Abstract
Background:
Somatic disorders and somatic symptoms are common in primary care populations; however, little is known about the prevalence in surgical populations. Identification of inpatients with high somatic symptom burden and psychological co-morbidity could improve access to effective psychological therapies.
Methods:
Cross-sectional analysis (n = 465) from a prospective longitudinal cohort study of consecutive adult admissions with non-traumatic abdominal pain, at a tertiary hospital in New South Wales, Australia. We estimated somatic symptom prevalence with the Patient Health Questionnaire-15 at three cut-points: moderate (⩾10), severe (⩾15) and ‘bothered a lot’ on ⩾3 symptoms; and psychological co-morbidity with the Patient Health Questionnaire-9 and Generalized Anxiety Disorder-7 at standard (⩾10) cut-points. We also examined gender differences for somatic symptoms and psychological co-morbidity.
Results:
Prevalence was moderate (52%), female predominance (odds ratio = 1.71; 95% confidence interval = [1.18, 2.48]), severe (20%), no gender difference (1.32; [0.83, 2.10]) and ‘bothered a lot’ on ⩾3 symptoms (53%), female predominance (2.07; [1.42, 3.03]). Co-morbidity of depressive, anxiety and somatic symptoms ranged from 8.2% to 15.9% with no gender differences.
Conclusion:
Somatic symptoms were common and psychological triple co-morbidity occurred in one-sixth of a clinical population admitted for abdominal pain. Co-ordinated surgical and psychological clinical intervention and changes in clinical service organisation may be warranted to provide optimal care.
Introduction
Somatic syndromes are a diagnostic challenge due to lack of objective signs of disease and the non-specificity of the symptoms, while diagnosis and treatment of these conditions are under-prioritised compared to other illnesses (Fink, 2017). Somatic symptoms are bodily symptoms, most commonly pain (back, abdominal, head, chest, limbs, coital or menstrual), dizziness, fainting spells, shortness of breath, palpitations, nausea and indigestion, which are often present over a period of months or more; are a source of patient distress and concern; and for which the sufferer often seeks frequent medical care (Kroenke et al., 2002). Somatic symptoms may or may not be associated with another medical condition; the Diagnostic and Statistical Manual of Mental Disorders (5th ed.; DSM-5) diagnosis of Somatic Symptom Disorder (SSD) and a medical illness may frequently occur together (American Psychiatric Association, 2013). A variety of syndromes of bodily distress or somatic syndromes are recognised, all of which can be costly for the health care system (diagnostic testing, fruitless treatment) (Barsky et al., 2005), for society (disability, lost productivity) (Konnopka et al., 2013) and for patients (health anxiety, frustration) (Duddu et al., 2008), who are additionally exposed to the risk of iatrogenic harms (false-positive tests, or unnecessary operations) (Fink, 1992). Systematic reviews have identified effective treatments for somatoform disorders with improved patient outcomes and reduced health service costs (Kroenke, 2007).
The classification of somatic disorders has changed over time with the most recent iterations being Somatic Symptom and Related Disorders for DSM-5 (American Psychiatric Association, 2013) and Disorders of Bodily Distress or Bodily Experience for International Classification of Diseases, 11th Revision (ICD-11; World Health Organization, 2018), with the key diagnostic exemplars being SSD and Bodily Distress Disorder (BDD), respectively (see Supplementary Table 1 for details). Alternatively, the concept of medically unexplained symptoms is well recognised and endorsed by the National Health Service (NHS, 2018) and the Royal College of Psychiatry (Royal College of Psychiatrists, 2015) in the United Kingdom. Medically unexplained (somatic) symptoms refer to symptoms that are disproportionate to identifiable physical disease (Sharpe, 2002); the advantages and disadvantages of the concept and its classification are recognised (Francis Creed et al., 2010).
Participant characteristics.
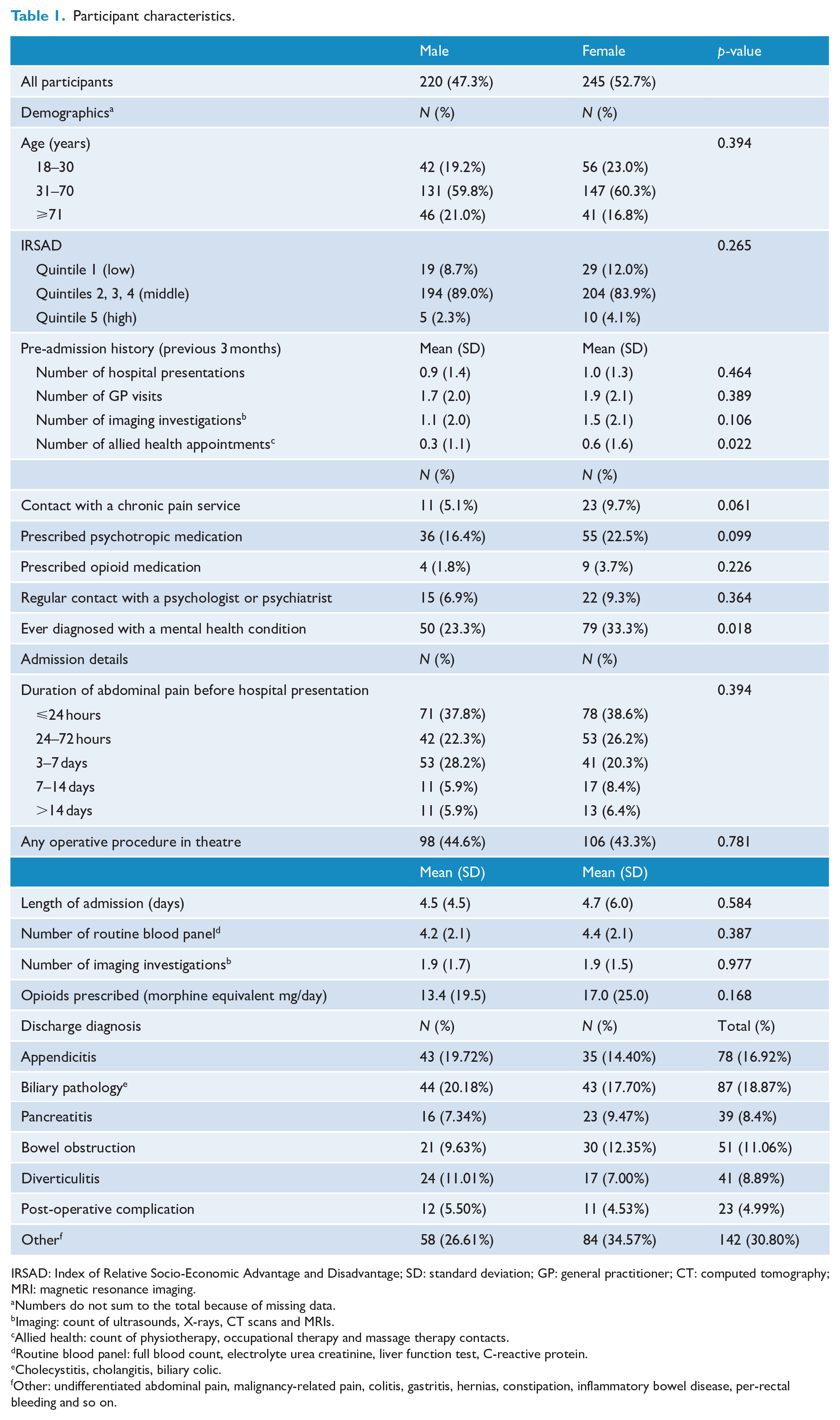
IRSAD: Index of Relative Socio-Economic Advantage and Disadvantage; SD: standard deviation; GP: general practitioner; CT: computed tomography; MRI: magnetic resonance imaging.
Numbers do not sum to the total because of missing data.
Imaging: count of ultrasounds, X-rays, CT scans and MRIs.
Allied health: count of physiotherapy, occupational therapy and massage therapy contacts.
Routine blood panel: full blood count, electrolyte urea creatinine, liver function test, C-reactive protein.
Cholecystitis, cholangitis, biliary colic.
Other: undifferentiated abdominal pain, malignancy-related pain, colitis, gastritis, hernias, constipation, inflammatory bowel disease, per-rectal bleeding and so on.
The classification of somatic disorders is one of the most disputed and controversial areas of psychiatric nosology (Creed, 2006) and so estimating the prevalence of somatic disorders is made difficult by the variety and complexity of diagnostic systems, instruments and populations studied. A recent systematic review of 32 primary care studies of somatoform disorders and medically unexplained symptoms reported a point prevalence of 0.8% for Diagnostic and Statistical Manual of Mental Disorders (4th ed.; DSM-IV) Somatisation Disorder and 38.1% for Diagnostic and Statistical Manual of Mental Disorders (3rd ed., rev.; DSM-III-R) Undifferentiated Somatoform Disorder while having at least one medically unexplained symptom ranged from 40% to 49% (Haller et al., 2015).
Regardless of diagnostic classification, somatic disorders have been variably reported as being more common in females. A systematic review for population studies reported five of six studies associated with female gender; and for primary care studies, eight showed a female association while six did not (Creed and Barsky, 2004). The reported association of somatic syndromes with age is also variable. A systematic review (k = 8 studies) found that somatoform disorders and medically unexplained symptoms are common in later life, with declining prevalence after age 65 years (Hilderink et al., 2013); while in another review, only one in six population studies found an association with older age; and for mostly primary care studies, seven were associated with older age, two with younger age and four found no association with age (Creed and Barsky, 2004).
‘Somatisation’ is associated with higher rates of disability pension and more frequent primary healthcare presentations in clinical populations (Hanel et al., 2009) and greater health care utilisation in community populations (Kohlmann et al., 2016). Somatic symptoms account for over half of all outpatient medical visits, are medically unexplained 33% of the time and are chronic or recurrent in 25% of patients (Kroenke, 2003).
One little studied clinical population is patients with abdominal pain. Improved understanding of the prevalence of somatic symptoms and psychological co-morbidity in this population would be useful in service planning and for the provision of appropriate psychological intervention services.
Aims
The aims of this study, in a surgical inpatient population with abdominal pain, were to:
Estimate the prevalence of somatic symptom severity and psychological co-morbidity;
Compare the prevalence of somatic symptom severity by gender;
Compare the prevalence of somatic symptom severity by gender stratified by age;
Compare the prevalence of psychological co-morbidity by gender.
Methods
Study design and population
This cross-sectional analysis was derived from a prospective cohort study conducted at a 694 adult bed tertiary hospital in NSW Australia (John Hunter Hospital, Newcastle). Consecutive adult (age ⩾ 18 years) patients admitted to the Acute General Surgical Unit (AGSU) with any form of non-traumatic abdominal pain were eligible to participate in the study. Exclusion criteria were participant age less than 18 years, inability or unwillingness to provide informed consent, significant developmental delay or being severely unwell (requiring ICU admission), or any patient with abdominal pain admitted under another speciality e.g. gynaecology, gastroenterology or general medicine. Recruitment was intended to continue until 1000 participants were recruited, however was prematurely terminated due to the hospital-wide suspension of clinical research activity soon after the onset of the COVID-19 pandemic. Only one admission per person during the study period was used for analysis in this study.
Study procedures
This study was approved by the Hunter New England Human Research Ethics Committee Regis SSA Reference No: 2018STE00509 on 20 May 2019.
After admission to the surgical ward, eligible patients were initially approached by a member of the clinical service and if interested in participation, the study and recruitment process was undertaken by either a surgical registrar or resident, who were members of the research team. Participants provided written informed consent for participation in the study, including the collection of personal data from the clinical record and the study instruments. Consenting participants were provided with an envelope containing the study questionnaires. The participants were encouraged to complete the questionnaires on their own if able; and if unable due to visual impairment, poor literacy or other reasons, participants were assisted by a research team member or nursing staff. Upon completion of the questionnaires, participants were encouraged to insert the materials into and seal the provided envelope and return it to research staff prior to discharge, or to mail the envelope to the research team following discharge. If participants wished to withdraw consent, they could do so at any point during their admission and the sealed envelope would be returned. The envelope was not opened during the index admission to maintain blindness of the research and surgical treatment team.
Research measures
Data were extracted for a range of variables, related to the admission and pre-admission period from the clinical records. The study questionnaires included a healthcare utilisation questionnaire developed by MS, which recorded patient-reported health service use variables during the 3 months prior to admission (see Supplementary Figure 1), and three validated mental health instruments. The Patient Health Questionnaire (PHQ) is a multiple-choice self-report version of the Primary Care Evaluation of Mental Disorders (PRIME-MD), which has been used as a screener and as a diagnostic tool for five mental health disorders: depression, anxiety, alcohol, eating and somatoform disorders (Spitzer et al., 1999). We used three sub-scales scored for somatoform (PHQ-15), depressive (PHQ-9) and anxiety (Generalized Anxiety Disorder-7 [GAD-7]) symptom severity and not as diagnostic categories.
Participant characteristics
We reported characteristics in three areas: demographic, pre-admission healthcare use and clinical admission details. Demographic: gender, age (by three sub-groups: 18–30, 31–70 and ⩾71 years) and socioeconomic status (categorical). Socioeconomic status was classified by the Socio-Economic Indexes for Areas (SEIFA) (Pink, 2011) based on postcode. We specifically used the Index of Relative Socio-Economic Advantage and Disadvantage (IRSAD) from SEIFA to categorise participants into five quintiles (1 = low to 5 = high) which we reported at three levels (quintile 1, quintiles 2–4, quintile 5), in order to demonstrate the proportion of patients at the extremes (high and low) of the socioeconomic spectrum in our sample.
Pre-admission service use was determined by participant response to the healthcare utilisation questionnaire. For 3 months prior to the index admission, participants reported the number of general hospital presentations, general practitioner (GP) visits, imaging investigations (continuous); any allied health visits, chronic pain service visits, psychologist or psychiatrist contacts; and history of any mental health diagnosis. We audited the accuracy of participant recall of hospital presentations in the 3 months before the index admission. We selected a sample of n = 20 participants, stratified by PHQ-15 scores ⩾15, to cross check the patient report against the digital institutional records for all hospitals in the Local Health District. Participant recollection was accurate for 19 out of 20 participants (95%). One minor discrepancy was noted of a participant recalling an admission for a procedure followed by a re-admission for a complication of that procedure, recalled as a single encounter. The clinical records were also used to determine any pre-admission medication use including any anti-depressants, anxiolytics and antipsychotics (analysed as a single variable of any psychotropic medications), and any opioid medication (categorical).
In-hospital service utilisation for the index admission was extracted for duration of presenting symptom (abdominal pain), any operative procedure in theatre (categorical), hospital length of stay (LOS) in days, number of routine blood tests (full blood count [FBC], liver function tests [LFT], electrolytes urea creatinine [EUC], C-reactive protein [CRP]), number of imaging investigations (X-ray, computed tomography [CT], magnetic resonance imaging [MRI], Ultrasound) and any opioid use in morphine milligram equivalents (MME) per day (continuous).
Discharge diagnosis (surgical) was extracted from the clinical record and classified into seven groups: Appendicitis, Biliary Pathology (Cholecystitis, Cholangitis, Biliary Colic), Pancreatitis, Bowel Obstruction, Diverticulitis, Post-operative Complication and Other (undifferentiated abdominal pain, malignancy-related pain, colitis, gastritis, hernias, constipation, inflammatory bowel disease, per-rectal bleeding, etc.)
Mental health instruments
The PHQ has sub-scales for somatoform, anxiety and depressive symptoms. Cut-points of 5, 10 and 15 represent mild, moderate and severe symptom levels on all three scales. A cut-point of 10 or greater is considered a ‘yellow flag’ on all 3 measures (possible clinically significant condition), while a cut-point of 15 is a considered a ‘red flag’ on all 3 measures (active treatment is probably warranted). A systematic review of the PHQ concluded that the PHQ-9, GAD-7 and PHQ-15 are brief well-validated measures for detecting and monitoring depression, anxiety and somatisation (Kroenke et al., 2010). The PHQ-15 has been validated as a screener for somatisation independent of medical co-morbidity (Barsky et al., 2005).
PHQ-15
The PHQ-15 is a 15-item subscale, which asks about 15 somatic symptoms commonly reported in outpatient populations. The items include the 15 most prevalent somatic symptoms (Kroenke et al., 2002) for DSM-IV Somatisation Disorder and include 4 pain symptoms – 2 gastrointestinal, 1 sexual and 1 pseudo-neurological symptom (Rockville, 2016). The PHQ-15 has also been shown to be a useful screener for DSM-5 SSD (Toussaint et al., 2020).
Patients are asked to rate how much they have been bothered by each symptom during the past month on a 0 (‘not at all’) to 2 (‘bothered a lot’) scale, for a total score from 0 to 30.
Cut-points of 5, 10 and 15 represent thresholds for mild, moderate and severe somatic symptom severity (Kroenke et al., 2010; Zijlema et al., 2013). The ⩾10 cut-point is the most frequently used: to report prevalence of ‘somatisation syndromes’ (Kocalevent et al., 2013), as a validated screener (de Vroege et al., 2012) and in diagnostic accuracy studies (Korber et al., 2011; van Ravesteijn et al., 2009). An alternative scoring of three or more symptoms scored as ‘bothered a lot’ has been used as a screener for the classification of ‘Multisomatoform’ disorder (Kroenke et al., 1997).
We reported ‘prevalence’ of somatic symptoms using three PHQ-15 cut-points: total score ⩾10 (moderate severity), ⩾15 (severe) and ‘bothered a lot’ on any 3 (or more) questions. We used the unadjusted total scores for males and females, without any differential accounting for item 4 ‘Menstrual cramps or other problems with your periods’, which may be optionally only scored for females.
PHQ-9
The PHQ-9 is a 9-item instrument that scores each of the DSM-IV diagnostic criteria for a Major Depressive Episode. For the preceding 2 weeks, each item is rated as a ‘0’ (not at all) to ‘3’ (nearly every day). Using the ⩾10 cut-point had a reported sensitivity of 88% and specificity of 88% for Major Depressive Episode (Kroenke et al., 2001). However, a recent individual patient meta-analysis in various populations showed that the pooled PHQ-9 ⩾ 10 prevalence (25%) was double that of pooled Structured Clinical Interview for the Diagnostic and Statistical Manual-IV Axis I Disorders (SCID) Major Depression prevalence (12%) (Levis et al., 2020), which suggest that the PHQ-9 might be best understood as a screener for a possible Major Depressive Episode in clinical populations. We used the PHQ-9 cut-points of ⩾10 to estimate ‘prevalence’ of Major Depressive Episode, reported as ‘depressive symptoms’.
GAD-7
The GAD-7 is a 7-item instrument developed to measure Generalized Anxiety Disorder (GAD) symptoms, which has established diagnostic accuracy for panic disorder, social anxiety and post-traumatic stress disorder (PTSD; Kroenke et al., 2010; Zijlema et al., 2013). Patients are asked how often, during the last 2 weeks, they were bothered by each symptom, with response options of ‘not at all’, ‘several days’, ‘more than half the days’ and ‘nearly every day’ scored as 0, 1, 2 and 3, respectively. A cut-point of ⩾10 has been shown to have a sensitivity of 89% and specificity of 82%, using the SCID for DSM-IV GAD (Spitzer et al., 2006). We used the GAD-7 cut-points of ⩾10 to estimate ‘prevalence’ of GAD, reported as ‘anxiety symptoms’.
Statistical analyses
Participant characteristics were disaggregated by gender and reported as number and percentage for categorical variables, and as means and standard deviation (SD) for continuous variables.
We calculated the proportion of patients with somatic symptoms, at three PHQ-15 cut-points, stratified by sex and age group. We also calculated the proportion of patients with somatic symptoms and anxiety and depression co-morbidities, stratified by gender. For each of these analyses, we calculated odds ratio (OR) and 95% confidence intervals (95% CIs) of the female vs male comparisons. Finally, we graphically examined the somatic symptom patterns endorsed by males and females, calculating the proportion of patients who were ‘bothered a little’ and ‘bothered a lot’ for each of the PHQ-15 items.
Results
Seven hundred and thirty-one eligible admissions were invited to participate in the study and 590 questionnaires were returned (participation rate 80.7% of admissions). After removal of 96 very incomplete data sets (no responses recorded) and 29 repeat admissions (only index admissions were used), 465 data sets were used in the final analysis of this study (response rate 63.6% of eligible admissions and 82.9% of participating individuals).
Participant characteristics
Of the 465 participants, 53% were female, 60% aged 30–70 years and 86% from the middle socioeconomic quintiles (Q2–Q4). Health service utilisation in the 3 months before admission showed a mean of just under one hospital admission, less than two GP visits, 1.3 imaging investigations and 0.5 allied health visits. 7.5% had contact with a pain service and 8.2% had contact with a psychologist or psychiatrist. Prescription of psychotropic medications was reported by 22.5% of females and 16.4% of males; prescription for opioids was reported by 3.7% of females and 1.8% of males. A mental health diagnosis was self-reported for more females (33.3%) than males (23.3%), which was the only statistically significant gender difference.
Most of the participants had a short duration of abdominal pain before admission (less than 72 hours), females 64.8% and males 60.1%. Nearly half had an operative procedure, with a mean LOS under 5 days. Participants received a mean of 4.3 routine blood panels (FBC, EUC, LFT, CRP) and 1.9 forms of imaging investigation (Ultrasound, CT scan, X-ray or MRI) throughout their admission. A mean of 15.3 MME per day was prescribed, equivalent to 20 mg of oxycodone. The most common surgical discharge diagnoses were appendicitis, biliary pathology and ‘other’. Details can be seen in Table 1.
Prevalence of somatic symptoms
Moderate somatic symptoms occurred in over half of the participants, with a significant female predominance (odds ratio [OR] = 1.71; 95% CI = [1.18, 2.48]), with a similar pattern for ⩾3 symptoms ‘bothered a lot’ (2.07; [1.42, 3.03]). Severe somatic symptoms occurred in 20% of participants with no gender difference. A similar pattern of results was seen in 31–70 years sub-group with a female predominance for moderate symptoms (1.69; [1.05, 2.73]) and for ⩾3 symptoms ‘bothered a lot’ (1.99; [1.23, 3.24]). With much smaller sample sizes and wider CIs, the 17- to 0-year sub-group had a similar pattern to the overall population; the ⩾71 sub-group had no significant gender differences and severe somatic symptoms in only 13%. Details can be seen in Table 2.
Somatic symptom prevalence and gender differences at different PHQ-15 cut-points and by age sub-groups.
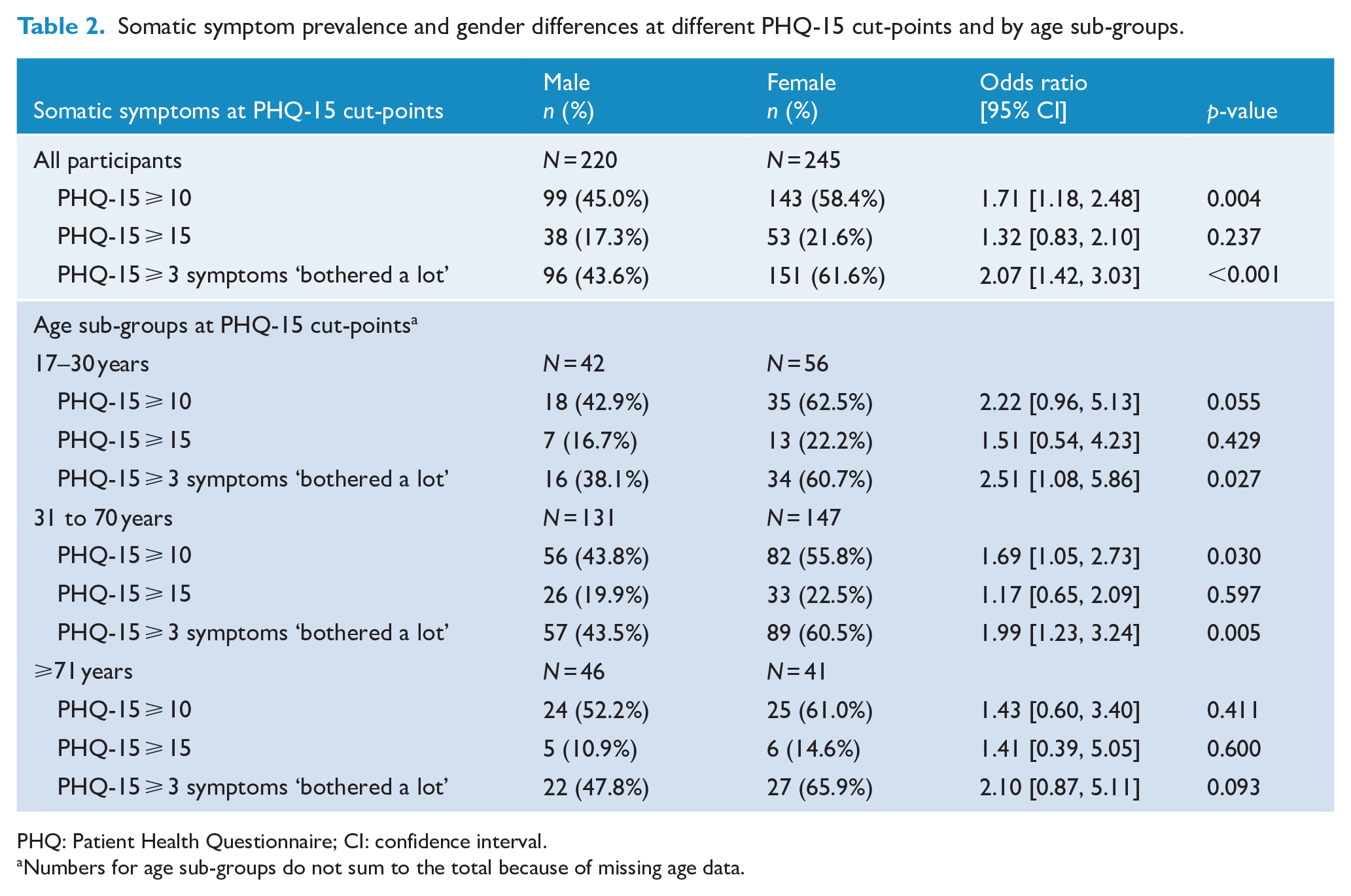
PHQ: Patient Health Questionnaire; CI: confidence interval.
Numbers for age sub-groups do not sum to the total because of missing age data.
Psychological co-morbidity
The prevalence of depressive symptoms (PHQ-9 ⩾ 10) was 33.1%, anxiety symptoms (GAD-7 ⩾ 10) was 21.7% and for both was 17.2%. Co-morbidity of depressive, anxiety and somatic symptoms (triple co-morbidity) ranged from 8.2% to 15.9%. There were no gender differences on any co-morbidity combinations. Details can be seen in Table 3. A graphical representation of the pattern of co-morbidity can be seen in the Venn diagrams for females in Figure 1(a) and males in Figure 1(b).
Prevalence and co-morbidity of depressive, anxiety and somatic symptoms at PHQ-15 cut-points.
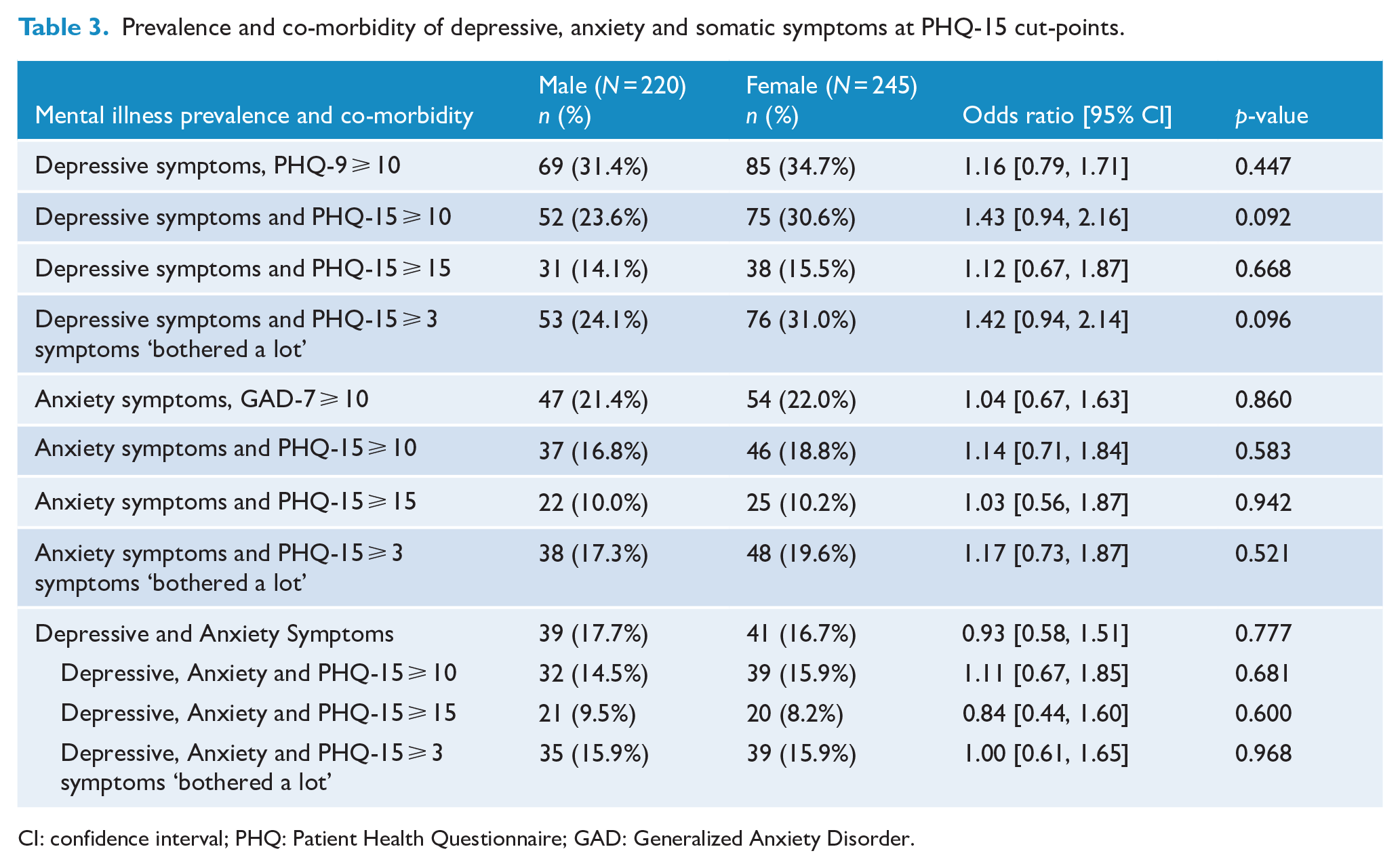
CI: confidence interval; PHQ: Patient Health Questionnaire; GAD: Generalized Anxiety Disorder.
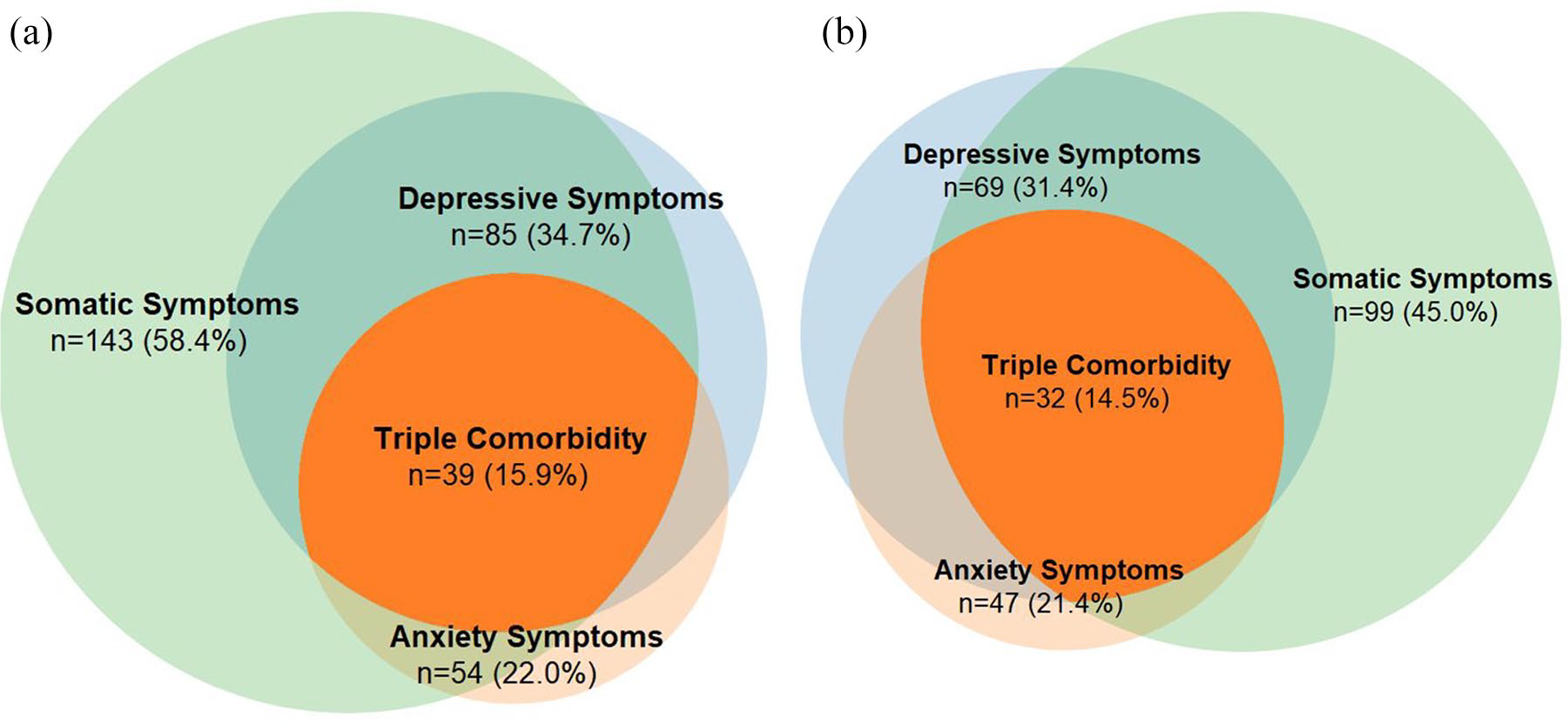
(a) Female: co-morbidity of anxiety (GAD-7 ⩾ 10), depressive (PHQ-9 ⩾ 10) and somatic symptoms (PHQ-15 ⩾ 10). (b) Males: co-morbidity of anxiety (GAD-7 ⩾ 10), depressive (PHQ-9 ⩾ 10) and somatic symptoms (PHQ-15 ⩾ 10).
Individual item endorsement for somatic symptoms
Figure 2 shows the proportion of participants scoring ‘bothered a little’ or ‘bothered a lot’ for each of the PHQ-15 items. The least frequently endorsed items at around 10% were ‘fainting spells’ and ‘pain or problems during sexual intercourse’ and the most frequently endorsed items at over 80% were ‘feeling tired or having low energy’ and ‘stomach pain’. The gender pattern was similar on the 14 items scored for both genders. Details can be seen in Figure 2.
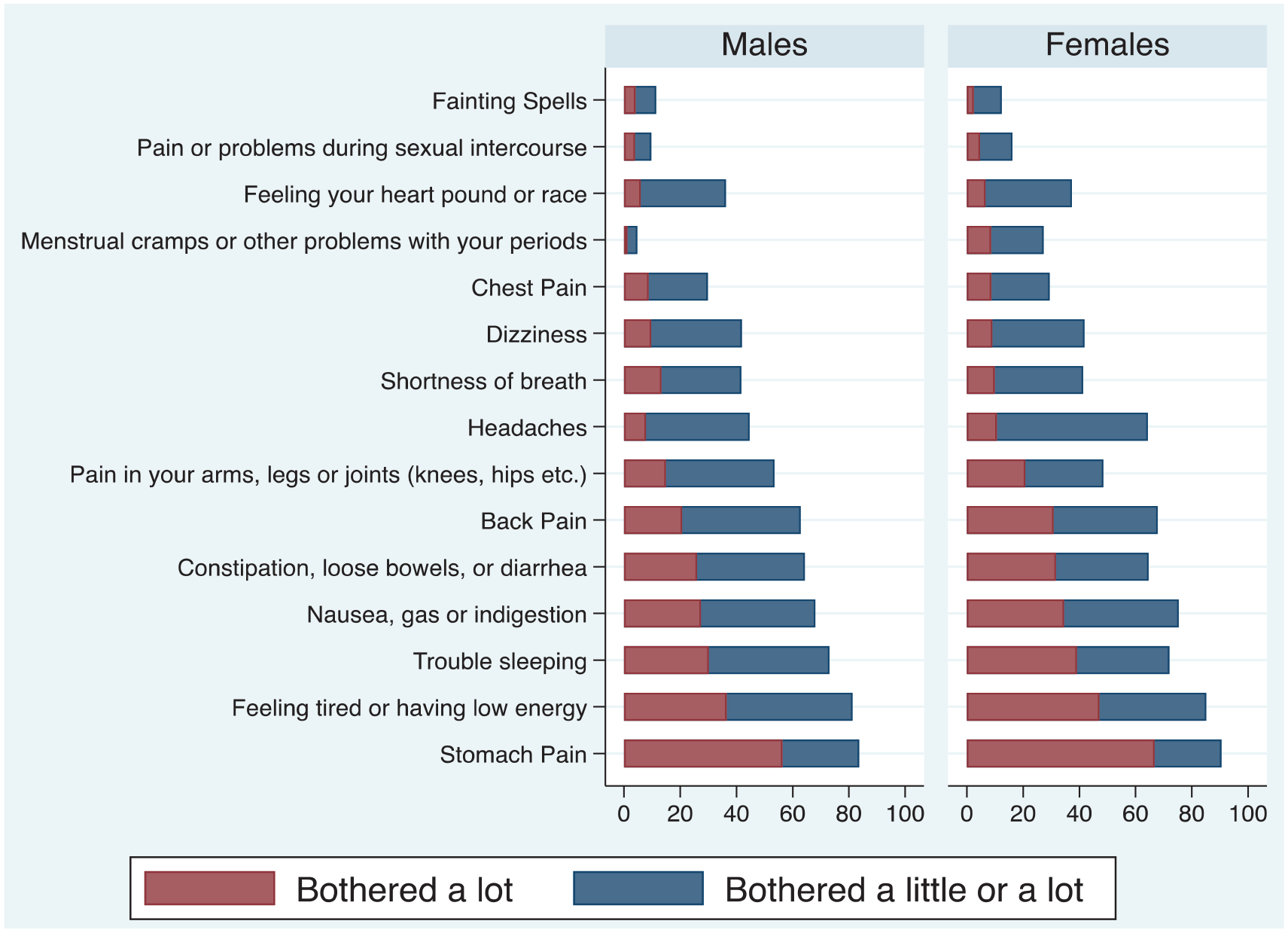
PHQ-15 individual item profile for males and females.
Discussion
Main findings
Clinically significant somatic symptoms (moderate severity or ⩾ 3 symptoms ‘bothered a lot’) are very common in this surgical population (over 50%), with a severe level of somatic symptoms for one in five and psychological co-morbidity on three measures for about one in six participants. Higher PHQ-15 scores have been associated with increased functional impairment, healthcare utilisation and symptom-related difficulty in activities and relationships, with scores ⩾15 reflecting considerable impairment and burden (Kroenke et al., 2002). PHQ-15 ⩾ 3 symptoms ‘bothered a lot’ has been associated with impairment in health-related quality of life, increased healthcare utilisation, more disability days and healthcare provider frustration (Kroenke et al., 1997).
Our results suggest a substantial level of need for further psychological assessment and intervention, which is probably not routinely offered during surgical inpatient admission.
Recommendations for the organisation of optimal clinical services include a multi-disciplinary team, with a re-conceptualisation of the nature of pain and pain management, avoidance of dichotomous biomedical explanations of mind and body, and a broad treatment plan encompassing biological and psychosocial factors (Henningsen, 2018). Inpatient treatment regimens should focus on symptomatic measures, physiotherapy, occupational therapy and psychotherapy (Henningsen, 2018). A recent call for a review of the approach to functional somatic syndromes presenting in various health settings identified several needs, including finding acceptable categories and labels for patients that promote a therapeutic partnership and the development of clinical services and research for people with severe disorders (Burton et al., 2020). Systematic reviews have identified effective specific treatments for somatoform disorders including cognitive behavioural therapy, antidepressant medications and a consultation letter advocating a management plan to the primary care physician. The primary outcomes from these intervention studies demonstrated benefits for somatic symptom scores, functional status, psychological distress and depression. For the secondary outcome of health care use and/or costs, 10 of the 11 studies assessing this outcome showed reduction in health care use (k = 5 trials) or costs (k = 5 trials) (Kroenke, 2007). We would suggest that improved case identification, patient acceptance and access to evidence-based interventions are warranted to reduce symptoms, improve function and reduce costs.
Prevalence rates
There are few similar studies of inpatient populations for direct comparison. Approximately half of our population experienced moderate somatic symptom burden, and one in five experienced severe somatic symptom burden. Medical inpatients (n = 392) screened using the Schedules Assessment in Neuropsychiatry (SCAN) interview reported 18.1% for an International Classification of Diseases, 10th Revision (ICD-10) somatisation disorder and 20.2% for DSM-IV somatisation disorder (Fink et al., 2004). Chinese general hospital inpatients screened using the PHQ-15 found 27.8% had a score greater than 10 (Zhang et al., 2016). The reason for the substantially higher somatic symptom burden in our inpatient population remains unclear.
Case identification and PHQ-15 scoring
We used the PHQ-15 to estimate the prevalence of somatic symptoms and did not use a diagnostic instrument or psychiatrist clinical assessment to determine specific diagnoses. The PHQ-15 can be used as a screener but has limitations for use as a diagnostic instrument (Fink, 2017), and a specific diagnosis is traditionally used as the basis to allocate treatment. The ⩾10 cut-point is the most frequently reported and has been used in diagnostic accuracy studies to yield a sensitivity of 80.2% and specificity of 58.5% for detecting somatoform disorders in the primary care setting (Korber et al., 2011). The sensitivity and specificity of the PHQ-15 at detecting somatoform disorders was found to be 78% and 71%, respectively, for the cut-point of PHQ-15 ⩾3 symptoms ‘bothered a lot’, using the Structured Clinical Interview for the Diagnostic and Statistical Manual IV Axis I disorders (SCID-I) as the reference standard (van Ravesteijn et al., 2009). PHQ-15 ⩾3 symptoms ‘bothered a lot’ was validated against the Diagnostic Interview Schedule (DIS), with a positive predictive value of 0.88% for DSM-IV Somatisation Disorder (Kroenke et al., 1998).
In our study, the prevalence estimates for PHQ-15 ⩾ 10 and ⩾ 3 symptoms ‘bothered a lot’ were similar, suggesting roughly equivalent prevalence for a ‘diagnosis’ of DSM-IV somatisation disorder or multisomatoform disorder, if using the PHQ-15 and these cut-points alone.
Psychological co-morbidity
Depressive and anxiety symptoms were common: depressive in 33.7% and anxiety in 21.7%. Co-morbidity with somatic symptoms was also common, around one in six participants scored over threshold (⩾10) for all three screener measures, which was greater than was reported in a large study of 3034 primary care patients screened using the PRIME-MD (depression 35.8%, anxiety 25.6%, somatoform 28.8%) with the ‘triple prevalence’ of 11.5% (Roca et al., 2009).
It is also important to recognise that abdominal pain, hospital admission, investigation and pending operation can cause warranted and non-pathological psychological distress, which may influence responses to depressive and anxiety symptom scores. The duration of abdominal pain was less than 72 hours for around 60% of the participants and recruitment was soon after admission for most, while the PHQ-9 and GAD-7 instruments refer to symptoms during a period of the preceding 2 weeks. We would expect that some of psychological co-morbidity prevalence estimates might be inflated by these non-pathological sources of psychological distress.
Female predominance of somatic symptoms
In our study, somatic symptom prevalence, psychological co-morbidity and somatic symptom patterns for females and males had few differences. We did find a female predominance of moderate symptom severity and ⩾3 ‘bothered a lot’, but no difference for severe somatic symptoms. Somatic symptom burden is often reported to be significantly higher in females than in males. One of the strongest early studies suggested gender was the most important demographic factor associated with symptom reporting even after adjusting for psychiatric co-morbidity. All somatic symptoms except one were reported more commonly by women, with the adjusted ORs (typically in the 1.5–2.5 range) (Kroenke and Spitzer, 1998). A review of mostly clinical populations in the last half of the 20th century found that females ‘somatise’ more than males; however, the authors suggest the results were based on largely flawed studies, characterised by unaccounted for confounding and observer bias (Wool and Barsky, 1994).
Many possible explanations have been offered for apparent female predominance. A systematic review of sex differences found higher pain prevalence and more sensitivity to experimentally induced pain in females (Fillingim et al., 2009) while oestrogen in females may increase painful stimuli experience (Goulden, 2019). Women may be more likely to report multiple somatic complaints, rather than experience more symptoms (Francis Creed, 2004; Wool and Barsky, 1994); and the societal stereotype that women tend to be more emotional, and thus more prone to catastrophising and hypervigilance, lends itself to attributing women’s physical experience to psychiatric illness by clinicians (Leung, 2012).
PHQ-15 studies have reported the same cut-points for both men and women despite one item referring to menstrual pain. No studies have clarified whether this unadjusted scoring results in inaccurate estimates of somatic symptoms in women. Notably, the PHQ-15 is a clinician-independent symptom severity score, reflecting the participant’s experience of symptoms. In this way, clinician bias in ‘diagnosis’ is removed, suggesting any female predominance might be attributed to differential symptom reporting or psychological or neurobiological differences in symptom experiences. It remains unclear whether the reported increased rates of somatic symptoms and disorders in females is a result of diagnostic practices, societal stigma or measurement bias; or whether it is related to genuine gender differences in the psychological and neurobiological experience of pain and other symptoms.
Somatic symptoms and age
Older participants (⩾71 years) had similar prevalence of moderate symptom severity compared to the population < 71 years, but had lower prevalence of high symptom severity. A systematic review of somatisation in older populations showed that in general, the prevalence of somatoform disorders in older age groups (⩾65 years) was 1.5–13% and the prevalence of medically unexplained symptoms was 4.6–18%, considerably lower than the rates for those under 65 years (Hilderink et al., 2013). The PHQ-15 has been validated as a screening tool for identifying ICD-10, DSM-IV and DSM-5 somatic disorders; however, these studies assessed validity across a diversely aged population and did not focus on elderly populations (Kroenke et al., 2010; Zijlema et al., 2013).
The lower reported prevalence of somatic symptoms in elderly populations has been thought to be due to a greater likelihood of attributing symptoms to underlying medical conditions and poorer recognition of somatic psychopathology. A systematic review of 40 somatic symptom questionnaires attempted to find the optimal screening questionnaire for elderly populations. The PHQ-15 score was not significantly associated with the presence of a somatoform disorder or with the severity of health anxiety in the elderly population. The authors concluded that the PHQ-15 had considerable overlap with common geriatric symptoms and probably overestimates the level of somatisation in older persons (van Driel et al., 2018).
Conclusion
This study highlights the high prevalence of clinically significant and under-recognised psychopathology in surgical inpatients with abdominal pain, a population which has been under-represented in previous studies exploring the prevalence of somatic symptoms. The PHQ-15 is a valid and easily applied screening instrument that can assess somatic symptom burden in this population, although it may not be optimal for patients over 65 years. Recognition of somatic symptoms in surgical inpatients is a necessary step before initiation of multi-disciplinary team treatment regimens, implementation of evidence-based specific interventions and primary care physician involvement, which could potentially reduce patient morbidity, tertiary care admissions and healthcare burden.
Strengths and limitations
This study of somatic symptoms in consecutive adult surgical inpatients used a cross-sectional analysis to estimate the prevalence and co-morbidity of somatic symptoms in an infrequently studied population. Our study used broad inclusion criteria, a large sample size and validated instruments for the key outcomes. Clinician treatment decisions were made blinded to results.
We initially planned to recruit 1000 participants, but recruitment was terminated prematurely due to the compulsory cessation of non-essential research activities across the local health district (LHD) due to the impact of COVID-19 during 2020. As a result, some of our analysis, especially when somatic symptom severity and psychological co-morbidity were stratified by age and gender, may be underpowered.
Screening for somatic symptoms while a patient is acutely unwell may lead to overestimation of prevalence, particularly for abdominal pain. The personally sensitive nature of the study instrument questions may lead to response bias. The validity of using the PHQ-15 in older population is questionable. We did not estimate long-term or lifetime prevalence of symptoms or estimate quality of life or function. We did not use any structured interviews or psychiatrist assessments to produce estimates of any specific somatic disorders. Our self-report measure of 3-month service utilisation has not been validated.
The recruitment processes meant some very brief overnight admissions may have been missed and some patients who were very unwell, especially those needing ICU, may have been excluded. We also excluded any patients whose hospital treatment was entirely within the Emergency Department without being admitted to a ward bed. This truncation of participants at both extremes, the well enough to go home almost immediately, and the very unwell, may have skewed the results of the study in an unpredictable manner and any generalisation of the results to these populations is not warranted. The study question arose from the general surgeons who were interested in providing improved care to the abdominal pain patients under their care. Generalisation of our results to other clinical populations (e.g. gastroenterology abdominal pain) should be done with caution.
Supplemental Material
sj-pdf-1-anp-10.1177_00048674211044639 – Supplemental material for Using the Patient Health Questionnaire to estimate prevalence and gender differences of somatic symptoms and psychological co-morbidity in a secondary inpatient population with abdominal pain
Supplemental material, sj-pdf-1-anp-10.1177_00048674211044639 for Using the Patient Health Questionnaire to estimate prevalence and gender differences of somatic symptoms and psychological co-morbidity in a secondary inpatient population with abdominal pain by Melissa Stieler, Peter Pockney, Cassidy Campbell, Vaisnavi Thirugnanasundralingam, Lachlan Gan, Matthew Spittal and Gregory Carter in Australian & New Zealand Journal of Psychiatry
Footnotes
Acknowledgements
The authors would like to thank and acknowledge the hard work of Angela Smith, the Calvary Mater Hospital Liaison Librarian, for her tireless assistance with our literature review. They would also like to thank all the staff at the John Hunter Hospital who supported this research project through patient support, assistance and collection of research materials. Finally, they wish to send their deepest thanks to the participants of this study, who selflessly gave their time and energy to this research.
Author contributions
M.S. contributed to conceptualisation; investigation; methodology; project administration; visualisation; writing – original draft; and writing – review and editing. C.C. contributed to investigation and writing – review and editing. V.T. contributed to investigation and writing – review and editing. L.G. contributed to investigation and writing – review and editing. P.P. contributed to conceptualisation; methodology; supervision; and writing – review and editing. M.S. contributed to data curation; formal analysis; visualisation; and writing – review and editing. G.C. contributed to conceptualisation; investigation; methodology; project administration; resources; supervision; visualisation; and writing – review and editing.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by the University of Newcastle School of Medicine and Public Health Higher Research Degree Student Support Allocation Funding: $1500. Matthew Spittal is a recipient of an Australian Research Council Future Fellowship (project number FT180100075) funded by the Australian Government.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
