Abstract
Objectives:
Evidence suggests that emotion regulation difficulty may play an important role in the association between life stress, sleep disturbance and depressive symptoms. We proposed two models depicting the possible moderating roles of prefrontal cortex activation during emotion regulation in the associations among these variables and tested them. We hypothesized that (1) the association between stress and sleep disturbance would differ across prefrontal cortex activation during emotion regulation (moderation model) and (2) the indirect effects of stress on depressive symptoms through sleep disturbance would depend on prefrontal cortex activation during emotion regulation (moderated mediation model).
Methods:
Forty-eight healthy adults without sleep disorders based on nocturnal polysomnography participated in this study. They received functional magnetic resonance imaging scans while performing an emotion regulation task. They also completed questionnaires assessing life stress, sleep disturbance and depressive symptoms. The proposed models were tested using the PROCESS macro for SPSS.
Results:
As hypothesized, there was a significant moderating effect of prefrontal cortex activation during emotion regulation on the association between life stress and sleep disturbance. Furthermore, right lateral prefrontal cortex activation had a moderating role in the indirect effect of life stress on depressive symptoms through sleep disturbance.
Conclusion:
These findings highlight the important role of prefrontal cortex function during emotion regulation in the associations between stress, sleep disturbance and depressive symptoms. Increasing lateral prefrontal cortex recruitment when regulating the emotional response to negative life events may be critical for the prevention and intervention of depression as well as sleep problems.
Introduction
Stress is a key risk factor for sleep problems, including long sleep latency and increased sleep fragmentation, and sleep disorders such as insomnia (Kalmbach et al., 2018; Kashani et al., 2012; Otsuka et al., 2017; Pillai et al., 2014; Vahtera et al., 2007). Despite evidence of a direct association between stress and sleep problems, it has been suggested that the impact of stress on sleep may be more dynamic and complicated (Pillai et al., 2014; van Schalkwijk et al., 2015). For example, not all individuals who experience life stress have sleep problems, with some factors making people more vulnerable to experiencing sleep problems. Therefore, it is essential to examine which factors moderate the association between life stress and sleep problems.
Several factors, including negative affectivity and coping style, appear to play moderating roles in the link between stress and sleep problems (Fortunato and Harsh, 2006; Kalmbach et al., 2018). Indeed, coping style moderated the association between stress and sleep quality (Sadeh et al., 2004). For instance, individuals with higher perceived stress levels reported more sleep problems (e.g. lower sleep quality) when they used high emotion-focused coping, which is known to be an ineffective coping strategy. Similar to coping style, emotion regulation, which is the ability to effectively manage emotional responses to stressful life events, may serve as an important factor that moderates the association between life stress and sleep problems. Emotion regulation plays an important role in dealing with negative responses to stress (Troy and Mauss, 2011) and is closely related to sleep disturbance (Kahn et al., 2013). However, a few studies have examined the moderating role of emotion regulation in the association between life stress and sleep disturbance. Emotion regulation may be particularly important for adjusting to high levels of life stress because stress impairs cognitive control, which is a necessary resource for emotion regulation. Cognitive control encompasses a wide range of cognitive processes, including attention, memory and inhibition to accomplish current tasks or goals (Botvinick and Braver, 2015). For example, people paid attention to information relevant to current tasks, but inhibited information irrelevant to these tasks. Emotion regulation requires some level of cognitive control, such as attention, action selection and inhibition (Joormann and Tanovic, 2015). Diminished cognitive control appears to be associated with difficulties in emotion regulation, whereas greater cognitive control is associated with more successful emotion regulation (Malooly et al., 2013; Zetsche et al., 2012).
Importantly, emotion regulation is often associated with activation in the prefrontal cortex (PFC) regions, including the lateral prefrontal cortex (LPFC) and medial prefrontal cortex (MPFC), which are implicated in cognitive control (Kober et al., 2008; Zilverstand et al., 2017). As noted above, emotion regulation requires some cognitive control that recruits PFC activation. This may indicate that emotion regulation and cognitive control share similar neural substrates, such as PFC regions. Furthermore, evidence suggests that stress causes changes in brain function, with a specific focus on PFC function (Arnsten, 2015; Scult et al., 2017). Given the negative impact of stress on cognitive control and PFC function, it is important to examine how PFC function involved in emotion regulation is associated with life stress and sleep problems. It is plausible that high levels of life stress may make it difficult for people to activate PFC regions, indicating difficulty in emotion regulation. Furthermore, life stress may have more detrimental effects on sleep problems when PFC activation is lower. That is, individuals who have more difficulty activating PFC regions when they encounter negative life events (i.e. high levels of life stress) are more likely to have greater sleep problems. However, relatively little is known about the moderating role of neurobiological factors (i.e. PFC activation) during emotion regulation in the link between life stress and sleep problems. Thus, this study examined whether PFC activation during emotion regulation moderated the association between life stress and sleep problems (moderation model).
Literature has shown that life stress is a significant risk factor for depression. For example, stressful life events predict the onset of depression (Vrshek-Schallhorn et al., 2015) and the severity of depressive symptoms in clinical and community samples (Muscatell et al., 2009; Sokratous et al., 2013). Sleep disturbance, including difficulty falling asleep and maintaining sleep, also appears to be a factor associated with an elevated risk of depression and greater depressive symptoms (Goldstone et al., 2020; Jackowska and Poole, 2017; Kalmbach et al., 2017; Maglione et al., 2014). Considering the associations among stress, sleep disturbance and depression, sleep disturbance may link stress and depression. Empirical studies have provided evidence that stress is linked to both physical (e.g. somatic symptoms) and mental health problems (e.g. depressive symptoms) through sleep disturbance (Chang et al., 2019; Mohr et al., 2003). Furthermore, deficits in emotion regulation and cognitive control as well as hypoactivity in PFC regions are known to be closely associated with depression (Joormann and Tanovic, 2015). More importantly, given our proposed moderation model that describes the role of PFC activation during emotion regulation in the association between life stress and sleep disturbance, we further proposed a moderated mediation model, in which PFC activation would moderate the indirect association between life stress and depressive symptoms through sleep disturbance (moderated mediation model).
Therefore, this study had two goals. First, we investigated whether the association between life stress and sleep disturbance differed across PFC activation during emotion regulation. That is, interactions between life stress and PFC activation could predict sleep disturbance (moderation model, Supplemental Figure S1). Second, we tested our expanded model, in which the association between life stress and depressive symptoms through sleep disturbance differed across PFC activation (moderated mediation model, Supplemental Figure S1). To achieve these goals, we assessed life stress levels, sleep disturbance and depressive symptoms using self-report measures and measured PFC activation during voluntary emotion suppression, which is an emotion regulation strategy that appears to activate prefrontal regions (Levesque et al., 2003; Ohira et al., 2006). Specifically, as used in Ohira et al. (2006), our emotion suppression strategy focused on suppressing the internal experience of negative emotions. To this end, our participants were instructed to suppress the internal and subjective emotional responses to negative pictures. We hypothesized that the association between life stress and sleep disturbance would differ across PFC activation during emotion regulation. More specifically, the association between life stress and sleep disturbance would be stronger when PFC activation is lower during emotion regulation. Furthermore, we predicted that the association between life stress and depressive symptoms through sleep disturbance would depend on the degree of PFC activation during emotion regulation. Therefore, significant indirect effects of life stress on depressive symptoms through sleep disturbance at lower PFC activation would be evident. However, such indirect effects would be weak or absent at higher PFC activation levels.
Method
Participants
Adult participants were recruited via advertisements at the Seoul National University Hospital. Participants were included if they were aged between 20 and 65 years. Adults with a wide range of life stress and sleep problems were also allowed to participate in this study to increase variability in scores of life stress and sleep disturbance. Participants were excluded if they were or had (1) a history of serious medical or neurological illness, (2) current medical or neurological illness, (3) Axis I psychiatric disorders, (4) sleep disorders, (5) shift workers, (6) borderline or antisocial personality disorders, (7) pregnant and (8) any contraindications (e.g. presence of metal objects including pacemakers and dental implants in the body) for magnetic resonance imaging (MRI) scans.
Procedures
This study was approved by the Institutional Review Board of Seoul National University Hospital. All participants provided written informed consent prior to participation. The participants visited our center on three occasions. During the first visit, they were assessed using the Structured Clinical Interview for Diagnostic and Statistical Manual of Mental Disorders–Fourth Edition (American Psychiatric Association, 1994) Axis I disorder (SCID) (First et al., 1997). Through the SCID, participants with psychiatric disorders were screened out. They were also asked to complete questionnaires assessing their life stress, sleep disturbance and depressive symptoms. During the second visit, participants underwent laboratory nocturnal polysomnography (PSG; Profusion PSG3; Compumedics, Abbotsford, Victoria, Australia). Participants with common sleep disorders such as obstructive sleep apnea (apnea-hypopnea index [AHI] >15) and periodic limb movement disorder (PLMD; periodic limb movement index [PLMI] >15/hour on PSG) were screened out based on the PSG assessment. During their third visit (within 2–3 weeks following the PSG), participants underwent a functional magnetic resonance imaging (fMRI) assessment while performing an emotion regulation task. Participants were given the task instructions and practiced the task prior to the fMRI assessment.
Self-report measures
Life stress was assessed using the Life Experiences Survey (LES) (Lee, 1993; Sarason et al., 1978) consisting of 47 items describing a wide range of stressful life events (e.g. death or illness of a family member, marital separation and financial problems) during the past 6 months. For each item, participants indicated whether they had experienced the life event and rated the degree of impact this event had on their life on a 7-point Likert-type scale ranging from extremely negative (−3) to extremely positive (+3). The impact ratings of negatively rated stressful life events were summed and reverse-coded. Thus, higher LES scores indicated higher levels of stress.
Sleep quality was assessed using the Pittsburgh Sleep Quality Index (PSQI) (Buysse et al., 1989; Sohn et al., 2012), which includes 19 items assessing several aspects of sleep based on seven components during the past month. The seven components are subjective sleep quality, sleep latency, sleep duration, habitual sleep efficiency, sleep disturbance, sleep medications and daytime dysfunction. The possible range of each component score was 0–3, and the sum of the component scores represents a PSQI global score of sleep quality ranging from 0 to 21. Thus, higher scores indicated lower sleep quality, such as greater sleep disturbance.
The Beck Depression Inventory (BDI) (Beck et al., 1961; Rhee et al., 1995) was used to assess depressive symptom severity during the previous 2 weeks. It is a self-report questionnaire with 21 items assessing various symptoms of depression, such as cognitive, emotional, physical and motivational symptoms using a 4-point Likert-type scale. Higher BDI scores indicated more severe depressive symptoms.
FMRI emotion regulation task
As in a previous study (New et al., 2009), each trial began with the presentation of a fixation cross for 1 second followed by negative socio-affective pictures or neutral pictures for 4 seconds. An emotion regulation cue was superimposed on the center of the picture for 1 second, and the picture continued to be displayed while emotions were regulated (i.e. suppressed or maintained) for the next 7 seconds. Then, the participants were asked to rate the intensity of their emotions using a scale ranging from 1 (slightly negative) to 3 (very negative) presented for 4 seconds, followed by a fixation dot for 4 seconds. In the ‘suppress’ condition, participants were asked to suppress/decrease their internal and subjective emotional responses to negative socio-affective pictures. In the ‘maintain’ condition, they were asked to maintain their responses to negative socio-affective and neutral pictures.
In total, 36 negative socio-affective pictures (18 pictures per each emotion regulation condition) and 18 neutral pictures (only for the ‘maintain’ condition) were used. These pictures, taken from the Korean Social Affective Visual Stimuli (K-SAVS) (Seok et al., 2018), have been validated as stimuli that induce socially relevant emotions in Koreans.
FMRI data acquisition and analyses
FMRI data acquisition and preprocessing
The fMRI data were acquired with a 3 T whole-body Tim Trio scanner (Siemens, Erlangen, Germany) using a 12-channel birdcage head coil and interleaved T2*-weighted echo planar imaging (repetition time [TR] = 2000 ms, echo time [TE] = 30 ms, flip angle = 90°, slice thickness = 4.0 mm, in-plane resolution = 3.4 mm × 3.4 mm, no gap, 33 axial slices, field of view [FOV] = 220 mm, 220 volumes). High-resolution structural images were acquired using a T1-weighted three-dimensional (3D) gradient-echo pulse sequence with magnetization-prepared rapid gradient-echo sequencing (TR = 1670 ms, TE = 1.89 ms, flip angle = 9°, slice thickness = 1.0 mm, in-plane resolution = 1.0 mm × 1.0 mm, FOV = 250 mm).
The fMRI data were preprocessed using SPM12 (Wellcome Trust Centre for Neuroimaging, London, UK). The data were slice time-corrected, motion-corrected, co-registered with the high-resolution structural image, spatially normalized to the Montreal Neurological Institute (MNI) template and smoothed using a 6-mm full-width at half-maximum Gaussian kernel. Co-registered and normalized fMRI data were visually inspected for quality control. Artifact Detection Tools (ART; www.nitrc.org/projects/artifact_detect/) were used to identify outlier volumes. Outliers with significant head motion for each participant were detected with >2-mm composite motion or larger global mean intensity (i.e. difference in global mean intensity across functional volumes >3 SD). The outlier volume did not exceed 15% of the total volume in any participant, so none were excluded from the final analyses. The outliers were also entered into the first-level general linear model (GLM) as nuisance regressors to remove possible artifacts.
FMRI data analyses
First-level GLM analyses were conducted for each participant. Five regressors pertaining to the presentation of pictures (two regressors: ‘looking at negative pictures’ and ‘looking at neutral pictures’) and the emotion regulation phase (three regressors: ‘suppressing emotion’, ‘maintaining emotion’ and ‘maintaining neutral’) were entered into the model. The fixation cross, rating phase and fixation dot were also modeled as regressors of no interest. Regressors were defined based on boxcar functions that were convolved with the canonical hemodynamic response function. Six head motion parameters and outliers were included in each participant’s GLM model to control for the effects of head motion and outliers. Based on previous research (Miller et al., 2018), the contrast image for emotion regulation was defined to identify brain regions showing greater activation when suppressing the emotional response to negative pictures compared to looking at negative pictures.
To test our moderation and moderated mediation models using PFC activation during emotion regulation, we extracted mean parameter estimates from each PFC region of interest (ROI) for the emotion regulation contrast (i.e. ‘regulating negative emotion > looking at negative pictures’ contrast). The PFC ROIs included the right LPFC, left LPFC and MPFC regions that were defined using automated anatomical labeling (AAL) (Tzourio-Mazoyer et al., 2002). The Bonferroni correction was used to correct for multiple comparisons for three PFC ROIs (i.e. corrected p = 0.05/3 ROIs = 0.017).
Statistical analyses
Statistical analyses were conducted using SPSS software (version 25.0; SPSS Inc., Chicago, IL, USA). First, correlation analyses and an independent sample t-test were conducted to test the association between age and each variable, as well as gender differences in the variables included in our models. Partial correlation analyses were conducted to explore whether the variables included in our models were correlated after controlling for age and gender. The PROCESS macro for SPSS (Hayes, 2012) was used to examine whether the association between life stress levels and sleep disturbance differed across PFC activation during emotion regulation (model 1; Supplemental Figure S1). Significant interactions between life stress and PFC activation were further tested using the Johnson–Neyman procedures implemented in PROCESS (Hayes, 2012). The moderated mediation model was also tested using the PROCESS macro to examine whether the indirect effects of life stress on depressive symptoms through sleep disturbance differed across PFC activation during emotion regulation (model 7; Supplemental Figure S1). All variables included in the two models, such as life stress and PFC activation, were mean centered prior to the analyses. Both models were tested after controlling for age and gender. These analyses were performed using bootstrapping (i.e. 95% bias-corrected bootstrap confidence intervals [CIs] for the indirect effects based on 10,000 bootstrap resamples). The indirect effect and index of moderated mediation were considered significant if the 95% bias-corrected CI did not include zero (Hayes, 2015; Preacher and Hayes, 2008).
Results
Participant characteristics
A total of 50 adult participants were initially recruited; however, two participants were excluded due to one having a sleep disorder (PLMD) and one due to poor imaging data quality. Thus, our final sample included 48 healthy adults (22 females; mean ± standard deviation (SD) age = 37.08 ± 11.56 years). Demographic characteristics and questionnaire scores are presented in Table 1. Age was positively correlated with PSQI scores (r = 0.38, p < 0.01). Older participants were more likely to report higher levels of sleep disturbance. There were significant gender differences in BDI scores and left LPFC activation, but not in any of the other variables. Female participants (M = 12.00, SD = 9.99) showed higher BDI scores than male participants (M = 5.35, SD = 5.58), t(46) = 2.78, p < 0.01. Female participants (M = 0.58, SD = 1.04) also showed less left LPFC activation during emotion regulation than male participants (M = 1.28, SD = 1.04), t(46) = 2.32, p < 0.05. The frequencies and percentages of the items (frequency ⩾1) in the LES are presented in Supplemental Table S1.
Demographic information and questionnaire scores.
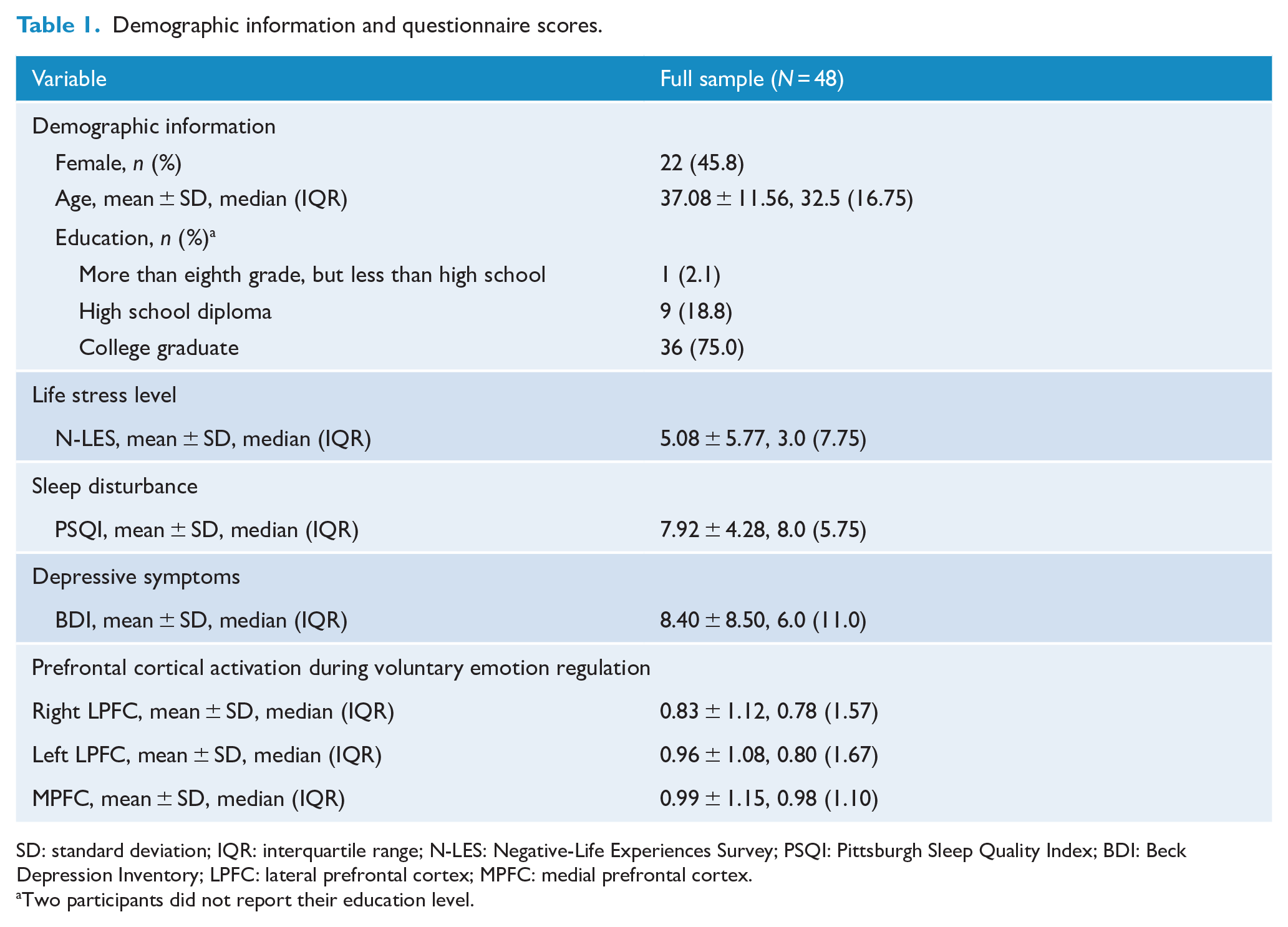
SD: standard deviation; IQR: interquartile range; N-LES: Negative-Life Experiences Survey; PSQI: Pittsburgh Sleep Quality Index; BDI: Beck Depression Inventory; LPFC: lateral prefrontal cortex; MPFC: medial prefrontal cortex.
Two participants did not report their education level.
Partial correlations between primary variables included in the models
The partial correlation coefficients between variables controlling for age and gender are presented in Supplemental Table S2. Life stress levels assessed by LES were positively correlated with PSQI scores (r = 0.56, p < 0.001) and BDI scores (r = 0.60, p < 0.001), respectively. Individuals with higher stress levels on the LES scale were more likely to have higher PSQI and BDI scores. PSQI scores were also positively associated with BDI scores (r = 0.60, p < 0.001). However, activation in the right and left LPFC, and MPFC during emotion regulation was not significantly associated with life stress levels on the LES scale, PSQI scores and BDI scores.
Moderation model result: the association between stress and sleep disturbance moderated by PFC activation during voluntary emotion regulation
As presented in Table 2a, right LPFC activation during emotion regulation significantly moderated the association between LES and PSQI scores after controlling for age and gender. This finding remained significant after correcting for multiple comparisons. Simple slope analyses using the Johnson–Neyman technique revealed that the association between life stress (LES) and sleep disturbance (PSQI scores) was stronger when right LPFC activation during emotion regulation was lower (−1D; b = 0.63, standard error (SE) = 0.11, p < 0.001) than when it was higher (+1D; b = 0.22, SE = 0.10, p = 0.03). Figure 1 depicts the interaction effect by presenting two simple slopes.
The results of the moderation analysis, controlling for age and gender.
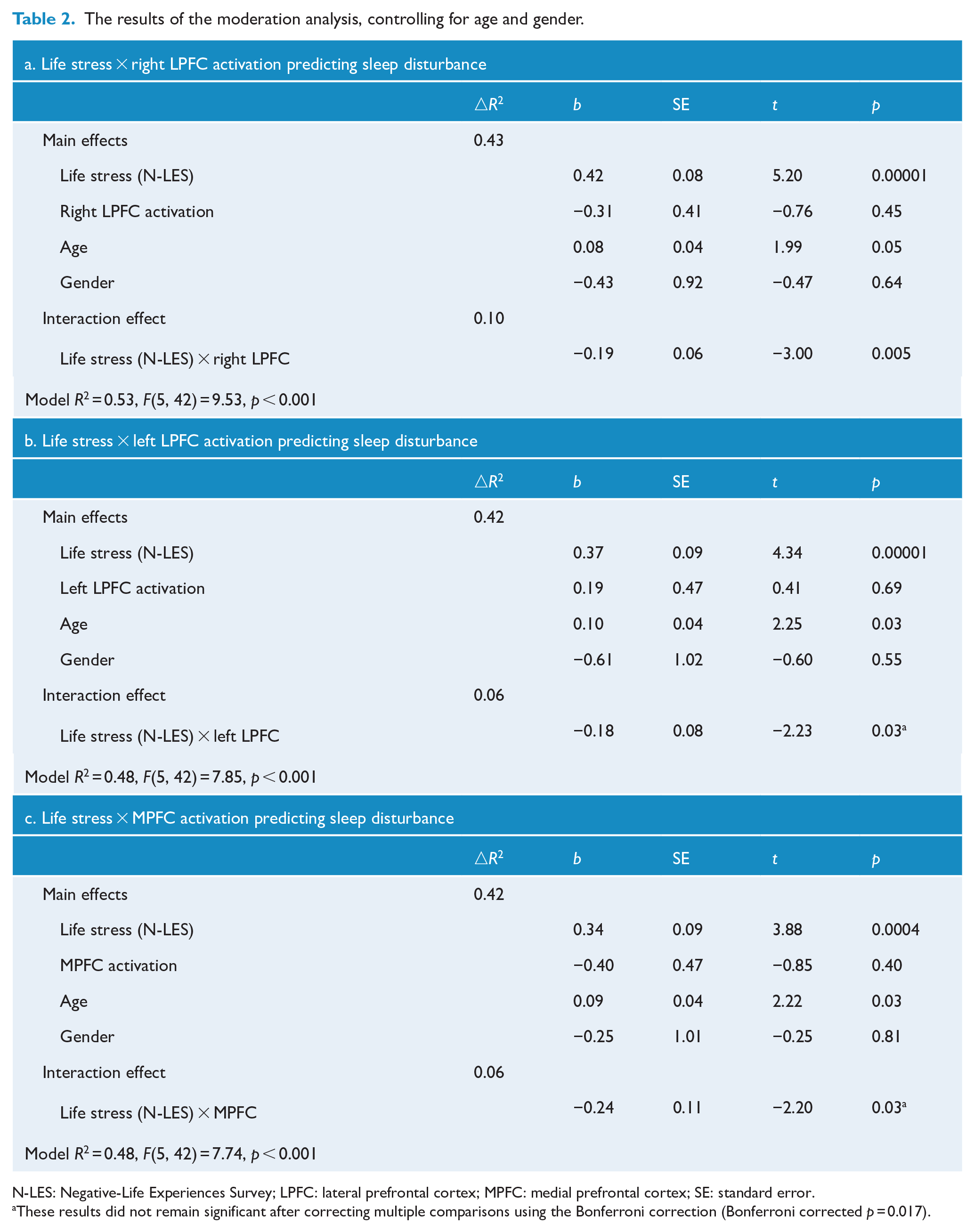
N-LES: Negative-Life Experiences Survey; LPFC: lateral prefrontal cortex; MPFC: medial prefrontal cortex; SE: standard error.
These results did not remain significant after correcting multiple comparisons using the Bonferroni correction (Bonferroni corrected p = 0.017).
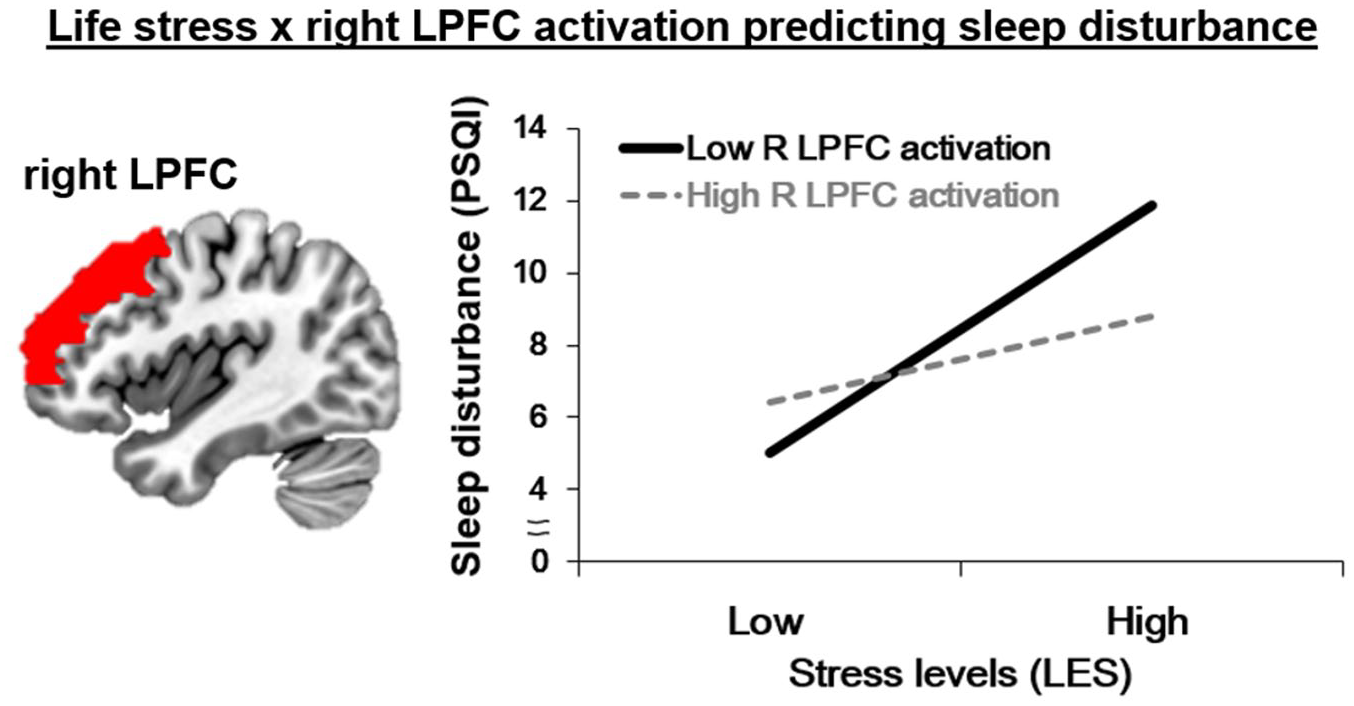
The results of the moderation model. Life stress × right PFC activation during voluntary emotion regulation predicting sleep disturbance.
Similar to the right LPFC, left LPFC and MPFC activation during voluntary emotion regulation significantly moderated the relationship between LES scores and PSQI scores after controlling for age and gender (Table 2b and 2c). However, these results were not significant after correcting for multiple comparisons. Simple slope analyses revealed similar interaction patterns. At lower left LPFC activation (−1 SD), higher levels of life stress were associated with greater sleep disturbance (b = 0.56, SE = 0.11, p < 0.001), but at higher left LPFC activation (+1 SD), life stress levels were not significantly associated with sleep disturbance (b = 0.18, SE = 0.13, p = 0.17) (Supplemental Figure S2(a)). At lower MPFC activation (−1 SD), higher levels of stress were associated with greater sleep disturbance (b = 0.62, SE = 0.13, p < 0.001), but at higher MPFC activation (+1 SD), life stress levels were not significantly associated with sleep disturbance (b = 0.07, SE = 0.17, p = 0.69) (Supplemental Figure S2(b)).
Moderated mediation model results: the indirect effects of life stress on depressive symptoms through sleep disturbance moderated by right LPFC activation
Given that only right LPFC activation moderated the association between LES and PSQI scores, the moderated mediation model was tested using right LPFC activation during voluntary emotion regulation. The results of the moderated mediation model are summarized in Table 3 and Figure 2. As hypothesized, the indirect effect of life stress (LES) on depression symptoms (BDI scores) through sleep disturbance (PSQI scores) was significantly moderated by right LPFC activation during voluntary emotion regulation (index of moderated mediation = −0.14, SE = 0.06, 95% CI = [−0.245, −0.020]). Given that the index of moderated mediation was negative, the indirect effect of life stress on depressive symptoms through sleep disturbance appeared to increase with decreasing right LPFC activation during emotion regulation. As shown in Table 3, the indirect effect was stronger when right LPFC activation was lower (0.48, 95% bootstrap CI [0.175, 0.804]) than when it was higher (0.12, 95% bootstrap CI [0.043, 0.415]) during emotion regulation.
The results of the moderated mediation analysis, controlling for age and gender.
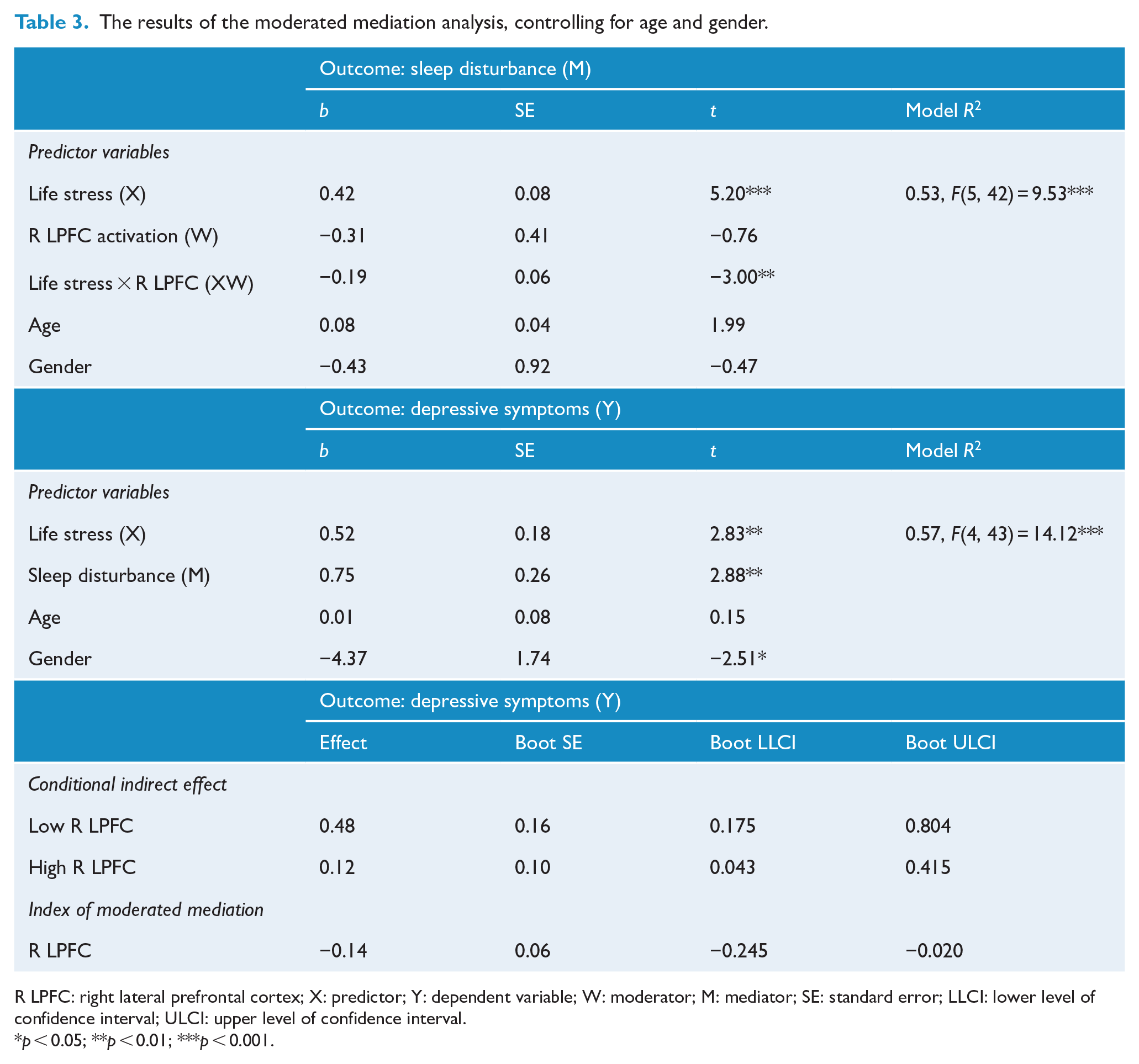
R LPFC: right lateral prefrontal cortex; X: predictor; Y: dependent variable; W: moderator; M: mediator; SE: standard error; LLCI: lower level of confidence interval; ULCI: upper level of confidence interval.
p < 0.05; **p < 0.01; ***p < 0.001.
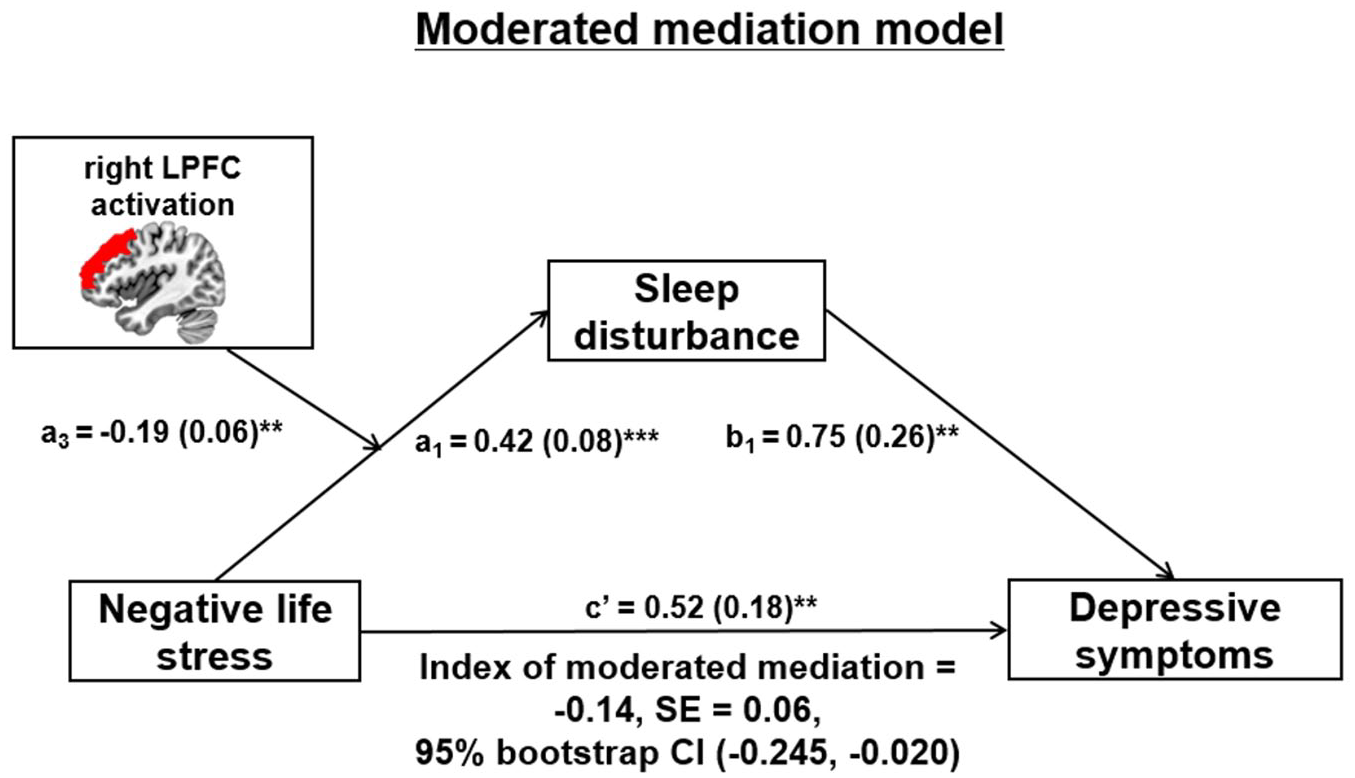
The results of the moderated mediation model. Unstandardized regression coefficients (standard errors [SE] in parentheses) were presented in the model.
Additional analyses
Behavioral ratings
A paired t-test was conducted to investigate whether subjective emotional responses to negative pictures after emotion regulation trials were lower than those after emotion maintenance trials. Lower emotional responses to negative pictures were reported for emotion regulation trials than for emotion maintenance trials, t(45) = 2.97, p < 0.01. This result indicated that emotion regulation used in this study contributed to successful emotion reduction in response to negative pictures.
Whole brain analysis
We conducted an exploratory whole brain analysis to examine whether PFC regions within our PFC ROIs showed greater activation during the regulation of emotions compared to looking at negative pictures. Multiple comparisons were corrected using the family-wise error rate (FWER) correction. This analysis revealed that the LPFC and MPFC regions were more activated when regulating emotions in response to negative pictures compared to looking at negative pictures (FWER corrected p < 0.05) (Supplemental Table S5 and Figure S3). This result confirmed that PFC regions were significantly activated by emotion regulation demands and provided supporting evidence for our PFC ROI selection.
Discussion
We examined the potential moderating roles of PFC function during emotion regulation in (1) the link between life stress and sleep disturbance (moderation model) and (2) the link between stress and depressive symptoms through sleep disturbance (moderated mediation model). We found that PFC activation during voluntary emotion regulation moderated the association between life stress and sleep disturbance, which was stronger when PFC activation was lower (i.e. greater emotion regulation difficulty) compared to when it was higher. Furthermore, right LPFC activation also moderated the indirect effect of life stress on depressive symptoms through sleep disturbance. The indirect effect was stronger when right LPFC activation was lower compared to when it was higher. Both results suggest that PFC activation during emotion regulation may play an important role in clarifying the complex associations between life stress, sleep disturbance and depressive symptoms.
The correlation results from this study support previous studies that showed significant associations between life stress, sleep problems and depression. First, consistent with the literature, life stress was significantly associated with greater sleep disturbance (Otsuka et al., 2017; Vahtera et al., 2007). Second, in line with past research that reported the negative impact of life stress on emotional and mental health problems such as depression, we found that life stress had a significant direct effect on depressive symptoms (Muscatell et al., 2009; Sokratous et al., 2013). These results emphasize that elevated stress due to negative life events may act as one of the important risk factors for both sleep disturbance and depression. Furthermore, there was a strong positive correlation between sleep disturbance and depressive symptoms. This is consistent with previous research showing that sleep disturbance is a critical risk factor for depression development and continuation (Jackowska and Poole, 2017; Kalmbach et al., 2017). Given that the associations between life stress and sleep disturbance, and between sleep disturbance and depressive symptoms were significant, we tested the mediation model that described the association between life stress and depressive symptoms through sleep disturbance. Consistent with the literature (Chang et al., 2019; Mohr et al., 2003), we found a significant indirect effect of life stress on depressive symptoms through sleep disturbance. This finding suggests that sleep disturbance may be one possible factor linking life stress and depressive symptoms in a healthy adult population.
In addition to such supporting evidence, we discovered novel findings that may contribute to the literature on the relationships between life stress, sleep disturbance and depressive symptoms. The novel findings were generated by examining the moderating roles of emotion regulation in (1) the connection between life stress and sleep disturbance and (2) the connection between life stress and depressive symptoms through sleep disturbance. Importantly, given that the LPFC and MPFC are interconnected and are core brain regions known to be involved in emotion regulation (Etkin et al., 2015; Rabinak et al., 2014), we assessed emotion regulation ability using neurobiological measures such as PFC activation during emotion regulation. First, in support of our hypothesis, we found that PFC activation during emotion regulation played a moderating role in the link between life stress and sleep disturbance. As hypothesized, life stress had a greater negative impact on sleep quality (i.e. greater sleep disturbance) when PFC activation was lower than when it was higher during emotion regulation. This result suggests that increasing emotion regulation capacity (i.e. PFC activation) when encountering negative life events may be important for decreasing sleep disturbance. However, given that the moderation effect of only right LPFC activation remained significant after applying stringent multiple comparisons correction and our relatively small sample size, our findings should be interpreted with caution.
Regarding the moderated mediation model, right LPFC activation during emotion regulation moderated the relationship between life stress and depressive symptoms via sleep disturbance. As predicted, the indirect effect of life stress on depressive symptoms through sleep disturbance differed across right LPFC activation during emotion regulation. This indirect effect was stronger at lower right LPFC activation than at higher right LPFC activation during emotion regulation. Individuals who experienced negative life events were more likely to have greater sleep disturbance and go on to have more severe depressive symptoms when they recruited lower right LPFC activation during emotion regulation. This finding underscores the role of emotion regulation assessed by PFC activation as a moderator in the relationship between life stress and depressive symptoms through sleep problems.
This study had some limitations. First, this study used a cross-sectional design that limited our ability to examine causal relationships between the variables, although the self-report measures assessed life stress, sleep quality and depressive symptoms during the preceding 6 months, 1 month and 2 weeks, respectively. To overcome this limitation, future research using a prospective longitudinal design is needed to investigate the causal relationships between these variables. Second, given that the LES measured life stress during the last 6 months, it is not clear whether the life stress that participants reported was acute or chronic. It has been suggested that the impact of acute life stress on health could be confounded by chronic life stress such as early life trauma (Hammen et al., 2009). Therefore, measuring both acute and chronic life stress may help to clarify similar or different impacts of acute and chronic stress on health problems such as sleep problems and depression. Third, we assessed sleep disturbance using the PSQI, which measures the subjective aspects of sleep quality. We collected one night of polysomnography as an objective measure of sleep quality. However, given the first-night effect of polysomnography (Newell et al., 2012), our polysomnographic data were not used to assess sleep quality, but to exclude participants with sleep disorders. There is evidence of subjective-objective sleep discrepancy (Rezaie et al., 2018); thus, future research is essential to replicate our findings by measuring both subjective (e.g. PSQI and sleep diaries) and objective sleep quality (e.g. actigraphy and consecutive nights of polysomnography). This may also help to deepen the understanding of how subjective and objective sleep quality is associated with life stress, depression and emotion regulation. Fourth, this study used emotion suppression as an emotion regulation strategy. Although the moderating role of PFC activation in the association between life stress, sleep disturbance and depressive symptoms may be true in the case of emotion suppression, further studies are needed to replicate our findings using other emotion regulation strategies, such as cognitive reappraisal. Finally, although PFC activation is supposed to represent emotion regulation capacity (Etkin et al., 2015; Rabinak et al., 2014), other measures of emotion regulation using self-report measures, such as the difficulties in emotion regulation scale, and psychophysiological measures, such as heart rate variability, may assist us in better understanding the role of emotion regulation as a moderator in the connection between life stress and sleep disturbance and in the connection of life stress to depression through sleep disturbance.
Despite these limitations, this study has several strengths. First, to the best of our knowledge, this is the first study to elucidate the moderating roles of PFC activation as emotion regulation in the associations between life stress and sleep disturbance, and between life stress and depressive symptoms via sleep disturbance. Thus, our findings underscore the importance of neurobiological factors measured using PFC activation during emotion regulation. Furthermore, this study helps clarify the complex relationships between life stress, sleep disturbance, depression and emotion regulation by testing two models. Finally, our findings have implications for both clinical and public health. First, to reduce the potential negative impact of life stress on sleep and emotional problems such as depression, emotion regulation should be given special attention. For example, individuals with high levels of sleep disturbance and depressive symptoms may need emotion regulation training and practice to deal with negative responses to stressful events. Repeated training and practice improves emotion regulation ability by engaging greater LPFC recruitment, which may be a promising neurobiological target for preventing and intervening in sleep and emotional problems. However, given that this study used one emotion regulation strategy (i.e. emotion suppression), caution is needed with respect to this conclusion and its clinical implications. Second, given that the LPFC is selected as a target region in neuromodulation treatments such as repetitive transcranial magnetic stimulation (Slotema et al., 2010), the moderating role of prefrontal activation may provide scientific evidence of such neuromodulation treatments that have been used for the treatment of stress-related sleep and emotional problems. Finally, during the COVID-19 pandemic crisis, which is now recognized as a major life stress, emotion regulation ability may be critical in the maintenance of good sleep, as well as physical and mental health. Overall, our results indicate the importance of maintaining or increasing the prefrontal function involved in emotion regulation and cognitive control during this global health crisis.
Supplemental Material
sj-pdf-1-anp-10.1177_00048674211025729 – Supplemental material for Life stress, sleep disturbance and depressive symptoms: The moderating role of prefrontal activation during emotion regulation
Supplemental material, sj-pdf-1-anp-10.1177_00048674211025729 for Life stress, sleep disturbance and depressive symptoms: The moderating role of prefrontal activation during emotion regulation by Kyung Hwa Lee, Ha Young Lee, Inkyung Park, Jeong Eun Jeon, Nambeom Kim, Seong Min Oh, Sehyun Jeon, Soohyun Kim, Yu Jin Lee and Seog Ju Kim in Australian & New Zealand Journal of Psychiatry
Footnotes
Data Availability Statement
The data that support the findings of this study are available from the corresponding authors upon reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by the Brain Research Program through the National Research Foundation of Korea (The Ministry of Science, ICT & Future Planning, Study no.: 2016M3C7A1904338) and the Technology Innovation Infrastructure Program through the Korea Institute for Advancement of Technology (The Ministry of Trade, Industry and Energy, Study no.: P0014279).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
