code in order to be supplied, so dosage level was used to distinguish between an indication for schizophrenia, i.e., chlorpromazine above 100 mg per day and haloperidol above 5 mg per day, rather than other use such as a sedative or anxiolytic.
Low doses (<100 mg chlorpromazine equivalent) of some SGAs commonly used for other indications (olanzapine, quetiapine, risperidone) were not included, as a diagnosis of schizophrenia could not be guaranteed. By contrast, all dispensed FGA long-acting injectable (LAI) antipsychotics were assumed to be used for schizophrenia. The full list of included treatments with a designated indication for schizophrenia is listed by dose and PBS item number in Supplemental Table 4. There were no additional exclusion criteria.
Antipsychotic group was based on formulation (oral or LAI) and generation (FGA or SGA) based on the index treatment. Clozapine was assessed as a separate group, given its special indication for treatment-resistant schizophrenia (TRS) only and the stringent processes and monitoring associated with its use. Patients were assigned to the five antipsychotic groups based on their index treatment which was the first supplied or dispensed treatment a patient received in the study period. It was recognised that an individual patient might be supplied more than one antipsychotic medication simultaneously (polypharmacy), and that patients might switch medications within an antipsychotic group, with group allocation dependent on the index treatment.
Primary care provider settings consisted of general practice, emergency medicine and community healthcare. Other specialist types were classified as secondary care providers.
Analyses
Descriptive statistics were used to present the patient characteristics and utilisation of antipsychotic supplies by quarter. Medication utilisation was assessed by evaluating the total supply volumes (new and repeat prescriptions) and the number of people dispensed antipsychotics for schizophrenia.
The median persistence time from antipsychotic initiation to discontinuation was evaluated using Kaplan–Meier methods. An adjusted Cox proportional hazard model was used to calculate the hazard ratio by antipsychotic group. The model was adjusted for gender, age group at index, state and provider setting (primary or secondary care) at initiation. Persistence was defined as the time from first (initiation or index) antipsychotic supply to the last persistent supply of an antipsychotic within the same treatment group + supply duration (30 days or 84 days for paliperidone 3-monthly injection). Thus, individuals who switched to a different antipsychotic within the same group were still deemed to be persistent, whereas those who moved to a different class or formulation were considered as non-persistent. Stopping antipsychotic treatment for three supply periods or more also indicated non-persistence. The final or last persistent supply was the one prior to a gap of three times the supply duration between supplies of an antipsychotic within the same group (30 days or 84 days for paliperidone 3-monthly injection). Individuals who were persistent with their medication at the end of the observation period were censored at the last supply. All analyses were conducted in SAS v9.4.
Results
Descriptive analyses
Of the 78,696 individuals with an antipsychotic supply for any indication in the observation period (1 June 2015 to 31 May 2020), 26,847 (34%) people aged 18 years or more received ⩾3 supplies of antipsychotics, had the indication of schizophrenia and were thus included in the study population. Of these, 7,589 patients began treatment in the study period, i.e., had their first antipsychotic supply between 1 January 2016 and 31 May 2019.
At the date of supply of the first antipsychotic in the observation period (index date), the mean (SD) age of the study population was 46 (17) years. They were more likely to be male (53.4%) and receive their prescription from a primary healthcare professional (61.5%). Within antipsychotic groups, the mean age ranged from 51 years for oral FGA to 44 years for SGA LAIs and clozapine, while the percentage of males ranged from 68% for clozapine to 46% for oral FGA. With the exception of clozapine, most prescriptions came from primary care (Table 1). The de novo patients on average were slightly younger (44 years) than the main study population and more likely to be male (51.8%). The majority of patients received their prescription from a secondary care provider (51.1%) (Supplemental Table 1).
Patient demographics by antipsychotic group.
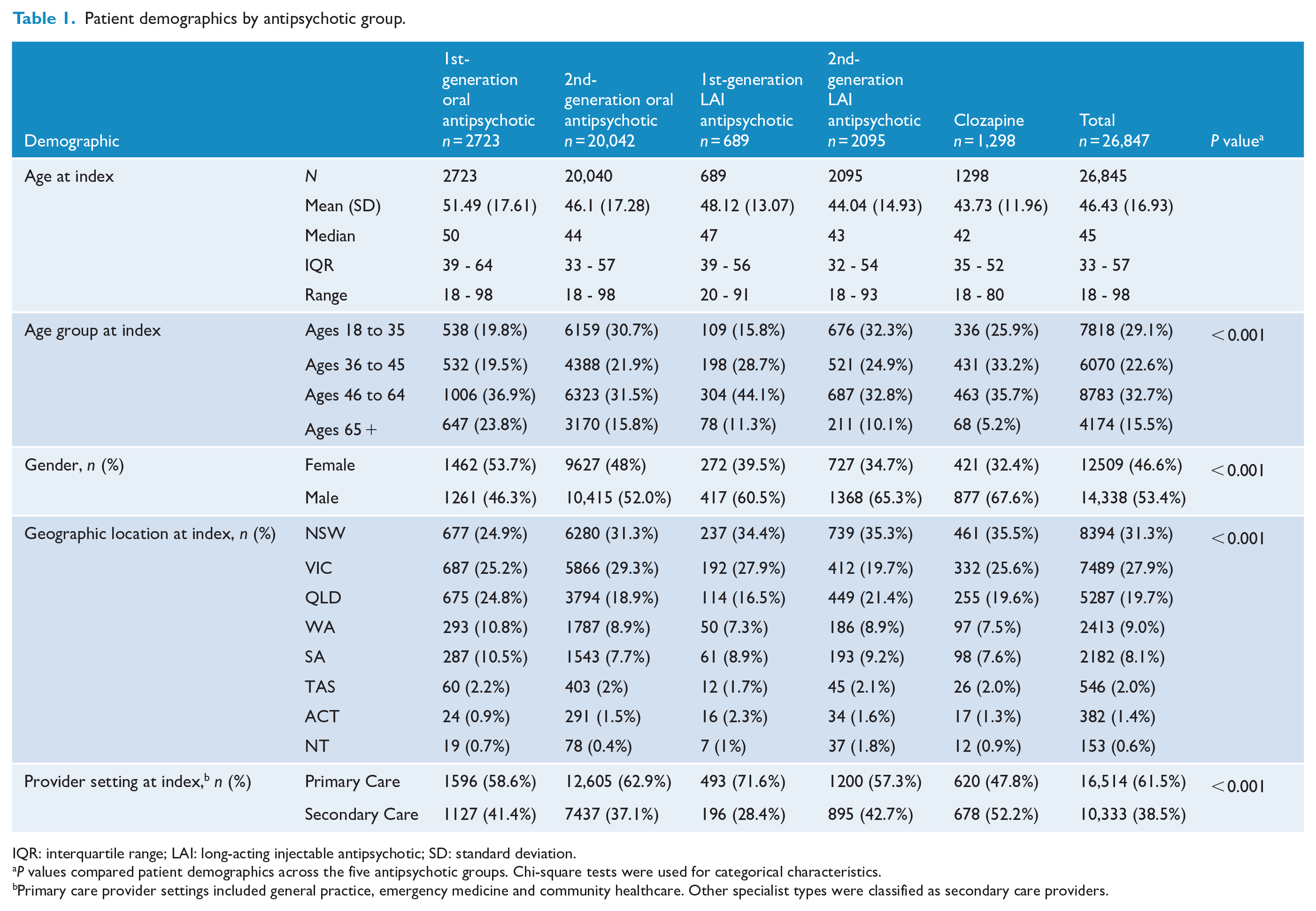
IQR: interquartile range; LAI: long-acting injectable antipsychotic; SD: standard deviation.
P values compared patient demographics across the five antipsychotic groups. Chi-square tests were used for categorical characteristics.
Primary care provider settings included general practice, emergency medicine and community healthcare. Other specialist types were classified as secondary care providers.
Oral SGAs were the most frequently dispensed antipsychotic group over the observation period. For instance, of the 16,906 patients receiving at least one antipsychotic in the last observation period of the study (the second 2020 quarter), 76% were dispensed an oral SGA, 16% SGA LAIs, 9% clozapine, 9% an oral FGA and 4.5% an FGA LAI. Antipsychotic volumes in the most recent quarter of the study were representative of volumes throughout the 5-year study period.
Over the 5-year study period, the utilisation of each antipsychotic group remained generally consistent (Figure 1).
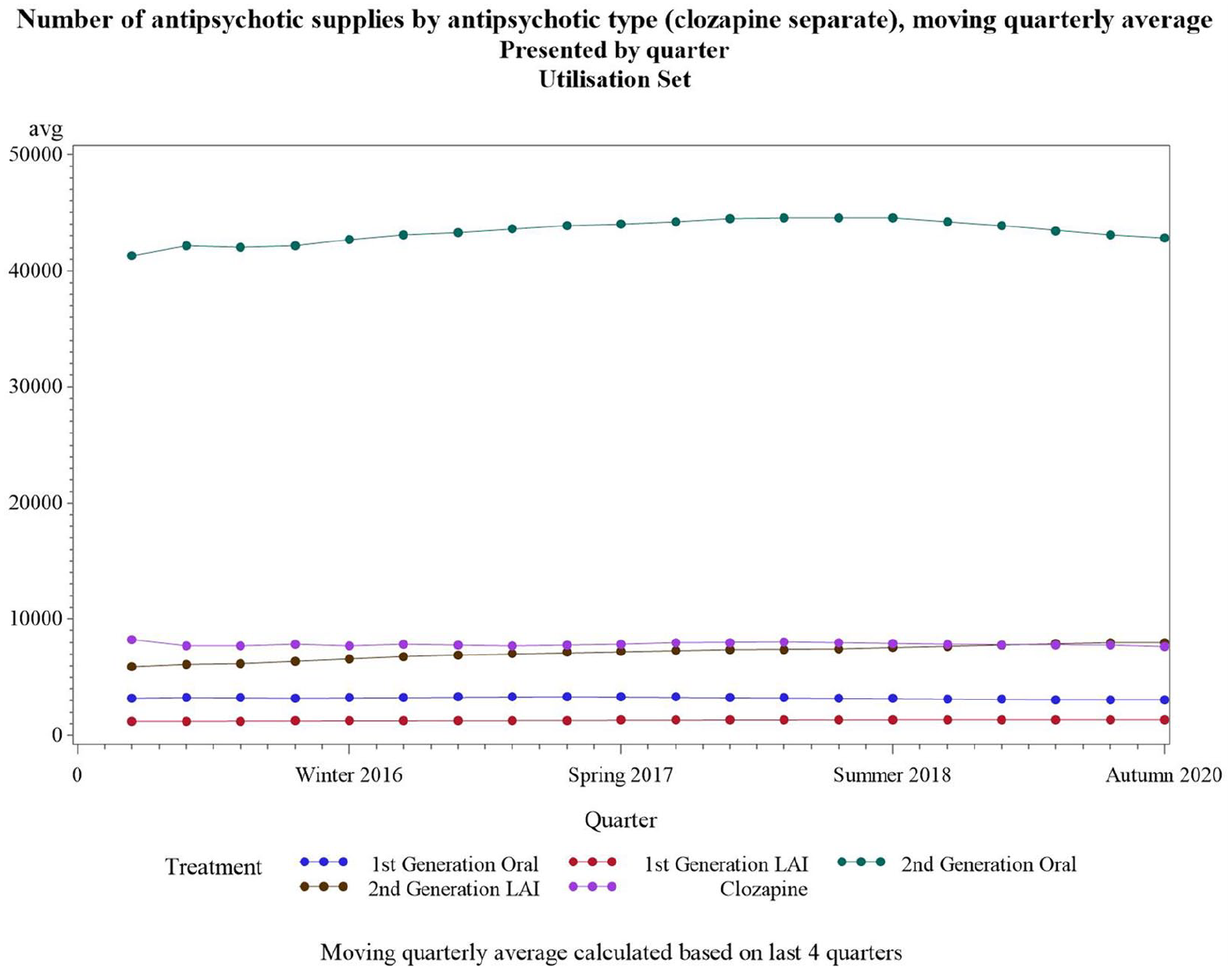
Number of antipsychotic supplies over time by antipsychotic group.
Some individuals were simultaneously or consecutively dispensed more than one antipsychotic in a quarter. In 2019, which is representative of the full observational period, 9.7% of all individuals studied were dispensed more than one antipsychotic concurrently each month for at least 3 months, with a mean of 2.07 antipsychotics (polypharmacy) per individual.
In the last quarter of observation in 2020, 68% of all supplies were oral SGAs, followed by SGA LAIs (13%), clozapine (12%), oral FGAs (5%) and FGA LAIs (2%). Overall, the 16,906 patients receiving at least one antipsychotic in the last observation period received a median of 3 supplies (Table 2).
Antipsychotic supplies by antipsychotic group.
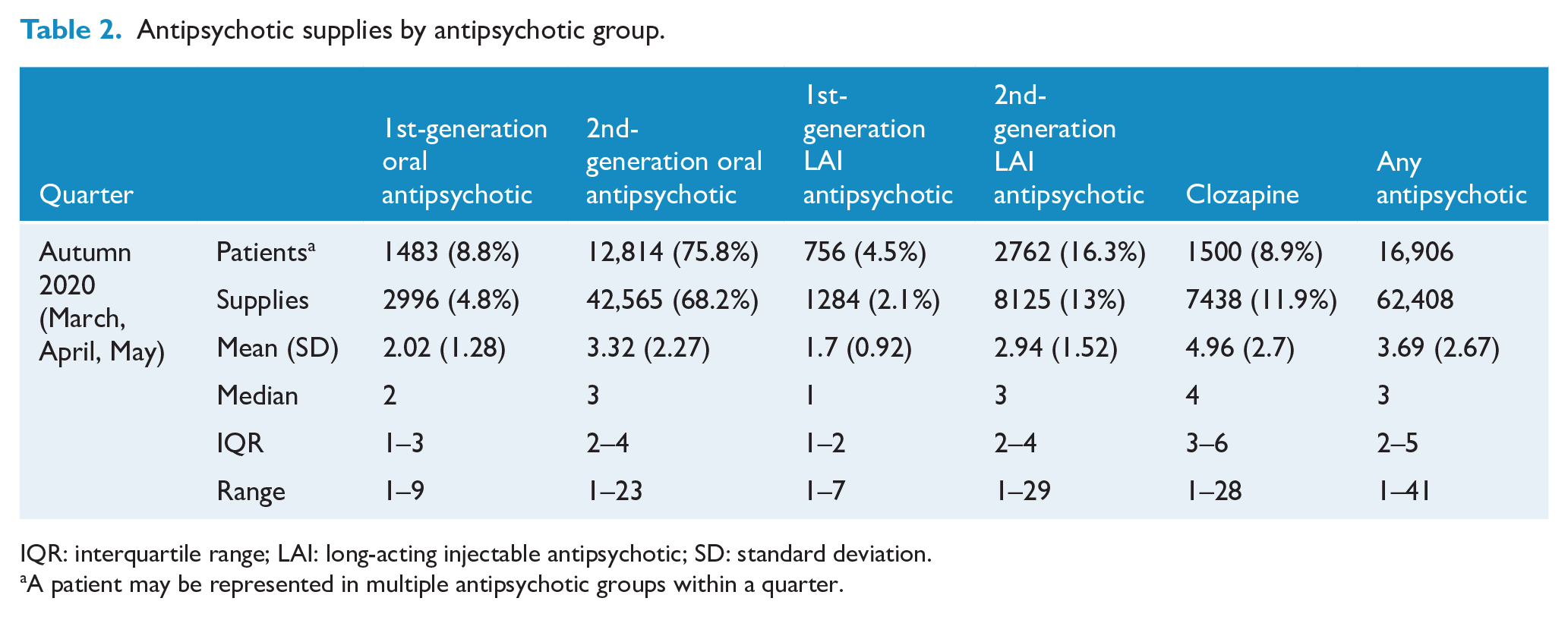
IQR: interquartile range; LAI: long-acting injectable antipsychotic; SD: standard deviation.
A patient may be represented in multiple antipsychotic groups within a quarter.
The median treatment persistence time was 18.3 months for SGA LAIs (95% confidence interval [CI]: [16.8, 20.1]) and 10.7 months for oral SGAs (95% CI: [10.3, 11.1]). The median treatment persistence was 5.2 months for FGA LAIs (95% CI: [4.2, 6.1]) and 3.7 months for oral FGAs (95% CI: [3.5, 3.9]). The median time on treatment (persistence) was the highest for clozapine, with 60% of people continuing clozapine over the 5-year study period (Figure 2 and Table 3).
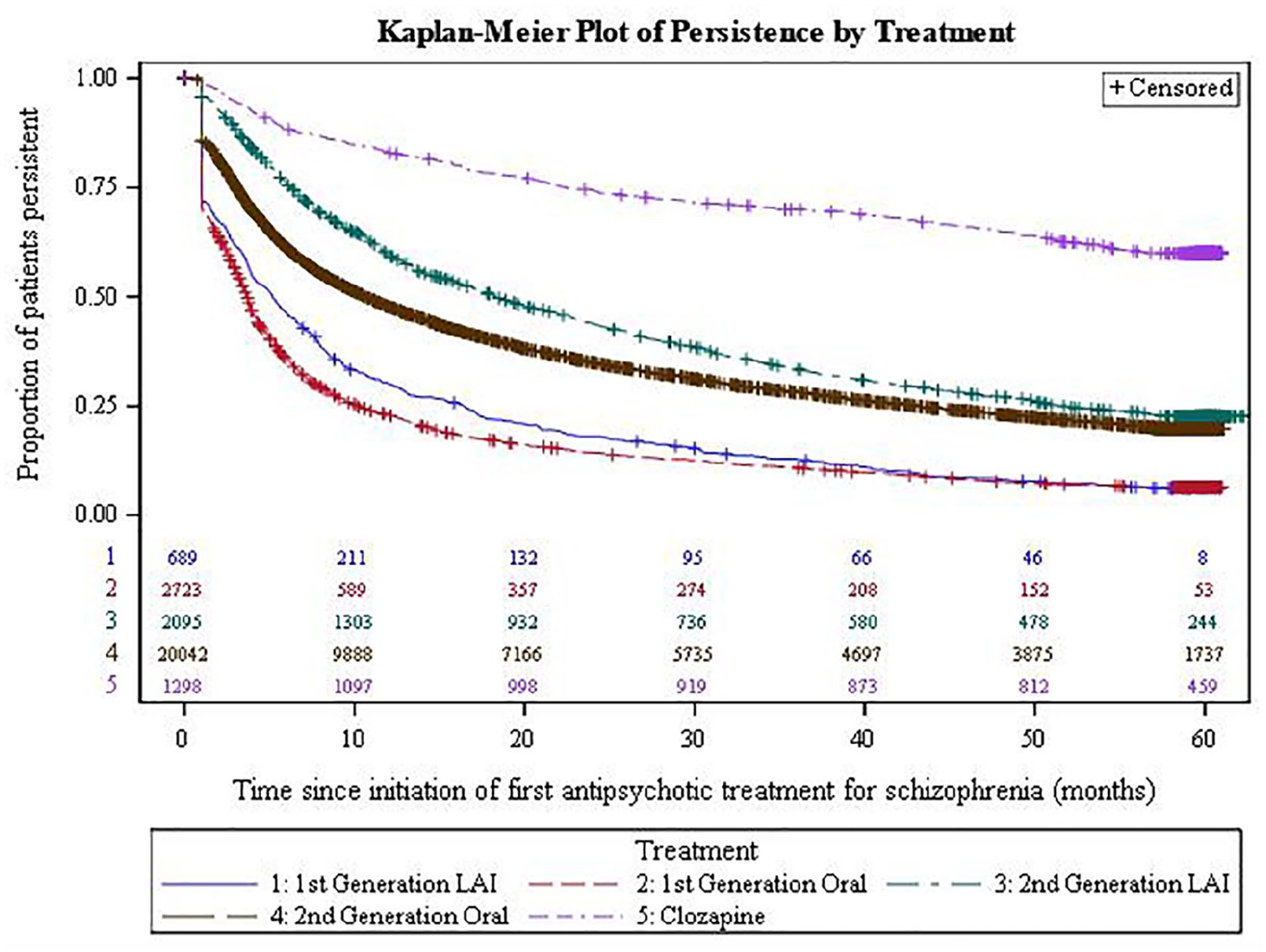
Kaplan–Meier plot of persistence by antipsychotic group.
Antipsychotic persistence by antipsychotic group.
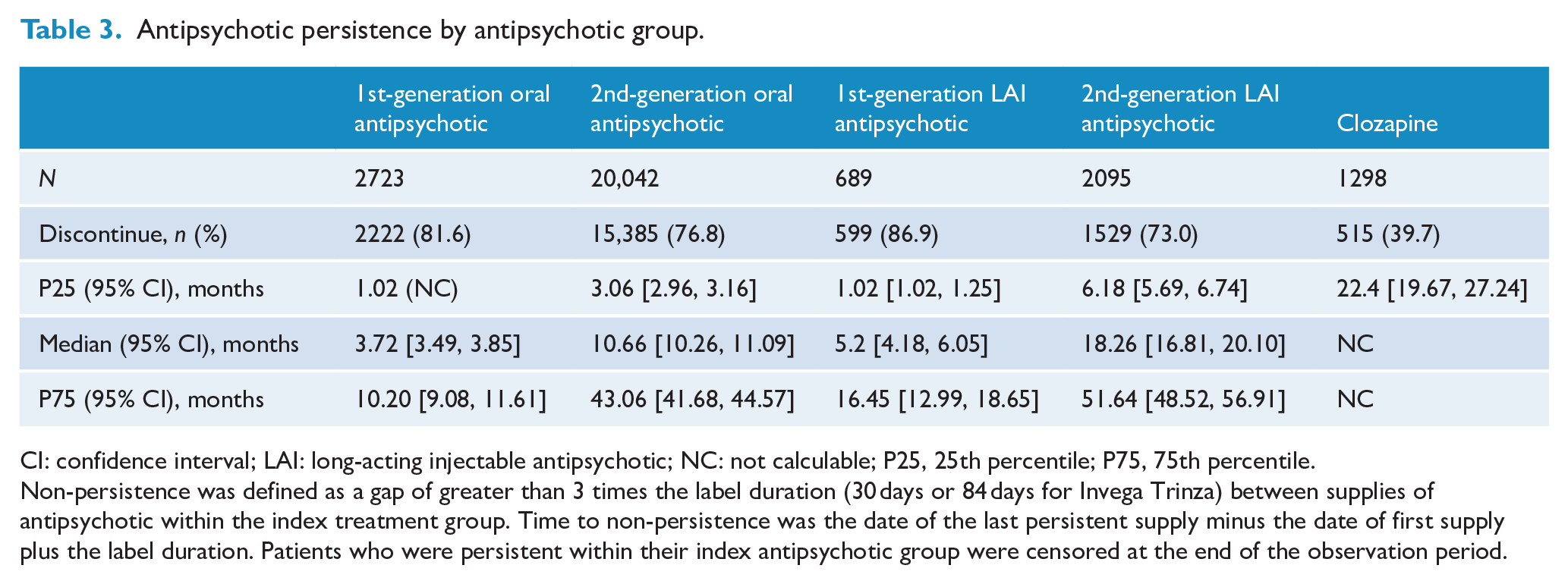
CI: confidence interval; LAI: long-acting injectable antipsychotic; NC: not calculable; P25, 25th percentile; P75, 75th percentile.
Non-persistence was defined as a gap of greater than 3 times the label duration (30 days or 84 days for Invega Trinza) between supplies of antipsychotic within the index treatment group. Time to non-persistence was the date of the last persistent supply minus the date of first supply plus the label duration. Patients who were persistent within their index antipsychotic group were censored at the end of the observation period.
In the de novo patients, the median treatment persistence time was the highest in the clozapine group (31.97 months, 95% CI: [17.27, NA]), followed by SGA LAIs (8.59 months, 95% CI: [7.57, 10.33]) and oral SGAs (4.54 months, 95% CI: [4.34, 4.74]). The median treatment persistence was considerably lower for FGA LAIs (3.67 months, 95% CI: [2.50, 4.41]) and oral FGAs (2.60 months, 95% CI: [2.27, 2.93]) (Supplemental Table 2).
Multivariate analyses
In the Cox regression model, individuals initially dispensed oral FGA (hazard ratio [HR] = 2.0, 95% CI: [1.9, 2.1], p < 0.0001) and FGA LAIs (HR = 1.8, 95% CI: [1.7, 2.0], p < 0.0001) as their index treatment were significantly more likely to be non-persistent with treatment when compared to the oral SGA group (Table 4). In addition, when compared to the oral SGAs, patients taking clozapine and SGA LAIs at index were significantly less likely to be non-persistent with treatment (HR = 0.30, 95% CI: [0.27, 0.32], p < 0.0001) and (HR = 0.79, 95% CI: 0.75 to 0.84, p < 0.0001), respectively. Similarly, patients on FGA LAIs were more than twice as likely to be non-persistent compared to those on SGA LAIs (HR = 2.3, 95% CI: [2.1, 2.5], p < 0.0001).
Cox proportional hazards model of time to non-persistence by antipsychotic group.
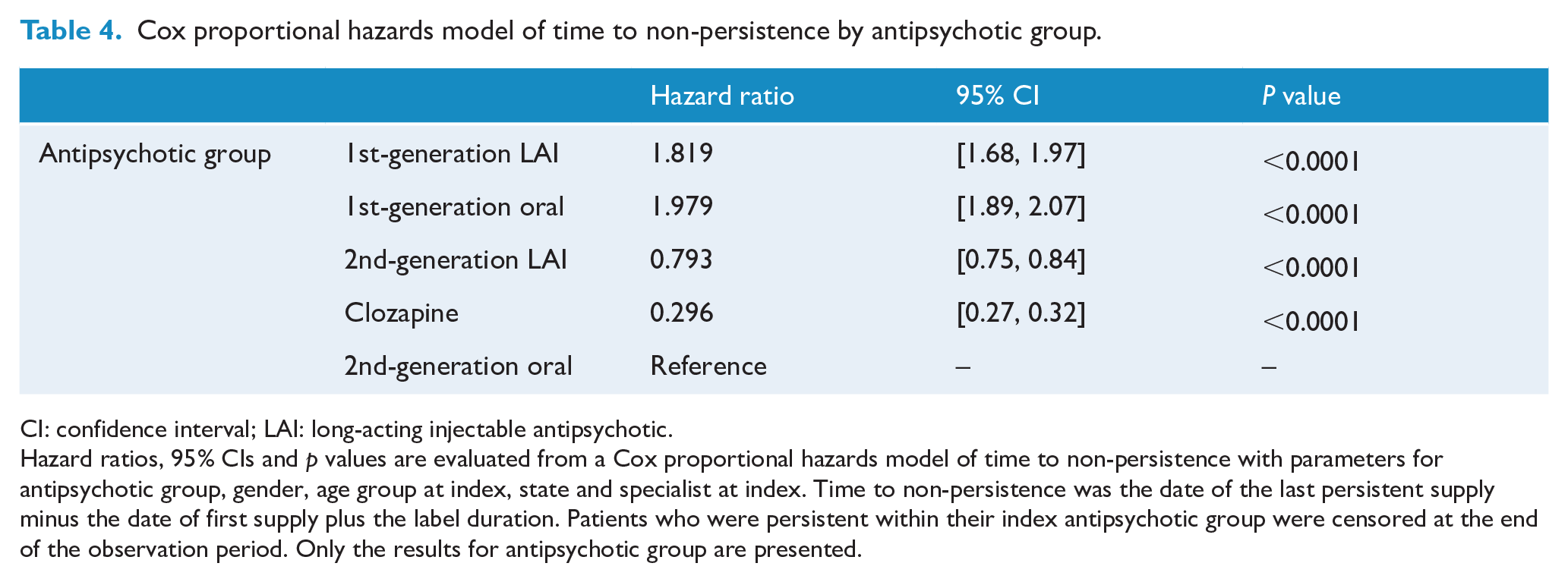
CI: confidence interval; LAI: long-acting injectable antipsychotic.
Hazard ratios, 95% CIs and p values are evaluated from a Cox proportional hazards model of time to non-persistence with parameters for antipsychotic group, gender, age group at index, state and specialist at index. Time to non-persistence was the date of the last persistent supply minus the date of first supply plus the label duration. Patients who were persistent within their index antipsychotic group were censored at the end of the observation period. Only the results for antipsychotic group are presented.
The Cox model of persistence in the de novo patient population showed very similar results albeit with smaller disparities between the FGAs and SGAs (Supplemental Table 3).
Discussion
Main findings
Between 2015 and 2020, dispensed antipsychotic volumes varied little. Most people with schizophrenia (76%) were dispensed oral SGAs, not including the 9% on clozapine. FGA usage was low at <14% for oral and LAIs combined. This difference between SGA and FGA use cannot simply be due to perceived better adverse side effect profiles, as the metabolic problems associated with SGAs are well known (Young et al., 2015). Australian (Royal Australian and New Zealand College of Psychiatrists [RANZCP]) clinical practice guidelines (Galletly et al., 2016) concerning the management of schizophrenia recommend that SGAs be used in preference to FGAs, which is the case here. The only other similar extant national data for schizophrenia come from Sweden and Finland (Taipale et al., 2020), where in 2016 oral SGAs were used at 65% and 57%, respectively, whereas oral FGA usage there was 17–18%.
Regular antipsychotic polypharmacy in Australia was not common, involving less than 10% of the whole study population. This is reassuring given that most clinical practice guidelines discourage regular antipsychotic polypharmacy, and reviews (Young et al., 2015) have concluded that polypharmacy is more likely to lead to an adverse side effect burden than monotherapy.
SGA LAIs were dispensed in 16% of people with schizophrenia, with an additional 4.5% receiving an FGA LAI. Total oral antipsychotic prescriptions (excluding clozapine) greatly outnumbered LAIs (73% vs 15%) despite evidence that LAI formulations dramatically reduce relapse rate in schizophrenia (Subotnik et al., 2015; Tiihonen et al., 2011). These levels of LAI prescription are lower than in the United Kingdom where just under 30% are dispensed an LAI (Patel et al., 2014) but similar to those in Scandinavia (Taipale et al., 2020) where between 13% and 20% received an SGA or FGA LAI from 2006 to 2016. In South East Asia (Tang et al., 2020), LAI use ranges from 0% to 44%. The underuse of LAIs is thus worldwide, and reasons include stigma, needle phobias and lack of patient and prescriber preference (Patel et al., 2009). Clinical guidelines (Galletly et al., 2016; Scottish Intercollegiate Guidelines Network, 2013) recommend LAIs in those who choose that formulation and in those whom the lack of medication adherence or persistence is a concern. Medication adherence is known to be a common (~45–50%) problem in people with schizophrenia (McCutcheon, 2020; Yaegashi et al., 2020).
During the 5 years of study here, only 9% of our study population were dispensed clozapine although up to one-third of people with schizophrenia (Mizuno et al., 2020) are thought to be treatment-resistant, and clozapine is the most efficacious antipsychotic available for TRS (Leucht et al., 2013). In addition, nearly 10% of all individuals studied here (in 2019) were on multiple simultaneous antipsychotic prescriptions, which possibly could also be an indication of treatment resistance. These results are consistent with findings that clozapine is underused internationally (Bachmann et al., 2017). Possible reasons include concerns about regular blood monitoring, a lack of case managers or clozapine nurses, and the genuinely significant side effects of clozapine (Young et al., 2015). In addition, people on clozapine need to have sufficient insight into their condition to take oral medication.
Nevertheless, our data reveal that clozapine has significantly superior persistence compared to other oral antipsychotics and even LAIs. This long persistence or duration of treatment may be due not only to its effectiveness but also to the intensity of required follow-up, and that clozapine is seen as a ‘last chance’ option for refractory cases. Given the paucity of long-term data on clozapine use, it is hard to contextualise a 60% persistence or continuation over 5 years, but this reflects the authors’ years of clinical experience with this population.
After clozapine, SGA LAIs were associated with the longest persistence (18 months). This compares to 11 months for oral SGAs and only 5 months for FGA LAIs. The difference between the older and newer LAIs is striking, particularly when contrasted with a large randomised controlled trial (RCT) of a newer SGA LAI (paliperidone) versus haloperidol LAI which failed to detect any significant difference in efficacy over 2 years (McEvoy et al., 2014). The contrast of SGA versus FGA LAIs was also seen in a recent network meta-analysis (Ostuzzi et al., 2021) which found that some newer LAIs such as paliperidone and aripiprazole outperformed other LAIs in terms of relapse prevention and acceptability. Comparable national studies to ours, from both Finland and Sweden, yield similar findings in that clozapine and LAIs were best at preventing relapse and re-hospitalisation (Tiihonen et al., 2011, 2017). This illustrates the benefit in having ‘real-world’ data such as this study alongside RCT results, which may be affected by the intensity of follow-up that is absent in everyday clinical settings (Haddad et al., 2011).
The comparison of persistence between the main study population and the de novo population shows that patients new to treatment are less persistent than treatment-experienced patients. Furthermore, the persistence estimates from the main study population are likely to be conservative as some treatment-experienced patients would have entered the study already receiving their treatment.
Clinical implications
These persistence data raise the important clinical question of ‘what is the optimum duration of antipsychotic treatment in schizophrenia?’ Perhaps surprisingly, there is no conclusive evidence-based answer to this question although some authors (Scottish Intercollegiate Guidelines Network, 2013; Correll et al., 2018) recommend between 1 and 5 years of continuous antipsychotic medication after a psychotic episode. The mean age of individuals here suggests our study population largely have established (rather than FEP) schizophrenia, so a medication persistence of 11 months or less with oral antipsychotics is less than recommended. Our treatment persistence data on SGA LAIs and clozapine, however, are more reassuring in terms of guideline concordance.
These results also highlight the value of initiating LAIs in people with schizophrenia. Psychiatrists often reserve the use of LAIs for non-adherent ‘revolving-door’ patients despite evidence of potential benefits for first-episode patients (Agid et al., 2010; Taylor and Ng, 2013). This may be due to misguided assumptions about patient preference of oral over LAI formulations (Patel et al., 2009; Waddell and Taylor, 2009).
In a systematic review (Waddell and Taylor, 2009) of patient satisfaction with LAIs, six studies reported a predominantly positive attitude, two were neutral and two were negative with acceptance of LAIs increasing with insight and experience of that form of medication. Where participants reported an unfavourable attitude, nearly half had felt forced to receive LAIs. This has important implications for clinicians initiating LAIs and the relevance of techniques such as Shared Decision Making (Nott et al., 2018) and Motivational Interviewing (Kisely et al., 2012). Similarly, in a ‘real-world’ study, 91% (213 of 234) of FEP and early-phase schizophrenia patients were willing to try at least one dose of an LAI when the option was presented in a supportive manner (Kane et al., 2019).
Strengths and limitations
This is a large representative national Australian sample followed up over a sufficient time period to adequately assess the effect of treatments like LAIs and clozapine. It is one of the few studies to directly contemporaneously compare SGA and FGA antipsychotics in both oral and LAI formulations. In our analysis, clozapine was separated from other treatments because of the distinct patient subgroup in which it is used and the intensive monitoring that is required during its administration. Given the likely differences in case-mix of people in the clozapine and non-clozapine groups, it may not be appropriate to compare the two. ‘Real-world’ observational studies of antipsychotics provide a more representative insight into treatment effectiveness than the more limited but rigorous RCTs but can also be prone to selection bias. We attempted to overcome this by using a random 10% selection of the national Australian PBS database.
We acknowledge several limitations to this study. Database studies show trends but do not permit causality to be deduced. It is also possible that not all antipsychotic prescriptions for schizophrenia were identified and that some antipsychotic prescriptions were not for schizophrenia due to the incorrect use of prescriber-generated Authority required codes. Furthermore, just because a pharmacy supplies an oral antipsychotic does not guarantee that the medication was actually taken, although the fact that we included only people who picked up 3 months’ supply of medication suggests usage for that period. These data lack information on legal status and any possible effect of compulsory community treatment on medication persistence. However, a Cochrane Review (Kisely et al., 2017) of three RCTs found no evidence that compulsory community treatment affected adherence to medication. Similarly, we have no information on other possible predicators of persistence such as significant relationships, case management and social supports, but it is assumed that these potential confounders were randomly distributed.
When interpreting these study results, one further explanation for ceasing medication (or being non-persistent) might be that the schizophrenia has satisfactorily resolved, and thus the patient and prescriber decide the medication is no longer necessary. Finally, the last quarter covered by the study included a period of the nationwide COVID lockdown and so may be atypical. However, a separate detailed analysis comparing this quarter with those of the same quarter in previous years failed to show any statistically significant difference in prescription rates (Kisely, 2021).
Conclusion
We suggest that persistence with treatment is a measure of adherence to medication, which in turn may be related to the effectiveness of a treatment in the ‘real world’. Oral SGAs accounted for over 68% of all antipsychotics dispensed during the 5-year study period, followed by 11% usage of SGA LAIs. The utilisation rate of both oral and LAI FGAs was low. SGA LAIs had significantly greater treatment persistence than oral SGAs and FGAs. Clozapine was only used in 9% of people with (treatment-resistant) schizophrenia despite having the longest persistence time, reflecting its special attributes and restrictions. Our data suggest prescribers and clinicians should give earlier consideration to the use of SGA LAIs and clozapine, with a view to ameliorating prognosis in schizophrenia.
Supplemental Material
sj-docx-1-anp-10.1177_1687814020966927 – Supplemental material for Antipsychotic utilisation and persistence in Australia: A nationwide 5-year study
Supplemental material, sj-docx-1-anp-10.1177_1687814020966927 for Antipsychotic utilisation and persistence in Australia: A nationwide 5-year study by Mark Taylor, Dante Dangelo-Kemp, Dennis Liu, Steve Kisely, Simon Graham, Jodie Hartmann and Sam Colman in Australian & New Zealand Journal of Psychiatry
Supplemental Material
sj-docx-2-anp-10.1177_1687814020966927 – Supplemental material for Antipsychotic utilisation and persistence in Australia: A nationwide 5-year study
Supplemental material, sj-docx-2-anp-10.1177_1687814020966927 for Antipsychotic utilisation and persistence in Australia: A nationwide 5-year study by Mark Taylor, Dante Dangelo-Kemp, Dennis Liu, Steve Kisely, Simon Graham, Jodie Hartmann and Sam Colman in Australian & New Zealand Journal of Psychiatry
Supplemental Material
sj-docx-3-anp-10.1177_1687814020966927 – Supplemental material for Antipsychotic utilisation and persistence in Australia: A nationwide 5-year study
Supplemental material, sj-docx-3-anp-10.1177_1687814020966927 for Antipsychotic utilisation and persistence in Australia: A nationwide 5-year study by Mark Taylor, Dante Dangelo-Kemp, Dennis Liu, Steve Kisely, Simon Graham, Jodie Hartmann and Sam Colman in Australian & New Zealand Journal of Psychiatry
Supplemental Material
sj-docx-4-anp-10.1177_1687814020966927 – Supplemental material for Antipsychotic utilisation and persistence in Australia: A nationwide 5-year study
Supplemental material, sj-docx-4-anp-10.1177_1687814020966927 for Antipsychotic utilisation and persistence in Australia: A nationwide 5-year study by Mark Taylor, Dante Dangelo-Kemp, Dennis Liu, Steve Kisely, Simon Graham, Jodie Hartmann and Sam Colman in Australian & New Zealand Journal of Psychiatry
Supplemental Material
sj-jpg-1-anp-10.1177_00048674211051618 – Supplemental material for Antipsychotic utilisation and persistence in Australia: A nationwide 5-year study
Supplemental material, sj-jpg-1-anp-10.1177_00048674211051618 for Antipsychotic utilisation and persistence in Australia: A nationwide 5-year study by Mark Taylor, Dante Dangelo-Kemp, Dennis Liu, Steve Kisely, Simon Graham, Jodie Hartmann and Sam Colman in Australian & New Zealand Journal of Psychiatry
Footnotes
Acknowledgements
The authors wish to acknowledge the early input of Dr Mona Taouk.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: Lundbeck/Otsuka funded the study but had limited control over study design or interpretation. M.T. has received hospitality and/or fees from Lundbeck, Janssen and Otsuka, and D.L. has received hospitality and/or fees from Lundbeck, Janssen, Otsuka, Seqirus and Servier in the last 3 years. S.K. has nothing to disclose. D.D.K. and S.C. are employees of Labcorp Drug Development, which has received funding from Lundbeck/Otsuka. S.G. and J.H. are employees of Lundbeck.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: Lundbeck/Otsuka, Australia, funded this study.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
