Abstract
Objective:
The effects of mood disorders on mortality may be mediated by their effects on the risk of dementia, and interventions to reduce the occurrence of dementia may reduce their overall mortality. This study aimed to investigate the direct effects of depressive and bipolar disorders on the 6-year risk of mortality and also their indirect effects on mortality due to their effect on the risk of dementia.
Methods:
A total of 5101 Koreans were selected from a community-based prospective cohort study, and 6-year risks of mortality and dementia in participants with depressive and bipolar disorders were estimated by Cox proportional hazard analysis. The direct and indirect effects of depressive and bipolar disorders on the risk of mortality were estimated using structural equation modeling.
Results:
The depressive and bipolar disorder groups showed 51% and 85% higher 6-year mortality, and 82% and 127% higher risk of dementia, respectively, compared to euthymic controls. The effects of depressive and bipolar disorders on mortality were mainly mediated by their effects on the risk of dementia in a structural equation model. The direct effects of each mood disorder on mortality were not significant.
Conclusion:
Both depressive and bipolar disorders increased the risks of mortality and dementia, and the effects of mood disorders on mortality were mainly mediated through dementia. As dementia occurs later in life than mood disorders, measures to prevent it may effectively reduce mortality in individuals with a history of mood disorders, as well as being more feasible than attempting to control other causes of death.
Introduction
Depressive disorder (DD) and bipolar disorder (BD) are major causes of mortality worldwide. A diagnosis of either disorder has been associated with increased mortality and a reduced lifespan of about 10 years (Walker et al., 2015). The influence of both disorders on death is significant, regardless of age (Osby et al., 2001), and some studies have suggested that the effect of DD may be more pronounced in older individuals (Markkula et al., 2016). DD and BD also increase the risk of dementia. Studies have shown that a diagnosis of DD or BD may result in a more than twofold increase in the risk of dementia including Alzheimer’s disease (AD) (da Silva et al., 2013; Ownby et al., 2006). Since dementia was found to increase the risk of mortality (Bae et al., 2019), the effects of DD and BD on mortality may be, at least in part, mediated by their effects on the risk of dementia. The significant overlap between the potential mechanisms, such as cardiovascular diseases and diabetes (Baumgart et al., 2015; Fiedorowicz et al., 2008; Raz et al., 2016; Thomas et al., 2004), neuroinflammation (Berk, 2009; Penninx, 2017; Raz et al., 2016), hypothalamic–pituitary–adrenal (HPA) axis function (Belvederi Murri et al., 2016; Milaneschi et al., 2019; Penninx, 2017; The Global Burden of Disease 2015 Obesity Collaborators, 2017) and autonomic function (An et al., 2020; Forte et al., 2019; Licht et al., 2008; Quintana et al., 2016; Thayer and Lane, 2007), underlying the effects of DD and BD on the risk of mortality and those on the risk of dementia may add weight to this argument. Furthermore, if this is true, then measures to prevent dementia may be a practical way to reduce overall mortality in individuals with a history of mood disorder. However, the mediatory role of dementia in the risk of mortality associated with DD and BD has not been directly investigated.
Our objective was to investigate the direct effects of DD and BD on the 6-year risk of mortality and also their indirect effects on mortality due to their effect on the risk of dementia in the randomly sampled, community-dwelling older individuals participating in the Korean Longitudinal Study on Cognitive Aging and Dementia (KLOSCAD).
Methods and materials
Study design and participants
We acquired data from the KLOSCAD (Han et al., 2018). The KLOSCAD is an ongoing, nationwide, multi-centered, prospective cohort study. In the KLOSCAD, 12,694 community-dwelling Koreans aged ⩾60 years who were randomly sampled from the residents of 13 districts across South Korea using the national residential roster at the end of 2009 were invited and 6818 (53.7%) completed the baseline assessment. The baseline assessment was conducted from November 2010 to October 2012, and follow-up assessments have been conducted every 2 years (the first follow-up assessment was from November 2012 to October 2014, the second follow-up assessment was from November 2014 to October 2016 and the third follow-up assessment was conducted from November 2016 to October 2018).
Among the 6818 participants in the baseline assessment of the KLOSCAD, we included 5101 in this study after excluding those who did not complete the baseline assessments on depression and other conditions employed as variables in the current analysis (n = 328), were diagnosed with dementia (n = 348), or did not respond to any follow-up assessments (n = 1041). Among the 5101 participants, 3012 had completed all the three follow-up assessments.
All procedures involving human subjects/patients were approved by the Institutional Review Board (IRB) of the Seoul National University Bundang Hospital (IRB No. B-0912-089-010). In all cases, a written statement of informed consent was obtained from either the participants or their legal guardians.
Assessments and diagnoses
Geriatric psychiatrists assessed all participants through face-to-face standardized diagnostic interviews using the Korean version of the Mini International Neuropsychiatric Interview (MINI) (You et al., 2006) and conducted neurological and physical examinations and laboratory tests. We obtained previous histories of mood disorders from the participants and/or their family members. Thereafter, a panel of geriatric psychiatrists evaluated the participants for current mood disorders, such as BD, major DD and minor DD, according to the Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition (DSM-IV) diagnostic criteria (American Psychiatric Association, 1994). Further, the presence of subsyndromal depression was assessed according to the following operational criteria: (a) two or more symptoms of depression occurring during the same 2-week period listed in criterion A for major depressive episode under DSM-IV; (b) at least one of the symptoms must be depressed mood or anhedonia; (c) each depressive symptom should be present >12 hours every day or for >7 days during the 2 weeks; and (d) not meet the diagnostic criteria for major or minor DDs (Jeong et al., 2012). We defined the DD group as participants currently experiencing a major DD, minor DD, or subsyndromal depression, or a previous history of major or minor DDs. We defined the BD group as participants currently experiencing a BD or a previous history of BD. Those who received a mood disorder diagnosis for the first time at the baseline assessment were further specified as ‘first-episode mood disorder’ cases, whereas those with a previous history were specified as ‘recurrent’ cases. All other subjects were included in the euthymic control (EC) group.
We evaluated the burden of comorbid chronic medical illnesses using the Cumulative Illness Rating Scale (CIRS) (Miller et al., 1992). The CIRS combines the morbidity of chronic medical problems affecting various organ systems into a single comprehensive score. It includes 14 organ systems, and the impact of the chronic conditions affecting each system is rated from 0 to 4; the sum of all ratings is the CIRS score. We defined ‘high comorbid disease burden’ as having a CIRS score of ⩾5 points, with at least one organ system receiving a score of 3 (severe) or 4 (extremely severe).
We administered the Korean version of the Consortium to Establish a Registry for Alzheimer’s Disease Clinical and Neuropsychological Assessment Battery (CERAD-K) (Lee et al., 2002) and the Mini Mental State Examination (MMSE) (Lee et al., 2004) to exclude dementia at the baseline assessment and to diagnose incident dementia at follow-up assessments. We diagnosed dementia according to the DSM-IV diagnostic criteria (American Psychiatric Association, 1994). We defined history of alcohol use and smoking as drinking or smoking any amount within the past 1 year. Of the 1980 participants who had used alcohol during the past year, five had a history of alcohol use disorder.
We identified the deaths of participants within 6 years from the baseline assessment using the National Mortality Database of Statistics Korea using personal identification numbers. In Korea, deaths are reported to the corresponding local governments and included in a national database of deaths in the country, which is managed by Statistics Korea. This database provides the date, place and cause of death, as confirmed by a physician.
Statistical analysis
We performed chi-square tests and one-way analysis of variance (ANOVA) to compare categorical and continuous variables, respectively. To examine the effect of BD and DD on mortality, we conducted a Cox proportional hazard analysis adjusting for age, gender, duration of education, history of smoking, history of alcohol use and high comorbid disease burden. To examine the effects of DD and BD on the future risk of dementia, we conducted a Cox proportional hazard analysis using the model explained above. We estimated the mean survival time to mortality and incident dementia using Kaplan–Meier analyses. Since dementia is also known to increase mortality (Bae et al., 2019), we used structural equation modeling to examine the direct effect of DD and BD on mortality and also their indirect effects on mortality due to their effect on the risk of dementia. We performed all statistical analyses using the dplyr, survival and lavaan packages in R version 4.0.0.
Results
At the baseline assessment, the participants’ mean age was 69.67 years and 56.8% of them were women. The proportion of recurrent cases was 27.5% in the DD group and 100% in the BD group. The demographic and clinical characteristics of the participants at baseline assessment are summarized in Table 1. The BD group had the highest 6-year mortality rate (8.97%), followed by the DD group (7.44%). The BD group also showed the highest risk of incident dementia during the follow-up period of 4.78 ± 1.58 years (6.84%), followed by the DD group (6.07%). Baseline characteristics of the participants included in this study were compared with those excluded due to incomplete assessments or the absence of any follow-up assessments (Supplemental Tables 1 and 2). The participants who did not complete the baseline assessment were more likely to have a past or current DD and showed higher rates of mortality and incident dementia compared to those included in this study. The participants who did not respond to any follow-up assessments showed higher mortality but were not more likely to have a past or current DD than those included in this study.
Baseline characteristics of the participants.
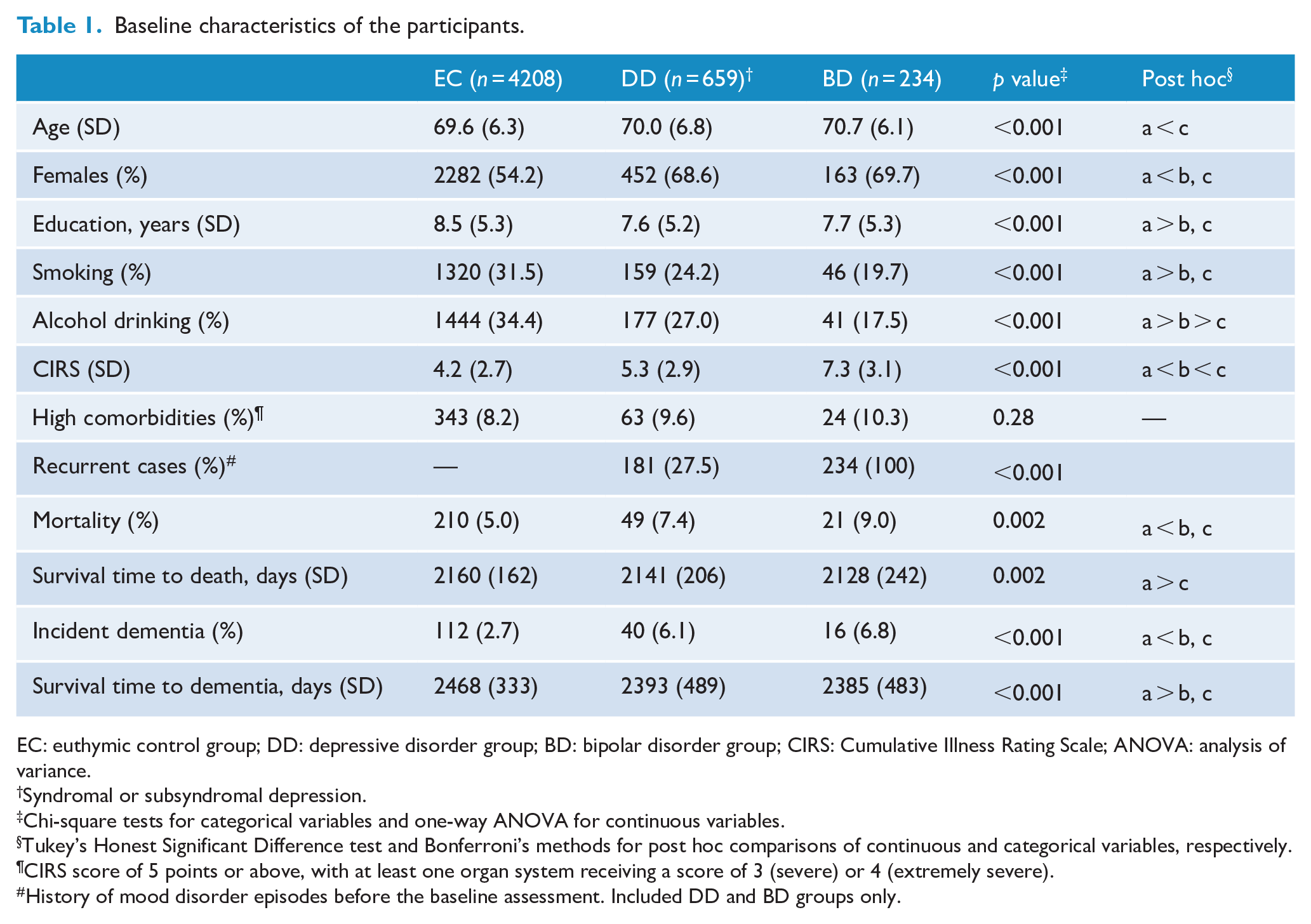
EC: euthymic control group; DD: depressive disorder group; BD: bipolar disorder group; CIRS: Cumulative Illness Rating Scale; ANOVA: analysis of variance.
Syndromal or subsyndromal depression.
Chi-square tests for categorical variables and one-way ANOVA for continuous variables.
Tukey’s Honest Significant Difference test and Bonferroni’s methods for post hoc comparisons of continuous and categorical variables, respectively.
CIRS score of 5 points or above, with at least one organ system receiving a score of 3 (severe) or 4 (extremely severe).
History of mood disorder episodes before the baseline assessment. Included DD and BD groups only.
Among the 4208 participants of the EC group, 149 (3.5%) developed dementia, and 32 (0.76%) of them passed away during the follow-up period. Among the 659 participants of the DD group, 54 (8.2%) developed dementia, and 6 (0.91%) of them passed away during the follow-up period. And among the 234 participants of the BD group, 19 (8.1%) developed dementia, and 2 (0.85%) of them passed away during the follow-up period. As shown in Table 2, the DD and BD groups showed 51% (95% confidence interval [CI] = [1.10, 2.07]; p = 0.012) and 85% (95% CI = [1.18, 2.90]; p = 0.008) higher 6-year mortality, respectively, compared to the EC group in the Cox proportional hazard analysis. The 6-year mortality was comparable between the DD and BD groups (p = 0.41). In this analysis, old age (hazard ratio [HR] = 1.14; 95% CI = [1.12, 1.16]; p < 0.001), men (HR = 2.75; 95% CI = [1.98, 3.81]; p < 0.001) and high comorbidity (HR = 1.63; 95% CI = [1.18, 2.26]; p = 0.003) were associated with the risk of mortality. The proportion of individuals who died during the follow-up period was comparable between the first-episode mood disorder and recurrent cases (37 [7.7%] deaths in the first-episode mood disorder cases and 33 [8.0%] deaths in the recurrent cases, p = 0.91).
Future risks of mortality and dementia by the presence of mood disorders.
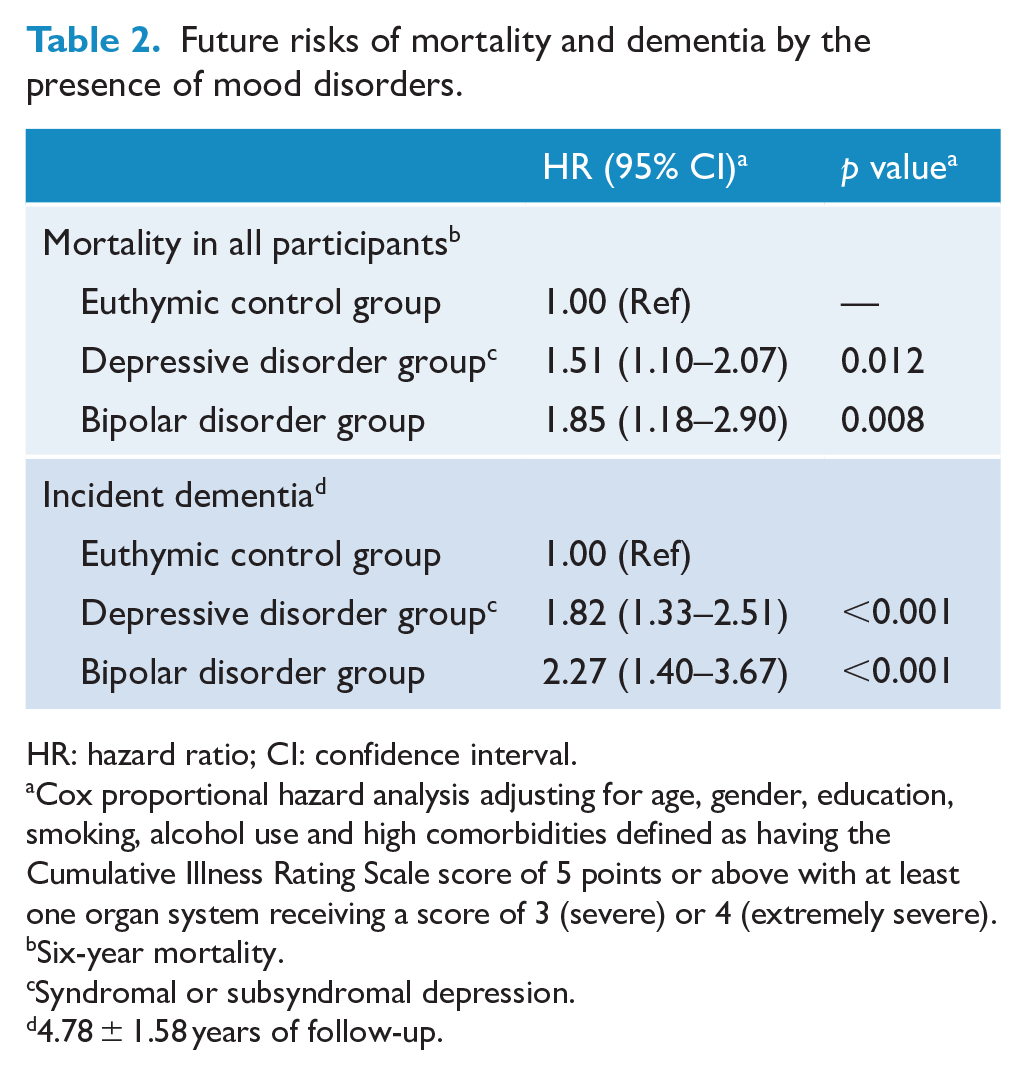
HR: hazard ratio; CI: confidence interval.
Cox proportional hazard analysis adjusting for age, gender, education, smoking, alcohol use and high comorbidities defined as having the Cumulative Illness Rating Scale score of 5 points or above with at least one organ system receiving a score of 3 (severe) or 4 (extremely severe).
Six-year mortality.
Syndromal or subsyndromal depression.
4.78 ± 1.58 years of follow-up.
The DD and BD groups also showed 82% (95% CI = [1.33, 2.51]; p < 0.001) and 127% (95% CI = [1.40, 3.67]; p < 0.001) higher risk of dementia, respectively, compared to the EC group in the Cox proportional hazard analysis. The risk of dementia was comparable between the DD and BD groups (p = 0.47). In this analysis, old age (HR = 1.16; 95% CI = [1.14, 1.19]; p < 0.001) increased, while higher education (HR = 0.95; 95% CI = [0.93, 0.98]; p = 0.002) and alcohol use (HR = 0.66; 95% CI = [0.46, 0.96]; p = 0.03) reduced the risk of dementia. Among the 222 incident dementia cases that occurred during the follow-up period, 166 and 56 were AD and non-AD (26 vascular dementia [VaD] and mixed dementia and 30 other dementia). In this study, the mortality of the participants with incident non-AD was higher than the mortality of those with incident AD (28.6% vs 14.5%, p = 0.017), which was in line with previous studies (Guehne et al., 2005). However, the distribution of the subtypes of incident dementia was comparable between the EC, DD and BD groups in this study (p = 0.75). In the DD group, the proportion of individuals who were diagnosed with dementia during the follow-up period was comparable between the first-episode mood disorder and recurrent cases (43 [9%] and 11 [6%] in the first-episode mood disorder and recurrent cases, respectively, p = 0.22).
Since dementia may also increase mortality (Bae et al., 2019), we developed a structural equation model to examine the direct and indirect effects of DD and BD on mortality (Figure 1). The effects of DD and BD on mortality were mediated by their effects on the future risk of dementia (unstandardized coefficient = 0.415; 95% CI = [0.259, 0.570]; p < 0.001 for the DD group, unstandardized coefficient = 0.410; 95% CI = [0.166, 0.653]; p = 0.001 for the BD group). However, the direct effects of DD and BD on mortality were not statistically significant (unstandardized coefficient = 0.060; 95% CI = [−0.106, 0.226]; p = 0.48 for the DD group, unstandardized coefficient = 0.163; 95% CI = [−0.087, 0.413]; p = 0.20 for the BD group).
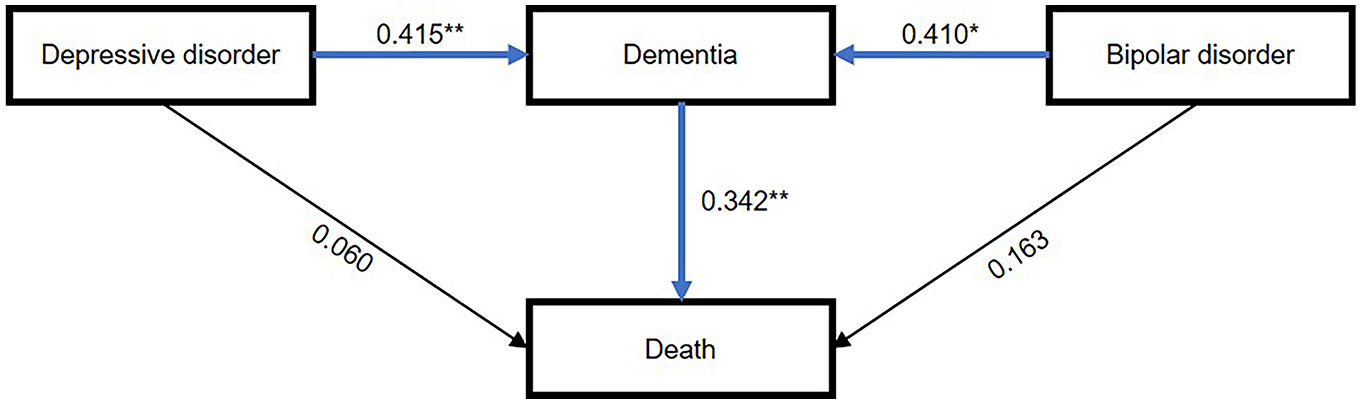
Structural equation model of depressive and bipolar disorders, dementia and death.
Discussion
This prospective cohort study showed that both DD and BD increased the risks of mortality and dementia, and that their effects on the risk of mortality were mediated by their effects on the risk of dementia. Although the strong associations of mood disorders with either dementia or mortality have been studied extensively (Bae et al., 2019; Berk, 2009; da Silva et al., 2013; Fiedorowicz et al., 2008; Gunde et al., 2011; Ownby et al., 2006; Thomas et al., 2004; Walker et al., 2015), this is the first prospective cohort study to reveal the structure of the relationship between mood disorders, dementia and death. Although none of the previous studies investigated the effects of DD and BD on the risk of mortality or dementia simultaneously, they consistently reported that individuals with DD or BD had a higher risk of mortality and dementia than controls.
DD and BD were found to increase the risk of all-cause mortality by 71% and 100%, respectively, in a systematic review of 43 studies (Walker et al., 2015). DD was also found to increase all-cause mortality in older adults by 62% in another meta-analysis of 10 studies (Brandao et al., 2019). BD usually occurs at around 20 years of age, follows a lifelong episodic course (Sadock et al., 2017) and only half of the course is spent in a euthymic state (Judd et al., 2002). On the other hand, DD usually occurs about 10 years later than BD, and about one-third of the patients do not experience recurrence (Sadock et al., 2017). BD may increase the risk of mortality more than DD because individuals with BD may have spent a longer time in symptomatic states than those with DD at any given age, although we could not directly examine the effect of the duration of the symptomatic phase on the differential association of BD and DD with the risks of dementia and mortality in this study because we did not obtain the age of onset of BD and DD.
The risk of mortality associated with DD or BD may be, at least in part, mediated by the risk of dementia associated with them. In many systematic reviews, both DD and BD increased the risk of dementia (Chan et al., 2019; Diniz et al., 2017; Ownby et al., 2006), which may also increase the risk of mortality (Bae et al., 2019). In the United States, more than one-third of the mortality in older adults may be attributable to AD, with a median time of 3.8 years from the diagnosis of AD to death (James et al., 2014). However, the mediatory role of dementia in the effects of DD and BD on mortality has never been directly investigated. In our structural equation model, DD and BD increased the risk of dementia by approximately 40%, and dementia, in turn, increased the risk of mortality by approximately 34% during the 6-year follow-up period. Thus, both DD and BD increased the risk of mortality indirectly by approximately 14% through their effects on the risk of dementia (p < 0.001). This indirect effect on the risk of mortality was comparable to the direct effect on the risk of mortality in the BD group and was larger than the direct effect in the DD group. About three-fourths of the participants in the DD group, and none in the BD group were primary cases. Since DD that begins in late life is more likely to be a prodromal stage of dementia than recurrent DD (Barnes et al., 2012), the mediatory role of dementia in the effect of DD on mortality might have been larger than that in the effect of BD on mortality.
The mediatory role of dementia in the effects of DD and BD on mortality can be explained in several ways. First, both DD (Thomas et al., 2004) and BD (Fiedorowicz et al., 2008) increase the risk of cardiovascular diseases and diabetes, which in turn, increase the risks of mortality (Mottillo et al., 2010) and dementing illnesses including AD (Baumgart et al., 2015; Raz et al., 2016). Second, both DD (Penninx, 2017) and BD (Berk, 2009) are associated with higher levels of inflammatory markers, which also increase the risks of mortality (Penninx, 2017) and dementia (Raz et al., 2016). Third, both DD (Milaneschi et al., 2019) and BD (Belvederi Murri et al., 2016) show HPA axis dysregulation. HPA axis dysregulation is associated with the risks of cardiovascular disease (Penninx, 2017) and obesity (Milaneschi et al., 2019) that increase mortality (The Global Burden of Disease 2015 Obesity Collaborators, 2017). HPA axis dysregulation also causes neuronal loss in the hippocampus (McEwen, 2007) and cortical gray matter thinning in other areas of the brain (The ENIGMA Major Depressive Disorder Working Group, 2017). Fourth, DD (Licht et al., 2008) and BD (Quintana et al., 2016) show autonomic dysregulation, such as reduced parasympathetic activity (An et al., 2020). Reduced parasympathetic activity is associated with the risks of cardiovascular mortality (Thayer and Lane, 2007) and cognitive impairment (Forte et al., 2019). Fifth, the relationships of DD with cardiovascular disease (Thomas et al., 2004) and neuroinflammation (Penninx, 2017) may be bi-directional, resulting in vicious cycles that accelerate the pathological processes leading from affective disorders to dementia to death. All these factors may also play in concert (Penninx, 2017; Thayer and Lane, 2007). Sixth, individuals with DD or BD are more likely to lead an unhealthy lifestyle (Morriss and Mohammed, 2005; Rahe et al., 2016), which is a significant risk factor for both dementia (Baumgart et al., 2015) and death (Loef and Walach, 2012). Seventh, another possible, albeit controversial, factor is treatment. Patients with DD or BD usually spend a significant amount of time taking medications, which may result in a lasting positive or negative effect on the risks of dementia and mortality. Currently, the impact of antidepressant use on the risks of dementia (Bartels et al., 2020; Chan et al., 2019) or mortality (Coupland et al., 2011; Sharma et al., 2016) is controversial. Lithium may mostly reduce (Cheng et al., 2017), while valproic acid may increase the risk for dementia (Tsai et al., 2016).
In the structural equation model, the direct effects of DD and BD on mortality were not statistically significant (p = 0.48 and 0.20, respectively). This lack of direct effects may be mainly attributable to the older age of the participants. In younger populations, the effects of DD and BD are more likely to be direct or mediated by diseases other than dementia, and both DD and BD are significant risk factors for death from external causes such as suicide and accidents in this age group (Khalsa et al., 2008; Osby et al., 2001; Reutfors et al., 2018). In addition, direct effects of DD or BD on the risk of mortality might have been found if the follow-up duration was longer. Since the KLOSCAD is ongoing, we may re-examine the direct effects of DD and BD on the risk of mortality in the future.
This study has several limitations. First, past histories of DD and BD were subject to recall biases because they were ascertained by self-reporting rather than medical records. However, recall biases were probably not large, as all participants were cognitively normal at baseline assessment. Second, the participants were aged ⩾60 years. Therefore, the current observations cannot be generalized to younger adults with DD or BD. Third, the follow-up duration was relatively short compared to the previous studies on the effects of mood disorders on mortality (Chan et al., 2019; Walker et al., 2015). Among the 68 studies included in the systematic review by Walker and colleagues (Walker et al., 2015), only 15 were community-based prospective cohort studies. Among them, six studies followed up their participants for more than 6 years. However, none of the 15 studies investigated the direct effect and indirect effect via dementia of DD and BD separately, and only one of them adjusted for the effect of dementia on mortality in their analysis on the effect of DD on the risk of mortality (Rapp et al., 2008). Fourth, we were unable to include some participants from the cohort due to incomplete assessments or the absence of any follow-up assessments. Participants with incomplete assessments showed higher risk of mortality and dementia compared to those included in the study, but were also more likely to have a current episode or past history of DD. (Supplemental Table 1) This suggests that the exclusion of these subjects likely reduced the difference in mortality and dementia risk between the EC and DD groups in our study. Subjects excluded from this study due to the absence of any follow-up assessments also had higher mortality than those that were included. (Supplemental Table 2) However, in this case, the proportion of DD or BD subjects was similar between the two groups. Therefore, the exclusion of these subjects probably did not have any significant effect on our results. Fifth, we did not examine the onset age of DD. Since DD can occur in late life as a prodrome of dementia, DD is more likely to be a prodrome of dementia when the ages of onset of DD and dementia are close. However, in this study, incident dementia did not occur more frequently in first-episode cases compared to recurrent cases.
In older adults, the effects of mood disorders on mortality are mainly mediated by their effects on the risk of dementia. As dementia occurs later in life than mood disorders, measures to prevent it may significantly reduce overall mortality in individuals with a history of mood disorders, as well as being more feasible than attempting to control other causes of death.
Supplemental Material
sj-docx-1-anp-10.1177_00048674211041937 – Supplemental material for Mood disorders increase mortality mainly through dementia: A community-based prospective cohort study
Supplemental material, sj-docx-1-anp-10.1177_00048674211041937 for Mood disorders increase mortality mainly through dementia: A community-based prospective cohort study by Hoyoung An, Hee Won Yang, Dae Jong Oh, Eunji Lim, Jin Shin, Dong Gyu Moon, Seung Wan Suh, Seonjeong Byun, Tae Hui Kim, Kyung Phil Kwak, Bong Jo Kim, Shin Gyeom Kim, Jeong Lan Kim, Seok Woo Moon, Joon Hyuk Park, Seung-Ho Ryu, Dong Woo Lee, Seok Bum Lee, Jung Jae Lee, Jin Hyeong Jhoo, Jong Bin Bae, Ji Won Han and Ki Woong Kim in Australian & New Zealand Journal of Psychiatry
Footnotes
Author Contributions
H.A. wrote the first draft and was mainly responsible for data analysis. H.W.Y., D.J.O., J.B.B. and J.W.H. were responsible for data management and contributed to data interpretation. All authors contributed to data acquisition and critical review of the manuscript. K.W.K. had full access to all the data in the study and had final responsibility for the decision to submit for publication.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This research was supported by a fund [Grant No. 2019-ER6201-01] by Research of Korea Centers for Disease Control and Prevention, and by a grant from the Korean Health Technology R&D Project, Ministry of Health and Welfare, Republic of Korea (grant no. HI09C1379 [A092077]).
Data Availability
The data that support the findings of this study are available from the corresponding author upon reasonable request. The data are not publicly available due to their containing information that could compromise the privacy of research participants.
Supplemental Material
Supplemental material for this article is available online.
