Abstract
Objectives(s):
To characterise the clinical profile, aetiology and treatment responsiveness of ‘Australian Lyme’, or Debilitating Symptom Complexes Attributed to Ticks.
Methods:
Single-centre retrospective case analysis of patients referred to the Infectious Diseases Unit at Austin Health – a tertiary health service in Heidelberg, Australia – between 2014 and 2020 for investigation and treatment of suspected Debilitating Symptom Complexes Attributed to Ticks. Patients were included if they had debilitating symptoms suggested by either themselves or the referring clinician as being attributed to ticks.
Results:
Twenty-nine Debilitating Symptom Complexes Attributed to Ticks cases were included in the analysis. Other than Lyme disease (83%), the most common prior medical diagnoses were Epstein–Barr virus (38%), chronic fatigue syndrome (28%) and fibromyalgia (24%). Prior histories of anxiety (48%) and depression (41%) were common. The most frequently reported symptoms included fatigue (83%), headache (72%) and arthralgia (69%). National Association of Testing Authorities/Royal College of Pathologists of Australasia–accredited serology was not diagnostic of acute infective causes, including Lyme disease, in any patient. Of 25 cases with available data, 23 (92%) had previously been prescribed antimicrobials, with 53% reporting benefit from them. The most common diagnoses made by our hospital were chronic fatigue syndrome (31%), migraines (28%) and fibromyalgia (21%). Only one patient’s symptoms were not accounted for by other diagnoses.
Conclusion:
This is the first case series of patients with Debilitating Symptom Complexes Attributed to Ticks. They had high rates of other medically unexplained syndromes, and no evidence of acute Lyme disease, or any common organic disease process. Debilitating Symptom Complexes Attributed to Ticks remains medically unexplained, and may therefore be due to an as yet unidentified cause, or may be considered a medically unexplained syndrome similar to conditions such as chronic fatigue syndrome.
Introduction
Debilitating Symptom Complexes Attributed to Ticks (DSCATT) is the diagnostic label adopted by the Australian Government in 2018 to describe a chronic syndrome of unclear aetiology affecting a cohort of Australians, commonly associated with tick bites and often believed to represent a form of Lyme Disease (Australian Government Department of Health, 2020; Commonwealth of Australia, 2016). While over 500 cases of this Australian ‘Lyme-like’ illness have been reported in the scientific literature (Australian Government Department of Health, 2020; Chalada et al., 2016; Collignon et al., 2016; Royal College of Pathologists of Australasia [RCPA], 2014), there is no evidence that the bacterial complex that causes acute Lyme disease – Borrelia burgdorferi sensu lato (B. burgdorferi s.l.) – exists in Australian ticks (RCPA, 2014).
Borrelia serology has low positive predictive value in non-endemic areas, with diagnoses of Lyme borreliosis among people in non-endemic areas often questionable due to a reliance on overseas laboratories that do not meet Australian standards (Chalada et al., 2016). This has rendered the aetiology of DSCATT open to controversy and speculation – whether it represents an infection by B. burgdorferi, an alternative tick-borne pathogen or an entirely different disease process (Collignon et al., 2016; Feder et al., 2007). The issue is complicated by the uncertain relationship, even in Lyme-endemic areas, of the ‘chronic Lyme disease’ syndrome with B. burgdorferi s.l. serology (Feder et al., 2007).
The Australian Government has not supported the syndrome as a locally acquired Lyme disease (Australian Government Department of Health, 2020). However, in response to patient advocacy groups (Brown, 2018; Lyme Disease Association of Australia, 2020), it formed the Clinical Advisory Committee on Lyme Disease in 2013, to ‘further research into identifying and characterising a causative agent in Australia’ (Clinical Advisory Committee on Lyme Disease, 2013). Following a 2015 Senate inquiry, the National Health and Medical Research Council released funding for this under the department of health’s aetiologically neutral new name, ‘Debilitating Symptom Complexes Attributed to Ticks’ (Brown, 2018; Commonwealth of Australia, 2016; National Health and Medical Research Council, n.d.).
Although DSCATT now has a name, its nature remains undefined. A 2020 review commissioned by the Australian Department of Health found no peer-reviewed published epidemiological or clinical studies of DSCATT in Australia (Allen & Clarke, 2020). Here, we present the first case series of patients with DSCATT, in an attempt to characterise the clinical profile, aetiology and treatment responsiveness of DSCATT.
Methods
We conducted a retrospective audit of the electronic medical records of patients referred to the Austin Health Infectious Diseases Unit between 2014 and 2020 for investigation and treatment of suspected DSCATT (Ethics approval: Audit/19/Austin/11). Patients were excluded from the analyses if they either did not have debilitating symptom complexes or did not have symptoms suggested by either themselves or the referring clinician as being possibly attributed to ticks.
The study protocol for data extraction and classification was piloted for five randomly selected cases which were independently extracted by two authors (G.O. and J.S.) and compared with disagreements reconciled by R.A.K. and data definitions established. These were iteratively tested on further randomly selected cases by G.O. and J.S. independently until 90% concordance was achieved (the final concordance achieved was 94%, after a total of seven cases) and a ‘data dictionary’ finalised (Supplement 1). The remaining 28 possible cases were then extracted by either of the authors (G.O. and J.S.) in accordance with the developed data dictionary.
Prior psychiatric and medical diagnoses were recorded if they were listed as confirmed diagnoses in the medical record, or described as a confirmed diagnosis by a medical officer, anywhere in cases’ medical records prior to the period of care under our infectious diseases service. Outcome variables, including clinical presentation, investigation findings, management and clinical assessments or new diagnoses made by any medical officer during the period of care under our infectious diseases service, were recorded based on the electronic medical records as ‘confirmed’, where records definitively identified a positive finding; ‘suspected’, where clinical records suggested but did not definitively identify a positive finding; ‘unknown/not assessed’ where data were not available; or ‘negative’, where records definitely excluded a positive finding. Specifically, the occurrence of a tick bite was coded ‘confirmed’ if the patient witnessed/reported a tick during the event. The occurrence of the characteristic erythema migrans rash was classified as ‘confirmed’ if the patient self-reported it, or it was observed or documented by a clinician.
In addition to the assessments reported by the treating clinicians, the serological and radiological data were reviewed by infectious diseases (K.B.G.) and neurology (R.M.) consultant authors, respectively, as noted in the results. Diagnostic laboratory testing guidelines proposed by the Royal College of Pathologists of Australasia (RCPA) were used to confirm or exclude diagnoses of Lyme disease; specifically, a diagnosis of Lyme disease required a positive enzyme immunoassay (EIA) (a highly sensitive screening test) followed by a positive immunoblot assay (a more specific test) from a National Association of Testing Authorities (NATA)/RCPA-accredited laboratory, where assays generally incorporate known specific antigens from both European and American stains of Borrelia burgdorferi s.l. known to cause Lyme Disease (RCPA, 2014). Consistent with the US Association of Public Health Laboratories (2021) guidance, for samples collected over 30 days post symptom onset, only the IgG results were interpreted. The medical and psychiatric diagnoses listed in medical records were defined as ‘active’ if they were either acutely symptomatic conditions or chronic conditions that were ongoing. Where detailed in their medical record, childhood adversity was defined as occurring prior to age 18 years, while adult trauma was defined as occurring after age 18, whether prior to or following the onset of DSCATT symptoms.
Symptoms were grouped into nine categories based on bodily systems prior to data extraction. Following data extraction, qualitative and quantitative data analyses were performed. Where multiple laboratory results for a single patient were available for the same investigation (e.g. C-reactive protein [CRP]) over the duration of care, the maximum value was used. Data were analysed using Microsoft Excel (Version 16.46, 2021 Microsoft).
Results
Of 35 possible DSCATT cases identified, 29 were included in the data analysis. The six excluded patients did not have debilitating symptom complexes, and five did not attribute their symptoms to ticks.
Case demographics
Of the 29 included cases, nearly two thirds (n = 18, 62%) were female. Age at presentation ranged from 18 to 73 years (median, 42 years). Over half (n = 16, 55%) were referred by a local ‘Lyme-literate’ general practitioner. Cases spent an average of 10 months under the care of our health service (median = 5 months, range = 1–49 months).
Medical and psychosocial history
The stated reason for referral in 16 out of 24 cases was a specialist opinion and/or management for ‘borreliosis’, ‘Lyme disease’ or ‘Lyme-like illness’. Other than a recorded (but not laboratory confirmed) diagnosis of Lyme Disease (n = 24; 83%), the most commonly reported prior medical and psychiatric diagnoses included a range of functional syndromes and affective and mood disorders (Figures 1 and 2). Notably, one case had a recently recorded diagnosis of mild Guillain–Barre Syndrome following a hospital admission with flu-like symptoms. The median number of medical and psychiatric diagnoses per case, as a subset of all recorded diagnoses, considered to be active conditions, was 3 (range = 0–25), while the median number of active psychiatric diagnoses was 1 (range = 0–5).
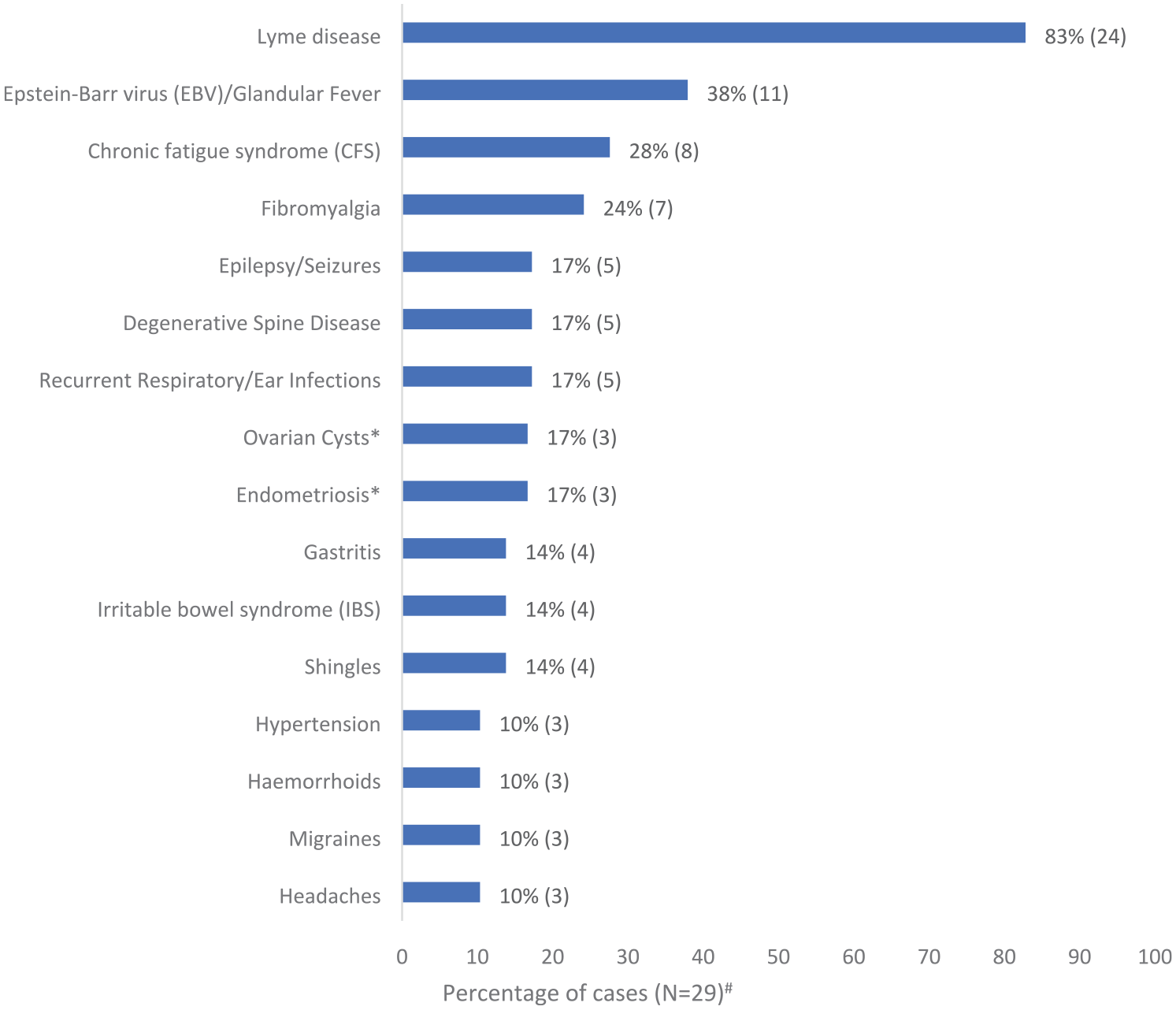
Commonly recorded medical diagnoses in DSCATT cases’ medical records.
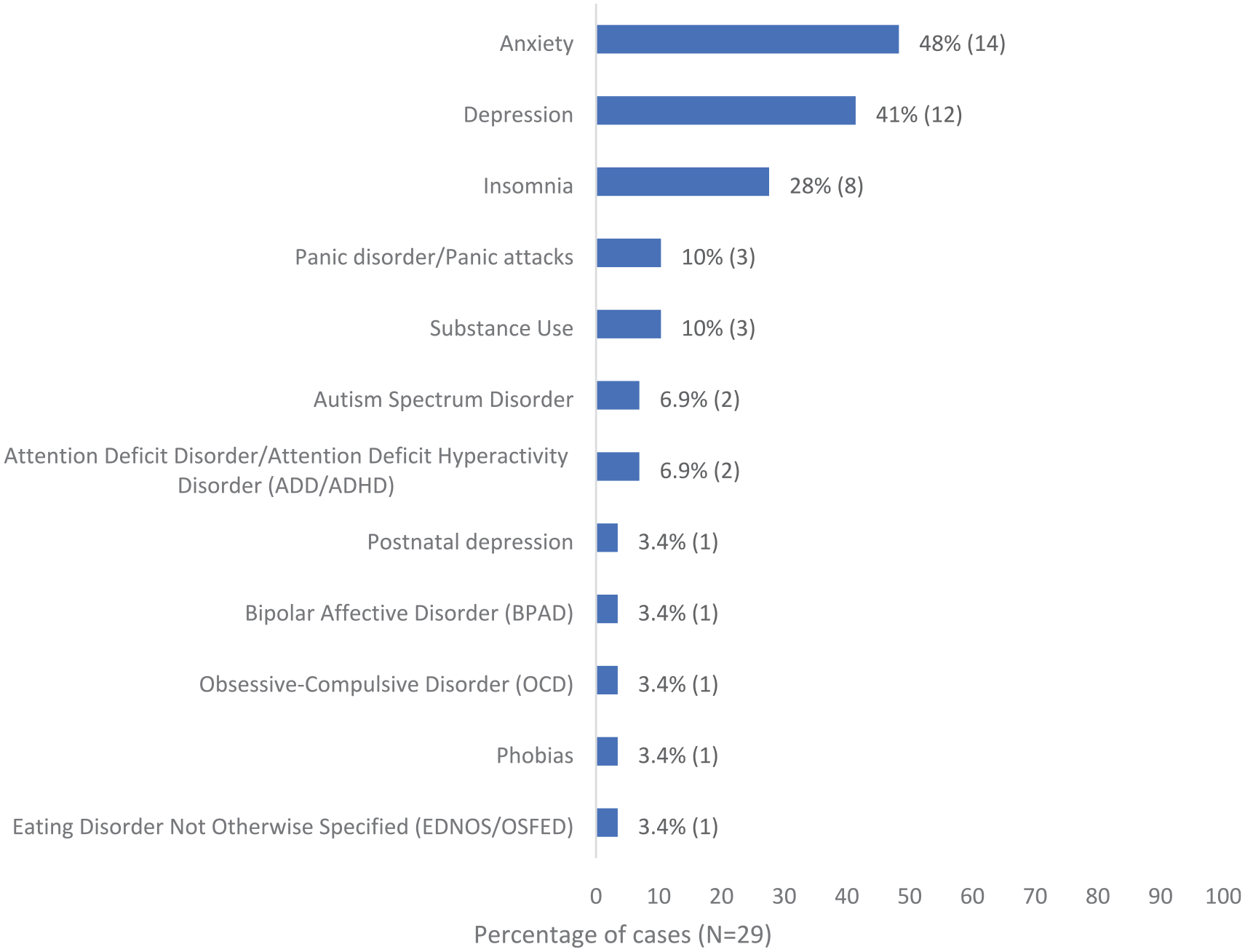
Commonly recorded psychiatric diagnoses in DSCATT cases’ medical records.
Nearly one quarter (n = 7, 24%) of cases had a confirmed current or past smoking history. One third (n = 9, 31%) of cases had a confirmed or suspected significant stressor preceding their illness, including familial illness or carer stress (n = 3, 10%); physical injury or illness (n = 2, 6.9%); or financial stress, workplace stress and relationship stress (n = 1 each). Nearly one quarter of cases (n = 7, 24%) had a history of childhood adversity, including death, illness or disability of a loved one (n = 5, 17%); bullying, harassment or assault at school (n = 3, 10%); parental alcoholism and/or domestic abuse (n = 2, 6.9%); and parental divorce or familial breakdown (n = 2, 6.9%). Two cases presented with symptoms similar to those of immediate relatives who had previously received diagnoses with Lyme disease and chronic brucellosis, respectively.
Commonly reported risk factors for tick-borne disease included overseas travel (n = 20, 69%), with 41% of cases (n = 12) reporting travel to areas with endemic Lyme disease, primarily North America and Europe. Only 38% (n = 11) of cases reported receiving a tick bite; of these, 45% (n = 5) were sustained overseas. Over one third of cases (n = 12, 41%) reported a history of other bites, most commonly from insects (mosquitoes, ants, bed bugs, mites), spiders, monkeys or dogs.
Clinical presentation
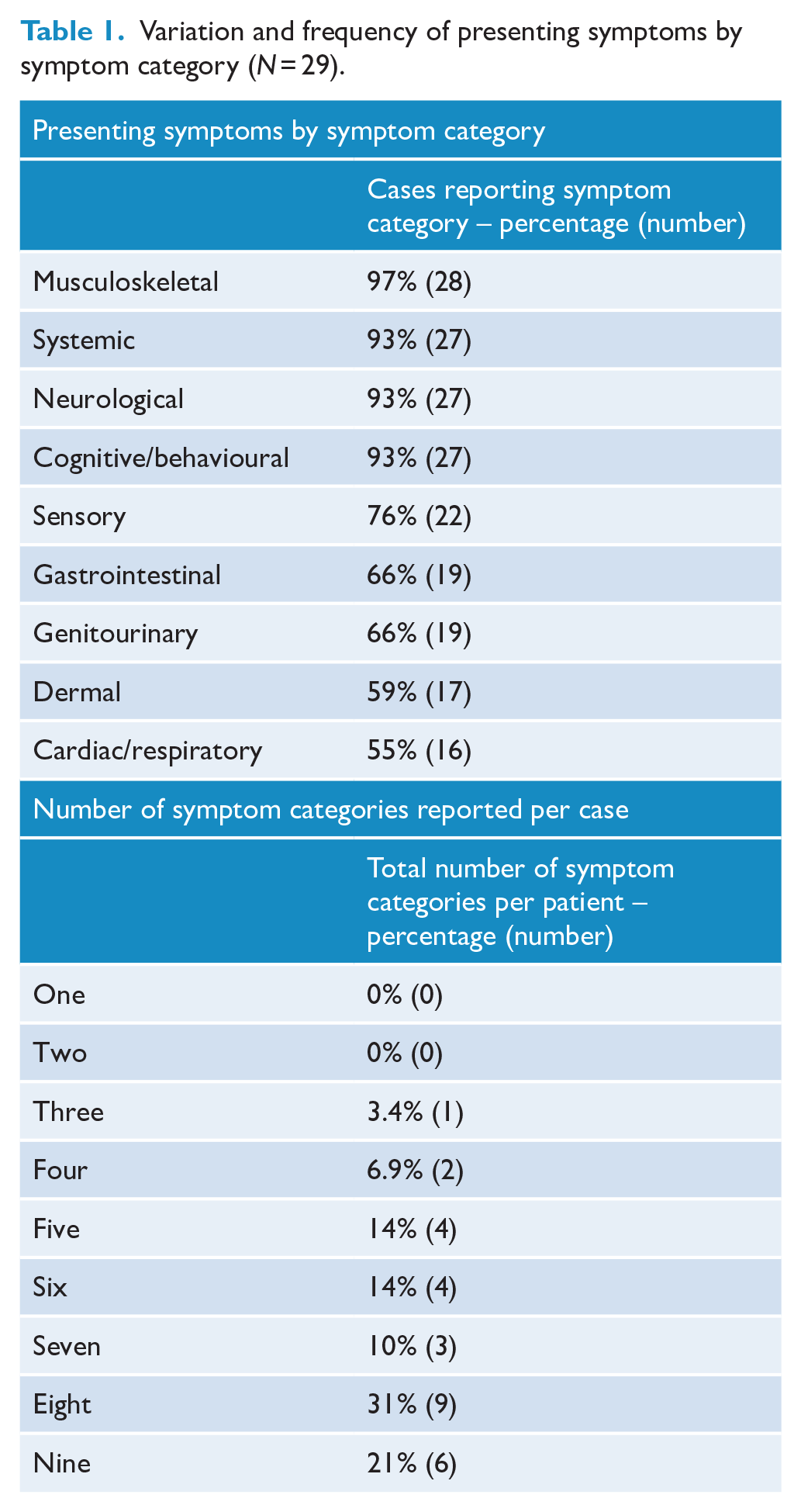
A range of 91 individual symptoms were reported across all bodily systems, with 90% of cases (n = 26) reporting symptoms across 5 or more symptom categories (Table 1). Constitutional symptoms were common (Figure 3), with a characteristic ‘bullseye’ rash reported in only two cases (6.9%) by either the patient or the referring medical practitioner, following their tick bite. One case was described by the treating clinician as reporting over 100 symptoms. Several cases also reported symptoms that either mirrored or subsequently occurred in immediate family members.
Variation and frequency of presenting symptoms by symptom category (N = 29).
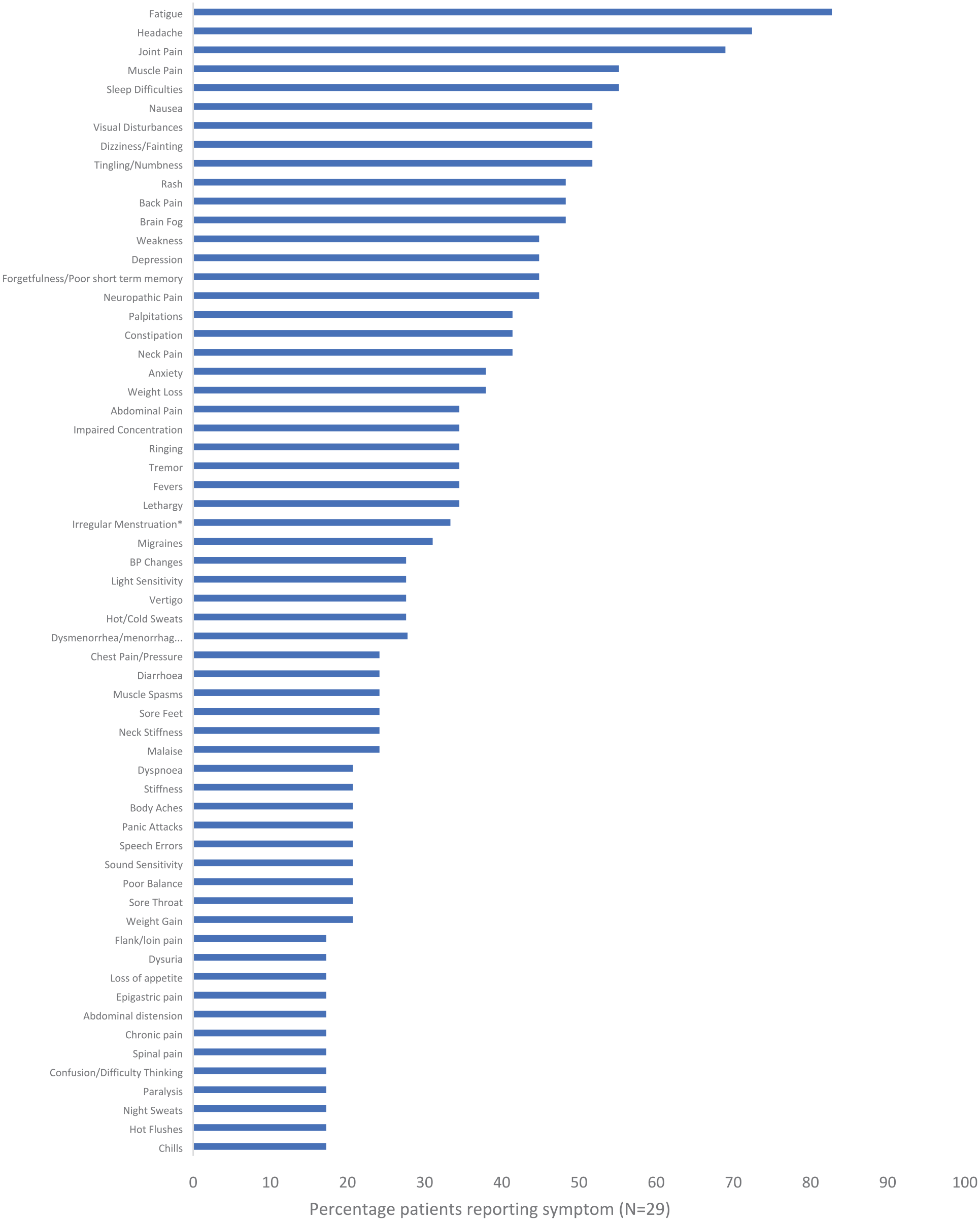
Presenting symptoms reported by cases.*#
Symptom severity ranged from mild to extremely debilitating, with some cases reporting severe episodes requiring presentation to an emergency department or hospitalisation. The duration of illness ranged from 1 month to 31 years (median = 4.5 years). The illness course varied between continuous (n = 13, 45%), episodic (n = 5, 17%) and mixed (chronic baseline symptoms with episodic flare-ups) (n = 11, 38%). Symptoms often varied with external stressors; one case experienced a sensation of disequilibrium immediately prior to a flight during the course of illness.
Burden of illness
A substantial proportion of cases reported severe impact due to their symptoms. Over half (n = 17, 59%) had either ceased employment or study, or had significantly impaired ability to continue in either of these domains. Over one third (n = 10, 35%) had impaired ability to conduct activities of daily living (ADLs), with 31% (n = 9) either bedbound or wheelchair bound/unable to walk. One case reported being bedbound for 6 months prior to presentation. Another reported believing their symptoms to be terminal due to their severity.
Six cases (21%) attributed financial stress to their DSCATT, including selling the family home or business. Four (14%) reported stress among their carers and two (6.9%) attributed separation from their partner to their illness. Seventeen cases (59%) sought overseas diagnostic testing and two (6.9%) sought overseas medical care. One case reported spending over $150,000 on medications and medical consultations.
Investigations
Laboratory
Laboratory investigation results, including inflammatory markers, were generally normal or non-diagnostic. One case had significantly elevated inflammatory markers in the context of urosepsis which have been excluded from the following description of laboratory results.
CRP results were available for 24 (83%) other cases, of which 4 (17%) recorded an elevated peak CRP value (normal range CRP < 5 mg/L), all < 15 (median = 1, range = 0.3–14.2) and considered clinically insignificant. Erythrocyte sedimentation rate (ESR, normal range = 5–12 mm/h) was recorded for 18 cases (62%) (median = 5, range = 2–42), of which 5 (28%) were elevated. The maximum result (ESR = 42) was recorded in a patient ultimately diagnosed with fibromyalgia, with subsequent results within normal limits.
Of 25 (86%) cases with recorded full blood examination (FBE) results, only 2 (6.9%) cases returned an elevated white cell count (WCC) at any stage (normal range 4.0 × 109/L–11.0 × 109/L), both < 15; one recorded WCC increases alongside sporadic abdominal pain suspicious for gastritis and the other had no identified cause, though did return magnetic resonance imaging of the brain (MRI-B) indicating chronic mastoiditis/eustachian tube dysfunction a year earlier, with alternative infective causes considered unlikely by their treating clinicians.
Of 22 (76%) cases with recorded thyroid function tests (TFTs), 3 (14%) had abnormal findings, 2 of which normalised on repeat testing. Of 21 (72%) cases with antinuclear antibody (Patel et al., 2018) results, 7 (33%) were abnormal (normal range < 1/80 units; median positive titre = 1/160, range 1/80–1/280).
Serology
Of 29 cases, 25 (86%) had undergone at least one diagnostic Lyme disease test. Of these cases, none met RCPA diagnostic criteria for confirmed acute Lyme disease (RCPA, 2014) (Box 1). Four (16%) returned at least one positive serological test, all on samples collected > 30 days after symptom onset, none of which were diagnostic of acute infection. One patient had positive Lyme IgM and IgG enzyme-linked immunosorbent assay (ELISA, also referred to as enzyme immunoassay, EIA) but a negative IgG Western Blot (also referred to as immunoblot); one patient had positive IgG by immunofluorescence assay (IFA, another highly sensitive screening test) and IgM Western blot but negative IgG Western blot and IgM by IFA, while culture and polymerase chain reaction (PCR) on blood was not detected; one patient had positive IgM Western blot but negative IgG Western blot and negative IgM and IgG ELISA, while PCR on blood was not detected; the final patient had positive IgG IFA but negative IgM IFA, and negative IgM and IgG on both ELISA and Western Blot (Supplement 2). No cases returned NATA/RCPA-accredited positive Lyme PCR, IFA IgM, IgM WB, cerebrospinal fluid (CSF) or culture results.
Approach to the diagnosis of acute Lyme Disease in Australian patients.

Source: Adapted from RCPA (2014).
NATA: National Association of Testing Authorities; RCPA: Royal College of Pathologists of Australasia.
Nineteen cases (66%) had available Lyme serology results from non-NATA/RCPA-accredited institutions, most frequently Igenex laboratories in California, USA (n = 14, 48%) and Infectolab in Germany (n = 14, 14%). Seven cases (24%) recorded positive Lyme IgM WB and IgG WB from Igenex laboratories, while 3 (10%) recorded positive IFA IgG/IgM/IgA from Igenex. Sixteen (84%) of the 19 cases tested at non-NATA/RCPA-accredited laboratories had at least one positive test result: 14/16 (88%) were also tested at a NATA/RCPA-accredited laboratory (all negative) while two were not re-tested at a NATA/RCPA-accredited laboratory (one case tested IgM positive and another tested IgG positive by immunoblot; Infectolab, Germany).
Non-Lyme serological investigations did not confirm any alternative acute infective causes. Serological results were consistent with past Epstein–Barr virus (EBV), cytomegalovirus (CMV) and Ross River virus infections in 12, 5 and 1 individual(s), respectively. One case returned an equivocal serology result for Ross River Virus IgM with a negative IgG, with the treating infectious diseases clinician in this case reporting no impression of an infectious diagnosis. Nine (31%) cases had positive but non-diagnostic rickettsial serology (titre = 128) to multiple rickettsial groups. These results could be due to recent or past infection or non-specific cross reactivity, requiring repeat testing with rising antibody titre to a single rickettsial group to confirm a recent infection. Of these nine, two were negative and two had unchanged titre on repeat testing, and five did not have repeat testing documented. Non-diagnostic serological results were noted for Bartonella in two individuals, one whose initial positive result (titre 128) became negative on repeat testing, and one with a single titre of 64.
All other serological investigations, including Brucella, dengue virus, malaria, HIV, tuberculosis (TB), syphilis, viral hepatitis and other zoonotic diseases, were negative.
Imaging
MRI-B was performed for 19 (66%) cases, of which 8 (42%) were reported as abnormal. Of these abnormal MRI-B scans, none were considered strongly suggestive of chronic manifestations of Lyme disease or neuroborreliosis by either the treating infectious diseases clinician or neurologist, or subsequent review of images and reports by R.M. Abnormal findings included chronic small vessel ischaemia, nonspecific white matter abnormalities, old atrophic lesions, sinusitis and possible small vascular malformations. One case was found to have a lesion most likely representing a dysembryoplastic neuroepithelial tumour (DNET). Computed tomography scans of the brain (CT-B) were available for 4 (14%) cases, of which 1 (25%) was abnormal, showing chronic mastoiditis/eustachian tube dysfunction.
MRI scans of other regions were performed on 11 (38%) cases, of which 5 (46%) were abnormal, with findings including elbow tendinopathy, disc herniation, osteomyelitis and spinal metastases, and cord signal abnormalities suggestive but not diagnostic of demyelination. No scans were suggestive of Lyme disease.
Management
The most commonly prescribed treatment was antibiotics. Of 25 cases (86%) for whom data were available, 23 (92%) were prescribed antimicrobials prior to initiating care with our health service (Supplement 4); the most commonly prescribed antimicrobials were doxycycline (n = 16, 64%), azithromycin (n = 8, 32%), penicillin (n = 6, 24%) and roxithromycin (n = 6, 24%). Data on antimicrobial use and duration were available for 19 cases (66%). The median number of antimicrobials prescribed was 2 (maximum 9), with a median duration of antimicrobial use of 3 months (maximum 24 months). Elaborate antimicrobial regimens were not uncommon. One patient was trialled on nine separate antibiotics over a 12-month period. Another case had been treated for a range of infections including Lyme disease, EBV, rickettsial infection, malaria, Mycoplasma pneumoniae, Chlamydia pneumoniae and Ross River virus infection over the course of their illness. One case incurred out-of-pocket expenses of over $1200 on atovaquone, a treatment for Pneumocystitis carinii pneumonia, while another had a vascular access port placed for long-term antimicrobial therapy.
For cases where antimicrobial use and duration data were available (n = 19, 66%), 10 (53%) were reported as benefitting from treatment (either self-reported or clinician-reported), with reported benefits subsiding with treatment cessation, and recurring on recommencement, while 2 (11%) denied benefit.
Eight cases (28%) were prescribed antimicrobials during the period of care with our health service. In this group, the median number of antimicrobials prescribed was 1.5 (maximum = 4). Median duration of antimicrobial use was 3 weeks (maximum = 3 months). Three cases (38%) reported benefitting from treatment (either self-reported or clinician-reported); two (25%) denied benefitting from treatment; with data not available for the remaining three cases (38%).
Other commonly prescribed medications included analgesics (n = 20, 69%), benzodiazepines (n = 12, 41%), hydroxychloroquine (n = 11, 38%) and antidepressants (n = 10, 35%). Nearly one quarter (n = 7, 24%) of cases utilised psychotherapy, while 21% (n = 4) utilised rehabilitation or physical therapies (including physiotherapy, myotherapy, chiropractic and exercise physiology services). Six patients (21%) were prescribed antihypotensive agents, including fludrocortisone and midodrine. Utilisation of several alternative therapies, including acupuncture, hyperthermia, ozone therapy, electromagnetic therapy, yoga, meditation, naturopathy and homoeopathy, was also reported.
Nearly all cases (n = 28, 97%) were seen in a specialist outpatient infectious diseases clinic. Other outpatient specialist involvement was sought from neurology (n = 12, 41%), psychiatry (n = 10, 35%), rheumatology (n = 8, 28%), gastroenterology (n = 5, 17%), gynaecology (n = 3, 10%), pain specialist (n = 2, 3%) and neuropsychology (n = 2, 3%) services. Over half (n = 16, 55%) of the cases presented to the emergency department at least once, with 14 cases (48%) requiring at least one inpatient admission.
Diagnoses
While no cases were diagnosed with acute Lyme disease, a range of alternative diagnoses were either formally made (‘confirmed’) or suggested (‘suspected’) by treating clinicians during the period of care at our health service (Figure 4; Supplement 5). The most common alternative diagnoses included several functional somatic syndromes.
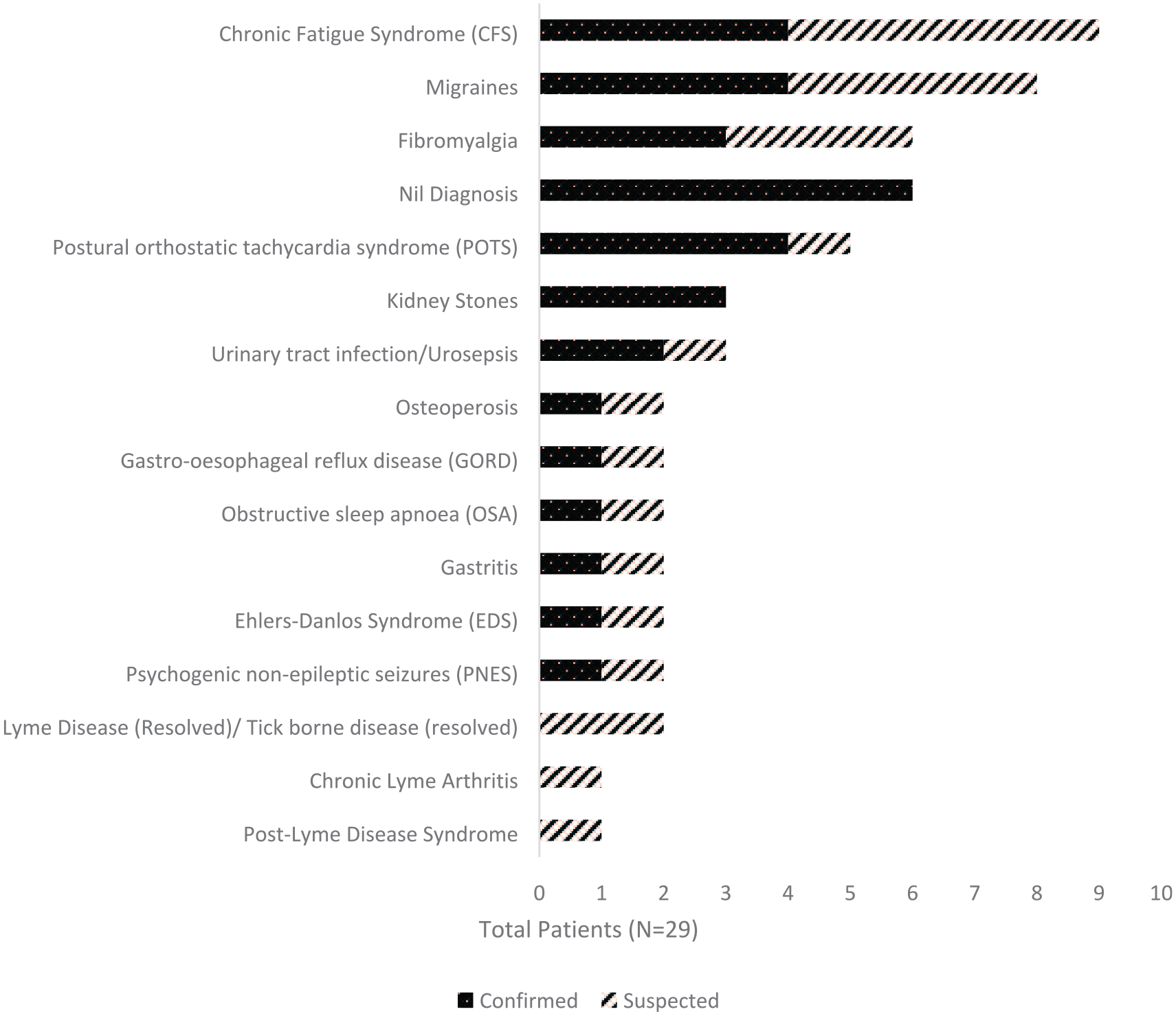
Confirmed and suspected diagnoses made during care at our health service.*
Two cases were considered to have possible diagnoses of tick-borne disease at some point in their clinical course. One was considered to have possibly had a rickettsial infection at the time of symptom onset, 7 years previously. The second case had a 7-year history of joint pain and stiffness following a tick bite in a Lyme-endemic area overseas, with 6 months of limited hand movement. This case’s symptoms had resolved by the time of review by the treating infectious diseases clinician. An episode of Lyme arthritis, now resolved, was considered possible. The first case returned negative rickettsial serology, while both cases returned negative NATA/RCPA accredited Lyme serology.
Another case reported an 8-year history of fatigue, musculoskeletal pain, visual disturbance and gastrointestinal symptoms following a tick bite in rural Australia. NATA/RCPA-accredited Lyme serology included positive ELISA IgG and IgM results with a negative Western Blot IgG, not suggestive of acute infection on interpretation. The treating infectious diseases specialist noted a possible post-Lyme disease syndrome, yet favoured either fibromyalgia or chronic fatigue syndrome (CFS) as a more likely cause.
Nearly half (n = 14, 48%) of all cases’ symptoms could be fully accounted for when both prior and new diagnoses were considered, with 14 (48%) partially accounted for and 1 (3.4%) unaccounted. When excluding functional somatic syndromes, only 7 (24%) cases had symptoms fully accounted for by prior and new diagnoses, with 19 (66%) partially accounted for and 3 (10%) unaccounted.
Discussion
To our knowledge, this is the first case series of patients with DSCATT (Chalada et al., 2016; Collignon et al., 2016). Our case series included 29 patients, the majority of whom had either been previously diagnosed elsewhere with Lyme disease or suspected elsewhere of having a ‘Lyme-like’ illness, who were referred to our service for diagnostic clarification and/or management of their condition. A prior history of chronic syndromes, including CFS, fibromyalgia, irritable bowel syndrome (IBS) and migraines, was common, as were histories of anxiety, depression and psychosocial stressors prior to illness onset. Risk factors for tick-borne disease, in particular tick bites in Lyme-endemic areas, were relatively infrequent given the illness attribution to tick bites. No cases had laboratory-confirmed Lyme disease.
While not observed in all cases, our findings suggest a common clinical syndrome involving a wide range of chronic, often non-specific symptoms. Cases reported a predominance of constitutional symptoms, in particular fatigue, pain, headaches, weakness, nausea, sleep difficulties and visual disturbances. Sensory and cognitive symptoms were also common. Nearly all cases experienced symptoms across multiple bodily systems, with a majority of cases reporting symptoms within each symptom category. While illness duration and course were varied, many cases suffered from chronic, continuous symptoms, at times resulting in severe physical impairment and financial and carer stress. Chronic healthcare resource utilisation, including high rates of emergency department presentations, admissions and outpatient specialist service attendances, was common, with many cases committing extensive time, resources and money to diagnosis and/or treatment, and several seeking overseas diagnostic services or care. A variety of treatments were sought for both symptomatic relief and curative intent, in particular antimicrobials, analgesics and benzodiazepines. A majority of patients reported benefitting from antimicrobial treatment, with perceived benefits often subsiding with treatment cessation.
While the majority of cases had had domestic and/or international Lyme serology performed, no cases met the RCPA diagnostic criteria for acute Lyme disease (RCPA, 2014). This is in keeping with several prior reviews that found no convincing diagnoses of locally acquired Lyme disease in Australian patients (Chalada et al., 2016; Collignon et al., 2016). All cases with at least one positive test result at a non-NATA/RCPA-accredited laboratory who were re-tested at a NATA/RCPA-accredited laboratory (14/16, 88%) went on to have negative serology; the two remaining cases were not re-tested at a NATA/RCPA-accredited laboratory, but this evidence suggests positive accredited tests would be unlikely.
We found no convincing evidence of common alternative infective causes (e.g. rickettsia), or of any occult infective or inflammatory state, on review of broader investigations. Imaging, in particular MRI, revealed either non-Lyme related changes or white matter lesions that are non-specific for Lyme disease and are commonly found with ageing and/or migraines. While we cannot exclude the possibility of an as yet unidentified Australian vector carrying B. burgdorferi, a different pathogen or a non-infectious aetiology, significant variation in clinical history and presentation, laboratory investigations and treatment responsiveness further suggest the absence of any unifying underlying inflammatory or infective process.
If DSCATT is not due to a single identified causative organism or other organic disease process, it may be considered as either a new medically unexplained syndrome or an umbrella term for a range of disease or symptom processes. Post-infective syndromes occur commonly following many viruses, with ‘long COVID’ being only the most high profile of these, and they remain unexplained. Our cases displayed high rates of pre-existing CFS, fibromyalgia, IBS and migraines on initial presentation to our health service, with a common clinical presentation characteristic of these syndromes and of post-treatment Lyme disease (Rebman and Aucott, 2020; Wessely et al., 1999). Anxiety, depression and psychosocial stressors were also common and may often overlap with, or increase risk of, other functional somatic syndromes (Henningsen et al., 2003). While a subset of patients received no new diagnoses during their period of care under infectious disease, many received confirmed or suspected new diagnoses, commonly chronic and/or functional syndromes including CFS, fibromyalgia, postural orthostatic tachycardia syndrome (POTS) and migraines. Nearly all patients’ symptoms were either fully or partially explained by their diagnoses, if other unexplained syndromes were included.
Indeed, previous reviews of other medically unexplained and/or functional somatic syndromes, including CFS, fibromyalgia and IBS, have found high rates of overlap between clinical presentations across these conditions, with debate as to whether these syndromes may represent variations of a single disorder (Aaron and Buchwald, 2001; Aaron et al., 2000; Kanaan et al., 2007; Wessely et al., 1999; Wessely and White, 2004). The high symptom overlap between these syndromes and low diagnostic stability in individual patients can be argued to support core transdiagnostic aetiological factors underlying these disorders and that common processes may perpetuate their symptoms and associated disability (Chalder and Willis, 2017). The fact that these syndromes commonly occur following infectious illnesses may suggest a multi-factorial response that, while incompletely understood, involves processes that may be universal (Chalder and Willis, 2017). In the case of DSCATT, tick bites may act as either an infective trigger or, potentially, through fear or anticipation of infection, as a psychological trigger.
Irrespective of aetiology, our findings depict a cohort with a significant burden of illness. Misunderstanding of complex Lyme serology by both patients and community medical officers (Moore et al., 2016), alongside the existence of commercial laboratories offering unvalidated testing for Lyme disease (Centres for Disease Control and Prevention, 2005), and the variable performance of some Lyme assays in different patient populations (Leeflang et al., 2016), may create diagnostic confusion and dissatisfaction for both doctors and patients with DSCATT. Variable responsiveness to antimicrobial treatments, as displayed in our cohort, may further potentiate uncertainty and dissatisfaction. While there is aetiological uncertainty, it may be best approached with transdiagnostic, aetiologically neutral rehabilitative therapies, which have proven effective for other unexplained syndromes (Chalder and Willis, 2017; White et al., 2011).
Limitations
Our study had several limitations. Data were often incomplete, and at times difficult to interpret, owing to a reliance on existing medical records and free-text information rather than prospectively designed data collection. Data quality may have been further impaired by recall and information bias, with occasional reliance on secondary sources, for example, review of investigations in clinical notes rather than primary investigation reports. Moreover, the small sample size, single site for case inclusion and a common source of referral for many cases may have increased selection and ascertainment bias and may limit generalisability of results to other settings.
Conclusion
Our study represents the first case series of patients with DSCATT. We found no evidence of acute Lyme disease or any separate underlying organic disease process common to a majority of cases in this cohort. DSCATT may therefore be due to an as yet unidentified cause or may be considered a medically unexplained syndrome similar to existing conditions such as CFS. Further investigation of potential causative agents should be prioritised as well as aetiologically neutral symptom-based treatment models as long as causes remain unclear.
Supplemental Material
sj-docx-2-anp-10.1177_00048674211043788 – Supplemental material for Characterising DSCATT: A case series of Australian patients with debilitating symptom complexes attributed to ticks
Supplemental material, sj-docx-2-anp-10.1177_00048674211043788 for Characterising DSCATT: A case series of Australian patients with debilitating symptom complexes attributed to ticks by Jesse Schnall, Georgina Oliver, Sabine Braat, Richard Macdonell, Katherine B Gibney and Richard A Kanaan in Australian & New Zealand Journal of Psychiatry
Supplemental Material
sj-docx-3-anp-10.1177_00048674211043788 – Supplemental material for Characterising DSCATT: A case series of Australian patients with debilitating symptom complexes attributed to ticks
Supplemental material, sj-docx-3-anp-10.1177_00048674211043788 for Characterising DSCATT: A case series of Australian patients with debilitating symptom complexes attributed to ticks by Jesse Schnall, Georgina Oliver, Sabine Braat, Richard Macdonell, Katherine B Gibney and Richard A Kanaan in Australian & New Zealand Journal of Psychiatry
Supplemental Material
sj-docx-4-anp-10.1177_00048674211043788 – Supplemental material for Characterising DSCATT: A case series of Australian patients with debilitating symptom complexes attributed to ticks
Supplemental material, sj-docx-4-anp-10.1177_00048674211043788 for Characterising DSCATT: A case series of Australian patients with debilitating symptom complexes attributed to ticks by Jesse Schnall, Georgina Oliver, Sabine Braat, Richard Macdonell, Katherine B Gibney and Richard A Kanaan in Australian & New Zealand Journal of Psychiatry
Supplemental Material
sj-docx-5-anp-10.1177_00048674211043788 – Supplemental material for Characterising DSCATT: A case series of Australian patients with debilitating symptom complexes attributed to ticks
Supplemental material, sj-docx-5-anp-10.1177_00048674211043788 for Characterising DSCATT: A case series of Australian patients with debilitating symptom complexes attributed to ticks by Jesse Schnall, Georgina Oliver, Sabine Braat, Richard Macdonell, Katherine B Gibney and Richard A Kanaan in Australian & New Zealand Journal of Psychiatry
Supplemental Material
sj-xlsx-1-anp-10.1177_00048674211043788 – Supplemental material for Characterising DSCATT: A case series of Australian patients with debilitating symptom complexes attributed to ticks
Supplemental material, sj-xlsx-1-anp-10.1177_00048674211043788 for Characterising DSCATT: A case series of Australian patients with debilitating symptom complexes attributed to ticks by Jesse Schnall, Georgina Oliver, Sabine Braat, Richard Macdonell, Katherine B Gibney and Richard A Kanaan in Australian & New Zealand Journal of Psychiatry
Footnotes
Acknowledgements
We thank Prof Paul Johnson, Austin Hospital, for assistance with identifying potential DSCATT cases.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical Approval
Austin Health ethics approval Audit/19/Austin/111; approved 15 October 2019.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This project was funded by an NHMRC targeted research call (TRC) grant (APP1169827). K.B.G. received salary support from NHMRC (APP1120816) and MRFF (MRF1193727).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
